Abstract
Purpose
Previous studies suggest that triglyceride-glucose index (TyG) index, as a marker of insulin resistance, may have associations with the risk of cardiovascular diseases (CVD) in elderly population. Given the paucity of data, it remains controversial, especially in general Chinese population. We aimed to further assess whether TyG index is an independent risk factor for CVD.
Methods
We conducted a prospective cohort study that enrolled a total of 96,541 participants from the Kailuan Study. TyG index was calculated as ln (fasting triglyceride [mg/dL] × fasting glucose [mg/dL]/2). Participants were divided into four groups (Q1, Q2, Q3, and Q4) by quartiles of the TyG index. Any CVD events occurred during 2006–2017 were recorded, including myocardial infarction (MI) and stroke. We assessed the association of TyG index with the risk of CVD and the subtypes of CVD by using Cox models estimated hazard ratios (HRs) and 95% confidence interval (CIs).
Results
During a median follow-up of 10.33 years, totally 6421 CVD events, 1493 MIs, and 5083 stroke events occurred. Multivariate Cox regression analysis showed that compared with Q1, HR (95% CI) for CVD events was 1.12 (95%, 1.03–1.21) in Q2, 1.28 (95%, 1.18–1.38) in Q3, and 1.34 (95%, 1.23–1.45) in Q4. In a time-dependent Cox Model we also found that compared with Q1, HR (95% CI) for CVD events was 1.09 (95%, 1.02–1.18) in Q2, 1.18 (95%, 1.09–1.27) in Q3, and 1.20 (95%, 1.11–1.30) in Q4. Similar results were showed in MI and stroke.
Conclusions
TyG index as a marker of insulin resistance was an independent risk factor for CVD. This may help in the early identification of people at high risk of CVD and be applicable to the primordial and primary prevention.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The latest report shows that cardiovascular disease (CVD) is the leading cause of death in the world’s population [1]. Nearly 290 million patients are now suffering from CVD in China and the prevalence of CVD is continuously rising [2]. The interventions and control for traditional risk factors of hypertension, smoking makes the prevention and control of CVD achieve initial results. However, the prevalence of CVD will continuously rise due to the emerging epidemic of obesity and diabetes and some uncovered factors.
Insulin resistance (IR) has been widely demonstrated to be an important risk factor for the emergence and development of CVD, because it leads to vascular damage [3]. Moreover, there is a causal relationship between triglycerides and CVD that higher levels indicating elevated risk in incidence [4,5,6]. The triglyceride-glucose (TyG) index, which was calculated as ln (fasting triglyceride [mg/dL] × fasting glucose [mg/dL]/2), has been a reliable surrogate marker of IR [7,8,9]. Zhao et al. has found that the TyG index was significantly associated with arterial stiffness and microvascular damage [10, 11]. Irace et al. found that the TyG index was associated with carotid atherosclerosis [12]. The VMCUN cohort study found greater predictive value of TyG index for CVD [13]. Nevertheless, studies on the relationship between TyG index and CVD remains insufficient, especially in the Chinese general population. We aimed to investigate the associations between TyG index and the risk of CVD through the Kailuan Study.
Methods
Study population
The Kailuan study is large, community-based and prospective cohort study, which followed the guidelines of Helsinki Declaration and was approved by the Ethics Committees of the Kailuan General Hospital. The detailed design of the Kailuan study have been described previously [14]. Health examinations were performed in 2-year intervals up since 2006, including questionnaire assessments, physical examination and laboratory test. 101,510 participants who participated in the survey in 2006 were enrolled. Exclusion criteria were as followed: (I) missing data of TG and FBG levels (n = 1305), (II) with history of cancer (n = 377), (III) with history of CVD (n = 3263), and (IV) Body mass index (BMI) >45 kg/m2 (n = 24). 96,541 participants were eventually enrolled in our study.
