Abstract
The potential mechanisms underlying the association between copper (Cu) exposure and impaired liver function are unclear. This study aimed to investigate the potential associations of dietary Cu intake and plasma Cu levels with liver function biomarkers. A cross-sectional study was performed to assess liver function biomarkers—namely, levels of total bilirubin (TBIL), direct bilirubin (DBIL), indirect bilirubin (IBIL), alanine transaminase (ALT), and aspartate transaminase (AST)—in 2376 subjects in Guangxi, China. Dietary Cu intake was determined from a food frequency questionnaire containing 109 common foods. Plasma Cu concentrations were determined by inductively coupled plasma‒mass spectrometry. Multiple linear regression and multivariate restricted cubic splines (RCS) were used to evaluate the correlations of plasma Cu levels and dietary Cu levels with liver function biomarkers. The covariate-adjusted results of the linear regression analysis showed that plasma Cu levels were significantly negatively correlated with serum IBIL (β = − 0.37), DBIL (β = − 0.22), and TBIL levels (β = − 0.32) (all p < 0.05), and dietary Cu was negatively correlated with serum AST levels (β = − 0.12, p < 0.05). The RCS analysis further indicated a negative linear relationship between dietary Cu levels and AST levels. In summary, our results suggested that the plasma Cu level is associated with serum bilirubin levels and that dietary Cu intake is associated with serum AST levels. Further studies are needed to validate these associations and elucidate the underlying mechanisms.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Liver disease is defined as pathological changes in the liver caused by multiple pathogenic factors, both internal and external [1]. Liver disease causes serious public health problems worldwide due to its high prevalence and poor long-term clinical outcomes [2]. In addition to the impact of viruses, bacteria, and nutritional factors on liver function, the association between metal exposure and biomarkers of liver function has received increasing attention [3]. Some metals, especially heavy metals, are easily absorbed by the blood and accumulate in the body, resulting in serious adverse health effects that are difficult to reverse [4]. Therefore, determining the effects of metal exposure on the health of the general population and elucidating the underlying mechanisms are important.
The liver is one of the main organs responsible for metal metabolism and detoxification [5]. Various blood protein or enzyme biomarkers have been used to assess hepatic function and diagnose liver diseases and liver injury [6, 7]. Commonly used clinical indicators of hepatocellular injury include levels of aspartate transaminase (AST), alanine transaminase (ALT), and bilirubin [8].
Copper (Cu) is one of the essential micronutrients for normal physiological functions, such as cellular respiration, protection against oxidative stress, and redox homeostasis [9, 10]. Additionally, Cu is a heavy metal and a frequent component of environmental pollutants. In the body, the liver is responsible for maintaining Cu homeostasis, and hepatocytes are the primary site of Cu metabolism within the liver, regulating Cu storage and excretion [11, 12]. Evidence of an association between environmental Cu exposure and alterations in biomarkers of liver function has accumulated in recent years. For example, positive correlations between blood Cu levels and elevated ALT and AST levels were reported in a study of rural residents in southwest China [13], and Wang et al. reported that urinary Cu levels were positively associated with serum ALT levels in the general population of city-dwelling Chinese adults [14].However, plasma Cu levels were negatively associated with total serum bilirubin (TBIL), AST, and ALT levels in a health cohort study of workers exposed to manganese [15]. Thus, the results of these studies are inconsistent. In addition, only a few population studies have reported an association between dietary Cu intake and liver injury or liver enzyme alterations [16,17,18]. Animal experiments have demonstrated that excessive or insufficient Cu intake causes liver injury [19,20,21,22]. Therefore, determining the appropriate dietary Cu intake will have important public health implications.
A paucity of studies has examined both dietary and plasma Cu levels, and evidence of linear or nonlinear dose‒response relationships between Cu exposure and liver function biomarkers is lacking. Therefore, we conducted a cross-sectional study to assess the relationships of dietary Cu intake and plasma Cu levels with liver function biomarkers.
Materials and Methods
Study Population
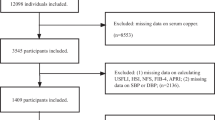
The study population was drawn from a rural population that participated in a health survey during 2018–2019 in Guangxi Yao Autonomous County in southwestern China. Study subjects who met the following criteria were included: (a) residing in the study area and (b) aged ≥ 30 years. The subjects were excluded based on the following criteria: (a) those who did not complete the questionnaire or provided incomplete information; (b) those with cardiovascular disease or liver diseases, such as liver cancer, liver fibrosis, or hepatitis virus; (c) those who took herbal medicine or antibiotics in the 2 weeks prior to the physical examination; and (d) those with abnormal metal values (defined as three times the 99th percentile of Cu levels). A total of 2376 subjects were included in the analysis. The study was approved by the Medical Ethics Committee of Guilin Medical University (no. 20180702–3). Written informed consent was provided by each participant.
