Abstract
The aim of this study was to investigate the association of trace element and toxic metal concentrations in blood and the outcome of in vitro fertilization (IVF). The study included 104 consecutive patients that underwent assisted reproductive technology (ART) procedures. The following parameters were determined: cadmium (Cd), mercury (Hg), arsenic (As), and lead (Pb); and copper (Cu), zinc (Zn), selenium (Se), and magnesium (Mg). Serum samples were obtained before commencing stimulation. Patients with smoking habit had significantly higher Pb concentrations (P = 0.022), as well as higher concentrations of As and Hg but not significantly. All subjects were divided into groups of pregnant and nonpregnant patients. Pregnant patients had lower mean values of Mg (P = 0.009), As (P < 0.05), and Pb (P = 0.034), compared to nonpregnant, and a significant correlation between pregnancy outcome and concentrations of Mg, Cd, and Pb was found. Women who had had delivered had lower Mg (P = 0.009) and Cd (P = 0.014) concentrations. There was a significant correlation of the negative outcome of IVF procedure with higher concentrations of Pb (P = 0.046) and Cd (P = 0.012). In conclusion, our results suggest that there is a difference in Mg, Pb, and Cd concentrations between pregnant and nonpregnant women. There was no association between toxic metals and number and quality of oocytes and embryos, while there was with fertilization rate. Concerning trace elements, we did not find the correlation of trace elements with oocyte number and quality, nor with a number of fertilized oocytes, except for Cu. Patients who were pregnant had lower concentrations of Mg.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Although pregnancy rates in in vitro fertilization (IVF) cycles are increasing, different factors that may influence the success of IVF procedures are still being investigated. Infertility has a multifactorial etiology with many genetic and environmental factors included [1]. Environmental and lifestyle factors are of particular interest, because unlike genetic causes, we can influence them in terms of treatment or prevention, which is significant in the treatment of infertility [1].
Toxic metals are widespread in the human environment, food, water, air, cigarettes, and alcoholic beverages and are often taken in trace concentrations over time through gastrointestinal system, respiratory system, and skin [2]. The studies suggest the toxic effects on the reproductive function, among other organic systems, acting directly on specific reproductive organs or indirectly to the neuroendocrine system [3]. Exposure to toxic metals can affect hormonal changes, ovulation, maturation of the oocyte, and the reduction or loss of fertility, and hence the achievement of pregnancy in women [4, 5]. Also, some of them, such as cadmium and lead, increase the rate of spontaneous abortion and affect fetal development [6]. Reproductive toxicity resulting from long-term exposure to in trace concentrations of toxic metals can have an impact on the success of IVF procedures [7]. In addition, studies demonstrated that deficiency of trace elements may have effect on reproductive health too [8]. Zinc and selenium play a role in sexual development, ovulation, and menstrual cycle [1]. There is an association between these nutrients and their derivatives and spontaneous abortions and congenital malformations [1]. Some studies found also a lack of Cu, Se, and Zn in IVF patients [1, 9, 10]. There are not many studies on serum Mg levels in IVF patients, but in one, it was suggested that there is a relationship between magnesium and sex steroid hormones [11]. Concentrations of toxic metals and trace elements are usually measured in the blood, urine, and/or follicular fluid in IVF studies, and the latest two studies used hair for assessment [12, 13]. In the study that made a comparison between blood and follicular fluid element concentrations, in IVF patients, the average blood concentrations were similar to those in follicular fluid of large follicles [9]; therefore, we decided to determine concentrations of heavy metals and trace elements in the blood only.
The aim of our study was to investigate the association of toxic metals and trace metals, in blood and IVF outcome, as well as its influence on the number of received oocytes, number of mature oocytes, embryo quality, fertilization, and pregnancy.
Materials and Methods
Study Subjects
The study included 104 consecutive female patients that underwent ART procedures as infertility treatment at the Clinic of Gynecology and Obstetrics, Clinical Center of Serbia, Belgrade. All patients agreed to participate in the study and signed an informed consent for all the undertaken procedures. The study was approved by The Ethics Committee of the Faculty of Medicine, University of Belgrade.
