Abstract
The diagnostic imaging contributes significantly to the diagnosis and management of sarcoidosis. Imaging techniques are widely employed in the assessment of thoracic and extra-thoracic involvement from sarcoidosis. For the diagnosis of sarcoidosis, chest radiograph has been the cornerstone of sarcoidosis since 1961, when Scadding proposed a standardized staging system. Currently, computed tomography (CT) represents the reference standard for the assessment of both mediastinal lymph nodes and pulmonary findings. In particular, high-resolution computed tomography (HRCT) is more accurate compared to chest radiography for the detection of subtle parenchymal involvement, and provides comprehensive overview of anatomical detail and abnormalities of lung structures. Notably, HRCT allows for accurate differentiation between reversible and irreversible lung disease, which is cornerstone of prognostication. Radionuclide imaging (gallium-67 and 18F-fluorodeoxyglucose) provides information about activity of the disease and is also useful for diagnostic workup of patients with unexplained persistent disabling symptoms. Magnetic resonance is sensitive for the detection of sarcoidosis granulomata within myocardium, thus providing detailed roadmap for biopsy. For the management of sarcoidosis, CT is of paramount importance in the detection and differential of most common complications, such as vascular disease and suspicious nodular lesions. Conversely, the role of CT in the follow-up of asymptomatic subjects is still under debate. This review focuses on the role of diagnostic imaging in the diagnosis and follow-up of sarcoidosis.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The diagnosis of sarcoidosis requires compatible clinical and imaging features, the identification of noncaseating granulomas in at least one organ, and the exclusion of other causes of granulomatous disorders [1]. Therefore, imaging contributes significantly to the diagnosis and, also, the management of patients with sarcoidosis. Chest radiograph may be often sufficient to establish the diagnosis of sarcoidosis [2]. However, the variable clinical-radiographic manifestations as well as the disease complications have substantially lead clinicians to increasingly use computed tomography (CT) [3]. Furthermore, the role of radionuclide imaging has reemerged in the assessment of the disease activity throughout the body [4]. Moreover, magnetic resonance imaging (MRI) is of great value in the detection of noncaseating granuloma, notably within myocardium [5]. This review focuses on the utility of these imaging tools in the diagnosis and follow-up of sarcoidosis.
Imaging Characteristics of Sarcoidosis
Chest Radiograph
The chest radiograph (CXR) can be sufficient for the diagnosis of sarcoidosis in many patients with suspected disease on the basis of consistent clinical context [1, 2]. Indeed, the chest radiograph of patients with sarcoidosis is abnormal in the vast majority of cases [6].
On CXR, sarcoidosis is most often characterized by bilateral, symmetrical hilar, and paratracheal lymphadenopathy (Fig. 1). Hilar lymph node enlargement ranges from the barely detectable to the massive and give the hila a lobulated and usually well-demarcated outline. Symmetry is the hallmark of hilar adenopathy associated with sarcoidosis, especially because this pattern is unusual in the major diagnostic alternatives such as lymphoma, tuberculosis, and metastatic disease [7, 8]. Nodal calcification may be noted, though its detection and characterization is better appreciated on CT [3].
a–b Postero-anterior chest radiograph in a patient with sarcoidosis. a Bilateral symmetric hilar enlargement and right paratracheal lymphadenopathy (stage I). b Bilateral hilar enlargement is seen along with upward retraction of hila. Reticular opacities and traction bronchiectasis are disclose chronic involvement of lung parenchyma (stage IV)
The characteristic patterns of parenchymal involvement include reticulonodular change, ground-glass opacity, alveolar opacities, large nodular opacities, and irreversible fibrotic change [9]. The clinical impact of radiographic assessment stands in the disclosure of signs of pulmonary fibrosis, such as coarse linear opacities or consolidation radiating from the hilum into the adjacent middle and upper zones, often associated with upward hilar retraction along with vascular and fissural distortion (Fig. 2) [10]. However, as with other interstitial diseases, a normal chest radiograph does not exclude pulmonary sarcoidosis (Fig. 3). Furthermore, radiographic findings are atypical in approximately 20 % of cases and are more frequent in patients over the age of 50 years [11, 12].