Detection of Liver Function
Fasting blood samples were collected from an elbow vein in the morning after at least 12 h of fasting and transported to the Laboratory of Gongcheng Yao Autonomous County People’s Hospital by cold-chain transport on the same day. Serum TBIL, direct bilirubin (DBIL), indirect bilirubin (IBIL), ALT, and AST levels were measured by a chemical autoanalyzer (Hitachi 7600–020, Kyoto, Japan) to assess liver function.
Dietary Cu Assessment
Diet was assessed with the Food Frequency Questionnaire (FFQ), with reference to the questionnaires designed by Liu [23] and Wang et al. [24]. The FFQ is a reasonable, valid, and reliable method for assessing long-term average dietary intake [25, 26] of 109 common foods. The subjects were asked to recall and report their average frequency of consumption and the amount consumed per serving over the past year. The FFQ was administered with a food album that included dietary items and measurement tools, and the weight of each portion could be determined by a provided standardized portable electronic kitchen scale, which measured food portions in grams and drink portions in milliliters. The intake of each food item was derived by multiplying the serving size by the weight of each serving. The energy and nutrient contents of foods were calculated in reference to the Chinese Food Composition Table (2019) [27]. We calculated the total daily energy intake and dietary Cu intake as daily intake = intake amount/frequency.
Plasma Cu Assessment
Plasma samples were stored at – 80 °C until further analysis. Cu concentrations in plasma samples were determined by inductively coupled plasma‒mass spectrometry (ICP–MS) (Thermo Fisher Scientific iCAPRQ01408, USA). The detailed method used to determine metal exposure levels is described elsewhere [28]. The experiment was performed in accordance with the instruction manual. We diluted 0.1 mL of plasma to 2.0 mL, with an acidic solution containing 0.01% Triton (Triton X-100, Inc.), 0.5% n-butanol (Across, Denmark), and 1% nitric acid (ultrapure). The standard solutions(100 mL, National Institute of Metrology, China) consisted of 43 and 6 elements in mixed standard solutions and single standard solutions of Cu, Zn, Fe, Mg, and Ca and 1% nitric acid solution. The accuracy of ICP‒MS was assessed by analyzing certified reference materials included in every 25 samples (ClinChek human plasma controls for trace metals level 1 [No. 8883] and level 2 [No. 8884]; Recipe Chemicals). Quality control of plasma metal content was achieved by three replicate measurements. We used mixed plasma sample-spiked recoveries (200 randomly selected samples) to assess the precision and accuracy of this method. In addition, the standard recovery of plasma Cu levels was controlled at 80–120%. The limit of detection (LOD) for Cu was 0.004 μg/L, and values lower than the LOD were replaced with the LOD/√2. The intra-assay and inter-assay coefficients of variation in plasma Cu levels were below 10%.
Assessment of Covariates
To collect information on sociodemographic characteristics and lifestyle habits, trained interviewers administered a face-to-face questionnaire to collect information on the following: including sex (male or female), age (30–59 years or ≥ 60 years), ethnicity (Han, Yao, or other), occupation (farmer or other), years of education (≤ 6 years or > 6 years), smoking (yes or no), and alcohol consumption (yes or no). Body mass index (BMI) was calculated from weight and height data as BMI = weight (kg)/height2 (m2). Smoking was defined as currently smoking at least one cigarette per day. Alcohol consumption was defined as consuming at least 50 g or more of alcohol per month [28]. Hypertension was defined as a systolic blood pressure ≥ 140 mmHg or diastolic blood pressure ≥ 90 mmHg or indicated by the current use of antihypertensive medication [29]. Hyperlipidemia was defined as total cholesterol > 5.72 mmol/L triglycerides > 1.70 mmol/L, or current use of lipid-lowering medication [30]. Type 2 diabetes was defined as a fasting blood glucose level ≥ 7.0 mmol/L or self-reported medication for diabetes [15].
Statistical Analysis
Descriptive statistics were calculated for all demographic and clinical characteristics of the study population. Continuous variables are expressed as the mean (standard deviation, SD) or median (interquartile range, IQR). Categorical variables are expressed as numbers (percentages). The levels of metals were log10 transformed to reduce skew. Spearman correlation analysis was used to evaluate the correlations of dietary Cu intake and plasma Cu levels with liver function biomarkers, and multiple linear regression and multivariate restricted cubic splines (RCS) were used to investigate the dose‒response relationships between dietary and plasma Cu levels and liver function biomarkers. Covariates that might affect liver function or Cu levels, including sex, age, ethnicity, years of education, occupation, BMI, smoking, alcohol consumption, hypertension, dyslipidemia, type 2 diabetes, and total energy intake, were adjusted. The five knots of the RCS were located at 5%, 27.5%, 50%, 72.5%, and 95% of the range of Cu levels. Demographic characteristics, such as sex, influence the distribution of Cu in the body [31]. Therefore, we stratified the analysis by sex (male vs. female). In addition, alcohol consumption is a major risk factor for liver disease; thus, we performed sensitivity analyses on the participants who did not drink alcohol to exclude the potential effects of alcohol consumption on dietary and plasma Cu levels. All statistical analyses were performed using SPSS 22.0 and R 4.0.3, and a two-sided p value < 0.05 was considered to indicate a statistically significant difference.
