Abstract
Today atherosclerosis is considered as a main cause of death in the worldwide. There is a significant association between heavy metal exposure and atherosclerosis. In this study, we discussed the scientific literature about the effect of mercury on the pathogenesis of atherosclerosis. We also considered the epidemiological studies on mercury as a risk factor for atherosclerosis. Web of Science, Google Scholar, Medline, PubMed, and Scopus were searched by using the following keywords to 2019: (cardiovascular diseases OR atherosclerosis OR endothelial dysfunction) AND (mercury). Mercury has the potential to act as one of the novel risk factors for atherosclerosis development. The findings have indicated the role of mercury in the pathogenesis of atherosclerosis, vascular endothelial dysfunction, oxidative stress, inflammation, and dyslipidemia. Mercury can induce atherosclerosis indirectly via increasing the total cholesterol, triglycerides, and LDL-C levels as well as decreasing the HDL-C level. Mercury can be considered as a risk factor in the atherosclerosis progression. However, more studies are required to find the exact mechanisms involved in the pathogenesis of atherosclerosis induced by mercury.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mercury is one of the major environmental pollutants that exerts toxic effects on the cardiovascular system [1]. The environmental health agencies identify mercury as a very toxic heavy metal that threatens public health. Mercury possesses toxic impacts on the ecosystem which widely spread in the world [2]. Mercury triggers endothelium lining in the blood vessel wall. The outcome of mercury exposure on cardiovascular is myocardial infarction, heart rate variability, high blood pressure, and atherosclerosis [3]. Although mechanisms underlying mercury-induced cardiovascular diseases are not fully studied, however, several studies have indicated endothelial dysfunction due to mercury-induced oxidative stress may be responsible for cardiovascular disorders [4]. Mercury exposure leads to over-production of reactive oxygen species (ROS), resulting in low-density lipoprotein cholesterol (LDL-C) oxidation, which can induce atherosclerotic plaque formation and progression [4]. Epidemiological studies have indicated an association between mercury exposure and atherosclerosis which focused on vascular endothelial dysfunction [5]. Atherosclerosis is initiated by various risk factors such as genetics, behavioral, and environmental factors [6]. The traditional risk factors for atherosclerosis include abnormal lipid profile (high total cholesterol, high LDL-C, and low high-density lipoprotein cholesterol (HDL-C)), age, male gender, hypertension, diabetes mellitus, and smoking [6]. Furthermore, the novel risk factors have been identified to enhance our ability to assess and control the global risk profile of atherosclerosis [7]. Impaired fasting glucose, triglycerides (TG), lipoprotein (a), homocysteine, and high-sensitivity C-reactive protein (CRP) have been recognized as new risk factors of atherosclerosis [7]. According to several clinical and pre-clinical studies, mercury is one of the important risk factors for atherosclerosis [8]. The present study was designed to review several experimental and epidemiological studies that evaluate the relationship between mercury exposure and atherosclerosis. In addition, several mechanisms have been discussed in the current review to explain this link.
Method
All available scientific publications about mercury exposure and atherosclerosis were gathered by searching several databases including Web of Science, Google Scholar, Medline, PubMed, and Scopus. The studies were selected by using the following keywords: (cardiovascular diseases OR atherosclerosis OR endothelial dysfunction) AND (mercury). Only published English language articles from beginning to 2019 were selected for providing the present review.
History
Metallic mercury and its inorganic salts have been known to man since antiquity and medical application of this element. Compounds of mercury were used as a reliable germicide [9], antiseptic [10], and treatment of fungal infection, psoriasis [11], and ocular conditions [12]. The organic mercury compounds with covalent bonds were first described in the nineteenth century [13]. Methyl mercury is stored in the lipid portion of muscle tissue of aquatic life and may lead to bioaccumulation over time. A well-publicized case of mercury bioaccumulation occurred between the 1930s and the 1960s in Japan, where thousands of people in and around the Minamata town were poisoned by eating fish contaminated with mercury dumped by a chemical factory into Minamata Bay [14]. Another major historical outbreak of mercury poisoning was reported from Iraq in which high exposure occurred as a result of the use of homemade bread of seed wheat exposed with a methyl-mercurial fungicide [15, 16].
Chemical Properties of Mercury
Mercury is a heavy metal that usually known with Hg as a symbolic name [17]. Mercury is also called liquid silver due to its liquid state in room temperature [17]. This metal has been effective as a medicine and a poisoning agent from ancient days [18]. Nowadays, mercury is used in fluorescent lamps, batteries, felt production, barometers, thermometers, and dental amalgams [19]. Recently, this metal has been focused as an important environmental pollutant that threatens human health [20]. The chemical forms, sources of exposure, toxicokinetics, and toxicity mechanism for mercury will be mentioned in the following.
Chemical Forms
There are three forms of mercury including metallic mercury (elemental mercury), inorganic mercury, and organic mercury [21]. Metallic mercury (Hg0) is a pure form of mercury that is a liquid at 20 °C [22]. Metallic mercury is applied in thermometers, latex paint, some electrical switches, and dental amalgams [22]. Metallic mercury may evaporate and enter the atmosphere at room temperature [22]. This agent in the vapor state is colorless and odorless that quickly absorbed by the respiratory system [22]. Inorganic mercury (Hg salts) is formed when it reacts with elements such as sulfur, acetate, chlorine, and oxygen [23]. Most mercury salts are white powders; mercuric sulfide is red and turns black following exposure to light [23]. Inorganic mercury is used in cosmetic products, laxatives, teething powders, antiseptics, and diuretics [24]. The formation of organic mercury compounds occurs following the interaction between mercury and carbon [24]. Methyl mercury (MeHg) and ethyl mercury (EtHg) are two of abundance organic mercury compounds in the environment [24]. The other organic mercury compound is dimethylmercury that is also used in some chemical analysis as a reference standard [24]. Methyl mercury and ethyl mercury are white powder, and dimethylmercury is a colorless liquid [24]. The use of methyl mercury and ethyl mercury compounds in antifungal agents has been forbidden from 1991 [25]. The addition of thimerosal-containing ethyl mercury to vials of the vaccine has been eliminated or decreased according to the guideline of Public Health Service agencies in 1999 [25].
