Abstract
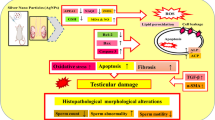
This study was performed to investigate whether the toxicity of nanoparticles (Ag NPs or TiO2 NPs) affected mitochondrial dynamics (mitochondrial fusion and fission mechanisms) in testicular cells of mice. Animals were assigned into three groups (ten mice per group): control group (distilled water), TiO2 NP group (5 mg/kg per dose), and Ag NP group (5 mg/kg per dose). NPs were administered intravenously (via tail vein) to mice with 3-day intervals. To determine the possible toxic effect of NPs on mitochondrial dynamics, the expression levels of mitochondrial fission (Drp1)- and fusion (Mfn1, Mfn2, OPA1)-related genes were analyzed. The results showed that both Ag NPs and TiO2 NPs entered the testis via the blood–testis barier and accumulated in mouse testis tissue. Experiments showed that administration of Ag NPs neither alters testicular weight and testicular index nor causes significant toxic effect on sperm parameters. RT-PCR analysis demonstrated that Ag NP treatment did not disrupt mitochondrial dynamics in testicular cells. Conversely, administration of TiO2 NPs (anatase, < 25 nm) decreased the sperm motility and the percentages of sperms with swollen tail. Furthermore, RT-PCR and western blot analyses showed that TiO2 NPs disrupted mitochondrial dynamics by causing excess mitochondrial fission (excess expression of Drp1 gene and DRP1 protein). This is the first report on the toxicity of nanoparticles on mitochondrial dynamics (fusion and fission mechanisms) in testicular cells.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mitochondria are dynamic organelles that constantly fuse with each other and then split apart. Fusion serves to mix and unify the mitochondrial compartments whereas fission generates morphologically and functionally distinct organelles [1]. Mitofusins (Mfn1 and Mfn2) and OPA1 are the most important proteins involved in the regulation of mitochondrial fusion. Mfn1 and Mfn2 are located on the outer mitochondrial membrane and essential for the fusion of the outer membranes. OPA1 is localized within the mitochondrial intermembrane space in soluble forms or attached to the inner mitochondrial membrane. It mediates inner mitochondrial membrane fusion [2, 3]. Drp1, also known as dynamin-related protein 1, mediates mitochondrial fission in mammalian cells. Inactive Drp1 is normally found in the cytosol of cells, whereas active Drp1 translocates from the cytosol to the mitochondrial outer membrane to mediates mitochondrial fission. Drp1 assembles into rings and spirals at division sites around the outer mitochondrial membrane in order to drive the fission process [2, 4].
In mammalian cells, mitochondrial fusion and fission proteins control mitochondrial morphology. The frequencies of fusion and fission events are balanced to maintain the overall morphology of the mitochondrial population. When this balance is experimentally perturbed, dramatic transitions in mitochondrial shape can occur [4]. Beyond the control of mitochondrial shape, fusion is also an important mechanism for the bioenergetic function of mitochondria. In cells lacking fusion, mitochondrial function is greatly diminished and oxygen consumption is compromised. Therefore, cells lacking mitochondrial fusion grow much more slowly than their wild-type counterparts. Mitochondrial fusion can also protect cells from the detrimental effects of mtDNA mutations by allowing functional complementation of mtDNA gene products [5]. Similar to fusion, mitochondrial fission also plays a key role in cell life and death. Mitochondrial fission helps the removal of damaged mitochondria via mitophagy and facilitates apoptosis in conditions of cellular stress [1]. Furthermore, mitochondrial fission has critical importance in cellular processes such as formation of synapses and dendritic spines in neurons as well as embryonic development [6]. However, it has been informed that overproduction of Drp1, namely excess fission, is linked to impaired regulation of mitochondrial dynamics (mitochondrial fusion and fission proteins), which control the balance in cells [7]. Since mitochondrial dynamics are involved in different biological processes, such as mitochondrial biogenesis, cell metabolism, calcium buffering, and cell survival and death, the disruptions in mitochondrial dynamics can cause neurodegenerative diseases, diabetes, aging, and cancer [8].