Results
Participant Characteristics
Table 1 shows the clinical and sociodemographic characteristics of the 2376 participants (932 males and 1444 females), 50.51% of whom were aged 60 years or older. The majority of the participants were from the Yao population, accounting for 74.62% of the total population; the subjects with ≤ 6 years of education accounted for 65.11% of the total population. The clinical median values (IQR) of serum ALT, AST, IBIL, DBIL, and TBIL levels were 17.00 U/L (14.00–23.00), 21.00 U/L (19.00–25.00), 9.30 µmol/L (7.50–11.60), 3.50 µmol/L (2.80–4.60), and 12.80 µmol/L (10.50–16.10). There were more nonsmokers than nondrinkers among the participants.
Correlations Between Liver Function Biomarkers and Cu Levels
The correlations of dietary Cu intake and plasma Cu levels with liver function biomarkers were analyzed with Spearman’s correlation analysis (Fig. 1). The results showed that dietary Cu intake was positively correlated with serum DBIL levels (r = 0.051, p < 0.05) and negatively correlated with serum AST levels (r = − 0.048, p < 0.05). Plasma Cu levels were negatively correlated with serum IBIL, TBIL, and DBIL levels, with correlation coefficients (r) ranging from − 0.15 to − 0.0096 (all p < 0.001). In the sex-stratified analysis, plasma Cu levels were positively correlated with serum AST levels in males (r = 0.066, p < 0.05). In females, dietary Cu intake was negatively correlated with serum AST levels (r = − 0.11, p < 0.001), while plasma Cu levels were positively correlated with serum AST levels (r = 0.078, p < 0.01) and negatively correlated with bilirubin levels, with correlation coefficients (r) ranging from − 0.14 to − 0.083 (all p < 0.01).
Spearman correlation analysis and scatter plots of log10-transformed trace copper and ln-transformed liver function biomarkers in 2376 participants. A Both sexes; B men; and C women. Below the diagonal are binary scatter plots with fitted lines, and above the diagonal are the correlation coefficients and the significance level (indicated by asterisks; *** indicate p < 0.001, ** indicate p < 0.01, and * indicates p < 0.05). Abbreviations: ALT, alanine transaminase; AST, aspartate transaminase; IBIL, indirect bilirubin; DBIL, direct bilirubin; TBIL, total bilirubin; Cu, copper
Multiple Linear Regression Analysis of the Relationships Between Liver Function Biomarkers and Dietary Cu Levels
The results of the linear regression analysis of liver function biomarkers and dietary Cu levels are shown in Table 2. In model 1, which did not adjust for covariates, dietary Cu levels were not correlated with ALT, AST, or bilirubin levels in both sexes. After adjusting for covariates, dietary Cu levels were significantly associated with AST levels in both sexes (β = − 0.12, p < 0.05) but not with serum bilirubin levels. In the sex-stratified analysis, dietary Cu intake was associated with AST levels in females before adjusting for covariates (β = − 0.11, p < 0.001), and this association persisted after adjusting for covariates (β = − 0.12, p < 0.05).In men, we did not observe an association between dietary Cu intake and liver function biomarkers.
Multiple Linear Regression Analysis of the Relationships Between Liver Function Biomarkers and Plasma Cu Levels
The results of the linear regression analysis of liver function biomarkers and plasma Cu levels are shown in Table 3. Before adjusting for covariates, plasma Cu levels were significantly associated with ALT (β = − 0.20), IBIL (β = − 0.41), DBIL (β = − 0.53), and TBIL levels (β = − 0.44) in all the participants (all p < 0.05). After adjusting for covariates, the association of plasma Cu levels with bilirubin levels persisted in both sexes, while the association of plasma Cu levels with ALT levels disappeared. In the sex-stratified analysis, plasma Cu levels were associated with IBIL levels in men (β = − 0.27) and women (β = − 0.46) (all p < 0.05). In women, DBIL (β = − 0.32) and TBIL (β = − 0.42) levels were significantly associated with plasma Cu levels (all p < 0.05).
Dose‒Response Relationships Between Liver Function Biomarkers and Dietary Cu Intake
Dose‒response relationships between liver function biomarkers and dietary Cu intake were analyzed using an RCS (Fig. 2). In Fig. 2, we present only the plots in which the results were significant. We found a negative linear relationship between dietary Cu intake and serum AST levels in both sexes (all Poverall association < 0.05, all Pnonlinear association > 0.05) (Fig. 2A), with consistent results observed in women (all Poverall association < 0.05, all Pnonlinear association > 0.05) (Fig. 2F). In men, there was a nonlinear relationship between dietary Cu intake and serum AST, IBIL, DBIL, and TBIL levels (all Poverall association < 0.05, all Pnonlinear association < 0.05) (Fig. 2B–E). However, we did not find a relationship between dietary Cu intake and serum ALT levels.