Participant recruitment and clinical protocols were previously described in detail [14]. Briefly, including criteria for patients were as follows: age 18–40 years; body mass index (BMI) from 18 to 30 kg/m2; regular menstrual cycles from 25 to 32 days; and without any medical disease, endocrinology disease, hydrosalpinx, or endometriosis stages III and IV. Infertility cause was categorized as male, tubal, ovarian, unknown, or combined. For all patients, age, BMI, years of treating infertility, smoking status (smoker/nonsmoker), and the cause of infertility were determined before commencing IVF procedure. In respect of supplements, all women were advised to use folic acid 400 μg per day, from 1 to 3 months before procedure. We divided patients into two groups, based on the outcome of IVF, those who failed to achieve pregnancy (nonpregnant) women and those who achieved pregnancy (pregnant) after IVF. Depending on age, ovarian reserve, and previous IVF cycle, patients were submitted to different protocols: short gonadotropin-releasing hormone (GnRH) antagonist protocol with contraceptive pretreatment (n = 19), or without it (n = 60), and long GnRH agonist protocol (n = 25). Methods of insemination were IVF, intracytoplasmic sperm injection (ICSI), or a combined method (IVF/ICSI). In assessing the quality of embryos, the Istanbul consensus clinical embryologist criteria were used as the reference frame [15]. Embryo transfer was performed on day 2 or 3 after the oocyte retrieval. For luteal support phase, patients received intramuscular progesterone, until the 12th week of gestation in cases of pregnancies. Pregnancy was diagnosed by positive serum β-hCG (> 25 MIU/mL) 14 days after the embryo transfer. Clinical pregnancies were confirmed by transvaginal ultrasound findings: gestational sac with a vital embryo and the 6-week gestation. Finally, we registered whether the examined women managed to maintain pregnancy and have a term delivery of a healthy child or they had a miscarriage.
Sample Collection and Analysis
Serum and whole blood specimens were obtained from each patient between the second and fourth day of the menstrual cycle, before commencing of stimulation, when the basal hormonal status was determined. The laboratory was blinded to clinical data and IVF procedure. After preparing a whole blood specimen, trace elements copper (Cu), zinc (Zn), selenium (Se), and magnesium (Mg) as well as toxic metals cadmium (Cd), mercury (Hg), arsenic (As), and lead (Pb) were determined. Blood samples for determining the content of trace elements and toxic metals (about 1 mL) were weighed in a Teflon vessel of microwave digestion apparatus (START D, Milestone, Sorisole, Italy) and then 8 mL conc. HNO3 for digestion mineralized by the microwave oven mineralization process. The conditions for the mineralization of the sample tested were set by adjusting the parameters of the digestion program, which was to achieve a temperature of 170 °C for 10 min, which was maintained for the next 10 min, then maintaining the ventilation for 15 min. The samples prepared in the manner described above were quantitatively transferred to normal vessels, and then in these solutions, the concentration of metals was determined by the inductively coupled plasma mass spectrometry (ICP-MS) (iCap Q mass spectrometer, Thermo Scientific, Bremen, Germany).
All methods for determining the metal concentration are validated, which covered linearity for the given range of concentrations, LOD and LOQ derivation, coefficient of variation, accuracy, and recovery. For the purpose of testing the method of determining metals by ICP-MS, the certified reference material (Seronorm™ Trace Elements Whole Blood, Serro AS, Billingstad, Norway) was used. Recovery rates from certified reference material analysis were 100.5, 99.6, 99.9, 98.7, 101.4, 100.2, 101.8, and 98.9% for Mg, Cu, Zn, As, Se, Cd, Hg, and Pb, respectively.
Statistical Analysis
Results were presented as arithmetic mean ± standard deviation for variables with normal distribution and as median and interquartile range for variables whose distribution is not normal. Testing of distribution was carried out by Kolmogorov-Smirnov analysis. Categorical variables are presented as relative or absolute frequency. Analysis of categorical values was performed using the chi-square test. Comparison of the mean values of independent groups of data was performed by ANOVA analysis. For parameters without normal distribution, test of significance between groups was performed using the Kruskal-Wallis test. The association of selected variables with outcome was assessed with the univariate and multivariate logistics regression analyses. A significance of 0.05 was required for a variable to be included into the multivariate model, whereas 0.1 was the cutoff value for exclusion. Odd ratios with the corresponding 95% confidence intervals were estimated. The sensitivity and specificity of identified parameters Mg and Pb blood concentrations for outcome prediction were evaluated with receiver operating characteristic curves. All analysis were performed using the Statistical Package for the Social Sciences (SPSS) 22, and differences were considered statistically significant at probability level less than 0.05.