Inorganic nanoparticles (NPs) are widely used in biological and biomedical studies, due to their specific properties such as small size, shape, high surface area, and special structure [9,10,11]. For example, Ag NPs can be used as antimicrobial and anti-inflammatory materials [12, 13]. Ag NPs have also the capacity to be used as a biosensor for detecting various abnormalities and diseases in the human body [13]. TiO2 NPs are used in crop production, dietary supplements, food colorants, pharmaceuticals, medicine, toothpastes, sunscreens, cosmetics, and wastewater treatment [9, 14, 15]. Although Ag NPs or TiO2 NPs are used as promising candidates for both industrial and biomedical applications, they are reported to have potential toxic effects on living organisms. For example, Ag NPs and/or TiO2 NPs can cause some adverse effects on various organs such as the lung, liver, brain, and kidney [13, 16]. TiO2 NPs and Ag NPs are also reported to cause harmful biological responses in the male and female reproductive system [10, 17,18,19].
The recent studies have revealed that the toxicity of some chemicals or nanoparticles is linked to the disruptions of mitochondrial dynamics (mitochondrial fusion and fission proteins) in cells such as neurons and hepatocytes [20,21,22]. However, the relation between the toxicity of nanoparticles and the disruption of mitochondrial dynamics (mitochondrial fission and fusion mechanisms) in reproductive system cells has not been investigated in any study.
Therefore, this study was performed to reveal whether intravenously administered Ag NPs or TiO2 NPs caused testicular toxicity in mice and whether the possible toxicity disrupted mitochondrial dynamics controlled by fusion and fission proteins.
Materials and Methods
Materials
Silver nanoparticles (Ag NPs) (99.9% purity, 20 nm, w/ ~ 0.3% PVP) were purchased from Nanostructured & Amorphous Materials, Inc. Titanium dioxide nanoparticles (TiO2 NPs) (anatase nanopowder, < 25 nm) were purchased from Sigma. TiO2 NPs and Ag NPs were separately suspended in sterile water and then sonicated for 25–30 min before use.
Animals and Treatments
Six-week-old male mice (BALB/c, 25–30 g) used in this study were obtained from the Laboratory Animal Center of Ataturk University, Erzurum, Turkey. The mice were acclimatized for 1 week before beginning the study in plastic cages at room temperature of 20–25 °C, relative humidity of 55 ± 5%, and 12 h light/dark cycle. After acclimatization, animals were weight-ranked and assigned randomly into three groups (ten mice per group) as follows: control group (distilled water), TiO2 NP-treated group (5 mg/kg per dose), and Ag NP-treated group (5 mg/kg per dose). Five doses containing 1 mL of NP suspension or distilled water was administered intravenously (via tail vein) to the mice with 3-day intervals. At the end of day 15, the mice were weighed and sacrificed by cervical dislocation. The testes were quickly removed and weighed.
Determination of NPs’ Presence in Testes After Treatment
The presence of TiO2 NPs or Ag NPs in testicular tissues of mice after intravenous application was analyzed using ICP (inductively coupled plasma spectrophotometer) (Perkin-Elmer, Optima 2100 DV, ICP/OES, Shelton, CT 06,484–4794, USA).
Epididymal Sperm Count and Sperm Morphology
The caudal epididymides were minced finely with scissors in 0.2 mL of 0.9% saline water in a Petri dish and then incubated at 37 °C for 30 min to allow the spermatozoa to swim out from the epididymal tubules. After diffusion, the cauda epididymides were removed from the medium. For the analysis of sperm motility, 10 µL of the sperm suspension was placed on a slide, covered by a coverslip, and incubated at 35 °C on a heating table. Afterwards, sperm motility was evaluated under × 400 magnification using a phase contrast microscope [23, 24].
Sperm viability was evaluated using eosin-nigrosin staining. For this purpose, 10 μL of sperm suspension was placed on a slide preheated at 35 °C on a heating table and then mixed with eosin (10 μL) and nigrosin (10 μL) solutions. Following this, a smear on the slide was produced using a glass rod and the slide was air-dried at room temperature.
Smears were observed using a light microscope with × 1000 magnification, and the percentage of live sperms (colorless or light pink) and dead sperms (red or dark pink color) was determined. In total, 200 sperms were counted in each slide, and viability percentages of sperm were calculated. Unstained sperms (with intact cell membrane) on smear were considered as living sperm, whereas purple-red stained sperms (with damaged membranes) were accepted as non-viable sperms [25]. The percentage of viable sperm and non-viable sperm was evaluated by counting a minimum of 200 spermatozoa.