Sources of Mercury Exposure
Mercury may enter into the environment within some natural processes such as volcanic outgassing and human activities such as mining [26, 27]. Metallic mercury, mercuric chloride, mercuric sulfide, and methyl mercury are the most known environmental mercury [28]. World Health Organization reported that occupational exposure, dental amalgam, and consumption of contaminated fish are the main familiar sources of mercury for human poisoning [28]. Mercuric sulfide and elemental mercury are the primary forms of mercury in the earth’s crust [29]. The mercury is released into the atmosphere through volcanic activity, coal combustion, mining, and solid waste [29]. Methyl and dimethylmercury are usually caused by the biological process [28]. Methyl mercury enters to human food chain when the organism containing methyl mercury is ingested by fish [28]. Dispersed mercury in water is converted into methyl mercury by microorganisms [29]. Dimethylmercury is found at dangerous waste sites, and it is very detrimental for human and animals [29]. The general population is usually exposed to metallic mercury through mercury released from dental amalgam fillings [29].
Toxicokinetics of Mercury Exposure
Elemental Mercury or Metallic Mercury Compounds
A liquid form of metallic mercury absorption is minimal. In fact, it is not considered a threat to human health [21]. About 80% of metallic mercuric is absorbed through the respiratory system in vapor form [30]. Metallic mercury has high lipophilicity, and thus mercury rapidly crosses the cell membranes, the blood-brain barrier, and placenta. Therefore, it is deposited in most tissue [31]. The complex of mercury at sulfhydryl group in proteins leads to toxic effects on the various tissues including vessels [32]. Although metallic mercury is rapidly changed to mercuric mercury form in the blood, the accumulation of metallic form has been found in the central nervous system [32]. The half-life of mercury is reported from few days to several months, depending on the rate of accumulation in tissue, the organ of deposition, and oxidation state [33].
Mercurous Mercury and Mercuric Mercury Compounds
The absorption of mercurous mercury in the form of Hg2Cl2 is very low, although some absorption occurs during oxidation to more absorbable forms [34]. It was proposed that the mercurous mercury is modified to metallic mercury and mercuric mercury in the body [34]. Mercuric chloride (HgCl2) has been considered as the most toxic and corrosive mercuric mercury compound which has been used as a preservative for the development of photographic film [34]. However, it results in death after accidental ingestion or eating for suicide purposes [34]. Similar to metallic mercury, mercuric mercury in the blood binds to sulfhydryl groups on glutathione, metallothionein, and erythrocytes [35]. Mercuric mercury does not pass the blood-brain barrier, but it can cross in the placenta and deposits in the fetal tissues [35]. Mercuric mercury is transported through amino acid transporters [35]. Its accumulation in the brain occurs via binding to cysteine [35]. Mercuric mercury is mainly deposited in the kidney and liver [35]. The cardiovascular toxicity has been reported after acute and chronic exposure to mercuric mercury [35]. It excretes through urine, stool, sweat, tears, breast milk, and saliva. Its effective half-life is 42 days for 80% of oral ingestion [35].
Organic Mercury Compounds
Among organic mercury compounds, methyl mercury is considered as a major threat to human health due to its consumption through polluted fish [36]. Methyl mercury is efficiently absorbed through lung, intestinal, and skin. This agent strongly binds to sulfhydryl-containing protein in the bloodstream [36]. The distribution of methyl mercury to peripheral tissues occurs through cysteine transporter [36]. It is distributed in the brain, kidney, liver, placenta, and fetus. The half-life of methyl mercury is about 70 days. It is usually exerted through stool and urine [36].
The Safety Levels for Human
The chemical form of mercury determines its toxic impact on several tissues [37]. Mercury compounds are classified in the neurotoxic agent groups [37]. Several studies have reported the safe recommended doses of different forms of mercury. FAO/WHO suggested that intake of 1.6 mg/kg of mercury did not cause neurological damage [38]. However, the neurotoxic effects of mercury at 1.6 mg/kg were found in the fetus. It was also reported that the blood mercury levels > 5 ng/mL are toxic for human [38]. The WHO confirmed the hair mercury levels < 6 μg/g are safe [38]. However, ethyl mercury even at levels below allowable concentrations causes neurological and cardiovascular diseases in children [38].
Mechanism of Mercury Toxicity
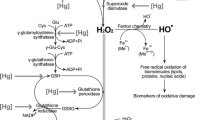
Mercury compounds induce toxic damages in the body through several mechanisms. Researchers have shown that the toxic mechanisms of all forms of mercury have high similarities [39]. The interaction of mercury with sulfhydryl groups is recognized as essential mechanisms with which altering the structure of proteins, especially low molecular weight thiols such as cysteine [40]. It was suggested that the binding of mercury to sulfhydryl groups led to modified DNA, RNA, and ribosomal proteins [40]. In addition, mercury compounds, particularly methyl mercury, have a high affinity for binding to sulfhydryl groups of microtubules which may lead to disturbances in cellular structural maintenance [40]. Mercury also causes damage to glutathione/amino acid transports and disturbs energy metabolism, amino acid metabolism, and membrane phospholipid structure [40]. Indeed, the toxicity of mercury may be mediated by the content of sulfhydryl groups including reduced glutathione (GSH), metallothioneins (MTs), and other sulfhydryl-containing constituents [41]. It is suggested that differences in the cellular sulfhydryl content may be responsible for the different sensitivity of various cells to the mercury toxicity [41]. Over-production of ROS is another important mechanism involved in the toxicity of mercury [41]. Mercury also decreases the activities of antioxidant enzymes by interacting with selenium (Se) [42]. Se is an essential element which mainly acts through selenoproteins such as glutathione peroxidase. Moreover, Se alleviates the toxicity of heavy metals. This element has been shown to react with Hg in the bloodstream by forming complexes containing the two elements at an equimolar ratio when selenite and mercuric chloride are co-administered. Se acts as a cofactor of the key antioxidant enzyme in which it contributes both catalytic activity and spatial conformation. Se also has the ability to protect the membrane lipids from oxidative damage. Se additionally increases the antioxidant status as indicated by the elevated glutathione peroxidase activities which will give protection against spontaneous lipid peroxidation [42]. Therefore, the outcomes of mercury-induced toxicity may be associated with a disturbed balance between the oxidant and antioxidant system [42]. The other mechanism which plays a main role in the toxicity induced by mercury is inflammation [33]. It was reported that mercury in the non-toxic levels induced proliferation and cytokine production from T lymphocytes [43]. Controversially, it has been shown that mercury inactivated nuclear factor B (NF-kB) [43]. NF-kB is the major transcription factor involved in the expression of several inflammatory and immunity components [43]. They suggested that the binding of mercury to sulfhydryl groups resulted in the inactivation of NF-KB [43]. However, numerous studies indicated that the toxic effects of heavy metals on the cellular function are related to the absorbed dose, route, and duration of exposure [44].
