Abstract
Doxorubicin is an effective chemotherapeutic agent prescribed to treat solid tumors (e.g., ovary, breast, and gastrointestinal cancers). This anti-cancer drug has various side effects, such as allergic reactions, cardiac damage, hair loss, bone marrow suppression, vomiting, and bladder irritation. The most dangerous side effect of doxorubicin is cardiomyopathy, leading to congestive heart failure. The exact mechanisms of doxorubicin-induced cardiotoxicity remain incompletely understood. Alteration in myocardial structure and functional cardiac disorders is provoked by doxorubicin administration; subsequently, cardiomyopathy and congestive heart failure can occur. Congestive heart failure due to doxorubicin is associated with mortality and morbidity. Probably, doxorubicin-induced cardiotoxicity starts from myocardial cell injury and is followed by left ventricular dysfunction. Many factors and multiple pathways are responsible for the creation of doxorubicin-induced cardiotoxicity. Inflammatory cytokines, oxidative stress pathways, mitochondrial damage, intracellular Ca2+ overload, iron-free radical production, DNA, and myocyte membrane injuries have critical roles in the pathophysiology of doxorubicin-induced cardiotoxicity. Unfortunately, there are currently a few medications for the treatment of doxorubicin-induced cardiotoxicity in clinical settings. Extensive basic and clinical researches have been carried out to discover preventive treatments. This review briefly discusses the basic and experimental approaches for treating or preventing doxorubicin-mediated cardiotoxicity based on its pathophysiological mechanisms.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Cardiovascular diseases and cancers are the most leading causes of morbidity and mortality in the world [1]. The reported incidence of new cancer cases is estimated at 19.3 million, with a mortality rate of 10 million in the world in 2020 [2]. Cardiac myocytes are potentially more susceptible to the long-term adverse effects of chemotherapy agents such as doxorubicin due to the less regeneration ability in cardiac cells [3]. Moreover, heart muscle dysfunction (cardiomyopathy) and cardiovascular complications may occur in cancer survivors even a year after chemotherapy or radiation therapy and influence their quality of life [4, 5].
Doxorubicin (Adriamycin; DOX), the anthracycline antibiotic, was first produced from Streptomyces peucetius var. caesius in 1960 [6]. DOX is represented as one of the most effective broad-spectrum anti-cancer anthracycline antibiotics either alone or in concomitant administration with other anti-cancer agents [3, 7]. It is determined widely efficient against various types of cancers, including solid malignancies (bladder, breast, lung, etc.) and hematologic neoplasia (Hodgkin’s disease and non-Hodgkin’s lymphomas and pediatric leukemia) [8, 9].
Intravenous injection of DOX produces high-plasma concentrations. Due to its rapid tissue distribution, the plasma concentration falls quickly after administration. Plasma protein binding of DOX is close to 50–85%. Nonetheless, repeated injections do not cause plasma accumulation. The volume of distribution (Vd) of DOX is in the range of 20 to 30 L/kg. DOX is quickly metabolized into the doxorubicinol, 13-hydoxy1 metabolite, aglycones, doxorubicinone, and 7-deoxydoxorubicinone. Metabolism of DOX occurs in red blood cells, liver, and kidney cells by cytoplasmic NADPH-dependent aldo–keto reductases. DOX and its metabolites are mainly excreted in the bile. Although, over 50% is eliminated under the liver first-pass effect. After seven days, cumulative fecal excretion of DOX has been estimated as 25 to 45% [10].
Doxorubicin can cause several side effects, ranging from cancer treatment’s common side effects, such as fever, nausea, and vomiting to lethal cardiac dysfunction. DOX-induced cardiotoxicity is an important clinical problem in managing different types of malignancies. The cardiac side effects of DOX during a chemotherapy regimen can be acute, chronic, or even gradually progressive and persistent after the termination of DOX therapy. In patients undergoing anthracycline therapy, presented symptoms were as follows: (1) reversible side effects, including cardiac rhythm changes, altering blood pressure, pericarditis, and myocarditis and (2) irreversible side effects, such as cardiomyopathy and congestive heart failure. The incidence of DOX-induced cardiotoxicity rises with the escalation in the total cumulative dose of doxorubicin during a day or a treatment cycle [11,12,13,14]. The mortality rate following DOX therapy ranged from 10 to 38% after a single or cumulative dose of 5–25 mg/kg. The mortality rate increases significantly to 50% within two years subsequent chemotherapy [12, 15]. Doxorubicinol, the main biometabolite of DOX, primarily accumulates in the heart during chronic DOX administration [16]. Doxorubicinol is markedly more potent than doxorubicin in compromising cardiac function and inhibiting the ion pumps activities. Collective cardiotoxicity of doxorubicin therapy is due to the accumulation of doxorubicinol in the heart. Doxorubicinol is nearly 30 times more potent at depressing cardiac contractile force than doxorubicin [17]. Given the poor prognosis of DOX-induced cardiomyopathy, different studies have suggested adjuvant therapies to reduce the severity or incidence of cardiomyopathy and maximize patients’ survival rates.
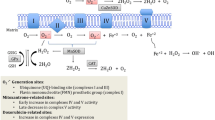
In several basic and clinical reports, anti-inflammatory or anti-oxidative medications are used as an adjuvant treatment to reduce DOX-induced cardiotoxicity. Various studies sought to explain the underlying cellular mechanism that participates in the manifestation of this medication’s undesirable effects (Fig. 1). So far, the studies agree with the multifactorial nature of DOX adverse effects, but the precise underlying mechanism of DOX-induced cardiotoxicity is not fully understood [18, 19]. Disruption of normal mitochondrial function is assumed to be a key factor involved in DOX-induced cardiotoxicity that eventually results in cardiac myocyte death. Mitochondrial damage, considerable demand for cardiac high-energy phosphate due to amplified oxidative phosphorylation, as well as decreased amount of anti-oxidant defense (catalase, superoxide dismutase, and peroxidase), make cardiomyocytes more susceptible to cardiotoxicity in comparison with other organs, such as the liver [18, 20]. Increased DOX concentration in mitochondria activates the redox cycle, leading to reactive oxygen species (ROS) production in both normal and tumor cells [21]. The presence of quinone in the chemical structure of DOX is responsible for the redox cycle [22]. In this regard, enzymatic cellular oxidoreductases (xanthine oxidase, NADH dehydrogenase, cytochrome P-450 reductase especially complex-1) and non-enzymatic pathway (complex with iron (Fe3+)) have been associated with mediating DOX-generated reactive oxygen and nitrogen compounds [23]. Besides, DOX is involved in the overproduction of nitric oxide (NO) by enhancing the expression of endothelial nitric oxide synthase (eNOS) and also inducible NOS (iNOS) mRNA [24, 25]. After DOX-induced injury in cardiac cells, an inflammatory cascade is activated through multiple inflammatory cytokines, including nuclear factor kappa B (NF-κB), tumor necrosis factor-alpha (TNF-α), and interleukin-1 beta (IL-1β) [26,27,28]. A transmembrane protein named connexin 43 (Cx43) plays an essential role in the electrical conduction system between the cardiac myocytes. The expression of Cx43 is reduced following DOX therapy and eventually, the electrical transmission delay occurs [29]. Other mechanisms associated with DOX cardiotoxicity are as follows: an imbalance in calcium hemostasis, targeting topoisomerase-IIβ (Top IIβ) and induced DNA damage [30], as well as a disturbance in the neuregulin /ErbB signaling pathway, which lead to apoptosis and mitochondrial dysfunction [31, 32].
The underlying cellular mechanism of DOX-induced cardiotoxicity and stages of cardiovascular dysfunction after DOX-induced cardiotoxicity. DOX chemotherapy can increase inflammatory and free radical factors and subsequently, DNA damage and cellular injury can occur. Cardiac cell death, cardiomyopathy, and heart failure are the outcomes of this process. In the early stage of chemotherapy, asymptomatic and then symptomatic cardiomyopathy can happen. After cellular injury and abnormal cell signaling development, cardiomyocyte apoptosis and severe heart failure are created. DOX doxorubicin
The present review aims to summarize the current adjuvant treatments that minimize DOX-induced cardiotoxicity in both in vivo and in vitro studies and propose the potential targets to enhance cardiac myocytes’ resistance against doxorubicin.
Study design
A systematic search was performed in Embase, MEDLINE, Scopus, Web of Science Core Collection, and Google Scholar for relevant references. Different combination of keywords including “Doxorubicin,” “Cardiotoxicity,” “Anthracycline,” “Cancer chemotherapy,” “Papillary contractile force,” “Cardiomyopathy,” “LVEF dysfunction,” “Calcium homeostasis,” “Ion disturbances,” “Connexin,” “Sirtuin,” “Rat model,” “Animal model,” “in vivo model,” “in vitro model,” “Inflammation,” “Oxidative stress,” “Mitochondrial damage,” and “Apoptosis” was searched in the “title/abstract” field in these databases. Related articles were selected manually. All articles which had an abstract available were included.
Classification of the Pathophysiology of Doxorubicin-Induced Cardiotoxicity and Its Experimental Therapeutic Approaches
The pre-clinical studies including in vivo and in vitro experiments are summarized in Table 1.
Mitochondrial Damage
Cardiomyocytes have a higher mitochondrion number in comparison to other tissues. Since the mitochondria play a critical role in producing ATP utilized by the cardiomyocytes, its damage is consequential for DOX-induced cardiotoxicity. Treatment with DOX increases reactive oxygen species (ROS), resulting in disruption of mitochondrial ATP synthesis. On the other hand, cardiolipin (mitochondrial inner membrane protein) creates a permanent complex with DOX. This combination intensifies ROS formation in the mitochondria [33]. Therefore, many basic and experimental studies focused on finding a way to reduce mitochondrial damage to decrease DOX-induced cardiotoxicity. DOX can induce mutations and defects in mitochondrial DNA (mtDNA) coupled with ROS elevation in mitochondria (Fig. 2). The mentioned events are associated with the onset of cardiomyopathy [34].
Disturbances of Ca2+ homeostasis and mitochondrial dysfunction after DOX administration. DOX can induce defects in mitochondrial function; as a result, elevation in ROS production of mitochondria can happen. On the other hand, because of the impairment of endoplasmic reticulum structure, an increase in Ca2+ release can occur. Moreover, ROS production can increase following the elevation in Ca2+ release from the endoplasmic reticulum. DOX-induced mitochondrion damage can initiate suppression of the SIRT3 expression. Attenuation of SIRT3 concentration in mitochondria can lead to ROS overproduction as well as exacerbation of mitochondrial dysfunction, mtDNA destruction, cellular injury, and heart failure. DOX doxorubicin; ROS reactive oxygen species; RER rough endoplasmic reticulum; mtDNA mitochondrial DNA; SIRT3 Sirtuin 3
In a previous report, dexrazoxane prevented DOX-induced cardiotoxicity and protected the cardiac mitochondria from functional damage through decreased mitochondrial ROS levels and increased normal mtDNA [35]. Moreover, another study showed improvement in LV function and isolated LV muscle contractile force via inhibition of mitochondrial permeability transition pore opening by cyclosporine A in a rat model of DOX-induced cardiotoxicity [36]. Also, it is hypothesized that DOX would specifically increase mitochondrial ROS production. Cardiomyocytes treated with resveratrol showed the role of Sirtuin 1 (SIRT1) signaling in cardioprotection against DOX-induced cardiotoxicity. Additionally, pre-treatment with resveratrol reduced basal DOX-induced mitochondrial ROS production [37]. Visnagin, a chief component of Ammi visnaga, exhibited protective effects against DOX-induced cardiotoxicity via suppressing malate dehydrogenase (MDH2) in mitochondria [38]. Sildenafil, a phosphodiesterase-5 (PDE-5) inhibitor, attenuates apoptosis by inhibiting caspase-3 and increasing the B-cell lymphoma protein 2 (Bcl-2) expression in DOX-induced cardiotoxicity. Furthermore, sildenafil exerted cardioprotective effects by regulating the NO signaling pathway, oxidative stress, and mitochondrial KATP channel activity [39]. Cardiac alpha-1 adrenergic receptors (α1-ARs) play a crucial cardioprotective role in cardiac injury. In an experimental model of DOX-induced cardiotoxicity, dabuzalgron, α1-ARs agonist, has been suggested as a desired candidate for inducing anti-apoptotic effects via inhibiting caspase-3 and cytochrome C expression. Activation of α1-ARs restores the deleterious effect of DOX by mediating apoptotic responses and inhibiting mitochondrial damage [40].