Dose‒response relationship between log10-transformed dietary copper intake and liver function biomarkers. The solid and dotted lines represent the fitting curves and 95% confidence intervals, respectively. Adjusted for age (30–59 years old or ≥ 60 years old), sex (male or female), ethnicity (Han, Yao, or other), BMI (< 18.5, 18.5–23.9, or ≥ 24 kg/m2),years of education (≤ 6 years or > 6 years), occupation (farmer or other), type 2 diabetes (yes or no), hyperlipidemia (yes or no), hypertension (yes or no), smoking (yes or no), alcohol consumption (yes or no), and total energy intake (continuous variable). A Both sexes; B–E men; and C women. Abbreviations: TBIL, total bilirubin; DBIL, direct bilirubin; IBIL, indirect bilirubin; AST, aspartate aminotransferase
Sensitivity Analysis
We excluded 813 participants who consumed alcohol for the sensitivity analysis. After adjusting for covariates, the results showed a negative association between dietary Cu intake and serum AST levels in both sexes (β = − 0.09) and in women (β = − 0.11) (all p < 0.05). The negative linear relationships of plasma Cu levels and serum IBIL (β = − 0.32), DBIL (β = − 0.19), and TBIL (β = − 0.28) levels persisted in both sexes (all p < 0.05) and in women after adjustment for covariates, as shown in Table 4.
Discussion
The variability in dietary Cu intake and plasma Cu levels in the study population was minimal, and the plasma Cu concentrations found were close to those reported by Yang et al. [13] in rural residents of Southwest China (910.00 μg/L). Furthermore, the daily dietary Cu intake of both men and women was broadly in line with the intake recommended by the US Food and Nutrition Board (recommended daily intake [RDI]: 0.9 mg/day) [32], indicating adequate Cu intake in the population. We observed negative correlations between plasma Cu levels and serum bilirubin levels as well as between dietary Cu levels and serum AST levels after adjusting for covariates. The RCS analysis further indicated a negative linear relationship between dietary Cu levels and serum AST levels. These findings have important public health implications for the identification of risk factors for liver injury.
Cu is an antioxidant [33] and plays a role in antioxidant defense, lipid peroxidation, and mitochondrial function [34]. In this study, dietary Cu intake was negatively associated with AST levels, and plasma Cu levels were negatively associated with serum bilirubin levels. In a prior health cohort study of workers exposed to manganese, a 1-SD increase in plasma Cu levels produced a 3.2% reduction in TBIL levels [15]. Similarly, plasma Cu levels were negatively correlated with serum bilirubin levels in a cross-sectional study [35]. Animal experiments have shown that Cu decreases blood levels of AST and ALT and enhances the healing of damaged hepatic tissue [36]. These findings are consistent with our current findings. Several studies have indicated that mitochondrial autophagy and apoptosis play a vital role in maintaining normal physiological function in the liver. Normal hepatic function depends on mitophagy. In addition, hepatic apoptosis eliminates infected hepatocytes, serving a protective function [37].Appropriate doses of the Cu supplement tribasic copper chloride (TBCC) promoted hepatic antioxidant activity and induced mitochondrial autophagy, maintaining intracellular homeostasis in the liver [21]; therefore, moderate dietary Cu intake may promote hepatic antioxidant activity and induce mitochondrial autophagy, underlying the decrease in AST levels observed in the present study. On the other hand, oxidative stress reflects an imbalance between the production of reactive oxygen species (ROS) and the scavenging capacity of the antioxidant system [38]. At high concentrations, ROS cause oxidative damage to cellular macromolecules (DNA, lipids, proteins, etc.), and the accumulation of damaged macromolecules induces liver injury [39, 40].Cu is also a cofactor for many antioxidant enzymes, which are important for the maintenance of normal metabolism [41]. Cu deficiency can thus directly or indirectly lead to oxidative damage to DNA, lipids, and proteins, mainly due to an increase in ROS formation and a reduction in the activity of several Cu-dependent antioxidant enzymes, i.e., superoxide dismutase (SOD) and ceruloplasmin (Cp) [42,43,44]. Copper-zinc-dependent superoxide dismutase (SOD1) is an important defense against oxidative stress [45]. Cu can be delivered to the radical-detoxifying enzyme SOD1 by the Cu chaperone of superoxide dismutase (CCS) to reduce ROS levels and thus protect liver function [46]. In addition, Cu is carried in the plasma by Cp, which assists in the maintenance of hepatocyte homeostasis [47, 48]. Cp is a major plasma antioxidant and also acts as a ferroxidase; deficiencies of Cp lead to iron overload, which further increases ROS production by the mitochondrial respiratory chain, leading to hepatic oxidative damage [49, 50]. Therefore, in the present study population, we assumed that moderate Cu intake protected liver function through the antioxidant effects of Cp as well as its ferroxidase activity. However, a cross-sectional study by Yang et al. [13] found that blood Cu levels were positively correlated with ALT and AST levels. Therefore, the relationships between Cu levels and biomarkers of liver function still need further elucidation in prospective studies with large sample sizes.