Sperm morphology was analyzed according to the Hypo-osmotic Swelling (HOS) test. For this purpose, 20 µL of sperm solution was added to 80 μL of HOS test solution. The mixture was incubated at 35 °C for 30 min. Then, 10 µL of incubated sperm solution was placed on a glass slide, covered with a coverslip, and evaluated under phase contrast microscope. At least 100 spermatozoa were evaluated per slide, and the percentages of sperms with swollen tails were calculated.
RNA Extraction and cDNA Synthesis
Total RNA was isolated from mouse testis using RNeasy Lipid Tissue Mini Kit (Cat No: 74804, Qiagen) according to the manufacturer’s instructions. The concentration and the purity of the isolated RNA were measured by a spectrophotometer (Thermo Scientific, Multiskan GO, USA). cDNA was synthesized according to the manufacturer’s instruction by utilizing ThermoScript™ RT-PCR System for First-Strand cDNA Synthesis Kit (Cat No:18080051, Invitrogen). All cDNAs were stored at − 20 °C for further studies.
Gene Expression Analysis by RT-PCR
The changes in expression levels of Mfn1, Mfn2, Opa1, and Drp1 genes were measured using quantitative real-time polymerase chain reaction (q-PCR). The relative abundance of mRNA species was assessed using the Taqman Probe method on an Qiagen (Applied Biosystems, Foster City, CA, USA). Gene-specific primers and probes were designed and purchased from Medsantek. β-Actin was used as a reference gene (housekeeping gene). The PCR reaction mixture containing 5μL of template cDNA, 1 μL of primers and probe mix and 10 μL of FastStart TaqMan Probe Master Mix, and 4 μL ddH2O was prepared in 20 μL final reaction volume for q-PCR. The PCR conditions were as follows: 50 °C for 2 min, 95 °C for 10 min, 45 cycles of 95 °C for 10 s, and at 60 °C for 1 min. β-Actin expression was used for normalization and relative quantification. We analyzed the relative gene expression data using the ΔCT method.
Protein Analysis by Western Blot
Testicular tissues samples were homogenized in RIPA buffer (150 mM of NaCl, 1% Triton X‐100, 0.5% sodium deoxycholate, 0.1% sodium dodecyl sulfate [SDS], 50 mM of Tris, pH 8.0, protease inhibitor cocktail [100 × , P8340; Sigma‐Aldrich]). The concentration of protein lysates was determined by using Bradford’s method [26]. Protein extract was separated by a 10% sodium dodecyl sulfate polyacrylamide gel and then transferred to polyvinylidene fluoride membrane for western blot analysis. The membranes were blocked with 5% nonfat dry milk powder in 1 × Tris-buffered saline and 0.05% Tween 20 (TBST) for 1 h at room temperature. The membranes were incubated at 4 °C overnight with DRP1 (ab184247; Abcam) antibodies at 1/1000 dilution and washed 3 times with TBST. After, the membranes were treated with horseradish peroxidase–conjugated goat antirabbit at room temperature. β‐Actin (sc‐47,778; Santa Cruz Biotechnology) was used as a housekeeping protein. After washing, the signals were visualized using an enhanced chemiluminescent substrate detection system (SuperSignal West Femto/Pico Maximum Sensitivity Substrate; Thermo Fisher Scientific) [27]. Densitometric results were analyzed with Image J software.
Statistical Analysis
Each group contains ten animals, and all measurements were triplicated for each animal. Results were compared statistically by utilizing GraphPad Prism Software version 7.0. Groups were compared by using the unpaired Student t-test. All data were presented as mean ± standard error of mean (SEM). p value below 0.05 was considered statistically significant and indicated by the * symbol. Symbol expressions are as follows: *P < 0.05 (significant); **P < 0.01 (very significant); and ***P < 0.001(very very significant).
Results
Determination of Ag NPs and TiO2 NPs in Testicular Tissue After Treatment
In this study, a total of 25 mg/kg TiO2 NPs or Ag NPs was administered in 5 doses (5 mg TiO2 NPs/kg tissue or 5 mg Ag NPs/kg tissue per dose). Elemental analyses demonstrated that 5.761 mg/kg tissue of TiO2 NPs and 2.953 mg/kg tissue of Ag NPs were deposited in the testes of mice after injection. Whereas, no TiO2 NPs or Ag NPs were detected in the testes of mice injected with saline water (control group) (Table 1).