Mercury and the Risk of Atherosclerosis: Epidemiological Observation
Numerous studies have indicated inconsistent results regarding the impact of mercury on the development of atherosclerosis. The relation between hair mercury levels and development of carotid atherosclerosis has been conducted by Salonen and coworkers (2000) on the 1014 men 40–60 years old from Kuopio Ischemic Heart Disease Risk Factor (KIHD) cohort study. The findings have indicated that there was an increase of 8 μm for each microgram of methylmercury in the hair in the 4-year increment in the mean common carotid artery intima-media thickness (CCA-IMT). It was indicated that the mean CCA-IMT was higher in the men with the highest hair mercury content versus the other subjects after adjustment for relevant confounders. The significant association was seen between the mean CCA-IMT, high hair mercury content, and cigarette smoking in participants [45]. The data have shown that the accumulated mercury in organs is connected to the development of carotid atherosclerosis. The connection between the level of mercury in urine and the thickness of carotid or the diameter of the intraventricular septum has been searched in 155 workers in the company with mercury production. The concentration of mercury in urine was 5.6 ± 12.2 μg/g creatinine in men (n = 125) and 1.9 ± 2.7 μg/g creatinine in women (n = 29). Additionally, the mercury concentration in the urine of smoker was higher than in the non-smokers. There was a positive relationship between the thickness of carotid and the concentration of mercury in urine, and also a negative linear correlation between the thickness of carotid and HDL-C. The negative dependence between the thickness of carotid and the antioxidant of serum (r = − 0.2142; p < 0.05), and the positive relation between the antioxidant of serum and HDL-C were exposed to be reliable for the all concerned groups. The lipid profile was normal in women; however, the level of TG was upper than the normal range in men. Since the control group was not selected, the findings were compared to the normal range. The marked differences in the serum lipid profiles and total antioxidant were not observed between the smokers and non-smokers.
It showed that the long exposure to the vapor of mercury might cause atherosclerosis in carotid. The relation between thickness in carotid and the elimination of mercury as a marker for metallic mercury in urine is documented in the non-smoking subjects. The protective mechanisms for preventing atherosclerosis in the non-smokers are tightly connected to HDL; however, these protective mechanisms are impaired in smokers [46]. A study conducted by You and coworkers on 477 Koreans (2011) has indicated there is a significant correlation between blood mercury and LDL-C as well as HDL-C levels. The blood mercury concentration in males was higher than in females, and the relation between mercury and cardiovascular disease was seen in the male. However, smoking status has not been related to blood methyl mercury levels [47]. Hong et al. (2013) studied the relation between the modification of lipid profile and that of mercury in hair in 236 non-smoking and smoking subjects 16–75 years old who had visited the center of health in hospital from January 2004 to January 2007. The data have shown that the concentration of methylmercury had a positive connection to the raised levels of total cholesterol, TG, LDL-C, and the reduced level of HDL-C. Furthermore, there was a rise in the concentration of mercury in the group of smoking. It has been proposed that smoking is positively connected to the accumulation of mercury, and high concentration of mercury is connected to the abnormal of serum lipid [48].
Eom et al. (2014) showed the importance of the level of methylmercury in blood on the lipid concentration in 2114 normal people that have not been involved in mercury in their jobs. The data have shown that the average concentration of mercury in the serum was 3.90 μg/L which was dramatically modified by residence area, alcoholic consumption, age, seafood intake, sex, and smoking [49]. Asgary and coworkers (2017) have indicated that mercury deteriorated lipid profile modification in coronary artery disease patients versus normal people. In the study of case-control, 65 angiographically coronary artery disease patients (35 females) 50–70 years old and 65 normal people (43 female) adjusted for age, sex, and living place were managed. They have shown that the mercury concentration of coronary artery disease patients was dramatically higher versus control people. The similar data were obtained after matching for risk factors of cardiovascular such as hypertension, diabetes mellitus, dyslipidemia, and age. The average levels of HDL-C, TC, and TC/HDL-C ratio were dramatically higher in coronary artery disease patients. However, there was no dramatic connection between HDL-C, TC, and TC/HDL-C ratio with the mercury concentration in serum. The obtained data have indicated that the concentration of methylmercury in serum is connected with coronary artery disease. The authors did not determine the smoking and smoking status and their prevalence since the main goal of their study was to evaluate the relationship between mercury levels and CAD [50].
The continued involvement with mercury acts in the progression of plaques in coronary atherosclerosis. The connections between risk factors of cardiovascular disease and the concentration of mercury and methyl mercury in adults were examined using information from the Nutrition Examination Survey (NHANES) and National Health, 2011–2012. The result for 1129 adults 12–19 years old who attended in the US NHANES 2011–2012 was evaluated. The statistical analysis was done to study the connection between risk factors of coronary artery disease and concentrations of methylmercury in blood. The direct connection between methyl mercury and mercury in blood and TC in adult in the matched model was seen. There are no connections with other risk factors of coronary artery disease were explored in all people. The analyzed data with sex linear correlation models showed that the serum TC level of girls exposed to the highest levels of methyl mercury increased 4.22% higher versus girls with the lowest levels of methyl mercury. The findings show the serum cotinine levels, indicator of cigarette exposure, were not related to mercury or methyl mercury levels. The results proposed that methyl mercury in the blood may be directly connected with the total cholesterol in adult girls [51].