Results
Demographic variables of 104 patients included in the study, as well as causes and duration of infertility, stimulation protocols, the method of insemination, and outcome of IVF, are shown in Table 1. Significantly, more patients who achieved pregnancy were less than 35 years old (P = 0.004), and most pregnant patients had had a delivery of a healthy baby (P = 0.000). Among pregnant patients, the most common cause of infertility was the male and unknown factor, and the ovarian cause was least presented (P = 0.031). Mean values with standard deviations and minimum and maximum values of clinical and cycle characteristics were shown in Table 2. Pregnant patients had significantly more oocytes retrieved (P = 0.026), as well as more fertilized oocytes (P = 0.032). Patients who achieved pregnancy had shorter duration of infertility (P = 0.050).
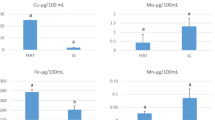
Patients who had positive outcome of IVF procedure had significantly lower Mg concentrations (94.4%), as well as lower concentrations of Cd (97.6%) and Pb (80.6%). We also compared mean concentrations of toxic metals and trace elements to IVF outcome (Table 3). In patients who achieved pregnancy, we found significantly lower mean concentrations of Mg (P = 0.009), as well as As (P = 0.050) and Pb (P = 0.034). Mean concentrations of Cd were higher in patients who failed to achieve pregnancy.
In Table 4, correlation of toxic metals and trace elements with cycle characteristics are shown. Analyzing interval of values and characteristics of IVF outcome, we found direct significant Pearson’s correlation of higher Cu concentrations and higher GT dose (P = 0.039), but also of lower Pb concentrations and higher GT dose (P = 0.008). There was a significant correlation between higher Cu concentrations and a higher number of fertilized oocytes (P = 0.041). Spearman correlation of toxic metal and trace element concentrations with IVF outcome is shown in Table 5, and there was a significant correlation between the positive outcome of IVF procedure and the lower concentrations of Mg (P = 0.010). In correlation with negative outcome were higher concentrations of Pb (P = 0.046) and Cd (P = 0.012). When embryo quality was correlated with the outcome of IVF, we found a significant correlation of pregnancy and better quality of embryos (P = 0.013), as well as with higher gonadotropin dose (P = 0.045).
Concerning the outcome of pregnancy, patients who had delivery had the correlation with lower Mg (P = 0.009) and Cd (P = 0.029) concentrations, while there was a significant correlation between miscarriages and higher concentrations of Pb (P = 0.029) (Table 6).
In univariate and multivariate logistic regression analysis, all the variables that were examined were included in the regression model. The parameters such as BMI, duration of infertility, insemination method, and fertilization rate did not show significant predictive power in the univariate logistic regression analysis in our investigation. In our study, patients older than 35 years indicate a 69% decrease of the positive outcome (P = 0.005). Unknown cause of infertility was a positive predictor for pregnancy (P = 0.037), while ovarian cause indicates 87.7% significantly decrease in the chance for a positive outcome (P = 0.010). Higher gonadotropin dose (P = 0.041), as well as higher number of fertilized oocytes (P = 0.040), were positive predictors of pregnancy. Lower embryo quality indicates a 79.4% decrease in the chance of positive outcome (P = 0.021). Higher Mg concentrations (P = 0.020), as well as higher Pb concentrations (P = 0.050), are associated with an 83.9 and 62.7% increased chance of negative outcome. Higher Cd concentrations indicate an 89.4% decreased chance for the positive outcome (P = 0.035) (Table 7). The results of multivariate regression analysis showed a decrease in chances of pregnancy in patients older than 35 years for 83.1% (P = 0.007). Higher gonadotropin dose is a predictor of positive outcome (P = 0.015). There was a decrease in chances of pregnancy in patients with lower embryo quality for 86% (P = 0.049). Increased Mg concentrations (P = 0.032), as well as Pb concentrations (P = 0.026), were associated with 88.3 and 77.6% chance for a negative outcome (Table 7).