Effect of Ag NPs and TiO2 NPs on Sperm Parameters
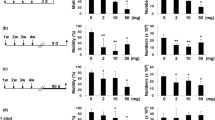
The results demonstrated that TiO2 NPs and Ag NPs did not reduce the testis weight and testis index (testis weight/body weight) values compared to the control group (Table 2) (P > 0.05). On the contrary, there was a slight increase in the testis weight of both TiO2 NP and Ag NP group compared to the control group. This was probably due to the accumulation of TiO2 NPs and Ag NPs in testes. Similarly, no significant difference for both sperm viability and sperm velocity was observed among Ag NPs, TiO2 NPs, and control groups. For example, percentages of non-viable sperms were 35.7, 37.9, and 39.2 in the control, TiO2 NPs, and Ag NPs groups, respectively. In the control, TiO2 NP group, and Ag NP groups, the percentages of sperm motility were 43.6, 35.7, and 39.8, and the percentages of sperms with swollen tails were 57.0, 44.1, and 56.8, respectively.
These results indicated that Ag NPs treatment slightly reduced the sperm motility and the percentages of sperms with swollen tails compared to the control group. Conversely, TiO2 NPs caused a significant reduction in sperm motility and the percentages of sperms with a swollen tail compared to the control.
Effect of Ag NPs and TiO2 NPs on Expression Levels of Mitochondrial Fission- and Fusion-Related Genes
RT-PCR analyses showed that intravenously applied AgNPs did not cause a significant change in the expression levels of Mfn1 and Drp1 compared to the control group. The expression level of Mfn 2 showed a significant increase in Ag NP group in comparison to the control group. Conversely, a significant decrease in the expression level of OPA was observed in Ag NP group compared to the control group (Fig. 1). The experiments indicated that there were no significant changes in expression levels of fusion genes (Mfn1, Mfn2, and Opa1) in TiO2 NP group when compared to the control. On the contrary, there was a significant increase in the expression level of the fission gene Drp 1 in TiO2 NPs group compared to the control group (Fig. 2).
Effect of Ag NPs and TiO2 NPs on Expression Level of Mitochondrial Fission Protein-DRP1
The western blot analysis showed that there was no increase in the concentration of DRP1 in the Ag NP group when compared to the control group. Conversely, TiO2 NPs were found to significantly augment the concentration of Drp1 in comparison with the control group (Fig. 3).
Discussion
The present experiments demonstrated that when TiO2 NPs and Ag NPs were administered intravenously to mice via tail vein, they could pass through the blood–testis barrier and accumulate in mouse testes. This result was in good agreement with those reported in the previous studies [10, 28, 29]. Since TiO2 NP and Ag NP were detected in testes, the subsequent stages of the present study were performed to investigate whether these NPs caused a toxic effect on testicular cells and sperm parameters, as well as mitochondrial dynamics (mitochondrial fusion and fission proteins).
There is limited data on the toxicities of Ag NPs based on in vivo experimental models, and the obtained results are controversial. That is, some researchers state that Ag NPs cause a toxic effect on human or animal models in vivo, while some researchers state that these nanoparticles do not cause any toxicity [29,30,31,32,33,34,35]. The present experiments demonstrated that Ag NPs did not cause a significant toxic effect on the reproductive system cells of male mice in vivo. This is because it was seen that intravenously applied AgNPs did not cause a significant change in sperm viability, sperm motility, sperm velocity, and the percentages of sperms with swollen tails as well as testis weight and testis index.
In the literature, it has been documented that TiO2 NPs can lead to some adverse effects in organs such as brain, lung, liver, and kidney in vivo models [15, 36]. TiO2 NPs are also reported to cause some toxic effects on reproductive system cells in vivo and/or in vitro models [9, 17, 18, 37, 38]. For example, Guo et al. [17] reported that when applied to male mice by intraperitoneal injection in vivo, TiO2 NPs decreased sperm numbers and motility, and caused germ cell apoptosis in testicular cells. Miura et al. [39] demonstrated that TiO2 NP injection significantly reduced sperm motility in mice. The similar toxic effect of intravenously applied TiO2 NPs on reproductive system cells of mice was also observed in this study. Namely, it was seen that although TiO2 NP treatment did not cause a significant change in sperm viability and sperm velocity as well as testis weight and testis index when compared to the control, it significantly reduced sperm motility and the percentages of sperms with swollen tails. It is known that approximately 40–50% of all infertility cases is due to “male factor” infertility, and defects in sperm parameters are important causes of male infertility [40]. Therefore, it is important to reveal that TiO2 NPs have a negative effect on sperm parameters.
