Abstract
Muscle strength, an independent predictor of metabolic disorders, disability, and mortality, reduces gradually with advancing age. Little is known about the influence of nutritional intervention on muscle strength in middle-aged. The aim of the present study is to examine whether magnesium could improve body composition and muscle strength in middle-aged overweight women. In this double-blinded, placebo-controlled, randomized trial, a total of 74 healthy middle-aged overweight women (25 ≤ BMI ≤ 30 kg/m2) received either 250 mg magnesium in the form of magnesium oxide or placebo daily for 8 weeks. Body composition was assessed using Bioelectrical Impedance Analysis (BIA). Handgrip strength and knee extension strength were measured with isometric dynamometry. Functional mobility was assessed using Time Get Up and Go Test (TGUG). A significant increase in mean lean body mass was observed (P = 0.05) accompanied with a significant decrease in fat mass (P = 0.02) solely in the magnesium group at the end of 8 weeks compared to baseline values but the changes did not reach significant as compared to placebo group. Handgrip strength and TGUG improved in the magnesium group compared to baseline but they were not significant compared to placebo. There were no significant differences in increasing knee extension strength in the magnesium group as compared with placebo. Baseline values of serum magnesium and muscle strength of participants did not indicate any influences on response to magnesium supplementation. Our findings indicate that magnesium as magnesium oxide, 250 mg/day, for 8 weeks do not lead to a significant greater gain in muscle strength and function compared to placebo.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Muscle strength is considered as an indicator of functional and nutritional status, which can predict osteoporotic fracture [1, 2], disability, and all-cause mortality in middle-aged and elderly [3–5]. Muscle strength has also been found to be a strong predictor of metabolic syndrome, independent of age, body size, insulin resistance, and abdominal fat [6–8], suggesting that interventions designed to prevent metabolic disorders should develop to improve muscle fitness in addition to reducing fatness.
Aging is generally accompanied by the gradual loss of muscle strength and muscle mass, leading to functional impairments, disability, and metabolic consequences [9, 10], which exert a significant burden on both individuals and the community. This age-related loss of muscle strength seems to be a greater public health concern for women because of an earlier development of muscle weakness, living longer and exhibiting higher rates of disabilities [11]. Declining muscle strength in some degree is as a part of normal aging, but it can be delayed or accelerated by a variety of factors including nutritional status, overweight, and physical activity [10]. Different rate of the reduction in muscle strength across the population also confirmed the important influences of modifiable behavioral factors on muscle strength [12]. Existing studies, which are mostly observational, indicate the potential effects of dietary patterns [13], quality of diet, and intakes of protein, vitamin D, and antioxidant nutrients on muscle strength [12].
Magnesium plays essential roles in various physiological processes involve in muscle function such as energy production, transmembrane transport, electrolyte balance, and muscle contraction and relaxation [14]. Despite the several dietary sources of magnesium, subclinical or marginal magnesium deficiency is prevalent worldwide [15]. Inadequate magnesium intake was related to decrease work economy during submaximal exercise; increased oxygen utilization to maintain ATP production [16]. Moreover, serum magnesium was positively associated with muscle strength in older persons, independent of disease and metabolic disturbances [17], but it is not clear whether magnesium supplementation can improve muscle strength. Therefore, the present study aimed to investigate the effect of magnesium supplementation on body composition, muscle strength, and functional mobility in apparently healthy middle-aged overweight women.
Methods
Participants
In this study, participants were recruited from staff at the Tehran University of Medical Sciences (TUMS) through advertisements. Volunteers could be included in the study if they were female, aged 40–55 years, had a body mass index 25 ≤ BMI ≤ 30 kg/m2, were not suffering from any chronic diseases, including diabetes mellitus or other endocrine disorder, liver and gastrointestinal disorder, hypertension, cardiovascular disease, and kidney disease, were not a smoker, and were not taking magnesium-containing medications, laxative, or hormone products.
Study Design
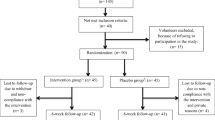
The study was a randomized, double-blinded and placebo-controlled trial with 8-week duration. The 74 volunteers met the criteria which were randomly allocated into two groups: magnesium group (n = 37) received one tablet of magnesium (250 mg magnesium oxide, equivalent to 150 mg of elemental magnesium) and placebo group received one placebo tablet daily with lunch. The placebo was made of corn starch, lactose, and stearic acid and had an identical appearance to the magnesium oxide. Participants were asked not to change their usual dietary intake and physical activity during the study. All participants were informed of the objectives and protocol of the study; written informed consent was obtained from them before their participation in the study. The study protocol was approved by the ethics committee of the School of Public Health of Tehran University of Medical Sciences, and was registered at the Iranian Registry of Clinical Trials (IRCT) as ID No. IRTC138805312365N1.