a–d Chest radiograph and CT showing parenchymal fibrosis. a Postero-anterior chest radiograph shows symmetrical bilateral hilar enlargement and parenchymal reticulation with predominant upper-mid lung involvement. b–d Transverse CT images of the chest show reticulonodular findings in the lung parenchyma, with involvement of fissures and bronchiovascular bundles. Notably, irregular vascular edge is observed in relation to small nodules reflecting granulomata along peribronchovascular lymphatics
a–d Negative chest radiograph and positive HRCT in patient with micronodular pattern. a Postero-anterior chest radiograph shows hilar enlarged lymph nodes; no parenchymal abnormalities can be noted. b–d Transverse CT images of the chest reveal perilymphatic miconodules distributed along fissures (arrows) and bronchovascular bundles
A variable combination of parenchymal and nodal involvement may be seen in sarcoidosis. Several radiographic staging systems for sarcoidosis have been reported. The most widely adopted system was developed by Scadding more than five decades ago [13] and modified by DeRemee in 1983 (Table 1) [14]. This scoring system has prognostic value as for the inverse relationship between stage at presentation and probability of recovery from sarcoidosis. Untreated patients in stage I reach out complete resolution of both symptoms and radiographic findings in up to 50–90 % of the cases, whereas this percentage decreases to 30–70 % for stage II, 10–20 % for stage III, and 0 % for stage IV [13, 15]. However, the major limitation of such staging is the poor interobserver concordance.
In the late 1990s, Muers et al. introduced a more articulated classification based on detailed morphology (e.g., reticulonodular, mass, confluent, and fibrosis) and extent and profusion of pulmonary findings. Muers’s classification is derived from the radiographic scoring system for pneumoconiosis from the International Labor Organization (ILO) and has higher agreement compared to Scadding score [16].
Computed Tomography
The role of chest CT in sarcoidosis is still somewhat controversial. Once the diagnosis is established, CT has no particular advantage over radiographs. However, there are a number of specific circumstances that should prompt referral to CT [3, 17]. In one leading center, the use of CT was reported in about 30 % of patients with sarcoidosis. Indications included atypical clinical and/or chest radiographic findings, discrepancy between the clinical and radiographic findings, and suspected complications of other lung diseases such as bronchiectasis, aspergilloma, infection, or malignancy [18]. CT is more sensitive than chest radiograph in detecting parenchymal abnormalities and identifying calcification pattern of lymph nodes (e.g., egg shell or “icing sugar” calcification) (Fig. 4) [17]. Furthermore, CT may guide transbronchial biopsy, thus enhancing its diagnostic yield. In this regard, disease extent on CT correlates with the positive predictive value of transbronchial biopsy [19]. CT is also required before endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) [20].
a–b Typical mediastinal lymph nodes calcification in sarcoidosis. a–b Transverse CT image of the chest with mediastinal window setting shows bilateral hilar and subcarinal lymphadenopathy. Two different patterns of lymph node calcification are seen: icing sugar (arrow in a) and egg shell (arrow in b)
The use of contrast medium is usually not indicated whereas the thin-section collimation (1- to 1.5-mm section thickness) and high-spatial-frequency reconstruction algorithm—that are used to generate high-resolution CT images—are mandatory for optimal detection and characterization of the parenchymal findings. In the appropriate clinical setting, a combination of mediastinal and parenchymal abnormalities on CT are virtually diagnostic of sarcoidosis. This is particularly the case of and bilateral hilar symmetric lymphadenopathy along with the typical perilymphatic distribution of nodules. The solid nodules are typically well defined with smooth or irregular margins and most commonly measure 2–5 mm. In keeping with the histological predilection of sarcoid granulomata for areas adjacent to the lymphatic vessels, the nodules are mainly clustered on CT along the bronchovascular bundles, interlobular septa, interlobar fissures, and subpleural regions. The nodules predominate in the mid to upper lung zones. Nodules that are beyond the resolution limits of CT may generate ground glass opacity or coalesce into larger opacities [9, 21].
Of note, a wide range of atypical findings are well-documented in sarcoidosis, thus leading radiologists to term sarcoidosis as “the great pretender” or “the great mimicker.” The CT description of these atypical findings is beyond the scope of this article and may be found elsewhere [22].
It was shown that neither the appearance nor the extent of disease on CT can be considered a good predictor of functional impairment or activity of disease (as assessed by serum angiotensin-converting enzyme levels and bronchoalveolar lavage findings) in any individual patient [23, 24].