Mitochondria can exist within the cell in many shapes and sizes. These different shapes are a result of the mitochondria’s ability to divide, fuse, and move within the cell. These processes are collectively referred to as mitochondrial dynamics. In the cells, mitochondrial dynamics are controlled by fusion and fission mechanisms [40]. The fusion process is regulated by the mitofusins Mfn1 and 2 proteins on the outer membrane, and OPA1 protein in the inner membrane, whereas, the major executor of the fission mechanism is the dynamin-related protein 1 (Drp1). Drp1 is mainly cytosolic, but translocates to the mitochondrial surface in order to mediate fisse ion of the organelle [41, 42]. As well as the control of mitochondrial morphology, fusion and fission mechanisms have also different functions in cells. For instance, mitochondrial fission is essential for cell growth and division, sustaining cell polarity, and aiding in eliminating damaged mitochondria [42]. However, it has been informed that excess fission causes the disruption of mitochondrial dynamics, which control the balance in cells [7]. Especially, overexpression of Drp1 is accepted as the most important indicator of toxicity and mitochondrial dysfunction in cells [43]. For example, Li et al. [21] demonstrated that rifampicin-induced hepatotoxicity caused the overexpression of Drp1 and mitochondrial dysfunction. Grohm et al. [7] reported that overexpression of Drp1 is associated with neuronal death in neurodegenerative diseases. Chen et al. [22] demonstrated that ropivacaine exclusively induced mitochondrial fission protein Drp1 and caused mitochondrial dysfunction in human neuronal SH-5Y5Y cells. In short, it has been shown in the literature that mitochondrial stress affects the expression levels of mitochondrial dynamics-related genes, especially the Drp1 gene. Therefore, in this study, the possible toxic effects of TiO2 NPs and Ag NPs on mitochondrial dynamics in testicular tissue of mice were also analyzed at the gene expression level. RT-PCR analysis demonstrated that Ag NPs did not lead to a significant change in the expression levels of Drp1 (mitochondrial fission gene) and Mfn1 (mitochondrial fusion gene). Ag NP treatment increased the expression level of the fusion gene Mfn2 but decreased the expression level of the other fusion gene OPA1. Based on these results, it was concluded that AgNP treatment did not significantly change the balance in the fusion mechanism because the expression level of one fusion gene (Mfn2) increased while that of another fusion gene (OPA1) decreased. Similarly, it was deduced that fission machinery was not significantly affected by Ag NP treatment, since no significant change was observed in the expression level of Drp1. In other words, it was deduced that no disruption in mitochondrial dynamics occurred after Ag NP treatment.
RT-PCR results also displayed that in comparison with the control group, there was a significant increase in the expression of Drp1 in TiO2 NP group but no significant change in expression levels of mitochondrial fusion genes (Mfn1, Mfn2, and Opa1). Namely, in TiO2 NP group, the expression level of only Drp1 was determined to change. Therefore, it was concluded that TiO2 NP treatment caused the disruption of mitochondrial dynamics in testicular cells by increasing mitochondrial fission.
In the literature, it has been shown that analyzing DRP1 protein by western blot technique is a good approach to reveal mitochondrial stress [44,45,46]. In the current study, the western blot analysis revealed that Ag NP treatment did not cause a significant change in the level of DRP1, whereas TiO2 NP treatment significantly increased the levels of DRP1. That is, in parallel with RT-PCR results, western blot results also revealed that TiO2 NP application increased DRP1 level in testicular tissue. Therefore, we concluded that, unlike Ag NPs, TiO2 NPs disrupted the mitochondrial dynamics in testicular cells by causing excess mitochondrial fission.