Randomization and Compliance
Participants were allocated randomly using a random number table; for this, a study leader who was not involved in study protocol created the randomization list assigning participants to the magnesium or the placebo group. Twenty-eight magnesium and placebo tablets were placed into 148 unlabelled identical containers; two containers per each subject were provided for treatment during two 4-week periods. The study leader labeled these containers with participant numbers using the randomization list. Compliance with treatment was assessed by pill counts. For this, all participants returned their two containers at the end of the each 4-week interval.
Serum Magnesium, Anthropometric, Dietary Intakes, and Physical Activity Measurements
Blood samples were collected after 10–12 h overnight fasting before and after the intervention and serum was obtained by centrifugation at 3,000–4,000 RPM for 10 min. Serum magnesium concentrations were measured using colorimetric method by an automatic analytical instrument (Hitachi 912; Roche Diagnostics). The sensitivity of the method allows for the determination of magnesium in amounts between 0.05 to 5 mg/dL. Inter- and intra-assay coefficients of variation for magnesium measurement were 1.43 % and 0.83 %, respectively.
Anthropometric, dietary intakes and physical activity variables were obtained twice, before and after intervention. Body weight was measured to the nearest 0.1 kg while the participants were minimally clothed and without shoes. Height was measured to the nearest 0.5 cm in standing position without shoes while the shoulders were in a normal position. Body mass index (BMI) was calculated as body weight (kg) divided by height squared (m2). Body composition including body fat mass (FM), lean body mass (LBM), and the percentage of FM and LBM were taken by Bioelectrical Impedance Analyzer (Quad scan 4000; Bodystat). Calibration of the Quad scan BIA analyzer was checked before testing according to the user’s guide instructions; the actual resistance obtained at 50 kHz from the analyzer current being run through a 500-Ω calibration resistor was tested. The resistance was within the calibration specification of 496–503 Ω.
Dietary intakes were assessed with a 24-h food recalls completed for 2 days (1 week day and 1 weekend day) by expert dietitians. Since the Iranian food composition table (FCT) is incomplete, and is limited to raw materials and few nutrients, nutrient intakes were calculated using nutritionist 4 software (N-squared Computing, San Bruno, CA). However, the Iranian FCT was used for some national foods that are not included in the FCT of USA. Physical activity levels were determined by the short form of the International Physical Activity Questionnaire (IPAQ) and expressed as Met-Min/Weeks [18].
Muscle Performance
Upper body strength was assessed by handgrip strength test, lower body strength was assessed by knee extension strength test, and functional mobility was assessed quantitatively using Time Get Up and Go Test (TGUG) before and after the intervention.
Handgrip strength was measured in kg in dominant hand using a calibrated hand-held dynamometer (digital hand dynamometer “DIGI-ІІ,” Korea). Participants were seated on standard armchair with shoulder adducted and neutrally rotated, elbow flexed to 90º, and the forearm and wrist were in a neutral position [19]. Participants were instructed to grip the dynamometer with maximum strength smoothly in response to a voice command, without rapid wrenching or jerking motion. The size of the grip was set to hand size of participants so that it fit comfortable [20]. Three measurements were made on dominant hand with 1 min rest between trials, and the maximum of these measurements was used for analyses. To exhibit the maximal force, the participants were strongly encouraged each time during the test. Test–retest reliability for handgrip strength test determined by using 10 participants measured 7 days apart showed a high correlation of 0.96 with no differences in mean strength levels.
Isometric knee extension strength was measured with a calibrated hand held dynamometer (Nicholas Manual Muscle Tester; Lafayette Inc) in kg in dominant leg. The dominant leg was determined by asking the participants which leg they use to kick a ball [21]. The participants were seated on straight-back standard chair with their hips and knees flexed 90º. The dynamometer was placed proximal to the ankle joint of participants, and they were asked to lift their leg [22]. During testing, the participants were strongly encouraged to increase the force to the greatest possible gradually while the tester was opposing. Knee extension strength was measured three times with a between measurement interval of 1 min, and the maximum of these measurements was used. Test–retest reliability for knee extension strength showed a correlation of 0.97 in 10 participants tested on 7 days apart. Hand-held dynamometers were properly “zeroed” before each measurement according the manufacturer’s instructions to insure calibration and accuracy.