Iron Homeostasis
Iron plays a vital role in forming dangerous free radicals with potentially harmful effects on the myocardial tissue. Numerous researches have suggested a role for metallic ions in DOX-mediated cardiac damage. DOX interacts with metallic ions, especially iron, which results in the creation of the DOX–iron complex [41, 42]. It has been proposed that DOX-mediated intracellular iron accumulation triggers increased oxidative stress, which is a major phase of DOX toxicity. Therefore, iron chelators are effective in reducing DOX-induced cardiotoxicity [43]. One study demonstrated that higher body iron content increases the cardiotoxic effects of DOX. This study showed that elevation in dietary iron loading caused an increase in DOX cardiotoxicity in rats [44].
The cardioprotective effect of the iron chelator such as desferrioxamine against DOX-induced cardiotoxicity in rats has been investigated. This positive effect was mediated through the transforming growth factor-β1 (TGF-β1)/Smad pathway [45]. After administration, DOX caused severe decreases in red blood cell (RBC) counts and hemoglobin concentrations. In another study, desferrioxamine administration protected the heart and blood elements from the toxic effects of DOX. This chelator agent improved levels of cardiac enzymes and RBC counts to normal range without significant myocardial lesions in a rat model [46]. Administration of deferiprone (iron chelator) remarkably reduced DOX-induced cardiac electrocardiographic and biochemical changes (malondialdehyde, glutathione, lactate dehydrogenase, and creatine kinase-MB) [47]. Dexrazoxane (an iron chelator) binds to intracellular free iron (Fe2+ and Fe3+) and eliminates iron from its complex with DOX, thereby reducing hydroxyl radicals and superoxide formation. A complex of DOX with an iron will remove when dexrazoxane is co-administered with DOX [35]. In many studies, the beneficial effects of flavonoids on the inflammatory and oxidative processes are demonstrated. Also, flavonoids probably have combined iron-chelating and anti-oxidant properties [48, 49]. In one study, the potential iron-chelating and oxygen radical scavenging properties of semi-synthetic flavonoid 7-monohydroxyetylrutoside (monoHER) were evaluated in both in vitro and in vivo models of DOX-induced cardiotoxicity [50]. Moreover, according to the results of phase I of a clinical study, it could be concluded that monoHER at the dose of 1500 mg/m2 is a possible and safe dose to be evaluated in a phase II study to examine the protective properties of monoHER against DOX-induced cardiotoxicity in cancerous patients [50, 51]. Electrophysiological changes and negative inotropic action after doxorubicin administration almost completely were protected with monoHER treatment in the electrically paced mouse left atrium model. This protective effect was related to the iron-chelating properties of monoHER [52].
Inflammatory Pathways
DOX-induced cardiotoxicity is associated with cardiac inflammation. DOX stimulates cytokine production, enhances natural killer (NK) cell activity, and stimulates cytotoxic T-lymphocyte (CTL) responses. All of these factors are essential components leading to direct cardiac damage [53, 54]. Also, inflammation-related signaling pathways have a crucial role in DOX-induced cardiotoxicity. It has previously been stated that DOX also provokes inflammatory responses by increasing the expression of nuclear factor kappa B (NF-κB) and induces the production of several proinflammatory cytokines, including TNF-α [55, 56] (Fig. 3).
Inflammatory cytokine production and oxidative stress activity in a cardiac muscle cell with DOX-induced cardiotoxicity. NF-κB (one of the main inflammatory transcription factors) is located in the cytosol tangled with the inhibitory protein IκB. DOX can convert the IκB/NF-κB complex to an active form of NF-κB. This protein can induce the production of inflammatory cytokines, such as TNF-α and IL-1β. These cytokines exhibit cardiotoxic effects via stimulation of other inflammatory pathways. Moreover, oxidative stress pathways and ROS production have an essential role in DOX-induced cardiotoxicity. An increase in ROS production and a decrease in the concentration of the anti-oxidant factor such as SOD, GSH, and CAT are involved in DOX-induced cardiomyopathy. Oxidative stress and lipid peroxidation can induce cellular damage, apoptosis, cardiomyocyte death, and finally, heart failure. DOX doxorubicin; NF-κB nuclear factor kappa B; IκB inhibitory kappa B; TNF-α tumor necrosis factor-alpha; IL-1β interleukin-1 beta; ROS reactive oxygen species; SOD superoxide dismutase; GSH glutathione; CAT catalase
Many basic studies could suppress DOX-induced cardiotoxicity by inhibiting inflammatory pathways and cytokines. In a new study, dapsone (as an anti-inflammatory compound) significantly reduced TNF-α tissue level and improved electrocardiographic (ECG) parameters (QRS complexes, RR, and QT intervals) in a rat model of DOX-induced cardiotoxicity [57]. Vitexin (30 mg/kg, p.o.) could markedly reduce the increased serum levels of IL-1β, TNF-α, IL-6, and NF-κB levels in the rat model of DOX-induced cardiotoxicity. Reducing cardiac inflammatory cytokines showed that vitexin could be an effective therapeutic compound to prevent DOX-induced cardiotoxicity [58]. A recent study confirmed that treatment with enoxaparin caused significant decreases in cardiac levels of TNF-α and IL-1β, suggesting a reliable cytoprotective mechanism for enoxaparin against DOX-induced cardiotoxicity in a rat model. These cardioprotective effects of enoxaparin are due to the inactivation of the NF-κB pathway and following cytokines release suppression [59]. In an earlier study, three anti-inflammatory drugs, aspirin, ibuprofen, and sulindac, were examined for their positive effects on the survival rate of the mouse model of DOX-induced cardiotoxicity. According to the result of this study, inhibition of cyclooxygenase could reverse the cardiotoxic effects of DOX [60]. DOX increases proinflammatory cytokines and causes a concomitant decrease in IL-10 levels, a well-known anti-inflammatory cytokine. IL-6 and TNF-α suggested a valuable predictor of cardiovascular morbidity and mortality in DOX cardiotoxicity.
Cardiomyocytes and endothelial cells usually produce IL-6, but the myocardial expression of IL-6 is associated with the progression of DOX-induced cardiotoxicity. The cardiac nitric oxide (NO) level enhances in DOX administration which is probably due to increased expression of iNOS. This pathway may be influenced by NF-κB activation [19]. The previous report evaluated the cytotoxicity of DOX in H9c2 cardiac cells. Based on extracted data from this study, a decrease in the levels of IL-1β, IL-6, and TNF-α was associated with MAPK/NF-κB pathway suppression. Exogenous hydrogen sulfide (H2S) attenuates DOX-induced inflammation and cytotoxicity by inhibiting p38 MAPK/NF-κB pathway. Interaction between the NF-κB pathway and IL-1β is essential in the induction of DOX-induced cardiotoxicity [61]. Several natural compounds (NCs) with anti-inflammatory effects were investigated for their ability to reduce DOX-induced cardiotoxicities, such as α-linolenic acid, asiatic acid, apigenin, β-lapachone, dioscin, ferulic acid, Ganoderma lucidum polysaccharides, curdione, genistein, ginsenoside Rg3, indole-3-carbinol, neferine, and p-coumaric acid. NCs may have protective effects against DOX-induced cardiac damage by suppressing inflammatory markers, such as inducing the Nuclear factor erythroid 2-related factor 2 (Nrf2) signaling pathway, reducing NF-κB expression, and inhibiting cyclooxygenase (COX) [62]. DOX excites macrophages and monocytes through subsequent TNF-α production, which activates the TNF receptor (TNFR). This action was probably related to DOX-induced cardiotoxicity [22].