Date collection and definitions
Information on demographic and clinical characteristics (age, sex, lifestyle, and past medical history, etc.) were collected using a self-reported questionnaire, as detailed elsewhere [15]. Education level was classified as primary school or below, middle school, and high school or above. Smoking and drinking status were classified as never, former and current. Active physical activity was defined as “>4 times per week and 20 min at time”. BMI was calculated as the weigh (kg)/height2 (m2).
Elbow venous blood samples of 5 mL were collected into an anticoagulant tube containing EDTA between 7:00–9:00 am after overnight fasting for at least 8 h, and the serum was collected after centrifugation at 3000 × g for 10 min. The supernatant was measured within 4 h. All biochemical measurement including total cholesterol (TC), TG, high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), high-sensitive C-reactive protein (Hs-CRP), FBG, and Uric acid (UA), and etc. was measured on the Hitachi 747 autoanalyzer (Hitachi, Tokyo, Japan).
Hypertension was defined as SBP ≥ 140 mmHg or DBP ≥ 90 mmHg, a self-reported history of hypertension, or any use of antihypertensive medication. Diabetes was defined as FBG ≥ 7.0 mmol/L, a self-reported history of diabetes, or use of antidiabetic medication. Dyslipidemia was defined as TC ≥ 5.17 mmol/L or TG ≥ 1.69 mmol/L or LDL-C ≥ 3.62 mmol/L or HDL-C ≤ 1.04 mmol/L, any self-reported history of dyslipidemia, or use of lipid-lowering drugs.
Calculation of TyG index
TyG index = Ln [TG (mg/dL) × FBG (mg/dL)/2] [16]. Since 2006, TG and FBG were measured every 2 years. The baseline TyG index was calculated from the TG and FBG values of the first health examination. We also entered the TyG index as a time-dependent explanatory variable, since it might change over time. The calculation of TyG index included all available data from baseline to the year prior to CVD occurred or to the end of follow-up. Antidiabetic medication and other time-dependent covariates such as BMI were entered into the model in the same way.
Definition of CVD and follow-up
According to the World Health Organization criteria, definition of CVD included myocardial infarction (MI) and stroke [17, 18]. Participants were followed from 2006 until CVD or death, whichever came first, otherwise until the end of the follow-up period, 2017. Assessment of CVD and death has been previously described in detailed [19]. CVD events are determined by a panel of three experienced physicians who review medical records and adjudicate cases annually. Information on deaths was collected from death certificates from the National Office of Vital Statistics.
Statistical analysis
Continuous, normally distributed variables were presented as the mean ± standard deviation (SD), and groups were compared using one-way ANOVA. Measurements of distribution anomalies were expressed as medians (Q25, Q75) and compared with Kruskal–Wallis rank sum test. Category variables were presented by number and percentage (%), with comparing between groups by chi-square test. Restricted cubic splines were performed to examine the nonlinear relationship between TyG index and CVD events with four knots (at the 5, 25, 75, and 95th percentiles) [20]. Participants were divided into four groups (Q1, Q2, Q3, and Q4) by TyG index quartiles and the Q1 group served as control.
The Cox model and the time-dependent Cox model were used to assess the association TyG index with the risk of CVD by calculating the HR and 95% CI. A Cause-specific hazard function (CS) model, one of competing risk model, was used to assess the association TyG index with the risk of CVD [21]. The HRs were also estimated for per-unit TyG index change in CVD. Model 1 adjusted for age and sex. Model 2 was adjusted as model 1, adding current smoking and drinking status, physical activity, education, BMI, hypertension, diabetes, HDL-C, LDL-C, hs-CRP, lipid-lowering drugs, antidiabetic drugs, and antihypertensive drugs. We conducted subgroup analysis on the basis of diabetes mellitus to assess the association between TyG index and CVD events. All the analyses were conducted using SAS version 9.4 (SAS Institute Inc., Cary, NC). A two-sided P < 0.05 was considered statistically significant.
Results
Baseline characteristics
The population included 96,541 participants in the current study. Among them, the mean age of the study participants was 51.19 ± 12.57 years, and 76,858 (79.61%) were men. There is increasing trend of the indicators except for the high school or above from Q1 to Q4 groups. Baseline characteristics of participants according to the quartile are presented in Table 1.