Receiver operating characteristic (ROC) curves were constructed for Mg and area under the curve (AUC) for the pregnancy. This relationship was confirmed (AUC of 0.651, P = 0.015). The optimal cutoff point for Mg was found to be 3.3 mg/dL resulting in a sensitivity of 94% and a specificity of 30% for positive status (Fig. 1). The AUC was 0.632 for the pregnancy. The cutoff point for Pb was 0.96 μg/dL resulting in a sensitivity of 92% and a specificity of 40% for pregnancy (Fig. 2) (P = 0.034) Using the Pb (μg/dL) cutoff value from the initial ROC curve, lower values Pb predicted pregnancy with a specificity of 92% and sensitivity of 40% (P = 0.034).
Discussion
Humans are exposed to toxic metals in trace concentrations through dietary sources or airborne pollution [16], and when presented in the blood or follicular fluid, they can have an effect on the reproductive health of women and consequently on the outcome of IVF procedures [2]. Although the data are controversial and for certain elements scarce, it is known that fertility is impaired in women professionally exposed to Hg, Cd, and Pb [17,18,19]. However, data from studies that examined the influence of exposure to those toxic metals in trace concentrations on reproductive function in women are not consistent [20,21,22,23].
In our study we wanted to find out if there was any effect of toxic metals and trace elements on any step in IVF procedure including on the outcome itself. We did not find correlation of toxic metal blood concentration either with oocyte number or with number of mature (MII) and fertilized oocytes. The finding is consistent with the study of Bloom et al. [24] in which there was no correlation of heavy metals with mature oocytes, while in the previous study [25], they found lower fertilization rate when Pb in follicular fluid was increased. In the study of Al-Selah et al. [26], concentrations of Pb, Cd, and Hg were determined in blood and follicular fluid in 619 women underwent IVF procedure. They also found a reverse proportional relationship between Pb concentrations and fertilization rate. But, interestingly, they found a positive correlation between Cd in follicular fluid and fertilization rate, which is hard to interpret. It could be supported by Henson and Chedrese [27] suggestion that Cd might have paradoxical effect on steroidogenesis, acting on ovarian and reproductive tract morphology, with extremely low dosages reported to stimulate ovarian luteal progesterone biosynthesis and high dosages inhibiting it.
Our data suggest that pregnant patients had lower concentrations of As and Pb, while nonpregnant patients had higher concentrations of Pb and Cd. Cd levels were lower in patients who had had delivery. Aforementioned toxic metals were predictors for pregnancy. Bloom et al. [24] found in their multivariable model 35 and 33% lower probability of clinical and biochemical pregnancies in correlation with Hg blood concentration, while there was no correlation with long-term exposure to Pb and Cd in female population. When they analyzed Cd concentrations in blood, they found 94 and 82% lower probability for clinical and biochemical pregnancies. Nonpregnant women had higher mean values of Hg and higher concentrations of Cd in blood; similar was found in our study too, while for Pb concentrations, results were reversed. Tolunay and et al. [2] determined concentrations of Cd, Pb, Hg, and As as well as of Cu, Zn, and Fe in blood and follicular fluid and they had two groups of patients, those with clinical pregnancies and those with miscarriages and biochemical pregnancies. Statistically significant negative correlation between concentrations of Pb in blood and MII oocytes, implantation rates, and clinical pregnancies was found. The result concerning pregnancies corresponds to our finding. Longitudinal study of infertility and environment (LIFE) did not found correlation between Hg levels in blood and biochemical pregnancies in 500 women [28], neither between concentrations of Pb and pregnancy rate, as well as in some other studies [7, 29]. The effect of As on female fertility has not been sufficiently investigated and is not clear. Pb and Cd have been shown to increase free radical species [30], demonstrate physiological estrogenic effect [31, 32], and alter progesterone synthesis [33] which could have deleterious effect with respect to pregnancy achievement. We should also mention that albeit higher Cd and Pb concentrations were found in nonpregnant patients, there was decreased chance for pregnancy in patients older than 35 years, longer duration of infertility, and ovarian cause of infertility that were more represented in nonpregnant group, indicating that many factors can influence the outcome of IVF procedure.
