Abstract
It is believed that cancer stem cells (CSCs) are precursors for the formation, development, and recurrence of malignant tumors. However, it has proven difficult to isolate and enrich these rare, undifferentiated cells from heterogeneous tumor masses. With some existing reports and preliminary results in mind, we hypothesized that the mitochondrial membrane potential within a tumor mass was heterogeneous and could be used as a tool to isolate and enrich CSCs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
It has long been thought that all tumor cells in a given population share similar characteristics, such as unlimited proliferation, loss of differentiation, and increased invasiveness and tumorigenicity [1, 2]. Recently this view was challenged by the discovery of tumor stem cells (TSCs) or cancer stem cells (CSCs) [3, 4]. The discovery of CSCs suggests that tumor cell populations are heterogeneous and exist in different states of differentiation, including immature CSCs, progenitors, and committed tumor cells. Moreover, research has shown that only a subset of undifferentiated CSCs present in many tumor types possesses the capacity of self-renewal and tumorigenicity [5–9]. Nonetheless, isolation of CSCs from tumor masses remains difficult due to their rarity among tumor cells.
Mitochondria are important organelles in eukaryotic cells. Not only do they provide energy to sustain the metabolic needs of cells, but also integrate death signals [10]. The mitochondrial membrane potential (∆ψm), which reflects the functional status of a mitochondrion, is associated with a cell’s differentiation status, tumorigenicity and malignancy. According to preliminary results presented here and elsewhere, we propose that ∆ψm heterogeneity might be useful as a tool to isolate and identify CSCs from tumor masses.
Evidence
∆ψm and Differentiation
There is a close relationship between a cell’s differentiation status and ∆ψm. When cells are induced to differentiate, ∆ψm may change significantly. For example, once myoblasts fuse into a myotube, ∆ψm increases by approximately 60 mV [11]. An increase in ∆ψm has also been detected during the differentiation of hippocampal neuroblasts induced by retinoic acid treatment [12]. During differentiation of bone marrow stromal cells into osteoprogenitor cells (OPC), elevated ∆ψm peaks are detected on days 3–4 and 10–11. Furthermore, when OPCs differentiate, a surge in ∆ψm during late matrix maturation could be a marker that heralds extracellular matrix mineralization [13, 14]. One can also induce an increase in ∆ψm in L6E9 myoblasts by treatment with growth factors [11]. Confocal imaging of differentiating cultured osteoblasts stained with a mitochondria-specific transmembrane potential-sensitive vital dye revealed that mature cells possess abundant amounts of high-transmembrane-potential mitochondria [15]. In contrast, an apparent decrease in ∆ψm is observed when HL-60 cells (human promyelocytic leukemia) differentiate into granulocyte-like cells [16] or when Friend erythroleukemia cells differentiate into erythroid-like cells [16, 17]. During re-differentiation of pancreatic cancer cells, ∆ψm is significantly reduced three days after the addition of retinoid [18]. In addition, the Ca2+-induced cell differentiation of epidermal HaCaT keratinocytes is accompanied by a significant decrease in ∆ψm [19]. Similar results were reported by Allombert-Blaise [20], who implied that a direct causal relationship between depolarization of ∆ψm and differentiation exists in other cell types as well.
Although the directional change of ∆ψm is not accordant, which may be attributed to differences in the studied cell types, and the change of ∆ψm between different node points of differentiation is not unilateral, these results support the hypothesis that mitochondrial activity plays a central role in regulating the differentiation of many types of cells.
∆ψm and Tumorigenicity
Recently, Toren Finkel and colleagues [21] described a relationship between the tumorigenicity of stem cells and their ∆ψm. It was found that stem cells with a high ∆ψm have a greater propensity to form tumors. They sorted mouse embryonic stem cells by their ∆ψm and found that, both visually and with regard to the expression of key stem cell markers, stem cells with high and low metabolic rates were indistinguishable. Yet, when transplanted into mice, these two types of cells had contrasting properties; cells with lower ∆ψm were more efficient at differentiating into other cell types, while cells with higher ∆ψm were more prone to continue dividing and form tumors. After the ∆ψm inhibitor rapamycin was administered to high membrane potential stem cells, their tumorigenicity decreased significantly. A human mtDNA-deficient breast cancer cell line, T47D rho(0), was found to exhibit a marked decrease in ∆ψm, a slower proliferation rate, and a severe impairment of tumorigenicity [22]. Reduction of lactate dehydrogenase A activity in tumor cells also resulted in a decrease in ∆ψm and compromised their ability to proliferate and form tumors [23]. On the contrary, expression of a mutant mitochondrial DNA polymerase-gamma in breast cancer cells results in depletion of mtDNA, decreased ∆ψm, and an increase in invasiveness/tumorigenicity [24].