CT has higher accuracy in discriminating between reversible and irreversible lung disease (Table 2), as compared to CXR. Knowledge of this may help define treatment goals and strategies (e.g., initiating or continuing potentially toxic treatments) [25]. The classic irreversible changes seen in fibrotic sarcoidosis consist of linear opacities (radiating laterally from the hilum), fissure displacement, bronchovascular distortion, bronchiectasis and honeycombing restricted to the upper zones of the lungs, especially in dorsal regions (Fig. 5). Such fibrotic CT pattern is indeed distinctive from those associated with other fibrosing lung diseases such as usual interstitial pneumonia (UIP) and nonspecific interstitial pneumonia (NSIP) [26].
Importantly, it was recently shown that both the visual assessment of the extent of lung fibrosis and of the main pulmonary artery diameter to ascending aorta diameter ratio (MPAD/AAD) on noncontrast CT may provide additional prognostic information. In particular, high clinical risk can be determined by integrating the composite physiologic index (i.e., a weighted index of pulmonary function variables) with CT measures of the pulmonary vasculature and interstitial disease [27].
There is plenty of evidence that tertiary centers perform CT at presentation in most patients with chest radiographic abnormalities to serve as a baseline in the often difficult assessment of change in disease severity [3, 27]. These views will not be reconciled until the use of serial CT in the detection of change has been validated in large patient cohorts (as discussed below).
Radionuclide Imaging
Gallium scanning has been used historically as a method for assessing sarcoid activity. However, its utility has been limited by low specificity, the absence of a clear definition of the amount of abnormal signal required to constitute “active” pulmonary disease, and the lack of an easy and reproducible method to quantify radioisotope uptake [18, 28]. 18F-Fluorodeoxyglucose positron emission tomography (18FDG-PET) is not recommended in the standard workup but may provide complementary information to routine techniques in very rare specific conditions. Notably, 18FDG-PET may help resolving some difficult diagnosis problems or decipher some signs of unclear significance with an impact on therapeutic decision. In particular, 18FDG-PET is a sensitive technique to assess inflammatory activity, also in sarcoidosis (Fig. 6). 18FDG-PET offers added value in sarcoidosis patients with unexplained persistent disabling symptoms [29]. 18FDG-PET activity of disease is seen in the 75–81 % of patients with persistent disabling symptoms. In these subjects, pulmonary involvement is reported in the 73–97 %, whereas extrapulmonary inflammation occurs in the 75–79 % of cases [30, 31]. Notably, 18FDG-PET is of great value in persistently symptomatic patients without serological signs of inflammatory activity, in patients with radiologic signs of fibrosis, and in the detection of active cardiac sarcoidosis. 18FDG-PET provides information about disease activity location and therefore highlights optimal targets for biopsy to obtain histological evidence for the diagnosis. Also, 18FDG-PET can reveal extrathoracic involvement for the explanation of symptoms and unexpected organ involvement may offer prognostic value [32]. 18FDG-PET identifies clinically silent uptakes in 15 % of patients with sarcoidosis and provides sites for biopsy and diagnosis in 3.8 % [33]. Several regimens have been described to properly minimize the physiological myocardial background uptake, including a long lasting period.
a–d 18FDG-PET scan and CT showing active inflammation. a–b Transverse 18FDG-PET image of the chest shows FDG-avid structures corresponding to active inflammation in axillary (8A) and mediastinal lymphadenopathy. c–d Transverse 18FDG-PET (8C) and CT (8D) of the chest show parenchymal consolidation in the right lung with active inflammation
Magnetic Resonance Imaging
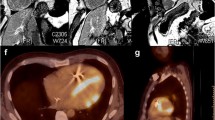
In sarcoidosis, MRI is probably the most sensitive tool for the detection of myocardium sarcoidosis. Cardiac involvement is mostly seen as focal hyperintense areas in T2-weighted images within the lateral wall of left ventricle and basal portion of the septum, up to dilative cardiomyopathy and sudden cardiac death [34, 35]. Late-enhancement pattern is also a very important sign even though it cannot discriminate between granulomatous inflammation and fibrosis. MRI assessment is also required to target biopsy within heterogeneously involved myocardium. The combination of 18FDG-PET and MRI could be helpful to differentiate active and inactive cardiac involvement in difficult cases [36]. The role of MRI has been reported also in the differential of mediastinal hilar masses between sarcoidosis and lymphoma, and the “dark lymph node sign” specific sign of mediastinal lymph node involvement has been proposed [8, 37].