In our study, we did not find the correlation of either trace element concentrations with oocyte number nor with a number of fertilized oocytes, except higher concentrations of Cu, were in direct correlation with a higher number of fertilized oocytes. Similar finding had one recent study by Ingle et al. [29] that examined correlation between trace elements in follicular fluid and urine and IVF outcome in 58 patients. In that study, mean number of retrieved oocytes was in correlation with higher concentrations of Cu in urine. Mg in follicular fluid was in negative correlation with MII oocytes, while Mg in urine was in positive correlation.
While in Ingle et al. [29] study Zn in follicular fluid was in reversed correlation with mean fertilization rate, and there was no correlation with implantation rate, pregnancy, and life birth rate. As an interesting finding, we detected lower concentrations of Mg in blood of pregnant patients, while mean concentrations of Cu were slightly higher and mean concentrations of Zn and Se were slightly lower in this group. Patients that had had delivered had lower concentrations of Mg and Cu in blood compared to those with miscarriages. Also both in univariate and multivariate regression analysis, predictors for positive outcome were lower Mg concentrations. Mg binds to cell membranes and stabilizes them, as well as proteins and nucleic acids [34]; also, it protects lipoproteins from reactive oxygen species [35], but there are no data in literature of its association with IVF outcome. In the recent study [13], it was shown that women who conceived after IVF procedure had lower levels of trace elements including Mg, compared to women with natural pregnancy. All that correspond partially to our results, but in our study, lower concentrations of Mg did not affect the pregnancy rate neither the delivery rate. Grossi et al. found the slight decline of total serum magnesium levels in women during controlled ovarian stimulation [36]. These findings point out that screening of infertile patients for magnesium levels before the start of IVF procedure and supplementation of Mg would be beneficial as probably the dietary intake is lower and requirements are higher during ovarian stimulation and pregnancy. In the study of Bloom et al. [37], Mg concentrations were higher in women with positive pregnancy test compared with those who had negative test results, while Zn concentrations were lower in women with a positive pregnancy test, but not significantly. Zinc has recently been recognized as an important factor in the completion of meiosis and oocyte activation in vitro [38,39,40,41], as well as in follicular rupture and completion of meiosis in vivo [41]. Tolunay et al. [2] determined concentrations of Cd, Pb, Hg, and As as well as of Cu, Zn, and Fe in blood and follicular fluid, and they found a lower pregnancy rate in patients with higher concentrations of Cu in follicular fluid. In our study, patients with higher concentrations of Cu had more fertilized oocytes and more pregnancies, but those who had had delivered had lower Cu concentrations compared with women that had miscarriage. Cu is involved in normal reproduction and it is necessary for different metabolic processes and enzymatic reactions [42], but possible negative effect on follicular maturation and embryo development was suggested when the exposure to Cu is chronic [2]. Since our results show Cu affecting positively fertility, more studies are needed to set the cutoffs. In our study, we did not find any significant difference in Se concentrations between pregnant and nonpregnant women. Data on Se in female fertility are scarce. It was reported that IVF patients had lower levels of Se than the control group [43, 44] and lower concentrations of Se were reported in women who experienced miscarriages compared to women with clinical pregnancies [45, 46].
Limitations and Strengths
Although the results are interesting, it would be more relevant if we had a larger number of patients. Also, our patients used folic acid supplementation, but they did not report use of other supplements and we also did not know their eating habits. Besides, reproduction requires a couple, and our analysis included the female partner only. It is known that toxic metals and trace elements impact semen parameters [47,48,49], so the future assessment will require incorporation of toxic metal and trace element concentrations from male partners too. This research is significant because there are time-specific vulnerable windows of human development when environmental factors, even small exposures, can alter developmental programming signals and trigger adverse health consequences that can manifest across the lifespan of individuals and generations [50,51,52,53]. There is a need for population-based, multidisciplinary research and also implementation of prevention.