Previous studies have shown that age, sex, BMI, and lifestyle factors are associated with biomarkers of liver function and heavy metal concentrations [51,52,53,54]. After adjusting for confounding factors, our study found negative associations of dietary Cu intake and plasma Cu levels in women with serum AST and bilirubin levels. Previous studies have shown that serum Cu levels are influenced by estrogen, which increases intracellular Cu concentrations and affects systemic and cellular responses to Cu [54, 55]. Serum Cu levels were higher in females than in males in our study; therefore, the influence of estrogen on Cu levels may provide a plausible explanation for this sex difference. More experimental and epidemiological evidence is needed to verify this relationship.
The main strength of the present study was that it demonstrated the effect of Cu exposure on serum levels of liver function biomarkers in terms of both dietary Cu intake and blood Cu levels. Second, we used RCS to investigate the dose‒response relationship between dietary Cu levels and serum levels of liver function biomarkers. Our current study also has some limitations. First, this cross-sectional study was unable to determine a causal relationship of Cu intake and plasma Cu levels with liver function biomarkers. Second, the biomarkers in our research included only serum bilirubin, AST, and ALT levels. Future studies should include more biomarkers of liver function, such as alkaline phosphatase, serum total protein, serum albumin, and serum globulin. Third, the effects of other nonpersistent and persistent environmental pollutants on liver injury were not considered. Further studies are needed to determine the relationships among Cu intake, plasma Cu levels, and markers of liver function, as well as the mechanisms underlying this relationship.
Conclusions
Plasma Cu levels were negatively associated with serum bilirubin levels, and dietary Cu levels were negatively associated with serum AST levels; these associations were sex specific. Further studies are needed to validate these associations and elucidate the underlying mechanisms.
References
Wang C, Ma C, Gong LH, Guo YQ, Fu K, Zhang YF, Zhou HL, Li YX (2021) Macrophage polarization and its role in liver disease. Frontiers in Immunology 12. ARTN 80303710.3389/fimmu.2021.803037
Wang FS, Fan JG, Zhang Z, Gao B, Wang HY (2014) The global burden of liver disease: the major impact of China. Hepatology 60(6):2099–2108. https://doi.org/10.1002/hep.27406
Zhao MD, Ge XY, Xu J, Li A, Mei YY, Yin GH, Wu JT, Liu XL, Wei LP, Xu Q (2022) Association between urine metals and liver function biomarkers in Northeast China: a cross-sectional study. Ecotox Environ Safe 231. ARTN 113163 https://doi.org/10.1016/j.ecoenv.2022.113163
Jarup L (2003) Hazards of heavy metal contamination. Br Med Bull 68:167–182. https://doi.org/10.1093/bmb/ldg032
Xu S, Pi H, Chen Y, Zhang N, Guo P, Lu Y, He M, Xie J, Zhong M, Zhang Y, Yu Z, Zhou Z (2013) Cadmium-induced Drp1-dependent mitochondrial fragmentation by disturbing calcium homeostasis in its hepatotoxicity. Cell Death Dis 4:e540. https://doi.org/10.1038/cddis.2013.7
Scheig R (1996) Evaluation of tests used to screen patients with liver disorders. Prim Care 23(3):551–560. https://doi.org/10.1016/s0095-4543(05)70347-x
Schmidt E, Schmidt FW (1993) Enzyme diagnosis of liver diseases. Clin Biochem 26(4):241–251. https://doi.org/10.1016/0009-9120(93)90123-n
Hamoud AR, Weaver L, Stec DE, Hinds TD Jr (2018) Bilirubin in the liver-gut signaling axis. Trends Endocrinol Metab 29(3):140–150. https://doi.org/10.1016/j.tem.2018.01.002
Uauy R, Olivares M, Gonzalez M (1998) Essentiality of copper in humans. Am J Clin Nutr 67(5 Suppl):952S-S959. https://doi.org/10.1093/ajcn/67.5.952S
Morrell A, Tallino S, Yu L, Burkhead JL (2017) The role of insufficient copper in lipid synthesis and fatty-liver disease. IUBMB Life 69(4):263–270. https://doi.org/10.1002/iub.1613