Complications
Vascular Diseases
Pulmonary arterial hypertension (PAH) is a substantial issue regardless of whether there is fibrosis or not [38]. Extrinsic compression of the central pulmonary vessels by lymphadenopathy or mediastinal fibrosis, destruction of the pulmonary vascular bed, intrinsic pulmonary vasoreactivity, pulmonary veno-occlusive disease, and intrinsic sarcoid vasculopathy have all been proposed to contribute to PAH pathophysiology in sarcoidosis [39]. Chest radiograph is relatively insensitive for the detection of PAH. Radiographic signs include dilatation of the pulmonary trunk and main pulmonary arteries with peripheral “pruning,” but they are difficult to identify particularly when enlarged lymph nodes are present [40].
CT signs of PAH have been investigated extensively in the setting of various diffuse lung diseases including idiopathic pulmonary fibrosis, but less is known about the accuracy of these signs in pulmonary sarcoidosis. Walsh et al. showed that the MPAD/AAD was predictive of mortality, independently of all other CT patterns. Furthermore, they suggested that the MPAD/AAD ratio might capture the prognostic effect of underlying vasculopathy, in sarcoidosis. Furthermore, visual estimates of the MPAD/AAD ratio on noncontrast CT scans correlated closely with electronic caliper measurements enhancing the clinical use of this finding [27].
Nevertheless, contrast CT may help specify the mechanisms in pulmonary hypertension by improving the identification of the compression or obstruction of the pulmonary vessels by mediastinal lymphadenopathy or fibrosis (e.g., fibrosing mediastinitis). Besides, pulmonary veno-occlusive disease (PVOD) should be investigated in patients presenting at CT with dilated central pulmonary arteries accompanied by smoothly thickened interlobular septa, ground-glass opacity and pleural effusion, and left heart chambers of normal size.
Furthermore, contrast CT should be performed when pulmonary embolism (PE) is suspected. By analyzing 46 million records of US decedents, it was found that the risk of PE among the 23,679 people coded with sarcoidosis was more than twofold greater than the risk of PE in the background population, regardless of gender, race, and age [41]. What is driving the risk of PE in sarcoidosis requires further exploration; meanwhile, PE should be strongly considered as a potential explanation for worsening of respiratory status in patients with chronic or severe sarcoidosis.
“Suspicious” Lung Parenchyma Lesions
There is an increased overall risk of lung cancer among sarcoidosis patients (Fig. 7a, b). In a recent nationwide cohort study in Denmark, a short-term increased risk of cancer in patients with sarcoidosis was potentially explained by increased surveillance or even initial misinterpretation of cancer as sarcoidosis [42]. Therefore, radiologists should be aware of this association and they should not overlook any larger mass in the context of diffuse perilymphatic nodules on CT, especially in the absence of any precise knowledge of the imaging features of lung cancer associated with sarcoidosis [43].
a–c Complications of sarcoidosis: mass-like findings. a, b Long time sarcoidosis complicated by the appearance of a solid nodule (a), which disproportionally grew in the follow-up CT controls (b), as compared to other solid nodules. The finding was later demonstrated to be a lung carcinoma. c Advanced fibrotic sarcoidosis with cystic lung lesion complicated by a mycetoma. Dependent consolidation partly fills a large cavity in the left upper lobe. Differential with lung cancer can be achieved with prone CT, which can show shift of the consolidation according to gravity (dependent leaning)
By contrast, the imaging features of mycetomas in sarcoidosis are better defined (Fig. 7c). Radiographically, mycetomas appear as a mass of soft tissue density within a lung cavity, most commonly located in the upper lobes, separated from the cavity wall by an air crescent [44]. Erosion of the fungal ball into the hypervascular wall causes hemoptysis, which in 5 % of patients may be life-threatening [45]. Thickening of the lateral pleural surface adjacent to the cavity wall is an early sign of mycetoma formation and may be detected on chest radiograph or CT [46].