∆ψm Between Normal Cells and Cancer Cells
Although the underlying mechanism remains unclear, hyperpolarization of ∆ψm is frequently seen in cancer cells [25]. It has been noted that most, if not all, carcinoma-derived cell lines possess a higher ∆ψm than normal epithelial cells [16, 25, 26].
The results of a six-year systematic study from Chen et al. overwhelmingly confirm that all normal epithelial cells tested have a low ∆ψm. In contrast, screenings of 200 cell lines/types derived from tumors of kidney, ovary, pancreas, lung, adrenal cortex, skin, breast, prostate, cervix, vulva, colon, liver, testis, esophagus, trachea, and tongue show that a great majority of adenocarcinoma, transitional cell carcinoma, squamous cell carcinoma, and melanoma cells have high ∆ψm. The difference in ∆ψm between normal epithelial cells and carcinoma cells is at least 60 mV [16]. In another important study, Bonnet compared the ∆ψm of A549 (non-small-cell lung cancer), M059K (glioblastoma), and MCF-7 (breast cancer) cancer cells to that of healthy, noncancerous, small airway epithelial cells, fibroblasts, and pulmonary artery smooth muscle cells. All cancer cell lines exhibited significantly hyperpolarized ∆ψm in comparison with normal cells, as seen by the increased fluorescence of the ∆ψm-sensitive positive dye tetramethyl rhodamine methyl ester (TMRM) [27]. In our preliminary studies, we also found that some lung cancer cell lines (A549, H446, SPC) possessed a higher ∆ψm than HBE cells, a normal bronchial epithelial cell type.
Hypothesis and Supporting Results
Hypothesis
Based on this evidence, we propose that ∆ψm heterogeneity, which is an indicator of transformation, differentiation status, and tumorigenicity, exists among different subpopulations of a tumor mass. In comparison with their differentiated descendants (progenitors and committed tumor cells), CSCs may possess a different ∆ψm. Furthermore, with instruments such as flow cytometry, heterogeneity (represented by varying intensities of a fluorescent dye, such as Rhodamine123 (Rh123), TMRM, JC-1, could be useful for isolating and/or enriching CSCs from a tumor population. In addition, given that cancer cells possess higher ∆ψm than normal epithelial cells, this heterogeneity may permit ∆ψm to be used in discriminating CSCs from normal cells.
Supporting Results from Other Researchers
Heerdt et al. subcloned and established two isogenic cell lines from the SW620 human colonic carcinoma cell line that display significant and stable differences in the intrinsic ∆ψm [28]. These differences in ∆ψm are linked to important tumorigenic properties of the cells. As compared to those with lower ∆ψm, cells with intrinsically higher ∆ψm exhibit significantly higher resistance to some apoptotic inducers. Despite the translocation of pro-apoptotic Bax and Bak to the mitochondria, fewer cells with elevated intrinsic ∆ψm exhibit concomitant cytochrome c release. Moreover, these cells undergo significantly lower levels of ∆ψm dissipation and apoptosis than do cells with low ∆ψm [29]. The subcloned cell lines with higher ∆ψm were more likely to exhibit elevated steady-state levels of vascular endothelial growth factor and matrix metalloproteinase 7 as well as increased invasive behavior (properties associated with tumor progression) than cells with lower intrinsic ∆ψm. Cells with elevated intrinsic ∆ψm have an enhanced capacity to respond to hypoxia by avoiding apoptosis and initiating angiogenesis. They also have the ability to escape anoikis and grow under anchorage-independent conditions. These data suggest that differences in the intrinsic ∆ψm of colonic carcinoma cells are likely to be tied to the subtle shifts in biochemical pathways and/or cell phenotypes that play fundamental roles in determining the probability of tumorigenesis and progression [30].