Suzuki Y, Ogra Y, Machida N, Watanabe I (2019) Changes in copper, zinc, and cadmium distributions in the liver of Formosan squirrels with characteristic high copper accumulation. Metallomics 11(10):1753–1758. https://doi.org/10.1039/c9mt00204a
Hellman NE, Gitlin JD (2002) Ceruloplasmin metabolism and function. Annu Rev Nutr 22:439–458. https://doi.org/10.1146/annurev.nutr.22.012502.114457
Yang Z, Li X, Tian L, Song Y, Zhang Y, Chen J, Zhang L (2021) Heavy metals exposure is associated with early liver dysfunction among rural residents aged 40–75 years in southwest China. J Appl Toxicol. https://doi.org/10.1002/jat.4276
Wang X, Bin W, Zhou M, Xiao L, Xu T, Yang S, Nie X, Xie L, Yu L, Mu G, Ma J, Chen W (2021) Systemic inflammation mediates the association of heavy metal exposures with liver injury: a study in general Chinese urban adults. J Hazard Mater 419:126497. https://doi.org/10.1016/j.jhazmat.2021.126497
Ge X, Liu Z, Hou Q, Huang L, Zhou Y, Li D, Huang S, Luo X, Lv Y, Li L, Cheng H, Chen X, Zan G, Tan Y, Liu C, Zou Y, Yang X (2020) Plasma metals and serum bilirubin levels in workers from manganese-exposed workers healthy cohort (MEWHC). Environ Pollut 258:113683. https://doi.org/10.1016/j.envpol.2019.113683
Lutsenko S, Barnes NL, Bartee MY, Dmitriev OY (2007) Function and regulation of human copper-transporting ATPases. Physiol Rev 87(3):1011–1046. https://doi.org/10.1152/physrev.00004.2006
Aigner E, Theurl I, Haufe H, Seifert M, Hohla F, Scharinger L, Stickel F, Mourlane F, Weiss G, Datz C (2008) Copper availability contributes to iron perturbations in human nonalcoholic fatty liver disease. Gastroenterology 135(2):680–688. https://doi.org/10.1053/j.gastro.2008.04.007
Song M, Vos MB, McClain CJ (2018) Copper-fructose interactions: anovel mechanism in the pathogenesis of NAFLD. Nutrients 10(11). https://doi.org/10.3390/nu10111815
Kumar V, Kalita J, Misra UK, Bora HK (2015) A study of dose response and organ susceptibility of copper toxicity in a rat model. J Trace Elem Med Biol 29:269–274. https://doi.org/10.1016/j.jtemb.2014.06.004
Yang F, Pei R, Zhang Z, Liao J, Yu W, Qiao N, Han Q, Li Y, Hu L, Guo J, Pan J, Tang Z (2019) Copper induces oxidative stress and apoptosis through mitochondria-mediated pathway in chicken hepatocytes. Toxicol In Vitro 54:310–316. https://doi.org/10.1016/j.tiv.2018.10.017
Yu W, Liao J, Yang F, Zhang H, Chang X, Yang Y, Bilal RM, Wei G, Liang W, Guo J, Tang Z (2021) Chronic tribasic copper chloride exposure induces rat liver damage by disrupting the mitophagy and apoptosis pathways. Ecotoxicol Environ Saf 212:111968. https://doi.org/10.1016/j.ecoenv.2021.111968
Aigner E, Strasser M, Haufe H, Sonnweber T, Hohla F, Stadlmayr A, Solioz M, Tilg H, Patsch W, Weiss G, Stickel F, Datz C (2010) A role for low hepatic copper concentrations in nonalcoholic fatty liver disease. Am J Gastroenterol 105(9):1978–1985. https://doi.org/10.1038/ajg.2010.170
Li S, Guo H, Liu Y, Wu F, Zhang H, Zhang Z, Xie Z, Sheng Z, Liao E (2015) Relationships of serum lipid profiles and bone mineral density in postmenopausal Chinese women. Clin Endocrinol (Oxf) 82(1):53–58. https://doi.org/10.1111/cen.12616
Wang CJ, Yang TF, Wang GS, Zhao YY, Yang LJ, Bi BN (2018) Association between dietary patterns and depressive symptoms among middle-aged adults in China in 2016–2017. Psychiat Res 260:123–129. https://doi.org/10.1016/j.psychres.2017.11.052
Barak F, Falahi E, Keshteli AH, Yazdannik A, Saneei P, Esmaillzadeh A (2015) Red meat intake, insulin resistance, and markers of endothelial function among Iranian women. Mol Nutr Food Res 59(2):315–322. https://doi.org/10.1002/mnfr.201400333
Esfahani FH, Asghari G, Mirmiran P, Azizi F (2010) Reproducibility and relative validity of food group intake in a food frequency questionnaire developed for the Tehran Lipid and Glucose Study. J Epidemiol 20(2):150–158. https://doi.org/10.2188/jea.je20090083
YX Y (2019) Ingredient list of Chinese food. ActaNutrimenta Sinica 41(05):426
Cai JS, Li Y, Liu SZ, Liu QM, Xu M, Zhang JL, Wei YF, Mo XT, Lin YX, Tang X, Mai TY, Mo CB, Luo TY, Huang SX, Lu HX, Zhang ZY, Qin J (2022) Associations between multiple heavy metals exposure and glycated hemoglobin in a Chinese population. Chemosphere 287. ARTN 132159 https://doi.org/10.1016/j.chemosphere.2021.132159