Conclusion
Our results suggest that there is a difference in some toxic metal and trace element concentrations between pregnant and nonpregnant women. There was no association between toxic metals and the number and quality of oocytes and embryos. Patients who were pregnant had lower concentrations of As and Pb, while nonpregnant women had higher concentrations of Pb and Cd, indicating a possible negative impact of toxic metals on IVF outcome. Concerning trace elements, we did not find the correlation of trace elements with oocyte number and quality, nor with a number of fertilized oocytes, except higher concentrations of Cu, was in direct correlation with a higher number of fertilized oocytes. Patients who were pregnant had lower concentrations of Mg. Larger studies in IVF population are required in order to find the association between the outcomes of the procedure and trace elements and toxic metals.
Abbreviations
- IVF:
-
In vitro fertilization
- ART:
-
Assisted reproductive techniques
- ICSI:
-
Intracytoplasmic sperm injection
- BMI:
-
Body mass index
- GnRH:
-
Gonadotropin-releasing hormone
- GT:
-
Gonadotropin
References
Ebisch IMW, Thomas CMG, Peters WHM, Braat DDM, Steegers-Theunissen RPM (2007) The importance of folate, zinc and antioxidants in the pathogenesis and prevention of subfertility. Hum Reprod Update 13(2):163–174
Tolunay HE, Şükür YE, Ozkavukcu S, Seval MM, Ateş C, Türksoy VA, Ecemiş T, Atabekoğlu CS, Özmen B, Berker B, Sönmezer M (2016) Heavy metal and trace element concentrations in blood and follicular fluid affect ART outcome. Eur J Obstet Gynecol Reprod Biol 198:73–77
Wong EW, Cheng CY (2011) Impact of environmental toxicants on male reproductive dysfunction. Trends Pharm Sci 32(5):290–299
Bloom MS, Parson PJ, Steuerwald JA et al (2010) Toxic trace metals and human oocytes during in vitro fertilization (IVF). Reprod Toxicol 23(3):298–305
Choi SM, Yoo SD, Lee BM (2004) Toxicological characteristics of endocrine-disrupting chemicals: developmental toxicity, carcinogenicity, and mutagenicity. J Toxicol Environ Health 7:1–24
Bellinger DC (2005) Teratogen update: lead and pregnancy. Birth Defects Res A Clin Mol Teratol 73:409–420
Bloom MS, Parsons PJ, Kim D, Steuerwald AJ, Vaccari S, Cheng G, Fujimoto VY (2011) Toxic trace metals and embryo quality indicators during in vitro fertilization (IVF). Reprod Toxicol 31:164–170
Kontic-Vucinic O, Sulovic N, Radunovic N (2005) Micronutrients in women’s reproductive health: II. Minerals and trace elements. Int J fertile Womens Med 51:116–124
Silberstein T, Saphier O, Paz-Tal O, Gonzalez L, Keefe DL, Trimarchi JR (2009) Trace element concentrations in follicular fluid of small follicles differ from those in blood serum, and may represent long-term exposure. Fertil Steril 91:1771–1774
Favier AE (1992) The role of zinc in reproduction. Hormonal mechanism. Biol Trace Elem Res 32:363–382
O'Shaughnessy A, Muneyyirci-Delale O, Nacharaju VL, Dalloul M, Altura BM, Altura BT (2001) Circulating divalent cations in asymptomatic ovarian hyperstimulation and fertilization patients. Gynecol Obstet Investig 52:237–242
Garcia-Fortea P, Cohen-Corcia I, Cordoba-Dona JA, Reche-Rosado A, Gonzales- Mesa E (2018) Toxic elements in hair and in vitro fertilization outcomes: a prospective cohort study. Reprod Toxicol 77:43–52
Skalny AV, Tinkov AA, Voronina I, Terekhina O, Skalnaya MG, Kovas Y (2018) Hair trace element and electrolyte content in women with natural and in vitro fertilization-induced pregnancy. Biol Trace Elem Res 181:1–9
Tulic L, Vidakovic S, Tulic I, Curcic M, Stojnic J, Jeremic K (2017) Oxidative stress markers in GnRH agonist and antagonist protocols in IVF. J Med Biochem 36:1–8