Imaging in the Follow-up
The chest radiograph is traditionally considered the cornerstone of follow-up in sarcoidosis patients, along with serial PFTs [1, 47]. Some studies reported good correlation between serial changes of chest radiographic stages and PFTs [16, 48]. However, Zappala et al. showed that change in radiographic extent is more applicable to routine monitoring in sarcoidosis than change in radiographic stage [49]. The same authors then showed a greater agreement between change in extent of disease on serial CT scan and significant PFT trends as compared to change in extent of disease on chest radiograph and significant PFT trends. Change in Dlco/alveolar volume displayed no overall linkage with change in disease extent on chest radiograph and only poor agreement with change in disease extent on CT scan [50].
It has been suggested that CT is more sensitive than pulmonary function tests in capturing the development of fibrotic abnormality and thus has the potential to influence disease management [51]. However, no study has systematically evaluated the additional information provided by serial CT over lung function tests and chest radiograph. CT may be too sensitive in some cases, revealing changes of limited, if any, clinical relevance. Therefore, follow-up CT cannot be recommended for routine use for monitoring disease, largely because of its increased radiation hazard in young patients [47]. Repeating chest radiograph along with clinical examination every 3–6 months is still regarded a good compromise for monitoring pulmonary sarcoidosis [52]. CT may be performed or repeated in subjects with unexplained worsening of respiratory symptoms, hemoptysis, disproportionately impaired lung function or airflow obstruction, uncertain chest radiographic abnormalities, aspergilloma, or pulmonary hypertension [3, 17].
References
Joint Statement of the American Thoracic Society (ATS), The European Respiratory Society (ERS) The World Association of Sarcoidosis and Other Granulomatous Disorders (WASOG) adopted by the ATS Board of Directors and by the ERS Executive Committee (1999) Statement on sarcoidosis. Am J Resp Crit Care Med 160:736–55
Mana J, Gomez-Vaquero C, Montero A et al (1999) Lofgren’s syndrome revisited: a study of 186 patients. Am J Med 107:240–245
Spagnolo P, Sverzellati N, Wells AU, Hansell DM (2014) Imaging aspects of the diagnosis of sarcoidosis. Eur Radiol 24:807–816
Keijsers RG, van den Heuvel DA, Grutters JC (2013) Imaging the inflammatory activity of sarcoidosis. Eur Respir J 41:743–751
Schatka I, Bengel FM (2014) Advanced imaging of cardiac sarcoidosis. J Nucl Med 55:99–106
Baughman RP, Teirstein AS, Judson MA et al (2001) Clinical characteristics of patients in a case control study of sarcoidosis. Am J Respir Crit Care Med 164:1885–1889
Nunes H, Brillet PY, Valeyre D, Brauner MW, Wells AU (2007) Imaging in sarcoidosis. Semin Respir Crit Care Med 28:102–120
Gumustas S, Inan N, Akansel G, Basyigit I, Ciftci E (2013) Differentiation of lymphoma versus sarcoidosis in the setting of mediastinal-hilar lymphadenopathy: assessment with diffusion-weighted MR imaging. Sarcoid, Vascul, Diffuse Lung Dis Offic J WASOG / World Assoc Sarcoid Other Granul Disord 30:52–59
Nishino M, Lee KS, Itoh H, Hatabu H (2010) The spectrum of pulmonary sarcoidosis: variations of high-resolution CT findings and clues for specific diagnosis. Eur J Radiol 73:66–73
Valeyre D, Nunes H, Bernaudin JF (2014) Advanced pulmonary sarcoidosis. Curr Opin Pulm Med 20:488–495
Rockoff SD, Rohatgi PK (1985) Unusual manifestations of thoracic sarcoidosis. AJR Am J Roentgenol 144:513–528
Conant EF, Glickstein MF, Mahar P, Miller WT (1988) Pulmonary sarcoidosis in the older patient: conventional radiographic features. Radiology 169:315–319