Associations between the quartiles of TyG index with the risk of CVD
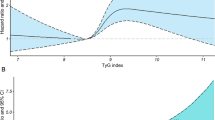
During a median follow-up of 10.33 years, we observed 6421 CVD events (MI events: 1493, Stroke:5083). From Q1 to Q4, the incidence of CVD increased per 1000 person-years, with Q4 was the highest (8.66/1000 person-years) (Table 2). Compared with Q1, the HRs (95% CIs) for CVD events were 1.12 (1.03–1.21) in Q2 group, 1.28 (1.18–1.38) in Q3 group, 1.34 (1.23–1.45) in Q4 group, after adjustment for confounding factors in the model 2 (Table 2). It appeared that the risk of CVD was higher in per-unit increasing of TyG index. Similar results were also appeared in MI, stroke and ischemic stroke except for a non-significantly increased risk of hemorrhagic stroke (Table 2). 7414 competing events (non-CVD deaths) occurred among participants during this follow-up. The result of the Cox model is similar with the competing risk model (Table 2). Multivariable adjusted spline regression model showed a nonlinear relationship between TyG index and the risk of CVD (Fig. 1).
Associations between the quartiles of TyG index and the risk of CVD with time-dependent variables
Some individuals originally in Q1 or Q2 groups at baseline might move to Q3 or Q4 during our follow-up, we entered quartiles of TyG index as a time-dependent Cox model, in which at each point in time, the last available TyG index was considered (Table 3). Lipid-lowering medication and other covariates changed over time were also entered into model as a time-dependent covariate. In this time-dependent Cox model, the HRs (95% CIs) for CVD were 1.09 (1.02–1.18) in Q2 group, 1.18 (1.09–1.27) in Q3 group, and 1.20 (1.11–1.30) for in group (Table 3). The risk of CVD was higher in per-unit increasing of TyG index. Similar results were found in MI, stroke and ischemic stroke and a non-significantly increased risk of hemorrhagic stroke (Table 3).
Subgroup analysis
In subgroup analysis, Cox models were recalculated in participants with and without diabetes mellitus (Supplementary Table 1). Higher quartiles of TyG index were associated with an increased risk of CVD in participants without diabetes mellitus. The highest quartile of TyG index were associated with an increased risk of CVD and ischemic stroke in participants with diabetes mellitus. There was no interaction between TyG index and diabetes mellitus status for the risk of outcomes (P for interaction >0.05).
Cox models were recalculated in participants who were younger than 60 years and 60 years or older (Supplementary Table 2). Highest quartiles of TyG index were associated with an increased risk of CVD in participants younger than 60 years and older than 60 years. We found similar results in MI and ischemic stroke. There was no interaction between TyG index and age for the risk of outcomes (P for interaction >0.05).
Discussion
We found that TyG index were associated with an increased risk of CVD (MI, stroke, and ischemic stroke). Similar results were found in the association of the time-dependent TyG index with CVD. It indicated both a high long-term TyG index and short-term TyG index were associated with the increase risk of CVD in general Chinese population.
In a 10.33-year follow-up, we indicated that TyG index was an independent risk factor of CVD, with its elevation indicated a higher risk of CVD. Classic Cox model normally identified outcomes as sole events without considering the competing risk of death, and whether participants died of CVD remaining unknown, hence the bias produced [22]. We then used competing risk model to assess the association TyG index with the risk of CVD. Relatively small difference showed between classic Cox models and competing risk models. The death might not make effects in the association of TyG index with CVD events in general.
In addition, we indicated that the short-term TyG index changes were an independent predictor of CVD. Since many participants, originally in low quartiles at baseline, might move to high quartiles during the follow-up, we entered TyG index as a time-dependent explanatory variable in the time-dependent Cox models. And the antidiabetic medication and other covariates (such as BMI) that changed over time were entered into the model as time-dependent covariates. Compared with the lowest level of TyG index (Q1), Q2, Q3, and Q4 of TyG index were found to be associated with 1.09-, 1.08-, and 1.20-fold risks of CVD, respectively. Classic Cox model focused on long-term effects on outcomes from the exposure information at baseline. However, the time-dependent Cox models focused more on short-term effects of time-varying exposures to outcomes, and could explore the association between exposure and outcomes through the changes of exposure information [23]. In conclusion, the TyG index represents an independent risk factor for CVD either long or short-term effects.