Alpha Scientists in Reproductive Medicine and ESHRE Special Interest Group of embryology (2011) The Istanbul Consensus workshop of embryo assessment: proceedings of an expert meeting. Hum Reprod:1–14
U.S. Centers of Disease Controls and Prevention (CDC) (2010) Fourth national report on human exposure to environmental chemicals—updated tables. Atlanta, GA
Macaluso M, Wright-Schnapp TJ, Chandra A, Johnson R, Satterwhite CL, Pulver A et al (2010) A public health focus on infertility prevention, detection, and management. Fertil Steril 93:16.e1–16.10
Vallombrosa Meeting Group (2006) Vallombrosa consensus statement on environmental contaminants and human fertility compromise. Semin Reprod Med 24:178–189
CDC (2012) Fourth national report on human exposure to environmental chemicals—updated tables, February 2012. Atlanta, GA: U.S. Centers for Disease Control and Prevention
Figà-Talamanca I (2006) Occupational risk factors and reproductive health of women. Occup Med 56:521–531
Mendola P, Messer LC, Rappazzo K (2008) Science linking environmental contaminant exposures with fertility and reproductive health impacts in the adult female. Fertil Steril 89:e81–e94
MacIntosh DL, Spengler JD, Ozkaynak H, Tsai LH, Barry Ryan P (1996) Dietary exposures to selected metals and pesticides. Environ Health Perspect 104:202–209
Satarug S, Garrett SH, Sens MA, Sens DA (2010) Cadmium, environmental exposure, and health outcomes. Environ Health Perspect 118:182–190
Bloom M, Fujimoto M, Steuerwalda A et al (2010) Background exposure to toxic metals in women adversely influences pregnancy during in vitro fertilization (IVF). Reprod Toxicol 34:471–481
Bloom MS, Parson PJ, Steuerwald JA et al (2010) Toxic trace metals and human oocytes during in vitro fertilization (IVF). Reprod Toxicol 23(3):298–305
Al-Saleh I, Coskun S, Mashhour A, Shinwari N, El-Doush I, Billedo G et al (2008) Exposure to heavy metals (lead, cadmium and mercury) and its effect on the outcome of in vitro fertilization treatment. Int J Hyg Environ Health 211:560–579
Henson MC, Chedrese PJ (2004) Endocrine disruption by cadmium, a common environmental toxicant with paradoxical effects on reproduction. Exp Biol Med 229:383–392
Buck Louis GM, Sundaram R, Schisterman EF, Sweeney AM, Lynch CD, Gore-Langton RE, Chen Z, Kim S, Caldwell KL, Barr DB (2012) Heavy metals and couple fecundity, the LIFE Study. Chemosphere 87:1201–1207
Ingle M, Bloom M, Parsons P et al (2017) Associations between IVF outcomes and essential trace elements measured in follicular fluid and urine: a pilot study. J Asist Reprod Genet 34:253–261
Ercal N, Gurer-Orhan H, Aykin-Burns N (2001) Toxic metals and oxidative stress. Part I: Mechanisms involved in metal-induced oxidative damage. Curr Top Med Chem 1:529–539
Dyer CA (2007) Heavy metals as endocrine-disrupting chemicals. In: Gore AC (ed) Endocrine-disrupting chemicals: from basic research to clinical practice. Humana Press, Totowa, NJ, pp 111–133
Iavicoli I, Fontana L, Bergamaschi A (2009) The effects of metals as endocrine disruptors. J Toxicol Environ Health B Crit Rev 12:206–223
Henson MC, Chedrese PJ (2004) Endocrine disruption by cadmium, a common environmental toxicant with paradoxical effects on reproduction. Exp Biol Med 229:383–392
Bara M, Guiet-Bara A, Durlach J (1990) Analysis of magnesium membraneous effects: binding and screening. Magnes Res 29:4121–4128
Rayssiguier Y, Gueux E, Bussiere L, Durlach J, Mazur A (1993) Dietary magnesium affects susceptibility of lipoproteins and tissues to peroxidation in rats. J Am Coll Nutr 12:133–137
Grossi E, Castiglioni S, Moscheni C, Antonazzo P, Cetin I, Savasi VM (2017) Serum magnesium and calcium levels in infertile women during a cycle of reproductive assistance. Magnes Res 30:35–41
Bloom MS, Buck Louis GM, Sundaram R, Kostyniak PJ, Jain J (2011) Associations between blood metals and fecundity among women residing in New York State. Reprod Toxicol 31(2):158–163