Oxidative Stress
Oxidative stress is produced due to an imbalance between ROS formation and endogenous anti-oxidants activation in reaction to cell injury, leading to myocardial toxicity. Increased oxidative stress and anti-oxidant factors deficiency serve important roles in DOX-induced cardiotoxicity. Previous studies reported that reduction in anti-oxidant enzymes such as superoxide dismutase (SOD) and catalase accompanied DOX-induced cardiotoxicity. Oxidative damages increase malondialdehyde (MDA) level, as a marker of oxidative and peroxidation process, in DOX-related cardiotoxicity [18, 63, 64]. Accordingly, many studies have increased the anti-oxidant level and decreased oxidative damage markers to suppress DOX-induced cardiotoxicity (Fig. 3). Moreover, the strong association between the created ROS and NF-kB activation exaggerates their harmful impacts on DOX-induced cardiac damage [65]. Extensive ROS generation induces lipid peroxidation and accumulation of the reactive electrophile, 4-HNE, a biomarker of DOX-induced oxidative stress [66]. Also, excess ROS generation in the mitochondria and ROS-induced nuclear p53 activation after DOX administration can lead to mitochondrial damage, including mutations in mitochondrial DNA, disturbances in mitochondrial membrane permeability, and disruption to Ca2+ homeostasis [67, 68]. Nuclear factor erythroid 2-related factor 2 (Nrf2) is a redox-sensitive transcription factor that regulates the detoxification process and cellular response to oxidative stress. Previously, it was reported that DOX administration decreases active nuclear Nrf2 in the heart. Moreover, Nrf2 upregulates the expression of several anti-oxidant genes, such as heme oxygenase-1 (HO-1) [69]. HO-1 activity suppresses vital cellular deleterious processes, including inflammation, apoptosis, and oxidative stress [70]. On the other hand, the inhibition of the Nrf2 pathway following DOX administration has been associated with suppressing the cardioprotective enzymes against DOX-induced oxidative damage [71]. Nrf2/HO-1 signaling pathway has been studied and evaluated in several studies on DOX-induced cardiotoxicity. Genistein, a phytoestrogen belonging to the class of isoflavones, could enhance the Nrf2 and HO-1 expression in an animal model of DOX-induced cardiotoxicity. The potential anti-oxidant properties of genistein may be because of its stimulatory effect on Nrf2/HO-1 signaling pathway and by this means shows cardioprotective effects from DOX-induced oxidative injury [72]. Another study confirmed that the Nrf2/HO-1 pathway also has anti-apoptotic action. DOX-induced cardiomyocyte apoptosis and oxidation were modulated with Nrf2/HO-1 signaling activation in DOX-treated H9c2 cells [73]. Pristimerin is a natural triterpenoid compound (extracted from Chinese traditional medicine) that showed strong anti-oxidant and anti-inflammatory activities, which effectively suppressed DOX-induced cardiotoxicity due to activation of the Nrf2 signaling pathway. Additionally, the HO-1 (Nrf2 target gene) level was significantly decreased in the hearts of DOX-treated animals. On the contrary, pristimerin administration significantly increased the activity of Nrf2 and the expression of the HO-1 gene. This compound caused a significant elevation of cardiac HO-1 level compared to the DOX-treated animals [74]. Caffeic acid protects cardiac tissue successfully from oxidant injury induced by DOX via elevation in SOD, catalase enzyme, and glutathione peroxidase levels in the heart of rats [75]. Significant enhancement in cardiac content of SOD and depletion of MDA after administration of dapsone could reverse cardiac papillary muscle damage and electrophysiological dysfunction induced by DOX in a rodent model [57]. Administration of garlic in DOX-received rats enhanced superoxide dismutase activities and reduced MDA production in plasma and heart tissue. Garlic protected the heart tissue against oxidative stress and apoptosis associated with DOX [76]. Sumatriptan, a 5-HT1B/D agonist, is involved in decreasing proinflammatory and oxidative factors production. Recently, therapeutic effects of sumatriptan in other conditions besides migraines have been studied [77,78,79]. A recent study demonstrated that repetitive administration of lower doses of sumatriptan is associated with significant protective effects against DOX-induced cardiotoxicity in a rat model. Sumatriptan could reduce the weight loss and mortality rate mainly by decreasing MDA levels and elevation in SOD activity. ECG disturbances, including QT interval and QRS complex widening, significantly modified in sumatriptan-treated rats. Also, papillary muscle contractile force and left ventricular function improved after administration of lower doses of sumatriptan [80]. Telmisartan, an angiotensin receptor blocker, could reduce the cardiotoxic effects of DOX through inhibition of cardiac lipid peroxidation and enhancement of heart level of glutathione (GSH) in rats. GSH has a crucial role in reducing toxic peroxides in all animal cells [76, 81]. Probucol is an anti-lipemic drugs with a beneficial anti-oxidant effects. Administration of probucol resulted in an escalation in GSH peroxidase (GSH-Px) and SOD activities as well as inhibition of lipid peroxidation process [82, 83]. The development of superoxide radical and subsequently lipid peroxidation from DOX are critical factors in the pathogenesis of DOX cardiotoxicity. Thymoquinone, the main constituent of the oil from Nigella sativa seeds, showed protective properties via superoxide radical scavenging action in a rat model of DOX-induced cardiotoxicity. The effect of thymoquinone on lipid peroxidation and scavenging of superoxide radical was like superoxide dismutase in this model [84]. In a mice model of DOX-induced cardiotoxicity, treatment with G-Rutin and luteolin (well-known flavonoid compounds) significantly inhibited lipid peroxidation destructive effects by elevation in GSH peroxidase activity. This study also suggested that oral administration of flavonoids or food intake, including flavonoids, has reduced the effect of DOX toxicity [85]. Luteolin significantly reduced MDA level and increased SOD activity in cardiotoxic rats, indicating luteolin exerted a protective effect against doxorubicin-induced cardiotoxicity not only through suppression of the ROS-mediated oxidative stress but also luteolin remarkably reduced Bcl-2-associated X (Bax) and caspase-3 protein expression. As a result, this flavonoid may be a novel, natural therapeutic agent for preventing DOX-induced cardiotoxicity [86]. Quercetin, a plant-derived flavonoid, has been established to comprise potent anti-oxidant and anti-inflammatory properties. Quercetin might inhibit DOX-induced ROS accumulation, diminishing oxidative stress and downregulating redox-related process [87]. Anti-oxidant properties and carbonyl reduction effect of different flavonoids such as quercetin against doxorubicin-induced cardiotoxicity have previously confirmed [88]. Moreover, a fine balance between the anti- and pro-oxidant properties of the flavonoids is well known [89]. 3′,4′-Dihydroxyflavonol (DiOHF), a recently industrialized potent synthetic flavonoid, effectively prevented the DOX-induced cardiotoxicity via reduction of ROS production, mitochondrial dysfunction, and apoptosis. Results confirmed that DiOHF is a strong anti-oxidant agent against the DOX-induced oxidative stress in vitro through inhibition of ROS generation and improving cell viability [90]. Ginsenoside Rh2, a main bioactive ginsenosides form Panax ginseng protects against DOX-induced cardiotoxicity by exerting anti-oxidative properties and restoring the balance between oxidative stress markers [91]. Schisandrin B (Sch B) is derived from Schisandra chinensis (Turcz.) and is capable of decreasing the adverse effects of DOX on excessive ROS formation through augmenting cardiomyocyte glutathione redox cycle as well as restoring mTOR signaling [92, 93]. There is a strong link between DOX-induced cardiotoxicity and 5′-adenosine monophosphate-activated protein kinase (AMPK) signaling pathway. DOX can neutralize the AMPK signaling transduction. AMPK is responsible for decreasing apoptosis and protecting the cardiac mitochondria by reducing oxidative stress activity as well as suppressing autophagy and fibrosis [94]. Metformin is a biguanide anti-diabetic drug used for type 2 diabetes mellitus. Along with its anti-hyperglycemic effect, metformin possesses protective effects against DOX-induced cytotoxicity via reduction of the oxidative stress activity. In addition, metformin has the potency to stimulate AMPK signaling pathway [95]. Another animal study demonstrated that metformin reduced the impaired autophagy process following DOX administration due to activation of AMPK signaling pathway. Additionally, the level of Troponin T and MDA remarkably decreased after metformin treatment [96]. Statins are identified as therapeutic agents in treating hyperlipidemia and cardiovascular disorders. Statins have been involved in protection against DOX-induced cardiotoxicity by activating AMPK signaling pathway. Regarding to their promising effects on lipoprotein metabolisms, statins can modulate Ras-related C3 botulinum toxin substrate (Rac1) and reactive nitrogen species (RNS) and trigger AMPK phosphorylation. For instance, in a study lovastatin significantly suppressed DOX-induced cardiotoxicity through reduction of the Rac1 signaling, DNA damage, and apoptosis [94, 97].
Apoptosis
Due to DOX administration, the imbalance between oxidative stress markers stimulates intrinsic and extrinsic apoptotic pathways, resulting in cardiac muscle cell death. DOX-induced enhancement of oxidative markers leads to activation of heat shock factor-1 (HSF-1) and subsequent activation of heat shock protein (HSP)-25 that provokes p53 protein, which is responsible for engendering proapoptotic agents [98, 99]. Following DOX administration, diverse HSPs are activated and affect cardiomyocytes differently. Expression of HSP-25 and HSP-70 in mice cardiac tissue results in inflammation, apoptosis, and fibrosis [100]. On the other hand, HSP-10, HSP-20, HSP-22, HSP-27, and HSP-60 exert protective effects against apoptosis and preserve the function of cardiac muscle cells [101]. Adiponectin, a protein hormone regulating glucose level and fatty acid breakdown, showed protective effects against DOX-induced cardiotoxicity through anti-apoptotic activities and reduced serum levels of creatin kinase and lactate dehydrogenase [102]. Erythropoietin is a glycoprotein hormone produced by the kidney to regulate red blood cells production and is used as an anti-anemia drug in patients undergoing chemotherapy. In addition to its protecting effect against anemia, it also protects against DOX-induced cardiotoxicity by decreasing apoptosis when administered as a prophylactic treatment [103]. Curcumin, a natural anti-inflammatory compound derived from turmeric, is able to potently induce anti-apoptotic mediators such as B-cell lymphoma 2 (Bcl-2) and reduce the expression of proapoptotic mediators like Bax that provoke mitochondrial cell death due to DOX administration. Curcumin attenuates DOX cardiotoxicity by mediating propyroptosis agents, e.g., NOD-, LRR-, and pyrin domain-containing protein 3 (NLRP3), inflammasome, IL-18, and caspase-1. Moreover, curcumin attenuates DOX-induced apoptosis and pyroptosis by activating Akt/mTOR signaling pathway [104, 105]. Carvedilol, a cardioselective β receptor blocker, is generally recommended to treat hypertension. Carvedilol displayed a wide range of pharmacological activities, including anti-oxidation, suppression of lipid peroxidation, and reduction in mitochondrial toxicity. In a mouse model of DOX-induced cardiotoxicity, carvedilol has the potency to improve DNA damage and cardiomyopathy through regulating apoptosis, mitochondrial damage (cytosolic cytochrome C), fibrosis, myocardial hypertrophy, and oxidative stress [106].
Calcium Homeostasis
Another mechanism of cardiac injury of DOX is impaired intracellular Calcium (Ca2+) homeostasis [107]. In cardiomyocytes, Ca2+ is the essential element for myocardial excitation and contraction. Disturbances in cellular Ca2+ concentration are accompanied by cardiac structural and functional alterations [108]. DOX increases the cytosolic Ca2+ primarily by releasing calcium from the endoplasmic reticulum (ER) [109]. Moreover, dysregulation of intracellular calcium levels is both a result and a cause of ROS formation. Thus, DOX-mediated ROS activation and apoptosis can be suppressed using a Ca2+ chelator [110].
Agmatine is a precursor for polyamine synthesis and can induce cytosolic Ca2+ uptake and cause an increase in the activity of calcium pumps. Agmatine may increase the therapeutic effects of DOX and it showed protective properties via modulating cellular Ca2+ disturbances against DOX-induced cardiotoxicity in rats. Agmatine could improve ventricular papillary muscle dysfunction and cardiac electrical disorders after DOX-induced cardiotoxicity in rats [111]. The arrhythmogenic effects of DOX, such as induction of QT interval prolongation, were significantly reversed via co-administration by magnesium sulfate. Mg2+ alleviates the intracellular Ca2+ through different mechanisms such as the effect on Ca2+ pumps on the sarcoplasmic reticulum surface and the change of Ca2+ in sarcoplasmic stores. Therefore, magnesium sulfate could improve electrophysiological disorders induced by DOX [112]. Some electrophysiological studies have confirmed that opioid receptors stimulation directly modulates ion channels by interfering with G-protein-coupled receptors. In the cardiac myocytes, opioid receptor stimulation induces mobilization of intracellular Ca2+ [113,114,115]. Morphine exerts cardiac protective effects via various receptors (κ and δ opioid receptors), which can be antagonized with naloxone. Additionally, morphine attenuates cardiac dysfunction and mortality induced by DOX via the aforementioned mechanisms. Yet, the tolerance and dependence impact of morphine in chronic treatments need to be taken into account [116, 117].
Gap Junctions
Gap junctions are aggregates of intercellular channels that facilitate direct cell–cell interactions and intercellular transmission of molecules and ions [118]. Gap junctions mediate the electrical connection between cardiomyocytes and maintain the normal cardiac rhythm. These junctions consist of groups of membrane channels that directly link the cytoplasmic parts of nearby cells. Each gap junctional channel is constructed from connexins. The most abundant isoform of connexin in the mammalian cardiac cells is connexin43 (Cx43), while other types of connexins are also expressed [119]. Changes in Cx43 expression and distribution are accompanied by myocardium diseases, such as hypertrophic cardiomyopathy, heart failure, and ischemia [120]. Cx43 generally localizes in gap junctions but has also been detected in mitochondria and it is considered vital for cardioprotection. Mitochondria-Cx43 has a crucial role in mitochondrial permeability under conditions of calcium overload and oxidative stress [120, 121]. Administration of DOX induces alterations in Cx43 expression and distribution. Therefore, electrical signal conduction between cardiomyocytes, intracellular calcium homeostasis, and mitochondrial function is disturbed. As a result, a part of the cardiotoxic effects of DOX is exerted by affecting the function of Cx43 [29, 122].