Investigators have reported that the toxic effect of inorganic NPs impairs mitochondrial dynamics by increasing mitochondrial fission in various cells. For example, Ke et al. [8] demonstrated that gold nanoparticles enhanced apoptotic and autophagic mitochondrial fission in NSCLC cells through the expression of Drp1. Zhao et al. [47] informed that TiO2 NP toxicity caused a mitochondrial dynamic imbalance in HT22 cells by increasing the expression of Drp1. Natarajan et al. [20] demonstrated that TiO2 NPs caused a strong oxidative stress on primary hepatocytes, and this stress resulted in a substantial loss in the fusion process (down-regulation of OPA-1 and Mfn-1 gene expression levels), thus impairing the mitochondrial dynamics and morphology. However, the toxicity of inorganic NPs, including TiO2 NPs on mitochondrial dynamics (mitochondrial fission and fusion mechanisms) in reproductive system cells, has been investigated for the first time in the present study. From the current results, it could be concluded that the toxicity of intravenously applied TiO2 NPs disrupted the mitochondrial dynamics in testicular cells by increasing mitochondrial fission. Finally, the disruption in the mitochondrial dynamics caused some defects in sperm parameters. Overall, it was found that there was a relationship between the toxicity of TiO2 NPs and the distribution of mitochondrial dynamics (mitochondrial fission and fusion mechanisms) in testicular cells.
It has been documented that deterioration in mitochondrial dynamics is associated with diseases such as Alzheimer’s, Parkinson’s, aging, heart failure, obesity, diabetes, and cancer [48,49,50,51]. This study points out that a disruption in mitochondrial dynamics of testicular cells may also cause male infertility. Therefore, when preparing TiO2 NP–conjugated drug formulations, the possible negative effects of TiO2 NPs on fertility should also be taken into account.
Conclusion
This study indicated that intravenously applied Ag NPs and TiO2 NPs could enter the testis via the blood–testis barier and accumulate in mouse testis. The accumulation of Ag NPs did not cause a significant toxic effect on mouse testes and did not disrupt the mitochondrial dynamics in the cells. Conversely, the accumulation of TiO2 NPs caused a significant toxic effect, and this toxic effect reduced the sperm motility and the percentages of swollen sperms. RT-PCR and western blot analyses revealed that TiO2 NPs disrupted the mitochondrial dynamics in testicular cells by causing excess mitochondrial fission.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Cagalinec M, Safiulina D, Liiv M et al (2013) Principles of the mitochondrial fusion and fission cycle in neurons. J Cell Sci 126:2187–2197
Suen DF, Norris KL, Youle RJ (2008) Mitochondrial dynamics and apoptosis. Genes Dev 22:1577–1590
Zorzano A, Liesa M, Sebastián D, Segalés J, Palacín M (2010) Mitochondrial fusion proteins: dual regulators of morphology and metabolism. In Seminars in cell & developmental biology. Academic Press, vol. 21, no. 6, pp. 566–574
Detmer SA, Chan DC (2007) Functions and dysfunctions of mitochondrial dynamics. Nat Rev 8:870–879
Chan DC (2006) Mitochondrial fusion and fission in mammals. Annu Rev Cell Dev Biol 22:79–99
Westermann B (2008) Molecular machinery of mitochondrial fusion and fission. J Biol Chem 283:13501–13505
Grohm J, Kim SW, Mamrak U et al (2012) Inhibition of Drp1 provides neuroprotection in vitro and in vivo. Cell Death Differ 19:1446–1458
Ke S, Zhou T, Yang P et al (2017) Gold nanoparticles enhance TRAIL sensitivity through Drp1-mediated apoptotic and autophagic mitochondrial fission in NSCLC cells. Int J Nanomed 12:2531
Jin CY, Zhu BS, Wang XF, Lu QH (2008) Cytotoxicity of titanium dioxide nanoparticles in mouse fibroblast cells. Chem Res Toxicol 21:1871–1877