Bernhardt ML, Kim AM, O'Halloran TV, Woodruff TK (2011) Zinc requirement during meiosis-meiosis II transition in mouse oocytes is independent of the MOS-MAPK pathway. Biol Reprod 84:526–536
Kim AM, Vogt S, O'Halloran TV, Woodruff TK (2010) Zinc availability regulates exit from meiosis in maturing mammalian oocytes. Nat Chem Biol 6:674–681
Kong BY, Bernhardt ML, Kim AM, O'Halloran TV, Woodruff TK (2012) Zinc maintains prophase I arrest in mouse oocytes through regulation of the MOSMAPK pathway. Biol Reprod 87(1):11 1–12
Tian X, Diaz FJ (2012) Zinc depletion causes multiple defects in ovarian function during the periovulatory period in mice. Endocrin 153:873–886
Michaluk A, Kochman K (2007) Involvement of copper in female reproduction. Reprod Biol 7:193–205
Paszkowski T, Traub AI, Robinson SY, McMaster D (1995) Selenium dependent glutathione peroxidase activity in human follicular fluid. Clin Chim Acta 236:173–180
Ozkaya MO, Naziroglu M, Barak C, Berkkanoglu M (2010) Effects of multivitamin/mineral supplementation on trace element levels in serum and follicular fluid of women undergoing in vitro fertilization (IVF). Biol Trace Elem Res 139:1–9
Barrington JW, Lindsay P, James D, Smith S, Roberts A (1996) Selenium deficiency and miscarriage: a possible link? Br J Obstet Gynaecol 103:130–132
Kocak I, Aksoy E, Ustun C (1999) Recurrent spontaneous abortion and selenium deficiency. Int J Gynaecol Obstet 65:79–80
Kiziler AR, Aydemir B, Onaran I, Alici B, Ozkara H, Gulyasar T, Akyolcu MC (2007) High levels of cadmium and lead in seminal fluid and blood of smoking men are associated with high oxidative stress and damage in infertile subjects. Biol Trace Elem Res 120:82–91
Camejo MI, Abdala L, Vivas-Acevedo G, Lozano-Hernández R, Angeli-Greaves M, Greaves ED (2011) Selenium, copper and zinc in seminal plasma of men with varicocele, relationship with seminal parameters. Biol Trace Elem Res 143:1247–1254
Sun J, Yu G, Zhang Y, Liu X, Du C, Wang L, Li Z, Wang C (2017) Heavy metal level in human semen with different fertility: a meta-analysis. Biol Trace Elem Res 176:27–36
Grandjean P, Bellinger D, Bergman A, Cordier S, Davey-Smith G, Eskenazi B, Gee D, Gray K, Hanson M, van den Hazel P, Heindel JJ, Heinzow B, Hertz-Picciotto I, Hu H, Huang TT, Jensen TK, Landrigan PJ, McMillen I, Murata K, Ritz B, Schoeters G, Skakkebaek NE, Skerfving S, Weihe P (2008) The faroes statement: human health effects of developmental exposure to chemicals in our environment. Basic Clin Pharmacol Toxicol 102(2):73–75
Woodruff TJ, Janssen SJ, Guillette LJ, Giudice LC (2010) Environmental impacts on reproductive health and fertility. Cambridge University Press, Cambridge
Newbold R, Heindel J (2010) Developmental exposures and implications for early and latent disease. In: Woodruff TJ, Janssen SJ, Guillette LJ Jr, Giudice LC (eds) Environmental impacts on reproductive health and fertility. Cambridge University Press, New York
American Academy of Pediatrics Council on Environmental Health (2012) Pediatric environmental health. (3rd ed.), American Academy of Pediatrics, Elk Grove Village, IL
Acknowledgments
The study is part of project no. III46009, Ministry of Science, Education and Technological Development of R. Serbia.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Conflict of Interest
The authors declare that they have no conflict of interests.
Rights and permissions
About this article
Cite this article
Tulić, L., Vidaković, S., Tulić, I. et al. Toxic Metal and Trace Element Concentrations in Blood and Outcome of In Vitro Fertilization in Women. Biol Trace Elem Res 188, 284–294 (2019). https://doi.org/10.1007/s12011-018-1421-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-018-1421-z