Using the cross-sectional analysis, they found the connections of dyslipidemia and its components with toenail mercury in the Yeungnam area in South Korea, including 269 women and 232 men. The average toenail methylmercury levels were 0.34 μg/g for women and 0.47 μg/g for men. After adjusting all the confounding including smoking status, subjects with the highest levels of toenail mercury had 2.24 times higher risk of dyslipidemia and 4.08 times higher risk of hyper-LDL-c than those in the lowest toenail mercury [52]. One cross-sectional study was conducted to evaluate the association between serum lipid profile (HDL-C, LDL-C, the ratio of APO B to APO-A, and TG levels), oxidized LDL (ox-LDL), and heavy metal (HM) content (numerous arsenic and mercury) of seafood consumption in 81 adults without risk factors for CVD. The smokers were not included in this study. The study indicated that the moderate amount consumption of shellfish was related to a pro-atherogenic alteration in lipid profile. This link is affected by the estimated exposure to arsenic and mercury in shellfish [53]. Controversially, the association between mercury exposure and incidence of atherosclerosis was not observed in the study conducted by Yoshizawa and coworkers (2002) on 33,737 individuals [54]. It was observed that the weak correlation between methylmercury exposure and fish consumption caused an increase in the risk of coronary heart disease. The highest percentage of current smokers being parallel to the lowest mercury level may be related to the lifestyles of subjects, that smoker healthy men may consume more fish [54]. Furthermore, the marked association was not observed between the urine levels of mercury and coronary heart disease in patients. It was suggested that nutrition (fish and alcohol eat) and smoking are important sources of mercury. However, this metal has a weak role in the pathogenesis of CAD versus the impact of blood pressure, glucose, and lipid profile in the selected participants [55]. Table 1 shows the epidemiological research on the connections of mercury levels of tissues with the atherosclerotic risk factors.
Possible Mechanism of Mercury-Induced Atherosclerosis
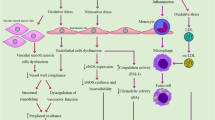
The involved mechanisms in the mercury-induced atherosclerosis are not precise, but vascular endothelial dysfunction, oxidative stress, inflammation as well as lipid profile abnormalities are suggested to play the main role in the toxic effects of mercury on the cardiovascular system [1].
Mercury, Vascular Endothelial Cells, and Atherosclerosis
Several hypotheses describe the initiation and progression of cardiovascular diseases. According to the present literature, “response to injury hypothesis” is the main mechanism that is responsible for atherosclerosis induced by mercury. This hypothesis indicates that primary endothelial damage and/or functional disruption causes atherosclerosis [56]. Endothelial damage is caused following the release of vasoconstrictor, proinflammatory, and prothrombic mediators. Although several pathways are involved in endothelial dysfunction, the over-production of free radicals has a main role in this process. Furthermore, NO and superoxide anions interaction leads to the peroxynitrite production which deteriorates lipid peroxidation and platelet aggregation. Mercury can affect the regulation of cellular redox, transcription factor, and gene expression related to the dysfunction of an endothelial cell. Mercury toxicity may damage the vascular endothelium cells by inducing oxidative stress [57]. ROS over-production following mercury exposure causes endothelial cell damage and dysfunction. Mercury also increased the production of superoxide anion (O2−) by inducing NADPH oxidase, resulting in the disruption of the mitochondrial membrane, which leads to endothelial damage [57].
Mercury induces a modification in the function of the endothelial barrier by the impairment of endothelial cell-cell adhesions and by inducing apoptosis in endothelial cell, which has been indicated in vivo and in vitro [55]. According to the “response to injury hypothesis,” the endothelial damage leads to elevated permeability to inflammatory cells and lipoproteins, and accumulation of LDL-c in the vessel wall. In vivo studies have indicated that lipids accumulate in various tissues such as a vascular wall in mercury-exposed animals [56]. Additionally, over-production of ROS by mercury causes ox-LDL that leads to the progression of atherosclerotic plaque [4]. The levels of mercury may be used as a predictor of the ox-LDL-c levels [4]. Other involved mechanism in the toxic mercury impact on the vascular endothelium is inhibition of the “paraoxonase” activity [4]. Paraoxonase decreases LDL oxidation, resulting in the inhibition of atherosclerosis progression [4]. It was observed that extracellular cells and matrix below the endothelium are exposed to factors in the bloodstream in endothelial damage induced by mercury [56]. Additionally, due to mercury-induced endothelial cells death in the vessel wall, cellular debris is released [56]. All these events induce cellular stress leading to secretion of inflammatory cytokines. Endothelial cell damage, oxidative stress, and inflammation have the main role in the onset and progression of atherosclerosis [56].
Mercury, Inflammation, and Atherosclerosis
Inflammations have an important role in the pathogenesis of CVD induced by mercury [58]. The association between mercury and inflammation of the brain and also systemic inflammation has been found, but the link between mercury and vascular inflammation is not fully understood [59]. Inflammation associated with vascular tissue remodeling demonstrates the presence of inflammation phenomenon induced by mercury [59]. In vitro studies have confirmed this hypothesis by indicating cytokines release, and also an expression of adhesion molecules intercellular adhesion molecule-1 (ICAM1) in cells exposed to mercury [60]. However, the link of serum mercury levels and CRP levels in the serum of humans has not been reported [60]. Controversially, some studies have indicated that mercury might also act as an anti-inflammatory agent. Research has shown that the low dose of mercury down-regulates the NF-Kβ expression [43]. One study indicated that the levels of IL-1β and TNF-α reduced in the workers exposed to mercury [61]. Due to the pro- and anti-inflammatory properties of mercury, more studies are needed to identify this issue [62]. It is suggested that the dose of mercury and duration of exposure are the major factors that cause the above “contradictory” findings [62].