Supporting Results from Our Group
The side population (SP) was first described by Goodell and defined as a small subpopulation of cells with enriched stem cell activity that possesses a distinct ability to extrude the fluorescent dye Hoechst33342 [31]. Adult stem cells can be identified by the SP phenotype, which may be a consequence of expressing an ATP-binding cassette (ABC) transporter in this subpopulation. In recent years, some studies have suggested that the SP may be useful for the identification and isolation of CSCs by fluorescence activated cell sorting (FACS) from human cancers, including acute myeloid leukemia (AML), neuroblastoma, glioma, and lung cancer [6, 32–34]. Interestingly, as illustrated in Fig. 1, our preliminary results indicate that SP A549 lung cancer cells (133.48 ± 29.33) display a fairly high ∆ψm as compared with their non-SP counterparts (111.37 ± 20.70).
Heterogeneity of ∆ψm between SP and non-SP tumor cells. A549 cells were seeded on to a coverslip at the density of 1 × 104 cells/mL. One week after culture, Hoechst33342 (2.5 μg/mL) was added for a 100 min incubation, and then Rh123 (1.0 μg/mL) was used for another 30 min staining. (A) Under a laser confocal microscopy, the heterogeneity of ∆ψm between SP (cells with dim nuclei at 450 nm and 625 nm wavelengths, denoted by white arrowhead) and non-SP (cells with bright nuclei at the same wavelengths, denoted by yellow arrow) tumor cells was determined. The ∆ψm was represented by the intensity of Rh123 (green). Nuclei were stained with Hoechst33342 (blue and red). Scale bar = 10 μm. (B) The intensity of Rh123 was analyzed using Image-Pro Plus 5.0 software. The results indicated that the ∆ψm in A549 SP cells (133.48 ± 29.33) was higher than that in the non-SP subpopulation (111.37 ± 20.70). Statistical significance was evaluated by a Student’s t test, n = 13, t = 2.22, *: P = 0.036. (C) The amount of mitochondria was normalized after the sequential incubation with Hoechst33342 (2.5 μg/mL, 100 min) and Mitotracker Green (a potential independent dye to indicate the mitochondria, 0.1 μg/mL, 30 min). Hoechst low/high cells were selected randomly under laser confocal microscopy. Then, the average intensity of Mitotracker Green was detected and analyzed to represent the average amount of mitochondria in SP and non-SP subpopulations. There was no significant difference of fluorescent intensity between these two subgroups (SP: 110.34 ± 17.93, non-SP: 106.03 ± 10.48; n = 10, t = 0.656, P = 0.52)
Reynolds and Weiss described the neurosphere method in 1992. Since its initial characterization, this method it has been used to test for the presence of neural stem cells in brain tissues in vitro [35]. This simple and convenient to enrich for neural stem cells in the adult mammalian brain has generated potential therapeutic strategies to facilitate brain regeneration after injury. In addition, it has been applied to the isolation of CSCs from many malignant tumor types including brain, breast, lung, and colon carcinoma. As shown in Fig. 2, our recent studies illustrate that high ∆ψm was also detected in sphere-forming lung cancer A549 cells (99.92 ± 18.27) when compared with adherent monolayer cells (53.53 ± 3.35) under serum-free stem cell culture condition.
Heterogeneity of ∆ψm between tumor spheres and monolayer tumor cells. Tumorspheres were obtained from the lung cancer A549 by serum-free stem cell medium containing DMEM/F12, B27 (1×), recombinant human epidermal growth factor (rhEGF, 20 ng/mL), basic fibroblast growth factor (bFGF, 20 ng/ml), and insulin (4 U/L). After the identification of self-renewal, multipotency, and tumorigenicity, tumorspheres and adherent non-sphere forming cells (monolayer cells) were collected and dissociated into a single cell suspension. Then, flow cytometry was used to detect the ∆ψm after the suspension was incubated with 1.0 μg/mL Rh123. Both populations were measured in the same experiment. (A) Tumorspheres and monolayer cells derived from lung cancer cell line A549 were imaged with optical microscopy. Tumorspheres are denoted by the blue arrowhead. (B) The ∆ψm in the A549 tumorsphere (90.92 ± 18.27) was higher than that in the monolayer subpopulation (50.53 ± 3.35). Statistical significance was evaluated by a Student’s t test, n = 6 × 104, t = 3.403, *: P = 0.014
CD133, a sorting marker derived from neural stem cells, is widely accepted for isolating CSCs from many different tumor populations, including lung cancer. After the A549 cells were sorted into populations with lowest 5% (∆ψmL) or highest 5% (∆ψmH) ∆ψm (represented by the intensity of Rh123) by flow cytometry, the expression of CD133 was analyzed under laser confocal microscopy. In most A549 cells with high ∆ψm (∆ψmH), CD133 expression could be obviously detected. However, the expression of this stem cell marker was nearly absent in another subpopulation (Fig. 3).