For TGUG test, the participants were seated with their back against the chair and arms resting on the chair. They were asked to stand upright on the word “go,” walk at normal pace 3 m to the marked point, turn to chair, and sit down. Participants got acquainted with procedure by doing a trial prior to data collection, then the test was performed twice and the mean value in seconds (s) was used[23, 24].
Statistical Analysis
The number of participants estimated for each group was 31 at 80 % power and α of 0.05 to detect a difference of 4 kg in handgrip strength between groups with an SD of 5.6 kg [25]. To allow for dropouts, it was decided to recruit 37 participants for each group.
Statistical analysis was performed with Statistical Package for Social Sciences (SPSS, Inc., Chicago, IL, USA) version 15.0. The Kolmogorov–Smirnov test was applied to assess normality of data. All quantitative variables had normal distribution except physical activity. Paired-samples t test was used to compare baseline and 4-week values in each group. Differences between two groups at baseline, at week 8, and its change after 8 weeks were examined with independent-samples t test. Comparison of non-normally distributed data of physical activity was conducted using Wilcoxon-signed ranks and the Mann–Whitney U test. Changes of variable after 8 weeks in each groups was calculated as week 8 values minus baseline values. The percentage of the change was also determined as (week 8 values − baseline values)/baseline values × 100. Data are presented as mean ± standard deviations (SD), unless stated otherwise. In addition, to examine whether baseline values of serum magnesium or muscle strength indices were effect modifiers for the effects of magnesium supplementation on muscle strength, a linear regression model was used with changes from baseline to 8 weeks in outcome measures as the dependent variable and treatment group, baseline values, and interaction terms of baseline value and treatment group as independent variables. A two-tailed P value ≤ 0.05 was considered significant statistically.
Results
Of 74 participants initially included in the study, 3 in the placebo group and 2 in the magnesium group withdrew because of non-compliance with the intervention. Compliance with treatment was approximately 93 % for each group. No significant differences between the groups were seen for age, weight, BMI, physical activity and serum magnesium at baseline (Table 1). In addition, no significant changes in weight, BMI, physical activity, and serum magnesium were observed within each group during the study.
The mean dietary intake of energy, protein, carbohydrate, total fat, fiber, selenium, and magnesium was not significantly different between the two groups at baseline and after 8 weeks (Table 1). Likewise, these dietary intake variables did not change significantly within each group during the study. Based on dietary intakes of magnesium at baseline, 91.4 % in the magnesium group and 94.1 % in the placebo did not consume the American Estimated Average Requirement (EAR) of 265 mg/day for magnesium.
Body composition variables were not different significantly between groups at baseline. At the end of the study, lean mass increased (1.6 %; P = 0.05) and fat mass decreased (−3.8 %; P = 0.02) in the magnesium group compared to baseline, while no significant changes were seen in any of these variables in the placebo group (Table 2). However, the changes did not reach significant compared with placebo group.
There were no significant differences between groups in regards of handgrip strength, knee extension strength, and TGUG test at baseline and after the end of 8-week intervention. Handgrip strength increased from 26.3 ± 5.5 to 27.8 ± 4.5 kg (P = 0.002) in the magnesium group and from 26.7 ± 5.0 to 27.8 ± 4.7 kg (P = 0.03) in the placebo group at the end of week 8 compared to baseline, but the increase was not significantly different between the two groups (Fig. 1a). Knee extension strength raised from 22.8 ± 6.1 to 23.8 ± 5.4 kg in the magnesium group and from 21.6 ± 5.5 to 22.8 ± 5.3 kg in the placebo group during the study, but none of these changes reached significant (Fig. 1b). The mean TGUG test decreased from 7.5 ± 1.1 to 7.3 ± 1.0 s in the magnesium group (P = 0.041) as compared to baseline, while no significant change was observed in the placebo group (Fig. 1c). No significant difference was observed in TGUG test between two groups at week 8.
The effects of baseline serum magnesium and muscle strength indices were examined to investigate whether those with low baseline serum magnesium or muscle strength had higher gain in muscle strength in response to magnesium supplementation. However, no significant interaction between treatment group and baseline muscle strength indices were observed for handgrip strength (P for interaction = 0.69) and knee extension strength (P for interaction = 0.86). In addition, baseline serum magnesium did not show any effect modifier for the effect of magnesium supplementation on hand grip strength (P for interaction = 0.74), knee extension strength (P for interaction = 0.37), and TGUG (P for interaction = 0.86).