Similar results were obtained in previous studies. In a cohort study including 6078 Chinese older adults (age 60 years or older), Li et al. investigated that Q3, Q4 of TyG index were associated with 1.33- and 1.72-fold risks of CVD compared with the lowest level of TyG index [24]. Since its study population comprised of adults aged 60 or older, who were originally at a higher CVD risk, the select bias was inevitable. Our study covered different ages range from the young to the old and found that TyG index was associated with the increased risk of CVD. And individuals younger than 60 years of age had a higher risk of CVD than individuals at age 60 or older. The prevalence of CVD for youth and middle age was increasing year on year from China Cardiovascular Diseases Report 2018. Another study showed that the association between type 2 diabetes and relative risk of CVD was stronger in younger type 2 diabetics [25]. In this way, TyG index could be used as an important indicator to assess the risk of CVD, especially in the young population, by identifying people at high risk of CVD. Lifestyle and pharmacological interventions may help prevent CVD.
Current studys have shown that TyG index is an independent risk factor for CVD, while we found that the association between TyG index and MI or stroke was different, the influence of TyG index on MI is higher than that on stroke. We also found that increasing risk of stroke was association with ischemic but not hemorrhagic stroke. This might be due to stronger IR related to endothelial dysfunction [26, 27], and it could lead to atherosclerosis and ischemic stroke through IR [28]. Therefore, energy balance and metabolism might be also effected by IR in the heart during vessel endothelial damage, which eventually resulted in lipotoxicity [29].
As living standards and lifestyle improve, the prevalence of diabetes and dyslipidemia is gradually increased. The prevalence of dyslipidemia has increased from 18.6% in 2002 year to 40.4% in 2012 year, and the prevalence of diabetes has increased from 4.5% in 2002 year to 10.4% in 2012 year. The status which glucose and lipid metabolism are abnormal and prevalence increase is significant. The control rate in dysglycemia and dyslipidemia in China is lower than in western developed countries. The prevention and treatment of dyslipidemia is more backward, the prevention and control of CVD is not optimistic.
The shortcomings of this study may have the following points. Firstly, we attempted to introduce as many potential confounders as possible into the analysis, but factors such as environmental varying and genetic factors were not collected due to the study design. Secondly, the lifestyle and drugs data were collected based on self-reported, which might lead to recall biases. Finally, due to unable to assess IR (HOMA), we could not compare the role of the TyG index and HOMA-IR in onset of CVD. The predictive effect of the TyG index and HOMA index on CVD need to be further compared in future.
The findings from the present show that the TyG index is an independent risk factor for CVD in the study with up to 10.33 years follow-up. When compared to different parameters including IR, the clinical application of the TyG index is suitable for real-life work due to be easy to measure and calculate. Hence, the TyG index may help in the early identification of people at high risk of CVD, intervention comprising lifestyle and medication in early life may contribute to prevent and control of CVD.
Data availability
De-identified data are available to researchers upon request by contacting with the corresponding author.