Heterogeneity of ∆ψm and CD133 expression. (A) After incubation with 1.0 μg/mL Rh123 for 20 min, the A549 cells were sorted by flow cytometry into populations with the lowest 5% (∆ψmL, R2 and M1) or highest 5% (∆ψmH, R3 and M2) ∆ψm (represented by the intensity of Rh123). (B) Then, the expression of CD133 was analyzed under laser confocal microscopy. In the ∆ψmH group, obvious CD133 expression (Red) could be detected. However, the expression of this stem cell marker was nearly absent in the ∆ψmL subpopulation. Nuclei were stained with DAPI (blue). Scale bar = 25 μm
Although additional investigations are needed to corroborate these findings in other cancer cell lines and tumor samples, they provide preliminary insights into the role of ∆ψm in CSCs.
Underlying Mechanism
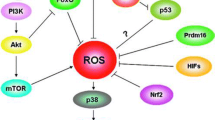
Reactive oxygen species (ROS), an important signaling molecule, participates in many intracellular physiopathological processes, including differentiation. It has been shown that neural and hematopoietic stem cells (HSC) contain lower levels of ROS than their more mature progeny and that these differences are essential for maintaining stem cell function [36–42]. Although, the connection between ROS and ∆ψm is not always straight forward, some sporadic results suggest that ROS is an important contributor to the decrease of ∆ψm [43]. The ∆ψm in those neural and HSCs, which possessed lower concentration of ROS, is fairly high indeed [44, 45]. Thy-1.1lowSca-1+Lin− mouse HSCs exhibit a higher ∆ψm than differentiated bone marrow cells, as detected by the intensity of Rh123 [44]. Plotnikov et al. studied the ∆ψm of neural precursor cells forming neurospheres in culture. Cell heterogeneity, as indicated by ∆ψm, increased with neurosphere enlargement during culturing. The population of cells exhibiting high ∆ψm responded to the addition of a nuclear dye by lowering their ∆ψm, which suggests that the ABCG2 complex is active in these cells, as would be expected of undifferentiated stem cells [45]. Do CSCs also share this property? Recently, results from Diehn et al. indicate that, similar to normal tissue stem cells, subsets of CSCs in some tumors also contain lower ROS levels and enhanced ROS defenses compared to their non-tumorigenic progeny [46]. Thus, one would suggest that a higher ∆ψm, which correlated with the lower ROS, might be another characteristic of these malignant stem cells. In another words, the lower concentration of ROS might be a molecular candidate for explaining the increased ∆ψm in CSCs.
Mitochondrial uncoupling proteins (UCPs) are members of a large protein family named mitochondrial anion carrier proteins (MACP). They separate oxidative phosphorylation from ATP synthesis with energy dissipated as heat, also referred to as the mitochondrial proton leak. The expression of UCPs is negatively correlated with ∆ψm in mammalian cells. Intriguingly, there is accumulating evidence to indicate a link between mitochondrial uncoupling proteins and cancer in recent years. Derda’k et al. address the potential role of UCP2 in cancer initiation [47]. They observe that UCP2−/− mice develop more aberrant crypt foci and colon tumors than UCP2 +/+ littermates. Another important study performed by Claire et al. show that UCP2−/− cells display enhanced colony formation and hypoxia resistance, increased oxygen consumption, and low ATP/cell content. In addition, the dysfunction of UCP2 does not result in a significant increase but a slight decrease of ROS production in these cells [48]. As we know, CSCs are the real initiators of tumorigenesis [1, 2]. At the same time, increased capacity of colony formation [2], hypoxia resistance [49], high rate of oxygen consumption, and low ATP content per cell are all valid indicators of stem cell competence [50]. Thus, one would presume that the expression level of UCPs might be another molecular candidate for the high ∆ψm in CSCs.