Mercury, Oxidative Stress, and Atherosclerosis
Mercury exposure increases the generation of reactive ROS during atherosclerosis through mitochondrial electron transport uncoupling, induction of oxidative enzymes, and some inflammatory mediators. Methyl mercury binds to the glutathione peroxidase (GPx) through thiol (–SH) and/or selenol (–SeH) groups from macromolecules [63]. Mercury has a high affinity to the thiol groups. This binding reduces glutathione peroxidase selenium-dependent activity [62]. GPx reduces hydrogen peroxide to water and oxygen [64]. GPx also reduces lipid peroxides to alcohols and oxygen by using glutathione [65]. Altogether, these molecules are the primary antioxidant defense system in cells [66]. Catalase and superoxide dismutase are the two antioxidant enzymes which combat against oxidative stress induced by mercury [67]. The increase of ROS and decrease of the antioxidant activity lead to the progression of cardiovascular disease including atherosclerosis [67]. Free radicals induce lipid peroxidation which is an autocatalytic process [68]. During lipid peroxidation, polyunsaturated fatty acids such as phospholipids in LDL in cell membranes decrease via a chain reaction [68]. The presence of ox-LDL in the artery walls attracts monocytes into the vessel endothelium, where they change into macrophages [57]. Macrophages remove ox-LDL particles and accumulate intracellular lipids in vessel endothelium [57]. Then macrophages release proinflammatory cytokines which induce monocyte recruitment and formation of foam cells. These are the main cells that produce early atherosclerotic lesions [57]. Mercury has been indicated to induce lipid peroxidation and atherosclerosis in animals and humans [69]. Mercury also increased myeloperoxidase activity in oxidant-induced tissue damage [69]. Mercury inactivates paraoxonase, an important HDL-C-bound antioxidative enzyme, which has been related to the risk of atherosclerosis [70]. The chronic exposure to mercury leads to increase endothelial dysfunction in vessels, because of the reduced nitric oxide (NO) bioavailability due to the elevation of superoxide anion (O2·−) generation from NADPH oxidase [71]. Inhibition of eNOS phosphorylation and expression by mercury are other mechanisms involved in the pathogenesis of atherosclerosis [71].
Mercury, Dyslipidemia, and Atherosclerosis
Several epidemiologic and experimental studies have indicated a link between dyslipidemia and progression of atherosclerosis [72]. Mercury has been found to increase the plasma levels of TC, TG, and phospholipids [72]. However, the underlying mechanism of dyslipidemia induced by mercury is not clear. It was reported that mercury exposure might cause dyslipidemia via inducing damage to enzymes related to lipid metabolism [73]. Furthermore, mercury binds to glutathione, leading to increased ROS production, which promotes lipid peroxidation [73, 74].
Epidemiological evidences have also confirmed that occupational exposure to mercury increased the prevalence of dyslipidemia [75, 76]. Regarding the main role of dyslipidemia in the pathogenesis of atherosclerosis, the screening of the blood lipid profile is necessary for preventing cardiovascular diseases [77]. More studies should be done to investigate the mechanisms involved in dyslipidemia induced by mercury.
Conclusion
Mercury is substantially present in our world. The pro-atherogenic effects of mercury versus the other common risk factors could cause a severe impact on human health. In the animal models, the toxic effects of mercury, especially the pathophysiological aspects of mercury on the classes have been found. The numerous epidemiological evidences have also confirmed the association between mercury exposure and atherosclerosis. Vascular endothelial cells have high sensitivity to oxidative stress which leads to endothelial dysfunction seen in atherosclerosis. The dose, time, and route of exposure to mercury play main roles in its toxic effects on the vascular endothelial cell. The mechanism of vascular toxicity of mercury is not fully understood, but the induction of oxidative stress is suggested as a primary mechanism. Mercury induces the atherosclerosis progression through vascular endothelial dysfunction, oxidation of lipoprotein, aggregation of platelets, inflammation in the vessels, vascular smooth muscle cell proliferation, and dyslipidemia. Additionally, mercury exerts toxic impact by disrupting several cellular signaling pathways that cause vascular inflammation and platelet aggregation. Most findings of the present study have indicated the association between the mercury level of blood, hair, urine, toenail, and atherosclerosis risk factors such as dyslipidemia and also coronary artery diseases. The studies have suggested that mercury can induce atherosclerosis indirectly via increasing the TC, TG, and LDL-C levels as well as decreasing HDL-C level. However, some studies have not confirmed the association between the mercury levels in biological samples and atherosclerosis.
Since the methylmercury form has suitable absorption in the gut and accumulates in epithelial tissues, this form of mercury was very common in the selected articles. Urinary mercury is the best index of exposure to elemental and inorganic mercury, while blood mercury has been focused on human studies as an index for methylmercury exposure through food in populations.
Limitation
Due to a low number of conducted studies on this field and also low scale size of intervention groups, the present review could not introduce mercury as one of the main risk factors of atherosclerosis. Additionally, the present study has other limitation: the confounding factors including age, gender, and smoking were different in the included studies. Most of the studies had reported various cigarette smoking in their participants. Interestingly, the studies have shown an association between cigarette smoking and blood mercury levels. Therefore, more research is needed to thoroughly understand the involved mechanisms in the mercury effects on the cardiovascular system.