References
GBD 2017 Causes of Death Collaborators. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 392(10159), 1736–1788 (2018). https://doi.org/10.1016/S0140-6736(18)32203-7
C. Han, F. Liu, X. Yang et al. Ideal cardiovascular health and incidence of atherosclerotic cardiovascular disease among Chinese adults: the China-PAR project. Sci. China Life Sci. 61(5), 504–514 (2018). https://doi.org/10.1007/s11427-018-9281-6
V. Ormazabal, S. Nair, O. Elfeky, C. Aguayo, C. Salomon, F.A. Zuñiga, Association between insulin resistance and the development of cardiovascular disease. Cardiovasc Diabetol. 17(1), 122 (2018). https://doi.org/10.1186/s12933-018-0762-4
R. Scicali, P. Giral, L. D’Erasmo et al. High TG to HDL ratio plays a significant role on atherosclerosis extension in prediabetes and newly diagnosed type 2 diabetes subjects. Diabetes Metab. Res Rev. 37(2), e3367 (2021). https://doi.org/10.1002/dmrr.3367
B.J. Arsenault, J.S. Rana, E.S. Stroes et al. Beyond low-density lipoprotein cholesterol: respective contributions of non-high-density lipoprotein cholesterol levels, triglycerides, and the total cholesterol/high-density lipoprotein cholesterol ratio to coronary heart disease risk in apparently healthy men and women. J. Am. Coll. Cardiol. 55(1), 35–41 (2009). https://doi.org/10.1016/j.jacc.2009.07.057
A. Varbo, M. Benn, A. Tybjærg-Hansen, A.B. Jørgensen, R. Frikke-Schmidt, B.G. Nordestgaard, Remnant cholesterol as a causal risk factor for ischemic heart disease. J. Am. Coll. Cardiol. 61(4), 427–436 (2013). https://doi.org/10.1016/j.jacc.2012.08.1026
M. Zhang, B. Wang, Y. Liu et al. Cumulative increased risk of incident type 2 diabetes mellitus with increasing triglyceride glucose index in normal-weight people: The Rural Chinese Cohort Study. Cardiovasc Diabetol. 16(1), 30 (2017). https://doi.org/10.1186/s12933-017-0514-x
W.Y. Su, S.C. Chen, Y.T. Huang et al. Comparison of the effects of fasting glucose, hemoglobin A1c, and triglyceride-glucose index on cardiovascular events in Type 2 diabetes mellitus. Nutrients 11(11), 2838 (2019). https://doi.org/10.3390/nu11112838
A. Sánchez-García, R. Rodríguez-Gutiérrez, L. Mancillas-Adame et al. Diagnostic accuracy of the triglyceride and glucose index for insulin resistance: a systematic review. Int J. Endocrinol. 2020, 4678526 (2020). https://doi.org/10.1155/2020/4678526
S.B. Lee, C.W. Ahn, B.K. Lee et al. Association between triglyceride glucose index and arterial stiffness in Korean adults. Cardiovasc. Diabetol. 17(1), 41 (2018). https://doi.org/10.1186/s12933-018-0692-1
S. Zhao, S. Yu, C. Chi et al. Association between macro- and microvascular damage and the triglyceride glucose index in community-dwelling elderly individuals: the Northern Shanghai Study. Cardiovasc. Diabetol. 18(1), 95 (2019). https://doi.org/10.1186/s12933-019-0898-x
C. Irace, C. Carallo, F.B. Scavelli et al. Markers of insulin resistance and carotid atherosclerosis. A comparison of the homeostasis model assessment and triglyceride glucose index. Int J. Clin. Pract. 67(7), 665–72 (2013). https://doi.org/10.1111/ijcp.12124
L. Sánchez-Íñigo, D. Navarro-González, A. Fernández-Montero, J. Pastrana-Delgado, J.A. Martínez, The TyG index may predict the development of cardiovascular events. Eur. J. Clin. Investig. 46(2), 189–97 (2016). https://doi.org/10.1111/eci.12583
X. Ma, H. Cui, M. Sun et al. Fasting blood glucose, cholesterol, and risk of primary liver cancer: the kailuan study. Cancer Res. Treat. (2021). https://doi.org/10.4143/crt.2020.817
M. Sun, W. Wang, X. Liu et al. Total cholesterol, alanine aminotransferase and the risk of primary liver cancer: a population-based prospective study. Medicine 100(18), e25746 (2021). https://doi.org/10.1097/MD.0000000000025746