In an animal model of DOX-induced cardiotoxicity, diazoxide could improve cardiac electrophysiological disturbances, intracellular Ca2+ signaling, and Cx43 expression and localization. Diazoxide exerts its cardioprotective effects by enhancing Cx43 in the sarcoplasmic membrane and mitochondria [122]. Ghrelin, an endogenous peptide (isolated from the stomach), showed cardioprotective properties in a rat model of DOX-induced cardiotoxicity. Treatment with ghrelin caused a significant improvement in electrophysiological parameters and myocardial histopathological damages. According to the results obtained from this study, the beneficial effects of ghrelin in DOX-induced cardiotoxicity may be mediated through an increase in myocardial Cx43 expression. Mechanistically, elevation in the level of Cx43 in the intercalated discs could reduce DOX cardiotoxic effects, such as cardiomyocytes’ calcium overload and ECG abnormities [123]. Hydrogen sulfide (H2S), as a unique gaseous mediator, plays a crucial role in many human organs, especially in the cardiovascular system. H2S donors such as S-diclofenac could lead to a significant increase in the number and density of cardiomyocyte gap junctions (Cx43 and Cx45 expression) in a mouse model of DOX-induced cardiotoxicity. On the other hand, H2S considerably attenuated DOX-related heart injury and left ventricular dysfunction via upregulation of cardiac gap junctions [124].
Sirtuins
Sirtuins are a well-known family of proteins found in most classes of organisms, including mammals. Sirtuins have many vital roles in mammalian physiology, and they may provide new targets for treating diseases associated with aging and perhaps increasing lifespan. Seven mammalian sirtuins, SIRT1–7, have been identified. Sirtuin 1 (SIRT1) and sirtuin 3 (SIRT3) are essential for regulating metabolic processes in mammalian. Various metabolic disorders are related to SIRT1 and SIRT3 deficiencies. The kidney, brain, and heart have the highest tissue levels of SIRT3. It is localized in the mitochondrial matrix and it is necessary for enzymatic activity and the expression of many mitochondrial proteins [125].
Moreover, mitochondrial sirtuins reduce myocardial ischemia–reperfusion injury, expansion of cardiac hypertrophy, and heart failure. Therefore, elevation in cardiac tissue levels of sirtuins for myocardial mitochondrial energetics is a novel approach in several cardiac disorders [126]. Sirtuins are activated by low energy levels and stimulate energy production by activating transcription factors and enzymatic regulators of cardiac energy metabolism. Moreover, DOX interferes with myocardial energetics; thus, a promising approach to reduce the cardiotoxic effects of DOX may be targeting mitochondria in order to improve metabolic function. DOX-treated neonatal rat cardiomyocytes have reduced mitochondrial membrane potential and SIRT3 expression. SIRT3 overexpression protects cardiomyocytes from DOX-induced mitochondrial damage by preventing mitochondrial destruction and cellular death in the heart (Fig. 2). Furthermore, SIRT3 expression reduced DOX-induced interstitial cardiac fibrosis and collagen expression. Also, SIRT1 overexpression in rat cardiomyocytes prevented DOX-induced ROS production, apoptosis, reduction in SOD1 expression, and the initiation of caspase-3 cleavage [127, 128].
Resveratrol attenuated DOX-induced cardiotoxicity via induction of SIRT1 expression. Resveratrol administration was associated with the prevention of SIRT1 suppression by DOX. On the other hand, elevation in the level of SIRT1 was associated with increased SOD1 expression, decreased apoptosis, and caspase-3 cleavage in DOX-induced cardiotoxicity in the rat [129, 130].
Honokiol, as an activator of SIRT3, protects the heart from DOX-induced cardiac damage in a rat model. Honokiol-mediated activation of SIRT3 prevented DOX-induced ROS creation, mitochondrial injury, and cell death in rat cardiomyocytes. Also, overexpression of SIRT3 by honokiol could effectively protect cardiomyocytes from DOX-induced apoptosis, cardiac hypertrophy, and the development of fibrosis [131].
Other Mechanisms
Troponin T (TnT) is expressed in skeletal muscle and cardiomyocytes. Troponin T binds to tropomyosin and helps position it on actin. This protein is a portion of the troponin complex, which is crucial for the contraction of cardiac and skeletal muscles [132, 133]. Cardiac troponin T is a very sensitive biomarker for detecting myocardial damages. Therefore, measuring serum levels of cardiac TnT as a biomarker for DOX-induced cardiotoxicity is becoming popular [134, 135]. Based on the importance of cardiac TnT in DOX-induced cardiotoxicity, it has been shown that lithium could prevent the elevation of cardiac troponin T and the development of myocardial injury. These results demonstrated the protective effects of lithium against DOX-induced cardiotoxicity in rat. In addition, papillary muscle contractility, electrophysiological dysfunction, and mortality rate could improve remarkably after lithium administration [136].
Novel Pre-clinical Model for DOX-Induced Cardiotoxicity Evaluation
The generation of human-induced pluripotent stem cells (hiPSCs) from somatic cells of the adult body opens new areas for discovering novel platforms for drug development. Researchers have shown enormous attention toward applying hiPSC technology to human disease modeling [137]. Also, the application of human iPSC-derived cells was previously recommended to evaluate drug-induced toxicity [138]. In this regard, hiPSCs are great tools for modeling and evaluating the DOX-induced cardiotoxicity since they replicate unlimitedly and have the capacity to differentiate into several human cell types, including cardiomyocytes. Advances in the differentiation and purification of hiPSC-derived cardiomyocytes have promoted strategies to use these cells for the cardiotoxicity assessment of drugs [139]. For this reason, the potential use of hiPSC-derived cardiac models in pre-clinical testing of drug-induced cardiotoxicity was previously confirmed. It is helpful to evaluate cardiotoxicity in 3 categories: electrophysiological abnormalities, contractile force dysfunction, and cardiac structural damage [140]. Primary mechanisms of DOX-induced cardiotoxicity in hiPSC-derived cardiomyocytes are DNA damage, ROS production, mitochondrial dysfunction, and disturbed calcium homeostasis [141]. Cellular and mitochondrial ROS production in hiPSC-derived cardiomyocytes increase 24 h after exposure with a low dose of DOX (0.01 μM). Moreover, mitochondrial dysfunction and reduced mitochondrial transmembrane potential were induced via a higher dose of DOX (5 μM) [142, 143]. One study demonstrated that hiPSCs-derived cardiomyocytes might be more sensitive to DOX because of higher expression of topoisomerase II; thus, they are more prone to severe DNA damage [144]. DOX potently induces apoptosis by activating death receptors (DRs), such as TNF receptor 1, Fas receptor, DR4, and DR5. One study showed that pre-treating hiPSC-derived cardiomyocytes completely inhibited DOX-induced apoptosis with a DR5 neutralizing antibody. However, this cardiotoxic effect was reversible after recovery for seven days [145, 146]. In contrast to these studies with acute exposure to DOX, another investigation showed that p53 could protect against chronic DOX cardiotoxic effect by counteracting mitochondrial DNA depletion after chronic exposure of hiPSC-derived cardiomyocytes to low doses DOX [147]. Treatment of cardiomyocytes with dexrazoxane is therefore expected to prevent cardiotoxicity. However, pre- and co-treatment of hiPSC-derived cardiomyocytes with dexrazoxane could not inhibit DOX-induced cardiotoxicity. This discrepancy may be correlated to the relative immature character of hiPSC-derived cardiomyocytes. Dexrazoxane might exert protective effects in more mature cardiomyocytes via depletion of topoisomerase IIβ [144, 148]. Moreover, studies with hiPSC-derived cardiomyocytes have revealed that cardiotoxic effects of DOX were dependent on various factors, such as lifestyle or genetic background of patient-derived hiPSC-cardiomyocytes. Taken together, the use of hiPSC-derived cardiomyocytes reduces the abrasion rate, costs, and time of drug development and facilitates the evaluation of drug toxicity, especially DOX-induced cardiotoxicity [141, 143]. Today, the maturity of hiPSC-derived cardiomyocytes is getting better by many efforts. However, there are also many limitations and challenges in using these cells to predict clinical cardiotoxicity [149, 150]. Although further studies are required, the safety and efficacy of drug therapy will be comprehended using hiPSCs in the near future [151].
Conclusion
Due to its cardiotoxic effects, DOX remains a critical drug in cancer treatment and a challenging chemotherapeutic agent. The mechanism of DOX-induced cardiotoxicity is multifactorial and occurs because of cell death and functional disturbances in cardiac cells. Based on its cytotoxic mechanisms, various strategies were proposed to prevent or attenuate the adverse cardiotoxicity effect of this drug. In this study, numerous compounds from different original studies with potential cardioprotective effects were mentioned that could reduce the DOX-induced cardiotoxicity by various mechanisms, such as anti-inflammatory and anti-oxidant effects or modification of intracellular ions homeostasis. We hope that these basic and animal studies can pave the way to develop novel therapies for the prevention or treatment of DOX-induced cardiotoxicity in various clinical settings in the future.
Data Availability
Available upon request.
Code Availability
Not applicable.
References
Najafi, M., Shayesteh, M. R. H., Mortezaee, K., Farhood, B., & Haghi-Aminjan, H. (2020). The role of melatonin on doxorubicin-induced cardiotoxicity: A systematic review. Life Sciences, 241, 117173.
Sung, H., Ferlay, J., Siegel, R. L., Laversanne, M., Soerjomataram, I., Jemal, A., & Bray, F. (2021). Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians, 71, 209–249.
Young, R. C., Ozols, R. F., & Myers, C. E. (1981). The anthracycline antineoplastic drugs. New England Journal of Medicine, 305, 139–153.
Narkiewicz, K., Ratcliffe, L. E., Hart, E. C., Briant, L. J., Chrostowska, M., Wolf, J., Szyndler, A., Hering, D., Abdala, A. P., & Manghat, N. (2016). Unilateral carotid body resection in resistant hypertension: A safety and feasibility trial. JACC: Basic to Translational Science, 1, 313–324.
Hardaway, B. W. (2019). Adriamycin-associated cardiomyopathy: Where are we now? Updates in pathophysiology, dose recommendations, prognosis, and outcomes. Current Opinion in Cardiology, 34, 289–295.
Arcamone, F., Cassinelli, G., Fantini, G., Grein, A., Orezzi, P., Pol, C., & Spalla, C. (1969). Adriamycin, 14-hydroxydaimomycin, a new antitumor antibiotic from S. Peucetius var. caesius. Biotechnology and Bioengineering, 11, 1101–1110.
Kalyanaraman, B. (2020). Teaching the basics of the mechanism of doxorubicin-induced cardiotoxicity: Have we been barking up the wrong tree? Redox Biology, 29, 101394.
Yu, J., Wang, C., Kong, Q., Wu, X., Lu, J.-J., & Chen, X. (2018). Recent progress in doxorubicin-induced cardiotoxicity and protective potential of natural products. Phytomedicine, 40, 125–139.
Wenningmann, N., Knapp, M., Ande, A., Vaidya, T. R., & Ait-Oudhia, S. (2019). Insights into doxorubicin-induced cardiotoxicity: Molecular mechanisms, preventive strategies, and early monitoring. Molecular Pharmacology, 96, 219–232.