References
Fernandes Azevedo B, Barros Furieri L, Peçanha FM, Wiggers GA, Frizera Vassallo P, Ronacher Simões M, Fiorim J, Rossi de Batista P, Fioresi M, Rossoni L, Stefanon I (2012) Toxic effects of mercury on the cardiovascular and central nervous systems. Biomed Res Int 2012:949048
Driscoll CT, Mason RP, Chan HM, Jacob DJ, Pirrone N (2013) Mercury as a global pollutant: sources, pathways, and effects. Environ Sci Technol 47(10):4967–4983
Roman HA, Walsh TL, Coull BA, Dewailly É, Guallar E, Hattis D, Mariën K, Schwartz J, Stern AH, Virtanen JK, Rice G (2011) Evaluation of the cardiovascular effects of methyl mercury exposures: current evidence supports development of a dose–response function for regulatory benefits analysis. Environ Health Perspect 119(5):607–614
Grotto D, Valentini J, Fillion M, Passos CJ, Garcia SC, Mergler D, Barbosa F Jr (2010) Mercury exposure and oxidative stress in communities of the Brazilian Amazon. Sci Total Environ 408(4):806–811
Stein JH, Korcarz CE, Hurst RT, Lonn E, Kendall CB, Mohler ER, Najjar SS, Rembold CM, Post WS (2008) Use of carotid ultrasound to identify subclinical vascular disease and evaluate cardiovascular disease risk: a consensus statement from the American Society of Echocardiography Carotid Intima-Media Thickness Task Force endorsed by the Society for Vascular Medicine. J Am Soc Echocardiogr 21(2):93–111
Long A, Long B, Koyfman A (2018) Non-traditional risk factors for atherosclerotic disease: a review for emergency physicians. Am J Emerg Med 36(3):494–497
Xiong XD, Xiong WD, Xiong SS, Chen GH (2017) Research progress on the risk factors and outcomes of human carotid atherosclerotic plaques. Chin Med J 130(6):722–729
Landmark K, Aursnes I (2004) Mercury, fish, fish oil and the risk of cardiovascular disease. Tidsskr Nor Laegeforen 124(2):198–200
Baker O (1889) A note on the use of perchloride of mercury gauze as a substitute for sponges in abdominal surgery. Ind Med Gaz 24(5):135
Lister J (1890) On two cases of long-standing dislocation of both shoulders treated by operation, with further observations on the cyanide of zinc and mercury. Br Med J 1(1514):1–4
Mapother ED (1891) The parasitic nature of psoriasis; its treatment by mercury. Br Med J 1(1568):110–112
Landolt E (1892) Clinical observations on the abuse of mercury in the treatment of diseases of the eyes. Br Med J 1(1630):650–651
Grandjean P, Satoh H, Murata K, Eto K (2010) Adverse effects of methylmercury: environmental health research implications. Environ Health Perspect 8; 118(8):1137–1145
Hogden RC (2012) Assessing the environmental literacy of intro environmental science students. University of Colorado at Denver
Damluji SF, Tikriti S (1972) Mercury poisoning from wheat. Br Med J 1(5803):804
Bakir F, Damluji SF, Amin-Zaki L, Murtadha M, Khalidi A, Al-Rawi NY, Tikriti S, Dhahir HI, Clarkson TW, Smith JC, Doherty RA (1973 Jul 20) Methylmercury poisoning in Iraq. Science. 181(4096):230–241
Bjørklund G, Dadar M, Mutter J, Aaseth J (2017 Nov 1) The toxicology of mercury: current research and emerging trends. Environ Res 159:545–554
Carvalho CM, Lu J, Zhang X, Arnér ES, Holmgren A (2011) Effects of selenite and chelating agents on mammalian thioredoxin reductase inhibited by mercury: implications for treatment of mercury poisoning. FASEB J 25(1):370–381
Sahani M, Sulaiman NS, Tan BS, Yahya NA, Anual ZF, Mahiyuddin WW, Khan MF, Muttalib KA (2016) Mercury in dental amalgam: are our health care workers at risk? J Air Waste Manage Assoc 66(11):1077–1083
Suvarapu LN, Baek SO (2017) Recent studies on the speciation and determination of mercury in different environmental matrices using various analytical techniques. Int J Anal Chem 2017:1–28
Dranguet P, Le Faucheur S, Slaveykova VI (2017 Dec 1) Mercury bioavailability, transformations, and effects on freshwater biofilms. Environ Toxicol Chem 36(12):3194–3205
Oliveira CS, Piccoli BC, Aschner M, Rocha JB (2017) Chemical speciation of selenium and mercury as determinant of their neurotoxicity. In: Neurotoxicity of metals. Springer, Cham, pp 53–83
Yuan B, Wang DX, Zhu LN, Lan YL, Cheng M, Zhang LM, Chu JQ, Li XZ, Kong DM (2019 Mar 6) Dinuclear HgII tetracarbene complex-triggered aggregation-induced emission for rapid and selective sensing of Hg2+ and organomercury species. Chem Sci 10(15):4220–4226
Carvalho LVB, Hacon SS, Vega CM, Vieira JA, Larentis AL, Mattos RCOC, Valente D, Costa-Amaral IC, Mourão DS, Silva GP, Oliveira BFA (2019) Oxidative stress levels induced by mercury exposure in Amazon juvenile populations in Brazil. Int J Environ Res Public Health 16(15)
Asano S, Eto K, Kurisaki E, Gunji H, Hiraiwa K, Sato M, Sato H, Hasuike M, Hagiwara N, Wakasa H (2000 Mar) Acute inorganic mercury vapor inhalation poisoning. Pathol Int 50(3):169–174
ATSDR (1999) Toxicological profile for mercury. U.S. Department of Health and Human Services. Public Health Service Agency for Toxic Substances and Disease Registry
USGS (2000) Mercury in the environment. https://www2.usgs.gov/themes/factsheet/14600/. (Accessed December 28th, 2017)
Genchi G, Sinicropi M, Carocci A, Lauria G, Catalano A (2017) Mercury exposure and heart diseases. Int J Environ Res Public Health 14(1):74