Conclusions and Perspectives
In recent years, mitochondrial and energy/metabolism-related features of adult/embryonic stem cells have sparked the interest of an increasing number of researchers [51–54]. It is thought that mitochondrial function and integrity may affect stem cell viability, proliferative and differentiation potential, and lifespan [54, 55]. Some well described features of stem cells include perinuclear mitochondrial arrangement, low ATP content, high rate of oxygen consumption, and low quantity of mitochondrial DNA. Departures from this profile indicate that cells are differentiating or perhaps becoming senescent [50–57]. Mitochondrial biogenesis and accumulation of much lower levels of ROS are required for adult HSCs to remain quiescent and retain their long-term self-renewal potential and multipotency [42, 58]. Studies during the past decade also have shed light on some of the peculiarities of mitochondrial function in cancer cells [59]. However, the intrinsic association between energy/metabolism and the biological characteristics of CSCs, the stem cell-like subpopulation in cancer cells, remains unknown. According to current research and some preliminary results, we suggest that the ∆ψm serves as an important indicator of intracellular metabolic status in undifferentiated and tumorigenic CSCs. In addition, it may differ from other subpopulations of tumor tissues, such as committed cancer cells mingled with normal epithelial cells (based on their ∆ψm, from high to low, we can order these three cell types like this: CSCs > committed cancer cells > normal epithelial cells). Consequently, by taking advantage of the cells’ own putative functional hallmark, heterogeneity in ∆ψm may provide an attractive new tool to isolate CSCs from a tumor mass. Indeed, this technology may lead to important new advances in developmental cell biology. For example, using ∆ψm to sort cells, one may utilize proteomics technology to analyze subpopulations for specific cellular markers and drug targets.
The dissipation of ∆ψm is also a critical event in apoptosis, [10] and the cells with high intrinsic ∆ψm possess a stronger resistance to apoptotic inducers than those cells with low ∆ψm [55]. Considering that CSCs are resistant to apoptosis [60, 61], one might ask whether such resistance correlates to CSCs’ intrinsic ∆ψm and how this value differs from that of committed or non-malignant cells. A greater reduction is needed for those cells with high intrinsic ∆ψm to reach the dissipation threshold. This might be another explanation for the results presented by Griguer that the proportion of CD133 positive glioma cells (termed glioma stem cells) is increased in the cell population after the treatment of rotenone and EB [62].
In conclusion, we have described some potential investigational and therapeutic uses of this technology. A complete investigation of the role of ∆ψ heterogeneity in cancer may lead to a wealth of additional therapeutic possibilities.
References
Weiss, L. (2000). Cancer cell heterogeneity. Cancer Metastasis Review, 19, 345–350.
Locke, M., Heywood, M., Fawell, S., et al. (2005). Retention of intrinsic stem cell hierarchies in carcinoma-derived cell lines. Cancer Research, 65, 8944–8950.
Reya, T., Morrison, S. J., Clarke, M. F., et al. (2001). Stem cells, cancer, and cancer stem cells. Nature, 414, 105–111.
Polyak, K., & Hahn, W. C. (2006). Roots and stems: stem cells in cancer. Nature Medicine, 12, 296–300.
Somervaille, T. C., & Cleary, M. L. (2006). Identification and characterization of leukemia stem cells in murine MLL-AF9 acute myeloid leukemia. Cancer Cell, 10, 257–268.
Ho, M. M., Ng, A. V., Lam, S., et al. (2007). Side population in human lung cancer cell lines and tumors is enriched with stem-like cancer cells. Cancer Research, 67, 4827–4233.
Yu, S. C., & Bian, X. W. (2009). Enrichment of cancer stem cells based on heterogeneity of invasiveness. Stem Cell Reviews and Reports, 5, 66–71.