Komatsu T, Tabata M, Kubo-Irie M, Shimizu T, Suzuki K, Nihei Y, Takeda K (2008) The effects of nanoparticles on mouse testis Leydig cells in vitro. Toxicol In Vitro 22:1825–1831
Schrand AM, Rahman MF, Hussain SM, Schlager JJ, Smith DA, Syed AF (2010) Metal-based nanoparticles and their toxicity assessment. WIREs Nanomed Nanobi 2:544–568
Tian J, Wong KK, Ho CM, Lok CN, Yu WY, Che CM, Chiu JF, Tam PK (2007) Tropical delivery of silver nanoparticles promotes wound healing. Chem Med Chem 2:129–136
Prabhu S, Poulose EK (2012) Silver nanoparticles: mechanism of antimicrobial action, synthesis, medical applications, and toxicity effects. Int Nano Lett 2:32
Weir A, Westerhoff P, Fabricius L, Hristovski K, von Goetz N (2012) Titanium dioxide nanoparticles in food and personal care products. Environ Sci Technol 46:2242–2250
Shi H, Magaye R, Castranova V, Zhao J (2013) Titanium dioxide nanoparticles: a review of current toxicological data. Part Fibre Toxicol 10:15
Shakeel M, Jabeen F, Shabbir S, Asghar MS, Khan MS, Chaudhry AS (2016) Toxicity of nano-titanium dioxide (TiO 2-NP) through various routes of exposure: a review. Biol Trace Elem Res 172:1–36
Guo LL, Liu XH, Qin DX, Gao L, Zhang HM, Liu JY, Cui YG (2009) Effects of nanosized titanium dioxide on the reproductive system of male mice. Zhonghua Nan Ke Xue 15:517–522
Gao G, Ze Y, Li B et al (2012) Ovarian dysfunction and gene-expressed characteristics of female mice caused by long-term exposure to titanium dioxide nanoparticles. J Hazard Mater 243:19–27
Lu T, Ling C, Hu M et al (2020) Effect of nano-titanium dioxide on blood-testis barrier and MAPK signaling pathway in male mice. Biol Trace Elem Res 1–11
Natarajan V, Wilson CL, Hayward SL, Kidambi S (2015) Titanium dioxide nanoparticles trigger loss of function and perturbation of mitochondrial dynamics in primary hepatocytes. PLoS One 10:e0134541
Li F, Zhou J, Li Y, Sun K, Chen J (2019) Mitochondrial damage and Drp1 overexpression in rifampicin-and ısoniazid-induced liver ınjury cell model. JCTH 7:40
Chen Y, Yan L, Zhang Y, Yang X (2019) The role of DRP1 in ropivacaine-induced mitochondrial dysfunction and neurotoxicity. Artif Cells Nanomed Biotechnol 47:1788–1796
Rezaei N, Chian RC (2005) Effects of essential and non-essential amino acids on in-vitro maturation, fertilization and development of immature bovine oocytes. Int J Reprod BioMed 3:36–41
Torabi F, Shafaroudi MM, Rezaei N (2017) Combined protective effect of zinc oxide nanoparticles and melatonin on cyclophosphamide-induced toxicity in testicular histology and sperm parameters in adult Wistar rats. Int J Reprod Biomed 15(7):403
Somfai T, Bodo SZ, Nagy SZ, Gocza E, Ivancsics J, Kovacs A (2002) Simultaneous evaluation of viability and acrosome ıntegrity of mouse spermatozoa using light microscopy. Biotech Histochem 77:117–120
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Ding H, Jiang N, Liu H et al (2010) Response of mitochondrial fusion and fission protein gene expression to exercise in rat skeletal muscle. BBA-Gen Subjects 1800:250–256
Gao G, Ze Y, Zhao X et al (2013) Titanium dioxide nanoparticle-induced testicular damage, spermatogenesis suppression, and gene expression alterations in male mice. J Hazard Mater 258:133–143
Castellini C, Ruggeri S, Mattioli S, Bernardini G, Macchioni L, Moretti E, Collodel G (2014) Long-term effects of silver nanoparticles on reproductive activity of rabbit buck. Syst Biol Reprod Med 60:143–150
Ji JH, Jung JH, Kim SS, Yoon JU, Park JD, Choi BS et al (2007) Twenty-eight-day inhalation toxicity study of silver nanoparticles in Sprague-Dawley rats. Inhal Toxicol 19:857–871
Gromadzka-Ostrowska J, Dziendzikowska K, Lankoff A, Dobrzyñska M, Instanes C, Brunborg G, Gajowik A, Radzikowska J, Wojewódzka M, Kruszewski M (2012) Silver nanoparticles effects on epididymal sperm in rats. Toxicol Lett 214:251–258
Zou J, Feng H, Mannerström M, Heinonen T, Pyykkö I (2014) Toxicity of silver nanoparticle in rat ear and BALB/c 3T3 cell line. J Nanobiotechnol 12:52