Bae HS, Dierberg FE, Ogram A (2019) Periphyton and flocculent materials are important ecological compartments supporting abundant and diverse mercury methylator assemblages in the Florida Everglades. Appl Environ Microbiol 85(13)
Chan TY (2011) Inorganic mercury poisoning associated with skin-lightening cosmetic products. Clin Toxicol 49(10):886–891
Barregård L, Sällsten G, Schütz A, Attewell R, Järvholm B (1992) Kinetics of mercury in blood and urine after brief occupational exposure. Arch Environ Health 47(3):176–184
Li Y, He B, Hu L, Huang X, Yun Z, Liu R, Zhou Q, Jiang G (2018) Characterization of mercury-binding proteins in human neuroblastoma SK-N-SH cells with immobilized metal affinity chromatography. Talanta. 178:811–817
Roos PM, Dencker L (2012) Mercury in the spinal cord after inhalation of mercury. Basic Clin Pharmacol Toxicol 111(2):126–132
Ozuah PO (2000) Mercury poisoning. Curr Probl Pediatr 30(3):91–99
Safari F, Akramian M, Salehi-Arjmand H, Khadivi A (2019) Physiological and molecular mechanisms underlying salicylic acid-mitigated mercury toxicity in lemon balm (Melissa officinalis L.). Ecotoxicol Environ Saf 183:109542
Khan F, Momtaz S, Abdollahi M (2019) The relationship between mercury exposure and epigenetic alterations regarding human health, risk assessment and diagnostic strategies. J Trace Elem Med Biol 52:37–47
IPCS (1991) Inorganic mercury. http://www.inchem.org/documents/ehc/ehc/ehc118.htm. (Accessed February 06, 2017)
JECFA (2006) Methylmercury. Summary and conclusions of the 67th The Joint FAO/WHO Expert Committee on Food Additives. Geneva, World Health Organization, International Programme on Chemical Safety. WHO Technical Report Series;(940) http://www.who.int/foodsafety/chem/en/
Guchhait R, Chatterjee A, Gupta S, Debnath M, Mukherjee D, Pramanick K (2018 Jan 1) Molecular mechanism of mercury-induced reproductive impairments in banded gourami, Trichogaster fasciata. Gen Comp Endocrinol 255:40–48
Bose-O'Reilly S, McCarty KM, Steckling N, Lettmeier B (2010 Sep 1) Mercury exposure and children’s health. Curr Probl Pediatr Adolesc Health Care 40(8):186–215
Herath HMLPB, Wickramasinghe PDSU, Bathige SDNK, Jayasooriya RGPT, Kim GY, Park MA, Kim C, Lee J (2017) Molecular identification and functional delineation of a glutathione reductase homolog from disk abalone (Haliotis discus discus): insights as a potent player in host antioxidant defense. Fish Shellfish Immunol 60:355–367
El-Demerdash FM (2001 Jun 30) Effects of selenium and mercury on the enzymatic activities and lipid peroxidation in brain, liver, and blood of rats. J Environ Sci Health B 36(4):489–499
Kim SH, Johnson VJ, Sharma RP (2002) Mercury inhibits nitric oxide production but activates proinflammatory cytokine expression in murine macrophage: differential modulation of NF-κB and p38 MAPK signaling pathways. Nitric Oxide 1;7(1):67–74
Tchounwou PB, Yedjou CG, Patlolla AK, Sutton DJ (2012) Heavy metal toxicity and the environment. In: Molecular, clinical and environmental toxicology. Springer, Basel, pp 133–164
Salonen JT, Seppänen K, Lakka TA, Salonen R, Kaplan GA (2000 Feb 1) Mercury accumulation and accelerated progression of carotid atherosclerosis: a population-based prospective 4-year follow-up study in men in eastern Finland. Atherosclerosis. 148(2):265–273
Skoczyńska A, Poręba R, Steinmentz-Beck A, Martynowicz H, Affelska-Jercha A, Turczyn B, Wojakowska A, Jędrychowska I (2009) The dependence between urinary mercury concentration and carotid arterial intima-media thickness in workers occupationally exposed to mercury vapour. Int J Occup Med Environ Health 22(2):135–142
You CH, Kim BG, Kim JM, Yu SD, Kim YM, Kim RB, Hong YS (2011) Relationship between blood mercury concentration and waist-to-hip ratio in elderly Korean individuals living in coastal areas. J Prev Med Public Health 44(5):218–225
Hong D, Cho SH, Park SJ, Kim SY, Park SB (2013) Hair mercury level in smokers and its influence on blood pressure and lipid metabolism. Environ Toxicol Pharmacol 1; 36(1):103–107
Eom SY, Choi SH, Ahn SJ, Kim DK, Kim DW, Lim JA, Choi BS, Shin HJ, Yun SW, Yoon HJ, Kim YM (2014) Reference levels of blood mercury and association with metabolic syndrome in Korean adults. Int Arch Occup Environ Health 87(5):501–513
Asgary S, Movahedian A, Keshvari M, Taleghani M, Sahebkar A, Sarrafzadegan N (2017) Serum levels of lead, mercury and cadmium in relation to coronary artery disease in the elderly: a cross-sectional study. Chemosphere. 180:540–544
Zhang Y, Xu C, Fu Z, Shu Y, Zhang J, Lu C, Mo X (2018) Associations between total mercury and methyl mercury exposure and cardiovascular risk factors in US adolescents. Environ Sci Pollut Res 25(7):6265–6272
Park K, Seo E (2017) Toenail mercury and dyslipidemia: interaction with selenium. J Trace Elem Med Biol 39:43–49
Aranda N, Valls RM, Romeu M, Sánchez-Martos V, Albaladejo R, Fernández-Castillejo S, Nogués R, Catalán Ú, Pedret A, Espinel A, Delgado MA (2017) Consumption of seafood and its estimated heavy metals are associated with lipid profile and oxidative lipid damage on healthy adults from a Spanish Mediterranean area: a cross-sectional study. Environ Res 156:644–651
Yoshizawa K, Rimm EB, Morris JS, Spate VL, Hsieh CC, Spiegelman D, Stampfer MJ, Willett WC (2002) Mercury and the risk of coronary heart disease in men. N Engl J Med 347(22):1755–1760