Mo XT, Cai JS, Lin YX, Liu QM, Xu M, Zhang JL, Liu SZ, Wei CM, Wei YF, Huang SX, Mai TY, Tan DC, Lu HX, Luo TY, Gou RY, Zhang ZY, Qin J (2021) Correlation between urinary contents of some metals and fasting plasma glucose levels: a cross-sectional study in China. Ecotox Environ Safe 228. ARTN 112976 https://doi.org/10.1016/j.ecoenv.2021.112976
Li ZY, Xu YL, Huang ZJ, Wei Y, Hou J, Long TF, Wang F, Hu H, Duan YY, Guo H, Zhang XM, Chen X, Yuan H, Wu TC, Shen MX, He MA (2019) Association between exposure to arsenic, nickel, cadmium, selenium, and zinc and fasting blood glucose levels. Environmental Pollution 255. ARTN 113325 https://doi.org/10.1016/j.envpol.2019.113325
Bjorklund G, Dadar M, Pivina L, Dosa MD, Semenova Y, Aaseth J (2020) The role of zinc and copper in insulin resistance and diabetes mellitus. Curr Med Chem 27(39):6643–6657. https://doi.org/10.2174/0929867326666190902122155
Trumbo P, Yates AA, Schlicker S, Poos M (2001) Dietary reference intakes: vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium, and zinc. J Am Diet Assoc 101(3):294–301. https://doi.org/10.1016/S0002-8223(01)00078-5
Ferns GA, Lamb DJ, Taylor A (1997) The possible role of copper ions in atherogenesis: the Blue Janus. Atherosclerosis 133(2):139–152. https://doi.org/10.1016/s0021-9150(97)00130-5
al-Othman AA, Rosenstein F, Lei KY, (1993) Copper deficiency increases in vivo hepatic synthesis of fatty acids, triacylglycerols, and phospholipids in rats. Proc Soc Exp Biol Med 204(1):97–103. https://doi.org/10.3181/00379727-204-43640
Peng YF, Wei YS (2017) Associations between serum bilirubin levels and essential trace elements status in an adult population. Oncotarget 8(46):81315–20. https://doi.org/10.18632/oncotarget.18351
Ayten R, Aygen E, Cerrahoglu YZ, Camci C, Ilhan YS, Girgin M, Ilhan N, Ozercan IH (2015) Effects of copper, zinc, and vitamin complex (Cernevit(R)) on hepatic healing in rats experimentally subjected to blunt hepatic trauma. Indian J Surg 77(Suppl 3):1045–1049. https://doi.org/10.1007/s12262-014-1128-0
Guicciardi ME, Malhi H, Mott JL, Gores GJ (2013) Apoptosis and necrosis in the liver. Compr Physiol 3(2):977–1010. https://doi.org/10.1002/cphy.c120020
Takaki A, Kawai D, Yamamoto K (2013) Multiple hits, including oxidative stress, as pathogenesis and treatment target in non-alcoholic steatohepatitis (NASH). Int J Mol Sci 14(10):20704–20728. https://doi.org/10.3390/ijms141020704
Rolo AP, Teodoro JS, Palmeira CM (2012) Role of oxidative stress in the pathogenesis of nonalcoholic steatohepatitis. Free Radic Biol Med 52(1):59–69. https://doi.org/10.1016/j.freeradbiomed.2011.10.003
Serviddio G, Bellanti F, Vendemiale G (2013) Free radical biology for medicine: learning from nonalcoholic fatty liver disease. Free Radic Biol Med 65:952–968. https://doi.org/10.1016/j.freeradbiomed.2013.08.174
Stern BR, Solioz M, Krewski D, Aggett P, Aw TC, Baker S, Crump K, Dourson M, Haber L, Hertzberg R, Keen C, Meek B, Rudenko L, Schoeny R, Slob W, Starr T (2007) Copper and human health: biochemistry, genetics, and strategies for modeling dose-response relationships. J Toxicol Environ Health B Crit Rev 10(3):157–222. https://doi.org/10.1080/10937400600755911
Klevay LM (2000) Cardiovascular disease from copper deficiency–a history. J Nutr 130(2S Suppl):489S-S492. https://doi.org/10.1093/jn/130.2.489S
Bertinato J, Iskandar M, L’Abbe MR (2003) Copper deficiency induces the upregulation of the copper chaperone for Cu/Zn superoxide dismutase in weanling male rats. J Nutr 133(1):28–31. https://doi.org/10.1093/jn/133.1.28
Majewski M, Ognik K, Juskiewicz J (2019) Copper nanoparticles modify the blood plasma antioxidant status and modulate the vascular mechanisms with nitric oxide and prostanoids involved in Wistar rats. Pharmacol Rep 71(3):509–516. https://doi.org/10.1016/j.pharep.2019.02.007
Song M, Zhou Z, Chen T, Zhang J, McClain CJ (2011) Copper deficiency exacerbates bile duct ligation-induced liver injury and fibrosis in rats. J Pharmacol Exp Ther 339(1):298–306. https://doi.org/10.1124/jpet.111.184325