Speth, P. A., van Hoesel, Q. G., & Haanen, C. (1988). Clinical pharmacokinetics of doxorubicin. Clinical Pharmacokinetics, 15, 15–31. https://doi.org/10.2165/00003088-198815010-00002
Von Hoff, D. D., Layard, M. W., Basa, P., Davis, H. L., Jr., Von Hoff, A. L., Rozencweig, M., & Muggia, F. M. (1979). Risk factors for doxorubicin-induced congestive heart failure. Annals of Internal Medicine, 91, 710–717. https://doi.org/10.7326/0003-4819-91-5-710
Steinherz, L. J., Steinherz, P. G., Tan, C. T. C., Heller, G., & Murphy, M. L. (1991). Cardiac toxicity 4 to 20 years after completing anthracycline therapy. JAMA, 266, 1672–1677. https://doi.org/10.1001/jama.1991.03470120074036
Ganz, P. A., Hussey, M. A., Moinpour, C. M., Unger, J. M., Hutchins, L. F., Dakhil, S. R., Giguere, J. K., Goodwin, J. W., Martino, S., & Albain, K. S. (2008). Late cardiac effects of adjuvant chemotherapy in breast cancer survivors treated on Southwest Oncology Group protocol s8897. Journal of Clinical Oncology, 26, 1223–1230. https://doi.org/10.1200/jco.2007.11.8877
Avagimyan, A., Mkrtchyan, L., Abrahomovich, O., Sheibani, M., Guevorkyan, A., Sarrafzadegan, N., Kozhukhov, S., Agati, L., Astengiano, R., Zaritska, V., & Jndoyan, Z. (2021). AC-mode of chemotherapy as a trigger of cardiac syndrome X: A case study. Current Problems in Cardiology. https://doi.org/10.1016/j.cpcardiol.2021.100994
Stark, C., Taimen, P., Savunen, T., & Koskenvuo, J. (2018). Pegylated and liposomal doxorubicin is associated with high mortality and causes limited cardiotoxicity in mice. BMC Research Notes, 11, 148. https://doi.org/10.1186/s13104-018-3260-6
Boucek, R. J., Kunkel, E. M., Graham, T. P., Brenner, D., & Olson, R. D. (1987). Doxorubicinol, the metabolite of doxorubucin, is more cardiotoxic than doxorubicin. Pediatric Research, 21, 187–187. https://doi.org/10.1203/00006450-198704010-00127
Olson, R. D., Mushlin, P. S., Brenner, D. E., Fleischer, S., Cusack, B. J., Chang, B. K., & Boucek, R. J., Jr. (1988). Doxorubicin cardiotoxicity may be caused by its metabolite, doxorubicinol. Proceedings of the National Academy of Sciences of the United States of America, 85, 3585–3589. https://doi.org/10.1073/pnas.85.10.3585
Lehenbauer Ludke, A. R., Al-Shudiefat, A.A.-R.S., Dhingra, S., Jassal, D. S., & Singal, P. K. (2009). A concise description of cardioprotective strategies in doxorubicin-induced cardiotoxicity. Canadian Journal of Physiology and Pharmacology, 87, 756–763.
Pecoraro, M., Del Pizzo, M., Marzocco, S., Sorrentino, R., Ciccarelli, M., Iaccarino, G., Pinto, A., & Popolo, A. (2016). Inflammatory mediators in a short-time mouse model of doxorubicin-induced cardiotoxicity. Toxicology and applied pharmacology, 293, 44–52.
Osataphan, N., Phrommintikul, A., Chattipakorn, S. C., & Chattipakorn, N. (2020). Effects of doxorubicin-induced cardiotoxicity on cardiac mitochondrial dynamics and mitochondrial function: Insights for future interventions. Journal of Cellular and Molecular Medicine, 24, 6534–6557.
Singal, P., Li, T., Kumar, D., Danelisen, I., & Iliskovic, N. (2000). Adriamycin-induced heart failure: Mechanisms and modulation. Molecular and Cellular Biochemistry, 207, 77–86.
Minotti, G., Menna, P., Salvatorelli, E., Cairo, G., & Gianni, L. (2004). Anthracyclines: Molecular advances and pharmacologic developments in antitumor activity and cardiotoxicity. Pharmacological Reviews, 56, 185–229.
Tokarska-Schlattner, M., Zaugg, M., Zuppinger, C., Wallimann, T., & Schlattner, U. (2006). New insights into doxorubicin-induced cardiotoxicity: The critical role of cellular energetics. Journal of Molecular and Cellular Cardiology, 41, 389–405.
Aldieri, E., Bergandi, L., Riganti, C., Costamagna, C., Bosia, A., & Ghigo, D. (2002). Doxorubicin induces an increase of nitric oxide synthesis in rat cardiac cells that is inhibited by iron supplementation. Toxicology and Applied Pharmacology, 185, 85–90.
Bahadır, A., Kurucu, N., Kadıoğlu, M., & Yenilme, E. (2014). The role of nitric oxide in Doxorubicin-induced cardiotoxicity: Experimental study. Turkish Journal of Hematology, 31, 68.
Nozaki, N., Shishido, T., Takeishi, Y., & Kubota, I. (2004). Modulation of doxorubicin-induced cardiac dysfunction in toll-like receptor-2-knockout mice. Circulation, 110, 2869–2874.
Riad, A., Bien, S., Gratz, M., Escher, F., Heimesaat, M. M., Bereswill, S., Krieg, T., Felix, S. B., Schultheiss, H. P., & Kroemer, H. K. (2008). Toll-like receptor-4 deficiency attenuates doxorubicin-induced cardiomyopathy in mice. European Journal of Heart Failure, 10, 233–243.
Hu, C., Zhang, X., Zhang, N., Wei, W. Y., Li, L. L., Ma, Z. G., & Tang, Q. Z. (2020). Osteocrin attenuates inflammation, oxidative stress, apoptosis, and cardiac dysfunction in doxorubicin-induced cardiotoxicity. Clinical and Translational Medicine, 10, e124.
Pecoraro, M., Sorrentino, R., Franceschelli, S., Del Pizzo, M., Pinto, A., & Popolo, A. (2015). Doxorubicin-mediated cardiotoxicity: Role of mitochondrial connexin 43. Cardiovascular Toxicology, 15, 366–376.
Lyu, Y. L., Kerrigan, J. E., Lin, C.-P., Azarova, A. M., Tsai, Y.-C., Ban, Y., & Liu, L. F. (2007). Topoisomerase IIβ–mediated DNA double-strand breaks: Implications in doxorubicin cardiotoxicity and prevention by dexrazoxane. Cancer Research, 67, 8839–8846.
Horie, T., Ono, K., Nishi, H., Nagao, K., Kinoshita, M., Watanabe, S., Kuwabara, Y., Nakashima, Y., Takanabe-Mori, R., & Nishi, E. (2010). Acute doxorubicin cardiotoxicity is associated with miR-146a-induced inhibition of the neuregulin-ErbB pathway. Cardiovascular Research, 87, 656–664.
Rohrbach, S., Muller-Werdan, U., Werdan, K., Koch, S., Gellerich, N. F., & Holtz, J. (2005). Apoptosis-modulating interaction of the neuregulin/erbB pathway with antracyclines in regulating Bcl-xS and Bcl-xL in cardiomyocytes. Journal of Molecular and Cellular Cardiology, 38, 485–493.
Renu, K., Abilash, V., & Arunachalam, S. (2018). Molecular mechanism of doxorubicin-induced cardiomyopathy–An update. European Journal of Pharmacology, 818, 241–253.
Lebrecht, D., Setzer, B., Ketelsen, U.-P., Haberstroh, J. R., & Walker, U. A. (2003). Time-dependent and tissue-specific accumulation of mtDNA and respiratory chain defects in chronic doxorubicin cardiomyopathy. Circulation, 108, 2423–2429.
Lebrecht, D., Geist, A., Ketelsen, U. P., Haberstroh, J., Setzer, B., & Walker, U. (2007). Dexrazoxane prevents doxorubicin-induced long-term cardiotoxicity and protects myocardial mitochondria from genetic and functional lesions in rats. British Journal of Pharmacology, 151, 771–778.
Montaigne, D., Marechal, X., Baccouch, R., Modine, T., Preau, S., Zannis, K., Marchetti, P., Lancel, S., & Neviere, R. (2010). Stabilization of mitochondrial membrane potential prevents doxorubicin-induced cardiotoxicity in isolated rat heart. Toxicology and Applied Pharmacology, 244, 300–307.
Danz, E. D. B., Skramsted, J., Henry, N., Bennett, J. A., & Keller, R. S. (2009). Resveratrol prevents doxorubicin cardiotoxicity through mitochondrial stabilization and the Sirt1 pathway. Free Radical Biology and Medicine, 46, 1589–1597.
Liu, Y., Asnani, A., Zou, L., Bentley, V. L., Yu, M., Wang, Y., Dellaire, G., Sarkar, K. S., Dai, M., Chen, H. H., Sosnovik, D. E., Shin, J. T., Haber, D. A., Berman, J. N., Chao, W., & Peterson, R. T. (2014). Visnagin protects against doxorubicin-induced cardiomyopathy through modulation of mitochondrial malate dehydrogenase. Science Translational Medicine, 6, 266ra170. https://doi.org/10.1126/scitranslmed.3010189
Fisher, P. W., Salloum, F., Das, A., Hyder, H., & Kukreja, R. C. (2005). Phosphodiesterase-5 inhibition with sildenafil attenuates cardiomyocyte apoptosis and left ventricular dysfunction in a chronic model of doxorubicin cardiotoxicity. Circulation, 111, 1601–1610. https://doi.org/10.1161/01.cir.0000160359.49478.c2
Beak, J., Huang, W., Parker, J. S., Hicks, S. T., Patterson, C., Simpson, P. C., Ma, A., Jin, J., & Jensen, B. C. (2017). An oral selective alpha-1A adrenergic receptor agonist prevents doxorubicin cardiotoxicity. JACC. Basic to translational science, 2, 39–53. https://doi.org/10.1016/j.jacbts.2016.10.006
Yourtee, D. M., Elkins, L. L., Nalvarte, E. L., & Smith, R. E. (1992). Amplification of doxorubicin mutagenicity by cupric ion. Toxicology and Applied Pharmacology, 116, 57–65.
Colombo, R., Dalle Donne, I., & Milzani, A. (1990). Metal ions modulate the effect of doxorubicin on actin assembly. Cancer Biochemistry Biophysics, 11, 217–226.
Shi, Y., Moon, M., Dawood, S., McManus, B., & Liu, P. (2011). Mechanisms and management of doxorubicin cardiotoxicity. Herz, 36, 296–305.
Panjrath, G. S., Patel, V., Valdiviezo, C. I., Narula, N., Narula, J., & Jain, D. (2007). Potentiation of doxorubicin cardiotoxicity by iron loading in a rodent model. Journal of the American College of Cardiology, 49, 2457–2464.
Al-Shabanah, O. A., Aleisa, A. M., Hafez, M. M., Al-Rejaie, S. S., Al-Yahya, A. A., Bakheet, S. A., Al-Harbi, M. M., & Sayed-Ahmed, M. M. (2012). Desferrioxamine attenuates doxorubicin-induced acute cardiotoxicity through TFG-/Smad p53 pathway in rat model. Oxidative Medicine and Cellular Longevity. https://doi.org/10.1155/2012/619185
Al-Harbi, M., Al-Gharably, N. M., Al-Shabanah, O. A., Al-Bekairi, A. M., Osman, A. M. M., & Tawfik, H. N. (1992). Prevention of doxorubicin-induced myocardial and haematological toxicities in rats by the iron chelator desferrioxamine. Cancer Chemotherapy and Pharmacology, 31, 200–204.