Sponder M, Fritzer-Szekeres M, Marculescu R, Mittlböck M, Uhl M, Köhler-Vallant B, Strametz-Juranek J (2014) Blood and urine levels of heavy metal pollutants in female and male patients with coronary artery disease. Vasc Health Risk Manag 10:311
Wolf MB, Baynes JW (2007) Cadmium and mercury cause an oxidative stress-induced endothelial dysfunction. Biometals. 1;20(1):73–81
Omanwar S, Fahim M (2015) Mercury exposure and endothelial dysfunction: an interplay between nitric oxide and oxidative stress. Int J Toxicol 34(4):300–307
Pant S, Deshmukh A, GuruMurthy GS, Pothineni NV, Watts TE, Romeo F, Mehta JL (2014) Inflammation and atherosclerosis—revisited. J Cardiovasc Pharmacol Ther 19(2):170–178
Lathe R, Sapronova A, Kotelevtsev Y (2014) Atherosclerosis and Alzheimer-diseases with a common cause? Inflammation, oxysterols, vasculature. BMC Geriatr 14(1):36
Jiang J, McCool BA, Parrish AR (2002) Cadmium-and mercury-induced intercellular adhesion molecule-1 expression in immortalized proximal tubule cells: evidence for a role of decreased transforming growth factor-β1. Toxicol Appl Pharmacol 179(1):13–20
Soleo L, Vacca A, Vimercati L, Bruno S, Di Loreto M, Zocchetti C, Di Stefano R, Candilio G, Lasorsa G, Franco G, Foa V (1997) Minimal immunological effects on workers with prolonged low exposure to inorganic mercury. Occup Environ Med 54(6):437–442
Virtanen JK, Rissanen TH, Voutilainen S, Tuomainen TP (2007) Mercury as a risk factor for cardiovascular diseases. J Nutr Biochem 18(2):75–85
Ghizoni H, de Souza V, Straliotto MR, de Bem AF, Farina M, Hort MA (2017) Superoxide anion generation and oxidative stress in methylmercury-induced endothelial toxicity in vitro. Toxicol in Vitro 38:19–26
Samarghandian S, Azimi-Nezhad M, Farkhondeh T, Samini F (2017) Anti-oxidative effects of curcumin on immobilization-induced oxidative stress in rat brain, liver and kidney. Biomed Pharmacother 87:223–229
Samarghandian S, Azimi-Nezhad M, Borji A, Samini M, Farkhondeh T (2017) Protective effects of carnosol against oxidative stress induced brain damage by chronic stress in rats. BMC Complement Altern Med 17(1):249
Samarghandian S, Ohata H, Yamauchi, N, Shibasaki, T (2003) Corticotropin-releasing factor as well as opioid and dopamine are involved in tail-pinch-induced food intake of rats. Neuroscience 116(2):519–524
Lemos NB, Angeli JK, de Oliveira Faria T, Junior RF, Vassallo DV, Padilha AS, Stefanon I (2012) Low mercury concentration produces vasoconstriction, decreases nitric oxide bioavailability and increases oxidative stress in rat conductance artery. PLoS One 7(11):e49005
Hajzadeh MAR, Rajaei Z, Shafiee S, Alavinejhad A, Samarghandian S, Ahmadi, M (2011) Effect of barberry fruit (Berberis Vulgaris) on serum glucose and lipids in streptozotocin-diabetic rats. Pharmacologyonline 1:809–817
Hagele TJ, Mazerik JN, Gregory A, Kaufman B, Magalang U, Kuppusamy ML, Marsh CB, Kuppusamy P, Parinandi NL (2007) Mercury activates vascular endothelial cell phospholipase D through thiols and oxidative stress. Int J Toxicol 26(1):57–69
Ayotte P, Carrier A, Ouellet N, Boiteau V, Abdous B, Sidi EA, Château-Degat ML, Dewailly É (2011) Relation between methylmercury exposure and plasma paraoxonase activity in Inuit adults from Nunavik. Environ Health Perspect 119(8):1077–1083
Wiggers GA, Furieri LB, Briones AM, Avendaño MS, Peçanha FM, Vassallo DV, Salaices M, Alonso MJ (2016) Cerebrovascular endothelial dysfunction induced by mercury exposure at low concentrations. Neurotoxicology. 53:282–289
Moreira EL, de Oliveira J, Dutra MF, Santos DB, Gonçalves CA, Goldfeder EM, de Bem AF, Prediger RD, Aschner M, Farina M (2012) Does methylmercury-induced hypercholesterolemia play a causal role in its neurotoxicity and cardiovascular disease? Toxicol Sci 130(2):373–382
Park K, Seo E (2016) Association between toenail mercury and metabolic syndrome is modified by selenium. Nutrients. 8(7):pii: E424
Aragao WA, da Costa NM, Fagundes NC, Silva MC, Alves-Junior SM, Pinheiro JJ, Amado LL, Crespo-López ME, Maia CS, Lima RR (2017) Chronic exposure to inorganic mercury induces biochemical and morphological changes in the salivary glands of rats. Metallomics. 9(9):1271–1278
Lim S, Chung HU, Paek D (2010) Low dose mercury and heart rate variability among community residents nearby to an industrial complex in Korea. Neurotoxicology. 1;31(1):10–16
Strandberg U, Palviainen M, Eronen A, Piirainen S, Laurén A, Akkanen J, Kankaala P (2016) Spatial variability of mercury and polyunsaturated fatty acids in the European perch (Perca fluviatilis)—implications for risk-benefit analyses of fish consumption. Environ Pollut 219:305–314
Rodriguez M, Diaz A, Blasco M, Mantilla T, Orera ML, Orera-Peña L (2017) Cardiovascular risk in patients with atherogenic dyslipidemia. Perspective of primary care physicians. Avanza study. Atherosclerosis. 263:e281
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
All the authors declared no conflict of interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Farkhondeh, T., Afshari, R., Mehrpour, O. et al. Mercury and Atherosclerosis: Cell Biology, Pathophysiology, and Epidemiological Studies. Biol Trace Elem Res 196, 27–36 (2020). https://doi.org/10.1007/s12011-019-01899-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-019-01899-w