Dongiovanni P, Lanti C, Riso P, Valenti L (2016) Nutritional therapy for nonalcoholic fatty liver disease. J Nutr Biochem 29:1–11. https://doi.org/10.1016/j.jnutbio.2015.08.024
Yamada T, Agui T, Suzuki Y, Sato M, Matsumoto K (1993) Inhibition of the copper incorporation into ceruloplasmin leads to the deficiency in serum ceruloplasmin activity in a Long-Evans cinnamon mutant rat. J Biol Chem 268(12):8965–8971
Fleming RE, Gitlin JD (1990) Primary structure of rat ceruloplasmin and analysis of tissue-specific gene expression during development. J Biol Chem 265(13):7701–7707
Medici V (2013) The evolving scenario of copper and fatty liver. Metab Syndr Relat Disord 11(1):4–6. https://doi.org/10.1089/met.2013.1501
Doguer C, Ha JH, Collins JF (2018) Intersection of iron and cpper metabolism in the mammalian intestine and liver. Compr Physiol 8(4):1433–1461. https://doi.org/10.1002/cphy.c170045
Chung SM, Moon JS, Yoon JS, Won KC, Lee HW (2020) The sex-specific effects of blood lead, mercury, and cadmium levels on hepatic steatosis and fibrosis: Korean nationwide cross-sectional study. J Trace Elem Med Biol 62:126601. https://doi.org/10.1016/j.jtemb.2020.126601
Lee MR, Lim YH, Lee BE, Hong YC (2017) Blood mercury concentrations are associated with decline in liver function in an elderly population: a panel study. Environ Health 16(1):17. https://doi.org/10.1186/s12940-017-0228-2
Li Y, Chen C, Lu L, Guo W, VanWagner LB, Shikany JM, Zhang S, Kahe K (2022) Cadmium exposure in young adulthood is associated with risk of nonalcoholic fatty liver disease in midlife. Dig Dis Sci 67(2):689–696. https://doi.org/10.1007/s10620-021-06869-8
Arredondo M, Nunez H, Lopez G, Pizarro F, Ayala M, Araya M (2010) Influence of estrogens on copper indicators: in vivo and in vitro studies. Biol Trace Elem Res 134(3):252–264. https://doi.org/10.1007/s12011-009-8475-x
Ayala M, Pizarro F, Mendez MA, Arredondo M, Araya M (2008) Copper and liver function indicators vary depending on the female hormonal cycle and serum hormone binding globulin (SHBG) concentration in healthy women. Biol Trace Elem Res 121(1):9–15. https://doi.org/10.1007/s12011-007-8029-z
Acknowledgements
The authors express their gratitude for all the participants and cooperating units who helped to complete this study.
Funding
The study was supported by the National Natural Science Foundation of China (grant nos. 81960583, 81760577, and 81560523), Major Science and Technology Projects in Guangxi (GKAA22399), Guangxi Science and Technology Development Project (grant nos. AD 17129003 and 18050005), Guangxi Natural Science Foundation for Innovation Research Team (2019GXNSFGA245002), and Guangxi Scholarship Fund of Guangxi Education Department of China.
Author information
Authors and Affiliations
Contributions
YW: writing—original draft preparation and writing—review and editing; JC: investigation, data supervision, and project administration; MX and XM: investigation, and data curation; QL, JZ, SL, and YL: methodology and formal analysis; JQ: software and validation; SH: data curation; JQ: conceptualization and resources; and ZZ: writing—original draft preparation, writing—review and editing, investigation, data supervision, and project administration. All the authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Ethics Approval
Approval was obtained from the Ethics Committee of Guilin Medical University (No: 20180702–3). The procedures used in this study adhered to the tenets of the Declaration of Helsinki.
Consent to Participate
Written informed consent was obtained from all the participants before the study.
Conflict of Interest
The authors declare no competing interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wei, Y., Cai, J., Mo, X. et al. Associations of Dietary and Plasma Copper Levels with Liver Function in a Chinese Population. Biol Trace Elem Res 201, 3268–3278 (2023). https://doi.org/10.1007/s12011-022-03445-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-022-03445-7