Scadding JG (1961) Prognosis of intrathoracic sarcoidosis in England. A review of 136 cases after five years’ observation. Br Med J 2:1165–1172
DeRemee RA (1983) The roentgenographic staging of sarcoidosis. Historic and contemporary perspectives. Chest 83:128–133
Iannuzzi MC, Rybicki BA, Teirstein AS (2007) Sarcoidosis. N Engl J Med 357:2153–2165
Muers MF, Middleton WG, Gibson GJ et al (1997) A simple radiographic scoring method for monitoring pulmonary sarcoidosis: relations between radiographic scores, dyspnoea grade and respiratory function in the British Thoracic Society Study of Long-Term Corticosteroid Treatment. Sarcoid, Vascul, Diffuse Lung Dis Offic J WASOG / World Assoc Sarcoid Other Granul Disord 14:46–56
Greco FG, Spagnolo P, Muri M et al (2014) The value of chest radiograph and computed tomography in pulmonary sarcoidosis. Sarcoid, Vascul, Diffuse Lung Dis Offic J WASOG / World Assoc Sarcoid Other Granul Disord 31:108–116
Keir G, Wells AU (2010) Assessing pulmonary disease and response to therapy: which test? Semin Respir Crit Care Med 31:409–418
de Boer S, Milne DG, Zeng I, Wilsher ML (2009) Does CT scanning predict the likelihood of a positive transbronchial biopsy in sarcoidosis? Thorax 64:436–439
Eckardt J, Olsen KE, Jorgensen OD, Licht PB (2010) Minimally invasive diagnosis of sarcoidosis by EBUS when conventional diagnostics fail. Sarcoidosis Vasc Diffuse Lung Dis 27:43–48
Criado E, Sanchez M, Ramirez J et al (2010) Pulmonary sarcoidosis: typical and atypical manifestations at high-resolution CT with pathologic correlation. Radiograph Rev Pub Radiol Soc North Am Inc 30:1567–1586
Hawtin KE, Roddie ME, Mauri FA, Copley SJ (2010) Pulmonary sarcoidosis: the ‘Great Pretender’. Clin Radiol 65:642–650
Remy-Jardin M, Giraud F, Remy J, Wattinne L, Wallaert B, Duhamel A (1994) Pulmonary sarcoidosis: role of CT in the evaluation of disease activity and functional impairment and in prognosis assessment. Radiology 191:675–680
Leung AN, Brauner MW, Caillat-Vigneron N, Valeyre D, Grenier P (1998) Sarcoidosis activity: correlation of HRCT findings with those of 67Ga scanning, bronchoalveolar lavage, and serum angiotensin-converting enzyme assay. J Comput Assist Tomogr 22:229–234
Brauner MW, Lenoir S, Grenier P, Cluzel P, Battesti JP, Valeyre D (1992) Pulmonary sarcoidosis: CT assessment of lesion reversibility. Radiology 182:349–354
Abehsera M, Valeyre D, Grenier P, Jaillet H, Battesti JP, Brauner MW (2000) Sarcoidosis with pulmonary fibrosis: CT patterns and correlation with pulmonary function. AJR Am J Roentgenol 174:1751–1757
Walsh SL, Wells AU, Sverzellati N et al (2014) An integrated clinicoradiological staging system for pulmonary sarcoidosis: a case-cohort study. Lancet Respirator Med 2:123–130
Kohn H, Klech H, Mostbeck A, Kummer F (1982) 67Ga scanning for assessment of disease activity and therapy decisions in pulmonary sarcoidosis in comparison to chest radiography, serum ACE and blood T-lymphocytes. Eur J Nucl Med 7:413–416
Mostard RL, van Kroonenburgh MJ, Drent M (2013) The role of the PET scan in the management of sarcoidosis. Curr Opin Pulm Med 19:538–544
Mostard RL, Verschakelen JA, van Kroonenburgh MJ et al (2013) Severity of pulmonary involvement and (18)F-FDG PET activity in sarcoidosis. Respir Med 107:439–447
Cremers JP, Van Kroonenburgh MJ, Mostard RL et al (2014) Extent of disease activity assessed by 18F-FDG PET/CT in a Dutch sarcoidosis population. Sarcoid, Vascul, Diffuse Lung Dis Offic J WASOG / World Assoc Sarcoid Other Granul Disord 31:37–45
Sobic-Saranovic D, Grozdic I, Videnovic-Ivanov J et al (2012) The utility of 18F-FDG PET/CT for diagnosis and adjustment of therapy in patients with active chronic sarcoidosis. J Nuclear Med Offic Pub Soc Nuclear Med 53:1543–1549