Yuan, X., Curtin, J., Xiong, Y., et al. (2004). Isolation of cancer stem cells from adult glioblastoma multiforme. Oncogene, 23, 9392–9400.
Yu, S. C., Ping, Y. F., Yi, L., et al. (2008). Isolation and characterization of cancer stem cells from a human glioblastoma cell line U87. Cancer Letters, 265, 124–134.
Brenner, C., & Kroemer, G. (2000). Apoptosis. Mitochondria-the death signal integrators. Science, 289, 1150–1151.
Duguez, S., Sabido, O., & Freyssenet, D. (2004). Mitochondrial-dependent regulation of myoblast proliferation. Experimental Cell Research, 299, 27–35.
Voccoli, V., & Colombaioni, L. (2009). Mitochondrial remodeling in differentiating neuroblasts. Brain Research, 1252, 15–29.
Klein, B. Y., Gal, I., Libergal, M., et al. (1996). Opposing effects on mitochondrial membrane potential by malonate and levamisole, whose effect on cell-mediated mineralization is antagonistic. Journal of Cellular Biochemistry, 60, 139–147.
Klein, B. Y., Gal, I., Hartshtark, Z., et al. (1993). Induction of osteoprogenitor cell differentiation in rat marrow stroma increases mitochondrial retention of rhodamine 123 in stromal cells. Journal of Cellular Biochemistry, 53, 190–197.
Komarova, S. V., Ataullakhanov, F. I., & Globus, R. K. (2000). Bioenergetics and mitochondrial transmembrane potential during differentiation of cultured osteoblasts. American Journal of Physiology Cell Physiology, 279, C1220–C1229.
Chen, L. B. (1988). Mitochondrial membrane potential in living cells. Annual Review of Cell and Developmental Biology, 4, 155–181.
Levenson, R., Macara, I. G., Smith, R. L., et al. (1982). Role of mitochondrial membrane potential in the regulation of murine erythroleukemia cell differentiation. Cell, 28, 855–863.
El-Metwally, T. H., & Pour, P. M. (2007). The retinoid induced pancreatic cancer redifferentiation-apoptosis sequence and the mitochondria: a suggested obligatory sequence of events. Journal of the Pancreas (Online), 8, 268–278.
Savignan, F., Ballion, B., Odessa, M. F., et al. (2004). Mitochondrial Membrane Potential (DeltaPsi) and Ca2+-Induced Differentiation in HaCaT Keratinocytes. Journal of Biomedical Science, 11, 671–682.
Allombert-Blaise, C., Tamiji, S., Mortier, L., et al. (2003). Terminal differentiation of human epidermal keratinocytes involves mitochondria- and caspase-dependent cell death pathway. Cell Death Differentiation, 10, 850–852.
Stefan, M. S., Ma, M. C., Cao, L., et al. (2008). Mitochondrial Metabolism Modulates Differentiation and Teratoma Formation Capacity in Mouse Embryonic Stem Cell. Journal of Biological Chemistry, 283, 28506–28512.
Yu, M., Shi, Y., Wei, X., et al. (2007). Depletion of mitochondrial DNA by ethidium bromide treatment inhibits the proliferation and tumorigenesis of T47D human breast cancer cells. Toxicology Letters, 170, 83–93.
Fantin, V. R., St-Pierre, J., & Leder, P. (2006). Attenuation of LDH-A expression uncovers a link between glycolysis, mitochondrial physiology, and tumor maintenance. Cancer Cell, 9, 425–434.
Singh, K. K., Ayyasamy, V., Owens, K. M., et al. (2009). Mutations in mitochondrial DNA polymerase-gamma promote breast tumorigenesis. Journal of Human Genetics, [Epub ahead of print]. doi:10.1038/jhg.2009.71.
Kroemer, G., & Pouyssegur, J. (2008). Tumor cell metabolism: cancer’s Achilles’ heel. Cancer Cell, 13, 472–482.
Hockenbery, D. M. (2002). A mitochondrial Achilles’ heel in cancer? Cancer Cell, 2, 1–2.
Bonnet, S., Archer, S. L., Allalunis-Turner, J., et al. (2007). A mitochondria-K+ channel axis is suppressed in cancer and its normalization promotes apoptosis and inhibits cancer growth. Cancer Cell, 11, 37–51.