Ammar, E.-S.M., Said, S. A., Suddek, G. M., & El-Damarawy, S. L. (2011). Amelioration of doxorubicin-induced cardiotoxicity by deferiprone in rats. Canadian Journal of Physiology and Pharmacology, 89, 269–276.
van Acker, S. A., van Balen, G. P., van den Berg, D. J., Bast, A., & van der Vijgh, W. J. (1998). Influence of iron chelation on the antioxidant activity of flavonoids. Biochemical Pharmacology, 56, 935–943.
Van Acker, S. A., Tromp, M. N., Griffioen, D. H., Van Bennekom, W. P., Van Der Vijgh, W. J., & Bast, A. (1996). Structural aspects of antioxidant activity of flavonoids. Free Radical Biology and Medicine, 20, 331–342.
Bast, A., Haenen, G. R., Bruynzeel, A. M., & Van der Vijgh, W. J. (2007). Protection by flavonoids against anthracycline cardiotoxicity: From chemistry to clinical trials. Cardiovascular Toxicology, 7, 154–159.
Willems, A. M., Bruynzeel, A. M., Kedde, M. A., Van Groeningen, C. J., Bast, A., & Van Der Vijgh, W. J. (2006). A phase I study of monohydroxyethylrutoside in healthy volunteers. Cancer Chemotherapy and Pharmacology, 57, 678–684.
Bast, A., Kaiserová, H., Den Hartog, G., Haenen, G., & Van Der Vijgh, W. (2007). Protectors against doxorubicin-induced cardiotoxicity: Flavonoids. Cell Biology and Toxicology, 23, 39–47.
Ehrke, M. J., Ryoyama, K., & Cohen, S. A. (1984). Cellular basis for adriamycin-induced augmentation of cell-mediated cytotoxicity in culture. Cancer Research, 44, 2497–2504.
Maccubbin, D. L., Wing, K. R., Mace, K. F., Ho, R. L., Ehrke, M. J., & Mihich, E. (1992). Adriamycin-induced modulation of host defenses in tumor-bearing mice. Cancer Research, 52, 3572–3576.
Nagai, K., Fukuno, S., Oda, A., & Konishi, H. (2016). Protective effects of taurine on doxorubicin-induced acute hepatotoxicity through suppression of oxidative stress and apoptotic responses. Anti-cancer Drugs, 27, 17–23.
Wang, S., Kotamraju, S., Konorev, E., Kalivendi, S., Joseph, J., & Kalyanaraman, B. (2002). Activation of nuclear factor-κB during doxorubicin-induced apoptosis in endothelial cells and myocytes is pro-apoptotic: The role of hydrogen peroxide. Biochemical Journal, 367, 729–740.
Sheibani, M., Nezamoleslami, S., Faghir-Ghanesefat, H., Hossein Emami, A., & Dehpour, A. R. (2020). Cardioprotective effects of dapsone against doxorubicin-induced cardiotoxicity in rats. Cancer Chemotherapy and Pharmacology, 85, 563–571.
Sun, Z., Yan, B., Yu, W. Y., Yao, X., Ma, X., Sheng, G., & Ma, Q. (2016). Vitexin attenuates acute doxorubicin cardiotoxicity in rats via the suppression of oxidative stress, inflammation and apoptosis and the activation of FOXO3a. Experimental and Therapeutic Medicine, 12, 1879–1884.
Shaker, R. A., Abboud, S. H., Assad, H. C., & Hadi, N. (2018). Enoxaparin attenuates doxorubicin induced cardiotoxicity in rats via interfering with oxidative stress, inflammation and apoptosis. BMC Pharmacology and Toxicology, 19, 1–10.
Inchiosa, M. A., Jr., & Smith, C. M. (1990). Effects of ibuprofen on doxorubicin toxicity. Research Communications in Chemical Pathology and Pharmacology, 67, 63–78.
Guo, R., Wu, K., Chen, J., Mo, L., Hua, X., Zheng, D., Chen, P., Chen, G., Xu, W., & Feng, J. (2013). Exogenous hydrogen sulfide protects against doxorubicin-induced inflammation and cytotoxicity by inhibiting p38MAPK/NFκB pathway in H9c2 cardiac cells. Cellular Physiology and Biochemistry, 32, 1668–1680.
Yarmohammadi, F., Rezaee, R., & Karimi, G. (2021). Natural compounds against doxorubicin-induced cardiotoxicity: A review on the involvement of Nrf2/ARE signaling pathway. Phytotherapy Research, 35, 1163–1175.
Xiong, C., Wu, Y. Z., Zhang, Y., Wu, Z. X., Chen, X. Y., Jiang, P., Guo, H. C., Xie, K. R., Wang, K. X., & Su, S. W. (2018). Protective effect of berberine on acute cardiomyopathy associated with doxorubicin treatment. Oncology Letters, 15, 5721–5729.
Asensio-López, M. C., Soler, F., Pascual-Figal, D., Fernández-Belda, F., & Lax, A. (2017). Doxorubicin-induced oxidative stress: The protective effect of nicorandil on HL-1 cardiomyocytes. PLoS ONE, 12, e0172803.
Morgan, M. J., & Liu, Z. G. (2011). Crosstalk of reactive oxygen species and NF-κB signaling. Cell Research, 21, 103–115. https://doi.org/10.1038/cr.2010.178
Zhao, Y., Miriyala, S., Miao, L., Mitov, M., Schnell, D., Dhar, S. K., Cai, J., Klein, J. B., Sultana, R., Butterfield, D. A., Vore, M., Batinic-Haberle, I., Bondada, S., & St Clair, D. K. (2014). Redox proteomic identification of HNE-bound mitochondrial proteins in cardiac tissues reveals a systemic effect on energy metabolism after doxorubicin treatment. Free Radical Biology & Medicine, 72, 55–65. https://doi.org/10.1016/j.freeradbiomed.2014.03.001
Guo, C., Sun, L., Chen, X., & Zhang, D. (2013). Oxidative stress, mitochondrial damage and neurodegenerative diseases. Neural Regeneration Research, 8, 2003–2014. https://doi.org/10.3969/j.issn.1673-5374.2013.21.009
Sardão, V. A., Oliveira, P. J., Holy, J., Oliveira, C. R., & Wallace, K. B. (2009). Doxorubicin-induced mitochondrial dysfunction is secondary to nuclear p53 activation in H9c2 cardiomyoblasts. Cancer Chemotherapy and Pharmacology, 64, 811–827. https://doi.org/10.1007/s00280-009-0932-x
Ma, Q. (2013). Role of nrf2 in oxidative stress and toxicity. Annual Review of Pharmacology and Toxicology, 53, 401–426. https://doi.org/10.1146/annurev-pharmtox-011112-140320
Loboda, A., Damulewicz, M., Pyza, E., Jozkowicz, A., & Dulak, J. (2016). Role of Nrf2/HO-1 system in development, oxidative stress response and diseases: An evolutionarily conserved mechanism. Cellular and Molecular Life Sciences: CMLS, 73, 3221–3247. https://doi.org/10.1007/s00018-016-2223-0
Singh, P., Sharma, R., McElhanon, K., Allen, C. D., Megyesi, J. K., Beneš, H., & Singh, S. P. (2015). Sulforaphane protects the heart from doxorubicin-induced toxicity. Free Radical Biology & Medicine, 86, 90–101. https://doi.org/10.1016/j.freeradbiomed.2015.05.028
Chen, M., Samuel, V. P., Wu, Y., Dang, M., Lin, Y., Sriramaneni, R., Sah, S. K., Chinnaboina, G. K., & Zhang, G. (2019). Nrf2/HO-1 mediated protective activity of genistein against doxorubicin-induced cardiac toxicity. Journal of Environmental Pathology, Toxicology and Oncology, 38, 143–152. https://doi.org/10.1615/JEnvironPatholToxicolOncol.2019029341
Cheng, X., Liu, D., Xing, R., Song, H., Tian, X., Yan, C., & Han, Y. (2020). Orosomucoid 1 attenuates doxorubicin-induced oxidative stress and apoptosis in cardiomyocytes via Nrf2 signaling. BioMed Research International, 2020, 5923572. https://doi.org/10.1155/2020/5923572
El-Agamy, D. S., El-Harbi, K. M., Khoshhal, S., Ahmed, N., Elkablawy, M. A., Shaaban, A. A., & Abo-Haded, H. M. (2019). Pristimerin protects against doxorubicin-induced cardiotoxicity and fibrosis through modulation of Nrf2 and MAPK/NF-kB signaling pathways. Cancer Management and Research, 11, 47–61. https://doi.org/10.2147/cmar.s186696
Fadillioglu, E., Oztas, E., Erdogan, H., Yagmurca, M., Sogut, S., Ucar, M., & Irmak, M. K. (2004). Protective effects of caffeic acid phenethyl ester on doxorubicin-induced cardiotoxicity in rats. Journal of Applied Toxicology: An International Journal, 24, 47–52.
Alkreathy, H., Damanhouri, Z. A., Ahmed, N., Slevin, M., Ali, S. S., & Osman, A.-M.M. (2010). Aged garlic extract protects against doxorubicin-induced cardiotoxicity in rats. Food and Chemical Toxicology, 48, 951–956.
Sheibani, M., Faghir-Ghanesefat, H., Dehpour, S., Keshavarz-Bahaghighat, H., Sepand, M. R., Ghahremani, M. H., Azizi, Y., Rahimi, N., & Dehpour, A. R. (2019). Sumatriptan protects against myocardial ischaemia-reperfusion injury by inhibition of inflammation in rat model. Inflammopharmacology, 27, 1071–1080. https://doi.org/10.1007/s10787-019-00586-5
Bazmandegan, G., Amirteimoury, M., Kaeidi, A., Shamsizadeh, A., Khademalhosseini, M., Nematollahi, M. H., Hassanipour, M., & Fatemi, I. (2019). Sumatriptan ameliorates renal injury induced by cisplatin in mice. Iranian Journal of Basic Medical Sciences, 22, 563–567. https://doi.org/10.22038/ijbms.2019.33620.8020
Eslami, F., Rahimi, N., Ostovaneh, A., Ghasemi, M., Dejban, P., Abbasi, A., & Dehpour, A. R. (2021). Sumatriptan reduces severity of status epilepticus induced by lithium-pilocarpine through nitrergic transmission and 5-HT(1B/D) receptors in rats: A pharmacological-based evidence. Fundamental & Clinical Pharmacology, 35, 131–140. https://doi.org/10.1111/fcp.12590
Mohammad, S., Hedyeh, F.-G., Yaser, A., Tahmineh, M., Hasan, Y. M., Roya Sattarzadeh, B., Amir Hossein, E., & Ahmad Reza, D. (2021). Anti-inflammatory and antioxidative effects of sumatriptan against doxorubicin-induced cardiotoxicity in rat. Acta Medica Iranica. https://doi.org/10.18502/acta.v59i7.7020
Iqbal, M., Dubey, K., Anwer, T., Ashish, A., & Pillai, K. K. (2008). Protective effects of telmisartan against acute doxorubicin-induced cardiotoxicity in rats. Pharmacological Reports, 60, 382.