Discussion
In the present study, daily intake of 250 mg magnesium in the form of magnesium oxide for 8 weeks did not lead to greater gains in handgrip strength and knee extension strength in the magnesium group as compared with placebo. The failure to show a significant improvement in muscle strength in the magnesium group compared with the placebo may be explained by several reasons:
The dose of magnesium might have been insufficient to exert any significant improvements in muscle strength. In a study of young untrained men, the only other clinical trial study to investigate magnesium supplementation on muscle strength in healthy subjects, magnesium supplementation as magnesium oxide in combination with strength training for 7 week leaded to a greater knee extension torque in the magnesium group as compared to the placebo. In that study, total magnesium intake, including diet and supplements, was 8 mg/kg body weight/day [26], while the mean intake of magnesium in the magnesium group of our participants with considering the magnesium intake from diet was less than 6 mg/kg. It has been suggested that to see an increase in muscle strength following creatine supplementation, reaching creatine to the specific level within muscle is necessary [27]. It is also possible that to observe any significant improvements in muscle strength, magnesium in muscle should be reached to an optimal level, which remains to be elucidated.
Bioavailability of magnesium from magnesium oxide could also affect responses of the participant to magnesium supplementation. Both elemental magnesium content in a magnesium preparation and its bioavailability can influence the response to the magnesium supplement. Relatively poor bioavailability of magnesium from magnesium oxide has been indicated [28, 29] in spite of the fact that this form of the magnesium supplement has higher elemental magnesium content (approximately 60 %) compared to other forms of supplements [30]. It has also been shown that magnesium bioavailability of magnesium oxide from the effervescent tablets is better than from its capsules [31].
Magnesium supplementation period may have been too short to increase muscle strength. The 8-week supplementation period in our study was based on the study by Brilla et al. [26], where magnesium supplementation combined with training for 7 weeks increased muscle strength significantly.
We also supposed that differences in baseline magnesium status or baseline muscle strength might have effects on the response to the magnesium supplementation; individuals with lower baseline serum magnesium or baseline muscle strength responded better to magnesium supplementation. However, we did not find any effect modifier for either baseline serum magnesium or baseline muscle strength in response to magnesium supplementation.
The TGUG test is the time needed to complete a series of functionally important tasks including stand up from a standard armchair, walk 3 m, turn down, and sit down and is an effective method of assessing functional mobility [24]. Samson et al. found higher correlations between muscle strength and TGUG tests in women than men [23], indicating that women may exert more mobility difficulties due to reducing muscle strength with age. In our study, a significant improvement on the TGUG in the magnesium group as compared to baseline values might be related to improved muscle strength but it does not seem to be significant clinically.
In present study, 250 mg magnesium in the form of magnesium oxide for 8 weeks had no effect on serum magnesium. No significant changes in serum magnesium during the study might reflect either the lack of significant response to the modest dose of magnesium supplementation or lack of validity of the serum magnesium. Serum magnesium concentrations are the most frequently used measure of magnesium status, even though it has been shown that it has slight responses to changes in magnesium intake [16]. Some studies suggest that serum magnesium did not alter linearly in response to magnesium supplementation though the patterns proposed for the response are not consistent [32–34]. In addition, it has been proposed that magnesium supplementation has no significant effect on total serum magnesium concentrations if participants have no evidence of magnesium deficiency [32, 35] while it can improve serum magnesium in individuals with hypomagnesemia (<1.8 mg/dL) [33, 36]. In our study, the total serum magnesium of the participants was within normal range of 1.8–2.6 mg/dL except one person in the magnesium group who had serum magnesium of 1.7 mg/dL. However, high dose of magnesium (600 mg) in the form of pidolate for 12 weeks could increase serum magnesium significantly in the participants with normal serum magnesium [37], suggesting that differences in form of magnesium salt and doses used might be also possible sources of divergence in serum magnesium response after magnesium supplementation between studies. To accurately address responses to magnesium supplementation, determinations of serum magnesium in combination with other indices of magnesium status at different intervals of time following magnesium supplementation needs to be considered in future research.