Heerdt, B. G., Houston, M. A., Wilson, A. J., et al. (2003). The intrinsic mitochondrial membrane potential (Deltapsim) is associated with steady-state mitochondrial activity and the extent to which colonic epithelial cells undergo butyrate-mediated growth arrest and apoptosis. Cancer Research, 63, 6311–6319.
Heerdt, B. G., Houston, M. A., & Augenlicht, L. H. (2005). The intrinsic mitochondrial membrane potential of colonic carcinoma cells is linked to the probability of tumor progression. Cancer Research, 65, 9861–9867.
Heerdt, B. G., Houston, M. A., & Augenlicht, L. H. (2006). Growth properties of colonic tumor cells are a function of the intrinsic mitochondrial membrane potential. Cancer Research, 66, 1591–1596.
Goodell, M. A., Brose, K., Paradis, G., et al. (1996). Isolation and functional properties of murine hematopoietic stem cells that are replicating in vivo. Journal of Experimental Medicine, 183, 1797–1806.
Wang, J., Guo, L. P., & Chen, L. Z. (2007). Identification of cancer stem cell-like side population cells in human nasopharyngeal carcinoma cell line. Cancer Research, 67, 3716–3724.
Patrawala, L., Calhoun, T., Schneider-Broussard, R., et al. (2005). Side population is enriched in tumorigenic, stem-Like cancer cells, whereas ABCG2+ and ABCG2− cancer cells are similarly tumorigenic. Cancer Research, 65, 6207–6219.
Hirschmann-Jax, C., Foster, A. E., Wulf, G. G., et al. (2004). A distinct “side population” of cells with high drug efflux capacity in human tumor cells. Proceedings of the National Academy of Sciences USA, 101, 14228–14233.
Reynolds, B. A., & Weiss, S. (1996). Clonal and population analyses demonstrate that an EGF-responsive mammalian embryonic CNS precursor is a stem cell. Developmental Biology, 175, 1–13.
Smith, J., Ladi, E., Mayer-Proschel, M., & Noble, M. (2000). Redox state is a central modulator of the balance between self-renewal and differentiation in a dividing glial precursor cell. Proceedings of the National Academy of Sciences USA, 97, 10032–10037.
Ito, K., Hirao, A., Arai, F., et al. (2004). Regulation of oxidative stress by ATM is required for self-renewal of haematopoietic stem cells. Nature, 431, 997–1002.
Tsatmali, M., Walcott, E. C., & Crossin, K. L. (2005). Newborn neurons acquire high levels of reactive oxygen species and increased mitochondrial proteins upon differentiation from progenitors. Brain Research, 1040, 137–150.
Ito, K., Hirao, A., Arai, F., et al. (2006). Reactive oxygen species act through p38 MAPK to limit the lifespan of hematopoietic stem cells. Nature Medicine, 12, 446–451.
Tothova, Z., Kollipara, R., Huntly, B. J., et al. (2007). FoxOs are critical mediators of hematopoietic stem cell resistance to physiologic oxidative stress. Cell, 128, 325–339.
Miyamoto, K., Araki, K. Y., Naka, K., et al. (2007). Foxo3a is essential for maintenance of the hematopoietic stem cell pool. Cell Stem Cell, 1, 101–112.
Chen, C., Liu, Y., Liu, R., et al. (2008). TSC-mTOR maintains quiescence and function of hematopoietic stem cells by repressing mitochondrial biogenesis and reactive oxygen species. The Journal of Experimental Medicine, 29, 2397–2408.
Levraut, J., Iwase, H., Shao, Z. H., et al. (2003). Cell death during ischemia: relationship to mitochondrial depolarization and ROS generation. American Journal of Physiology. Heart and Circulatory Physiology, 284, H549–H558.
Hüttmann, A., Liu, S. L., Boyd, A. W., et al. (2001). Functional heterogeneity within rhodamine123lo Hoechst33342lo sp primitive hemopoietic stem cells revealed by pyronin Y. Experimental Hematology, 29, 1109–1116.
Plotnikov, E. Y., Marei, M. V., Podgornyi, O. V., et al. (2006). Functional activity of mitochondria in cultured neural precursor cells. Bulletin of Experimental Biology and Medicine, 141, 142–146.