Siveski-Iliskovic, N., Hill, M., Chow, D. A., & Singal, P. K. (1995). Probucol protects against adriamycin cardiomyopathy without interfering with its antitumor effect. Circulation, 91, 10–15. https://doi.org/10.1161/01.cir.91.1.10
Siveski-Iliskovic, N., Kaul, N., & Singal, P. K. (1994). Probucol promotes endogenous antioxidants and provides protection against adriamycin-induced cardiomyopathy in rats. Circulation, 89, 2829–2835. https://doi.org/10.1161/01.cir.89.6.2829
Nagi, M. N., & Mansour, M. A. (2000). Protective effect of thymoquinone against doxorubicin-induced cardiotoxicity in rats: A possible mechanism of protection. Pharmacological Research, 41, 283–289.
Sadzuka, Y., Sugiyama, T., Shimoi, K., Kinae, N., & Hirota, S. (1997). Protective effect of flavonoids on doxorubicin-induced cardiotoxicity. Toxicology letters, 92, 1–7.
Zhang, Y., Ma, C., Liu, C., & Wei, F. (2020). Luteolin attenuates doxorubicin-induced cardiotoxicity by modulating the PHLPP1/AKT/Bcl-2 signalling pathway. PeerJ, 8, e8845.
Chen, J.-Y., Hu, R.-Y., & Chou, H.-C. (2013). Quercetin-induced cardioprotection against doxorubicin cytotoxicity. Journal of Biomedical Science, 20, 1–11.
Kaiserová, H., Šimůnek, T., van der Vijgh, W. J., Bast, A., & Kvasničková, E. (2007). Flavonoids as protectors against doxorubicin cardiotoxicity: Role of iron chelation, antioxidant activity and inhibition of carbonyl reductase. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease, 1772, 1065–1074.
Laughton, M. J., Halliwell, B., Evans, P. J., Robin, J., & Hoult, S. (1989). Antioxidant and pro-oxidant actions of the plant phenolics quercetin, gossypol and myricetin: Effects on lipid peroxidation, hydroxyl radical generation and bleomycin-dependent damage to DNA. Biochemical Pharmacology, 38, 2859–2865.
Chang, D., Li, H., Qian, C., & Wang, Y. (2019). Diohf protects against doxorubicin-induced cardiotoxicity through ERK1 signaling pathway. Frontiers in Pharmacology, 10, 1081.
Wang, H., Yu, P., Gou, H., Zhang, J., Zhu, M., Wang, Z. H., Tian, J. W., Jiang, Y. T., & Fu, F. H. (2012). Cardioprotective effects of 20(S)-ginsenoside Rh2 against doxorubicin-induced cardiotoxicity in vitro and in vivo. Evidence-Based Complementary and Alternative Medicine, 2012, 506214. https://doi.org/10.1155/2012/506214
Li, L., Pan, Q., Han, W., Liu, Z., Li, L., & Hu, X. (2007). Schisandrin B prevents doxorubicin-induced cardiotoxicity via enhancing glutathione redox cycling. Clinical Cancer Research, 13, 6753–6760. https://doi.org/10.1158/1078-0432.Ccr-07-1579
Zhu, W., Soonpaa, M. H., Chen, H., Shen, W., Payne, R. M., Liechty, E. A., Caldwell, R. L., Shou, W., & Field, L. J. (2009). Acute doxorubicin cardiotoxicity is associated with p53-induced inhibition of the mammalian target of rapamycin pathway. Circulation, 119, 99–106. https://doi.org/10.1161/CIRCULATIONAHA.108.799700
Timm, K. N., & Tyler, D. J. (2020). The role of AMPK activation for cardioprotection in doxorubicin-induced cardiotoxicity. Cardiovascular Drugs and Therapy, 34, 255–269. https://doi.org/10.1007/s10557-020-06941-x
Kobashigawa, L. C., Xu, Y. C., Padbury, J. F., Tseng, Y.-T., & Yano, N. (2014). Metformin protects cardiomyocyte from doxorubicin induced cytotoxicity through an AMP-activated protein kinase dependent signaling pathway: an in vitro study. PLoS ONE, 9, e104888.
Zilinyi, R., Czompa, A., Czegledi, A., Gajtko, A., Pituk, D., Lekli, I., & Tosaki, A. (2018). The cardioprotective effect of metformin in doxorubicin-induced cardiotoxicity: The role of autophagy. Molecules (Basel, Switzerland). https://doi.org/10.3390/molecules23051184
Huelsenbeck, J., Henninger, C., Schad, A., Lackner, K. J., Kaina, B., & Fritz, G. (2011). Inhibition of Rac1 signaling by lovastatin protects against anthracycline-induced cardiac toxicity. Cell Death & Disease, 2, e190. https://doi.org/10.1038/cddis.2011.65
Rawat, P. S., Jaiswal, A., Khurana, A., Bhatti, J. S., & Navik, U. (2021). Doxorubicin-induced cardiotoxicity: An update on the molecular mechanism and novel therapeutic strategies for effective management. Biomedicine & Pharmacotherapy, 139, 111708. https://doi.org/10.1016/j.biopha.2021.111708
Vedam, K., Nishijima, Y., Druhan, L. J., Khan, M., Moldovan, N. I., Zweier, J. L., & Ilangovan, G. (2010). Role of heat shock factor-1 activation in the doxorubicin-induced heart failure in mice. American Journal of Physiology, Heart and Circulatory Physiology, 298, H1832–H1841. https://doi.org/10.1152/ajpheart.01047.2009
Liu, P., Bao, H. Y., Jin, C. C., Zhou, J. C., Hua, F., Li, K., Lv, X. X., Cui, B., Hu, Z. W., & Zhang, X. W. (2019). Targeting extracellular heat shock protein 70 ameliorates doxorubicin-induced heart failure through resolution of toll-like receptor 2-mediated myocardial inflammation. Journal of the American Heart Association, 8, e012338. https://doi.org/10.1161/jaha.119.012338
Shan, Y. X., Liu, T. J., Su, H. F., Samsamshariat, A., Mestril, R., & Wang, P. H. (2003). Hsp10 and Hsp60 modulate Bcl-2 family and mitochondria apoptosis signaling induced by doxorubicin in cardiac muscle cells. Journal of Molecular and Cellular Cardiology, 35, 1135–1143. https://doi.org/10.1016/s0022-2828(03)00229-3
Zhao, D., Xue, C., Li, J., Feng, K., Zeng, P., Chen, Y., Duan, Y., Zhang, S., Li, X., & Han, J. (2020). Adiponectin agonist ADP355 ameliorates doxorubicin-induced cardiotoxicity by decreasing cardiomyocyte apoptosis and oxidative stress. Biochemical and Biophysical Research Communications, 533, 304–312.
Ramond, A., Sartorius, E., Mousseau, M., Ribuot, C., & Joyeux-Faure, M. (2008). Erythropoietin pretreatment protects against acute chemotherapy toxicity in isolated rat hearts. Experimental Biology and Medicine, 233, 76–83. https://doi.org/10.3181/0706-RM-152
Man, S. M., Karki, R., Briard, B., Burton, A., Gingras, S., Pelletier, S., & Kanneganti, T.-D. (2017). Differential roles of caspase-1 and caspase-11 in infection and inflammation. Scientific Reports, 7, 45126. https://doi.org/10.1038/srep45126
Yu, W., Qin, X., Zhang, Y., Qiu, P., Wang, L., Zha, W., & Ren, J. (2020). Curcumin suppresses doxorubicin-induced cardiomyocyte pyroptosis via a PI3K/Akt/mTOR-dependent manner. Cardiovascular Diagnosis and Therapy, 10, 752–769. https://doi.org/10.21037/cdt-19-707
Chen, Y. L., Chung, S. Y., Chai, H. T., Chen, C. H., Liu, C. F., Chen, Y. L., Huang, T. H., Zhen, Y. Y., Sung, P. H., Sun, C. K., Chua, S., Lu, H. I., Lee, F. Y., Sheu, J. J., & Yip, H. K. (2015). Early administration of carvedilol protected against doxorubicin-induced cardiomyopathy. The Journal of Pharmacology and Experimental Therapeutics, 355, 516–527. https://doi.org/10.1124/jpet.115.225375
Dulhunty, A., & Casarotto, B. (2011). The ryanodine receptor: A pivotal Ca2+ regulatory protein and potential therapeutic drug target. Current Drug Targets, 12, 709–723.
Dewenter, M., von der Lieth, A., Katus, H. A., & Backs, J. (2017). Calcium signaling and transcriptional regulation in cardiomyocytes. Circulation Research, 121, 1000–1020.
Aziz, A. U. R., Geng, C., Li, W., Yu, X., Qin, K.-R., Wang, H., & Liu, B. (2019). Doxorubicin induces ER calcium release via Src in rat ovarian follicles. Toxicological Sciences, 168, 171–178.
Octavia, Y., Tocchetti, C. G., Gabrielson, K. L., Janssens, S., Crijns, H. J., & Moens, A. L. (2012). Doxorubicin-induced cardiomyopathy: From molecular mechanisms to therapeutic strategies. Journal of Molecular and Cellular Cardiology, 52, 1213–1225.
Yarmohmmadi, F., Rahimi, N., Faghir-Ghanesefat, H., Javadian, N., Abdollahi, A., Pasalar, P., Jazayeri, F., Ejtemaeemehr, S., & Dehpour, A. R. (2017). Protective effects of agmatine on doxorubicin-induced chronic cardiotoxicity in rat. European Journal of Pharmacology, 796, 39–44.
Khalilzadeh, M., Abdollahi, A., Abdolahi, F., Abdolghaffari, A. H., Dehpour, A. R., & Jazaeri, F. (2018). Protective effects of magnesium sulfate against doxorubicin induced cardiotoxicity in rats. Life Sciences, 207, 436–441.
Gross, R. A., Moises, H. C., Uhler, M. D., & Macdonald, R. L. (1990). Dynorphin A and cAMP-dependent protein kinase independently regulate neuronal calcium currents. Proceedings of the National Academy of Sciences, 87, 7025–7029.
North, R. A., Williams, J. T., Surprenant, A., & Christie, M. J. (1987). Mu and delta receptors belong to a family of receptors that are coupled to potassium channels. Proceedings of the National Academy of Sciences, 84, 5487–5491.
Lashgari, N. A., Roudsari, N. M., Zandi, N., Pazoki, B., Rezaei, A., Hashemi, M., Momtaz, S., Rahimi, R., Shayan, M., Dehpour, A. R., & Abdolghaffari, A. H. (2021). Current overview of opioids in progression of inflammatory bowel disease; pharmacological and clinical considerations. Molecular Biology Reports, 48, 855–874. https://doi.org/10.1007/s11033-020-06095-x
Zamanian, G., Shayan, M., Rahimi, N., Bahremand, T., Shafaroodi, H., Ejtemaei-Mehr, S., Aghaei, I., & Dehpour, A. R. (2020). Interaction of morphine tolerance with pentylenetetrazole-induced seizure threshold in mice: The role of NMDA-receptor/NO pathway. Epilepsy & Behavior, 112, 107343. https://doi.org/10.1016/j.yebeh.2020.107343
Kelishomi, R. B., Ejtemaeemehr, S., Tavangar, S. M., Rahimian, R., Mobarakeh, J. I., & Dehpour, A. R. (2008). Morphine is protective against doxorubicin-induced cardiotoxicity in rat. Toxicology, 243, 96–104.