Mohamed El Mahdy M, Salah T, Sayed Aly H, Mohammed F, Shaalan M (2015) Evaluation of hepatotoxic and genotoxic potential of silver nanoparticles in albino rats. Exp Toxicol Pathol 67:21–29
Hassanen EI, Khalaf AA, Tohamy AF, Mohammed ER, Farroh KY (2019) Toxicopathological and immunological studies on different concentrations of chitosan-coated silver nanoparticles in rats. Int J Nanomed 14:4723
Hassanen EI, Ragab E (2021) In vivo and in vitro assessments of the antibacterial potential of chitosan-silver nanocomposite against methicillin-resistant Staphylococcus aureus–induced infection in rats. Biol Trace Elem Res 199:244–257
Hong F, Yu X, Wu N, Zhang YQ (2017) Progress of in vivo studies on the systemic toxicities induced by titanium dioxide nanoparticles. Toxicol Res 6:115–133
Alaee S, Ilani M (2017) Effect of titanium dioxide nanoparticles on male and female reproductive systems. JAMSAT 3:3–8
Wang R, Song B, Wu J, Zhang Y, Chen A, Shao L (2018) Potential adverse effects of nanoparticles on the reproductive system. Int J Nanomed 13:8487
Miura N, Ohtani K, Hasegawa T, Yoshioka H, Hwang GW (2019) Biphasic adverse effect of titanium nanoparticles on testicular function in mice. Sci Rep 9:1–8
Kumar N, Singh AK (2015) Trends of male factor infertility, an important cause of infertility: a review of literature. J Hum Reprod Sci 8:191–196
Serasinghe MN, Chipuk JE (2016) Mitochondrial fission in human diseases. Handb Exp Pharmacol 240:159–188
Ren L, Chen X, Chen X, Li J, Cheng B, Xia J (2020) Mitochondrial dynamics: fission and fusion in fate determination of mesenchymal stem cells. Front Cell Dev Biol 8:580070
Qi X, Qvit N, Su YC, Mochly-Rosen D (2013) A novel Drp1 inhibitor diminishes aberrant mitochondrial fission and neurotoxicity. J Cell Sci 126:789–802
Manczak M, Reddy PH (2012) Abnormal interaction between the mitochondrial fission protein Drp1 and hyperphosphorylated tau in Alzheimer’s disease neurons: implications for mitochondrial dysfunction and neuronal damage. Hum Mol Genet 21:2538–2547
Gawlowski T, Suarez J, Scott B et al (2012) Modulation of dynamin-related protein 1 (DRP1) function by increased O-linked-β-N-acetylglucosamine modification (O-GlcNAc) in cardiac myocytes. J Biol Chem 287:30024–30034
Perdiz D, Lorin S, Leroy-Gori I, Poüs C (2017) Stress-induced hyperacetylation of microtubule enhances mitochondrial fission and modulates the phosphorylation of Drp1 at 616 Ser. Cell Signal 39:32–43
Zhao H, Chen L, Zhong G et al (2019) Titanium dioxide nanoparticles induce mitochondrial dynamic imbalance and damage in HT22 cells. J Nanomater 2019:1–16
Ganguly G, Chakrabarti S, Chatterjee U, Saso L (2017) Proteinopathy, oxidative stress and mitochondrial dysfunction: cross talk in Alzheimer’s disease and Parkinson’s disease. Drug Des Dev Ther 11:797–810
De Rosa M, Gambardella J, Shu J, Santulli G (2018) Dietary fat is a key determinant in balancing mitochondrial dynamics in heart failure: a novel mechanism underlying the obesity paradox. Cardiovasc Res 114:925–927
Dai W, Jiang L (2019) Dysregulated mitochondrial dynamics and metabolism in obesity, diabetes, and cancer. Front Endocrinol 10:570
Amartuvshin O, Lin CH, Hsu SC et al (2020) Aging shifts mitochondrial dynamics toward fission to promote germline stem cell loss. Aging Cell 19:e13191
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All animal procedures were approved by the Animal Experiments Committee of Ataturk University (25 July 2014, Approval No: 2014/116).
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Arslan, N.P., Keles, O.N. & Gonul-Baltaci, N. Effect of Titanium Dioxide and Silver Nanoparticles on Mitochondrial Dynamics in Mouse Testis Tissue. Biol Trace Elem Res 200, 1650–1658 (2022). https://doi.org/10.1007/s12011-021-02763-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-021-02763-6