Teirstein AS, Machac J, Almeida O, Lu P, Padilla ML, Iannuzzi MC (2007) Results of 188 whole-body fluorodeoxyglucose positron emission tomography scans in 137 patients with sarcoidosis. Chest 132:1949–1953
Giesbrandt KJ, Bolan CW, Shapiro BP, Edwards WD, Mergo PJ (2013) Diffuse diseases of the myocardium: MRI-pathologic review of cardiomyopathies with dilatation. AJR Am J Roentgenol 200:W274–W282
Kim JS, Judson MA, Donnino R et al (2009) Cardiac sarcoidosis. Am Heart J 157:9–21
Soussan M, Brillet PY, Nunes H et al (2013) Clinical value of a high-fat and low-carbohydrate diet before FDG-PET/CT for evaluation of patients with suspected cardiac sarcoidosis. J Nuclear Cardiol Offic Pub Am Soc Nuclear Cardiol 20:120–127
Chung JH, Cox CW, Forssen AV, Biederer J, Puderbach M, Lynch DA (2014) The dark lymph node sign on magnetic resonance imaging: a novel finding in patients with sarcoidosis. J Thorac Imaging 29:125–129
Handa T, Nagai S, Miki S et al (2006) Incidence of pulmonary hypertension and its clinical relevance in patients with sarcoidosis. Chest 129:1246–1252
Resten A, Maitre S, Humbert M et al (2004) Pulmonary hypertension: CT of the chest in pulmonary venoocclusive disease. AJR Am J Roentgenol 183:65–70
Matthay RA, Schwarz MI, Ellis JH Jr et al (1981) Pulmonary artery hypertension in chronic obstructive pulmonary disease: determination by chest radiography. Invest Radiol 16:95–100
Swigris JJ, Olson AL, Huie TJ et al (2011) Increased risk of pulmonary embolism among US decedents with sarcoidosis from 1988 to 2007. Chest 140:1261–1266
Sogaard KK, Svaerke C, Thomsen RW, Norgaard M (2014) Sarcoidosis and subsequent cancer risk: a Danish nationwide cohort study. Eur Respir J 45(1):269–72. doi:10.1183/09031936.00084414
Kumar S, Baghdadi S, Cale AR (2006) Concurrence of sarcoidosis and lung cancer: a diagnostic dilemma. Thorax 61:1100
Wollschlager C, Khan F (1984) Aspergillomas complicating sarcoidosis. A prospective study in 100 patients. Chest 86:585–588
Franquet T, Muller NL, Gimenez A, Guembe P, de La Torre J, Bague S (2001) Spectrum of pulmonary aspergillosis: histologic, clinical, and radiologic findings. Radiographics 21:825–837
Sansom HE, Baque-Juston M, Wells AU, Hansell DM (2000) Lateral cavity wall thickening as an early radiographic sign of mycetoma formation. Eur Radiol 10:387–390
Nunes H, Uzunhan Y, Gille T, Lamberto C, Valeyre D, Brillet PY (2012) Imaging of sarcoidosis of the airways and lung parenchyma and correlation with lung function. Eur Respir J 40:750–765
Baughman RP, Shipley R, Desai S et al (2009) Changes in chest roentgenogram of sarcoidosis patients during a clinical trial of infliximab therapy: comparison of different methods of evaluation. Chest 136:526–535
Zappala CJ, Desai SR, Copley SJ et al (2011) Optimal scoring of serial change on chest radiography in sarcoidosis. Sarcoid, Vascul, Diffuse Lung Dis Offic J WASOG / World Assoc Sarcoid Other Granul Disord 28:130–138
Zappala CJ, Desai SR, Copley SJ et al (2014) Accuracy of individual variables in the monitoring of long-term change in pulmonary sarcoidosis as judged by serial high-resolution CT scan data. Chest 145:101–107
Gafa G, Sverzellati N, Bonati E et al (2012) Follow-up in pulmonary sarcoidosis: comparison between HRCT and pulmonary function tests. Radiol Med 117:968–978
Valeyre D, Prasse A, Nunes H, Uzunhan Y, Brillet PY, Muller-Quernheim J (2014) Sarcoidosis. Lancet 383:1155–1167
Conflict of Interest
Mario Silva, Hilario Nunes, Dominique Valeyre, and Nicola Sverzellati declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Silva, M., Nunes, H., Valeyre, D. et al. Imaging of Sarcoidosis. Clinic Rev Allerg Immunol 49, 45–53 (2015). https://doi.org/10.1007/s12016-015-8478-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12016-015-8478-7