Goodenough, D. A., & Paul, D. L. (2009). Gap junctions. Cold Spring Harbor Perspectives in Biology, 1, a002576.
Severs, N. J. (1994). Pathophysiology of gap junctions in heart disease. Journal of Cardiovascular Electrophysiology, 5, 462–475.
Siti, H. N., Jalil, J., Asmadi, A. Y., & Kamisah, Y. (2020). Effects of quercetin on cardiac function in pressure overload and postischemic cardiac injury in rodents: A systematic review and meta-analysis. Cardiovascular Drugs and Therapy, 16, 1–15.
Srisakuldee, W., Makazan, Z., Nickel, B. E., Zhang, F., Thliveris, J. A., Pasumarthi, K. B., & Kardami, E. (2014). The FGF-2-triggered protection of cardiac subsarcolemmal mitochondria from calcium overload is mitochondrial connexin 43-dependent. Cardiovascular Research, 103, 72–80.
Pecoraro, M., Ciccarelli, M., Fiordelisi, A., Iaccarino, G., Pinto, A., & Popolo, A. (2018). Diazoxide improves mitochondrial connexin 43 expression in a mouse model of doxorubicin-induced cardiotoxicity. International Journal of Molecular Sciences. https://doi.org/10.3390/ijms19030757
Elhadidy, M. G., Elmasry, A., Rabei, M. R., & Eladel, A. E. (2020). Effect of ghrelin on VEGF-B and connexin-43 in a rat model of doxorubicin-induced cardiomyopathy. Journal of Basic and Clinical Physiology and Pharmacology. https://doi.org/10.1515/jbcpp-2018-0212
Zhang, H., Zhang, A., Guo, C., Shi, C., Zhang, Y., Liu, Q., Sparatore, A., & Wang, C. (2011). S-diclofenac protects against doxorubicin-induced cardiomyopathy in mice via ameliorating cardiac gap junction remodeling. PLoS ONE, 6, e26441.
Michan, S., & Sinclair, D. (2007). Sirtuins in mammals: Insights into their biological function. Biochemical Journal, 404, 1–13.
Tomaselli, D., Steegborn, C., Mai, A., & Rotili, D. (2020). Sirt4: A multifaceted enzyme at the crossroads of mitochondrial metabolism and cancer. Frontiers in Oncology, 10, 474.
Dolinsky, V. W. (2017). The role of sirtuins in mitochondrial function and doxorubicin-induced cardiac dysfunction. Biological Chemistry, 398, 955–974.
Cheung, K. G., Cole, L. K., Xiang, B., Chen, K., Ma, X., Myal, Y., Hatch, G. M., Tong, Q., & Dolinsky, V. W. (2015). Sirtuin-3 (SIRT3) protein attenuates doxorubicin-induced oxidative stress and improves mitochondrial respiration in H9c2 cardiomyocytes. Journal of Biological Chemistry, 290, 10981–10993.
Liu, M. H., Shan, J., Li, J., Zhang, Y., & Lin, X. L. (2016). Resveratrol inhibits doxorubicin-induced cardiotoxicity via sirtuin 1 activation in H9c2 cardiomyocytes. Experimental and Therapeutic Medicine, 12, 1113–1118.
Ruan, Y., Dong, C., Patel, J., Duan, C., Wang, X., Wu, X., Cao, Y., Pu, L., Lu, D., & Shen, T. (2015). SIRT1 suppresses doxorubicin-induced cardiotoxicity by regulating the oxidative stress and p38MAPK pathways. Cellular Physiology and Biochemistry, 35, 1116–1124.
Pillai, V. B., Kanwal, A., Fang, Y. H., Sharp, W. W., Samant, S., Arbiser, J., & Gupta, M. P. (2017). Honokiol, an activator of Sirtuin-3 (SIRT3) preserves mitochondria and protects the heart from doxorubicin-induced cardiomyopathy in mice. Oncotarget, 8, 34082.
Needham, D. M., Shufelt, K. A., Tomlinson, G., Scholey, J. W., & Newton, G. E. (2004). Troponin I and T levels in renal failure patients without acute coronary syndrome: A systematic review of the literature. The Canadian Journal of Cardiology, 20, 1212–1218.
Jin, J.-P. (2016). Evolution, regulation, and function of N-terminal variable region of troponin T: Modulation of muscle contractility and beyond. International Review of Cell and Molecular Biology, 321, 1–28.
Bleuel, H., Deschl, U., Bertsch, T., Bölz, G., & Rebel, W. (1995). Diagnostic efficiency of troponin T measurements in rats with experimental myocardial cell damage. Experimental and Toxicologic Pathology, 47, 121–127.
Mair, J., & Apple, F. (1997). Progress in myocardial damage detection: New biochemical markers for clinicians. Critical Reviews in Clinical Laboratory Sciences, 34, 1–66.
Rahimi Balaei, M., Momeny, M., Babaeikelishomi, R., Ejtemaei Mehr, S., Tavangar, S. M., & Dehpour, A. R. (2010). The modulatory effect of lithium on doxorubicin-induced cardiotoxicity in rat. European Journal of Pharmacology, 641, 193–198. https://doi.org/10.1016/j.ejphar.2010.05.046
Chang, E. A., Jin, S. W., Nam, M. H., & Kim, S. D. (2019). Human induced pluripotent stem cells: Clinical significance and applications in neurologic diseases. Journal of Korean Neurosurgical Society, 62, 493–501. https://doi.org/10.3340/jkns.2018.0222
Matsui, T., Miyamoto, N., Saito, F., & Shinozawa, T. (2020). Molecular profiling of human induced pluripotent stem cell-derived cells and their application for drug safety study. Current Pharmaceutical Biotechnology, 21, 807–828. https://doi.org/10.2174/1389201021666200422090952
Devalla, H. D., & Passier, R. (2018). Cardiac differentiation of pluripotent stem cells and implications for modeling the heart in health and disease. Science Translational Medicine. https://doi.org/10.1126/scitranslmed.aah5457
Blinova, K., Dang, Q., Millard, D., Smith, G., Pierson, J., Guo, L., Brock, M., Lu, H. R., Kraushaar, U., Zeng, H., Shi, H., Zhang, X., Sawada, K., Osada, T., Kanda, Y., Sekino, Y., Pang, L., Feaster, T. K., Kettenhofen, R., … Gintant, G. (2018). International multisite study of human-induced pluripotent stem cell-derived cardiomyocytes for drug proarrhythmic potential assessment. Cell Reports, 24, 3582–3592. https://doi.org/10.1016/j.celrep.2018.08.079
Schwach, V., Slaats, R. H., & Passier, R. (2020). Human pluripotent stem cell-derived cardiomyocytes for assessment of anticancer drug-induced cardiotoxicity. Frontiers in Cardiovascular Medicine. https://doi.org/10.3389/fcvm.2020.00050
Maillet, A., Tan, K., Chai, X., Sadananda, S. N., Mehta, A., Ooi, J., Hayden, M. R., Pouladi, M. A., Ghosh, S., Shim, W., & Brunham, L. R. (2016). Modeling doxorubicin-induced cardiotoxicity in human pluripotent stem cell derived-cardiomyocytes. Science and Reports, 6, 25333. https://doi.org/10.1038/srep25333
Burridge, P. W., Li, Y. F., Matsa, E., Wu, H., Ong, S. G., Sharma, A., Holmström, A., Chang, A. C., Coronado, M. J., Ebert, A. D., Knowles, J. W., Telli, M. L., Witteles, R. M., Blau, H. M., Bernstein, D., Altman, R. B., & Wu, J. C. (2016). Human induced pluripotent stem cell-derived cardiomyocytes recapitulate the predilection of breast cancer patients to doxorubicin-induced cardiotoxicity. Nature Medicine, 22, 547–556. https://doi.org/10.1038/nm.4087
Cui, N., Wu, F., Lu, W. J., Bai, R., Ke, B., Liu, T., Li, L., Lan, F., & Cui, M. (2019). Doxorubicin-induced cardiotoxicity is maturation dependent due to the shift from topoisomerase IIα to IIβ in human stem cell derived cardiomyocytes. Journal of Cellular and Molecular Medicine, 23, 4627–4639. https://doi.org/10.1111/jcmm.14346
Zhao, L., & Zhang, B. (2017). Doxorubicin induces cardiotoxicity through upregulation of death receptors mediated apoptosis in cardiomyocytes. Science and Reports, 7, 44735. https://doi.org/10.1038/srep44735
McSweeney, K. M., Bozza, W. P., Alterovitz, W.-L., & Zhang, B. (2019). Transcriptomic profiling reveals p53 as a key regulator of doxorubicin-induced cardiotoxicity. Cell Death Discovery, 5, 102. https://doi.org/10.1038/s41420-019-0182-6
Li, J., Wang, P. Y., Long, N. A., Zhuang, J., Springer, D. A., Zou, J., Lin, Y., Bleck, C. K. E., Park, J. H., Kang, J. G., & Hwang, P. M. (2019). p53 prevents doxorubicin cardiotoxicity independently of its prototypical tumor suppressor activities. Proceedings of the National Academy of Sciences of the United States of America, 116, 19626–19634. https://doi.org/10.1073/pnas.1904979116
Deng, S., Yan, T., Jendrny, C., Nemecek, A., Vincetic, M., Gödtel-Armbrust, U., & Wojnowski, L. (2014). Dexrazoxane may prevent doxorubicin-induced DNA damage via depleting both Topoisomerase II isoforms. BMC Cancer, 14, 842. https://doi.org/10.1186/1471-2407-14-842
Ahmed, R. E., Anzai, T., Chanthra, N., & Uosaki, H. (2020). A brief review of current maturation methods for human induced pluripotent stem cells-derived cardiomyocytes. Frontiers in Cell and Developmental Biology. https://doi.org/10.3389/fcell.2020.00178
Pang, L. (2020). Toxicity testing in the era of induced pluripotent stem cells: A perspective regarding the use of patient-specific induced pluripotent stem cell–derived cardiomyocytes for cardiac safety evaluation. Current Opinion in Toxicology, 23–24, 50–55. https://doi.org/10.1016/j.cotox.2020.04.001
Soma, Y., Morita, Y., Kishino, Y., Kanazawa, H., Fukuda, K., & Tohyama, S. (2021). The present state and future perspectives of cardiac regenerative therapy using human pluripotent stem cells. Frontiers in Cardiovascular Medicine. https://doi.org/10.3389/fcvm.2021.774389
Funding
This study was supported by Iran National Sciences Foundation (INSF).
Author information
Authors and Affiliations
Contributions
ARD and MS designed the study. SN, MS, and YA performed the literature search and data analysis. MS and YA drafted the manuscript and edited it. MHF prepared all figures. MS prepared Table 1. FE and SN made critical revision and edited the manuscript. ARD, MS, and MS made final editions to the paper prior to the submission. All authors have read and agreed to the submitted version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Ethical Approval
The protocol of this study was approved by the Ethics Committee of Tehran University of Medical Sciences.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Additional information
Handling Editor: Y. James Kang.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sheibani, M., Azizi, Y., Shayan, M. et al. Doxorubicin-Induced Cardiotoxicity: An Overview on Pre-clinical Therapeutic Approaches. Cardiovasc Toxicol 22, 292–310 (2022). https://doi.org/10.1007/s12012-022-09721-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12012-022-09721-1