X. Ma, L. Dong, Q. Shao et al. Triglyceride glucose index for predicting cardiovascular outcomes after percutaneous coronary intervention in patients with type 2 diabetes mellitus and acute coronary syndrome. Cardiovasc. Diabetol. 19(1), 31 (2020). https://doi.org/10.1186/s12933-020-01006-7
Stroke-1989, Recommendations on stroke prevention, diagnosis, and therapy. Report of the WHO task force on stroke and other cerebrovascular disorders. Stroke 20(10), 1407–31 (1989). https://doi.org/10.1161/01.str.20.10.1407
K. Thygesen, J.S. Alpert, A.S. Jaffe et al. Fourth universal definition of myocardial infarction (2018). J. Am. Coll. Cardiol. 72(18), 2231–2264 (2018). https://doi.org/10.1016/j.jacc.2018.08.1038
S. Wu, S. An, W. Li et al. Association of trajectory of cardiovascular health score and incident cardiovascular disease. JAMA Netw. Open 2(5), e194758 (2019). https://doi.org/10.1001/jamanetworkopen.2019.4758
L. Desquilbet, F. Mariotti, Dose-response analyses using restricted cubic spline functions in public health research. Stat. Med. 29(9), 1037–57 (2010). https://doi.org/10.1002/sim.3841
B. Lau, S.R. Cole, S.J. Gange, Competing risk regression models for epidemiologic data. Am. J. Epidemiol. 170(2), 244–56 (2009). https://doi.org/10.1093/aje/kwp107
S.D. Berry, L. Ngo, E.J. Samelson, D.P. Kiel, Competing risk of death: an important consideration in studies of older adults. J. Am. Geriatr. Soc. 58(4), 783–7 (2010). https://doi.org/10.1111/j.1532-5415.2010.02767.x
S. Wu, Y. Song, S. Chen et al. Blood pressure classification of 2017 associated with cardiovascular disease and mortality in young Chinese adults. Hypertension 76(1), 251–258 (2020). https://doi.org/10.1161/HYPERTENSIONAHA.119.14239
S. Li, B. Guo, H. Chen et al. The role of the triglyceride (triacylglycerol) glucose index in the development of cardiovascular events: a retrospective cohort analysis. Sci. Rep. 9(1), 7320 (2019). https://doi.org/10.1038/s41598-019-43776-5
M. Zhao, L. Song, L. Sun et al. Associations of Type 2 diabetes onset age with cardiovascular disease and mortality: the Kailuan study. Diabetes Care. 44(6), 1426–32 (2021). https://doi.org/10.2337/DC20-2375
A. Janus, E. Szahidewicz-Krupska, G. Mazur, A. Doroszko, Insulin resistance and endothelial dysfunction constitute a common therapeutic target in cardiometabolic disorders. Mediators Inflamm. 2016, 3634948 (2016). https://doi.org/10.1155/2016/3634948
M. Montagnani, I. Golovchenko, I. Kim et al. Inhibition of phosphatidylinositol 3-kinase enhances mitogenic actions of insulin in endothelial cells. J. Biol. Chem. 277(3), 1794–9 (2002). https://doi.org/10.1074/jbc.M103728200
J.A. Kim, M. Montagnani, K.K. Koh, M.J. Quon, Reciprocal relationships between insulin resistance and endothelial dysfunction: molecular and pathophysiological mechanisms. Circulation 113(15), 1888–904 (2006). https://doi.org/10.1161/CIRCULATIONAHA.105.563213
Y.T. Zhou, P. Grayburn, A. Karim et al. Lipotoxic heart disease in obese rats: implications for human obesity. Proc. Natl Acad. Sci. USA. 97(4), 1784–9 (2000). https://doi.org/10.1073/pnas.97.4.1784
Acknowledgements
The authors thank all the members of the Kailuan Study Team for their contribution and the participants who contribution their data. All authors revised and approved the article as submitted.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics approval
This study followed the Declaration of Helsinki and was approved by the Ethics Committee of the Kailuan Medical Group.
Consent to participate
All participants signed an informed consent.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Liu, Q., Cui, H., Ma, Y. et al. Triglyceride-glucose index associated with the risk of cardiovascular disease: the Kailuan study. Endocrine 75, 392–399 (2022). https://doi.org/10.1007/s12020-021-02862-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-021-02862-3