Diehn, M., Cho, R. W., Lobo, N. A., et al. (2009). Association of reactive oxygen species levels and radioresistance in cancer stem cells. Nature, 45, 780–783.
Derdák, Z., Fülöp, P., Sabo, E., et al. (2006). Enhanced colon tumor induction in uncoupling protein-2 deficient mice is associated with NF-kappaB activation and oxidative stress. Carcinogenesis, 27, 956–961.
Pecqueur, C., Bui, T., Gelly, C., et al. (2008). Uncoupling protein-2 controls proliferation by promoting fatty acid oxidation and limiting glycolysis-derived pyruvate utilization. FASEB Journal, 22, 9–18.
Li, Z., Bao, S., Wu, Q., et al. (2009). Hypoxia-inducible factors regulate tumorigenic capacity of glioma stem cells. Cancer Cell, 15, 501–513.
Lonergan, T., Brenner, C., & Bavister, B. (2006). Differentiation-related changes in mitochondrial properties as indicators of stem cell competence. Journal of Cellular Physiology, 208, 149–153.
Nesti, C., Pasquali, L., Vaglini, F., et al. (2007). The role of mitochondria in stem cell biology. Bioscience Reports, 27, 165–171.
Bavister, B. D. (2006). The mitochondrial contribution to stem cell biology. Reproduction, Fertility and Development, 18, 829–838.
Lonergan, T., Bavister, B., & Brenner, C. (2007). Mitochondria in stem cells Mitochondrion, 7, 289–296.
Parker, G. C., Acsadi, G., & Brenner, C. A. (2009). Mitochondria: determinants of stem cell fate? Stem Cells and Development, 18, 803–806.
Pietilä, M., Lehtonen, S., Närhi, M., et al. (2009). Mitochondrial function determines the viability and osteogenic potency of human mesenchymal stem cells. Tissue Engineering Part C Methods, Oct 19. [Epub ahead of print]. doi:10.1089/ten.tec.2009.0247.
Van, B. J. (2008). Mitochondria as regulatory forces in oocytes, preimplantation embryos and stem cells. Reproductive Biomedicine Online, 16, 553–569.
Facucho-Oliveira, J. M., & St John, J. C. (2009). The relationship between pluripotency and mitochondrial DNA proliferation during early embryo development and embryonic stem cell differentiation. Stem Cell Reviews and Reports, 5, 140–158.
Chen, C., Liu, Y., Liu, Y., et al. (2009). The axis of mTOR-mitochondria-ROS and stemness of the hematopoietic stem cells. Cell Cycle, 8, 1158–1160.
Gogvadze, V., Orrenius, S., & Zhivotovsky, B. (2008). Mitochondria in cancer cells: what is so special about them? Trends in Cell Biology, 18, 165–173.
Yajima, T., Ochiai, H., Uchiyama, T., et al. (2009). Resistance to cytotoxic chemotherapy-induced apoptosis in side population cells of human oral squamous cell carcinoma cell line Ho-1-N-1. International Journal of Oncology, 35, 273–280.
Bertrand, J., Begaud-Grimaud, G., Bessette, B., et al. (2009). Cancer stem cells from human glioma cell line are resistant to Fas-induced apoptosis. International Journal of Oncology, 34, 717–727.
Griguer, C. E., Oliva, C. R., Gobin, E., et al. (2008). CD133 is a marker of bioenergetic stress in human glioma. Public Library of Science One, 3, e3655.
Acknowledgments
This study was supported by grants from the National Natural Science Foundation of China (No. 30670919, 30700863) and the National Basic Research Program of China (973 Program, No. 2010CB529403).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Guang-Hui Wang and Xiao-Qun Ye contributed equally to this study.
Rights and permissions
About this article
Cite this article
Ye, XQ., Wang, GH., Huang, GJ. et al. Heterogeneity of Mitochondrial Membrane Potential: A Novel Tool to Isolate and Identify Cancer Stem Cells from a Tumor Mass?. Stem Cell Rev and Rep 7, 153–160 (2011). https://doi.org/10.1007/s12015-010-9122-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12015-010-9122-9