Data from our study showed a significant mean increase in LBM of 0.7 kg, and a significant reduce in FM of 1 kg in the magnesium group. In this study, all participants were instructed not to eat or drink 4 to 5 h prior to the test, not to consume coffee or alcohol 24 h prior to the test, and not to exercise 12 h prior to testing in order to reduce the influences of hydration status and other factors that might affect the results of the test. Since dietary intakes and physical activity did not change during the study and the changes in body composition were significant only in the magnesium group, the changes in these variables might be attributable to magnesium supplementation. These changes in body composition is clinically important, since both body fat mass and fat-free mass have been associated with metabolic disorders as well as muscle strength [38, 39]. If confirmed that magnesium intake causes the favorable changes in body composition in further studies, it might be one of the mechanism that magnesium intake can reduce several diseases.
Magnesium can influence muscle function and performance through its roles in energy production, muscle contraction and relaxation, and oxygen uptake. Growing studies indicate that magnesium is involved in modulation of anabolic hormones secretion [40, 41] and inflammation [42, 43]; the known leading causes of declining muscle strength [44, 45]. Therefore, it is possible that magnesium may have indirect or long-term impacts on muscle strength and muscle mass through its effect on inflammation and anabolic hormone which deserve investigations.
There were some limitations in the present study. First, the dose of magnesium administered was rather modest. Second, response to magnesium supplementation was assessed by serum magnesium and ionized serum magnesium or intracellular magnesium concentrations were not determined. Third, in order to confirm treatment compliance determined by pill count, urinary magnesium concentrations were not measured.
In conclusion, 250 mg magnesium in the form of magnesium oxide for 8 weeks in middle-aged overweight women had no significant effects on upper and lower muscle strength compared with placebo. These results might have been a factor of insufficient loading of magnesium at skeletal muscle due to inadequate dose of magnesium, poor bioavailability of magnesium oxide, short period of supplementation, or the combination of these factors. This study provides some evidence indicating that magnesium might affect both fat and lean body mass. Further study is required, in which magnesium supplemented in another form or combined with resistance exercise, to determine the effects of magnesium supplementation on body composition and muscle performance indices in both young and old individuals. The long-term association between magnesium intake and muscle strength regarding to its relationship with inflammation and anabolic hormones are also needed to be investigated.
References
Sirola J, Rikkonen T, Tuppurainen M, Jurvelin JS, Alhava E, Kroger H (2008) Grip strength may facilitate fracture prediction in perimenopausal women with normal BMD: a 15-year population-based study. Calcif Tissue Int 83:93–100
Cheung CL, Tan KC, Bow CH, Soong CS, Loong CH, Kung AW (2012) Low handgrip strength is a predictor of osteoporotic fractures: cross-sectional and prospective evidence from the Hong Kong Osteoporosis Study. Age 34:1239–1248
Rantanen T, Guralnik J, Foley D, Masaki K, Leveille S, Curb J, White L (1999) Midlife Hand Grip Strength as a Predictor of Old Age Disability. JAMA 281:558–560
Rantanen T, Harris T, Leveille S, Visser M, Foley D, Masaki K, Guralnik J (2000) Muscle Strength and Body Mass Index as Long-Term Predictors of Mortality in Initially Healthy Men. Journal of Gerontology: MEDICAL SCIENCES 55A:M168–M173
Newman A, Kupelian V, Visser M, Simonsick E, Goodpaster B, Kritchevsky S, Tylavsky F, Rubin S, Harris T (2006) Strength, but not muscle mass, is associated with mortality in the health, aging and body composition study cohort. Journal of Gerontology: MEDICAL SCIENCES 61A:72–77
Jurca R, Lamonte MJ, Barlow CE, Kampert JB, Church TS, Blair SN (2005) Association of muscular strength with incidence of metabolic syndrome in men. Med Sci Sports Exerc 37:1849–1855
Atlantis E, Martin SA, Haren MT, Taylor AW, Wittert GA (2009) Inverse associations between muscle mass, strength, and the metabolic syndrome. Metabolism 58:1013–1022
Aoyama T, Asaka M, Ishijima T, Kawano H, Cao ZB, Sakamoto S, Tabata I, Higuchi M (2011) Association between muscular strength and metabolic risk in Japanese women, but not in men. J Physiol Anthropol 30:133–139
Lauretani F, Russo C, Bandinelli S, Bartali B, Cavazzini C, Iorio A, Corsi A, Rantanen T, Guralnik J, Ferrucci L (2003) Age-associated changes in skeletal muscles and their effect on mobility : an operational diagnosis of sarcopenia. J Appl Physiol 95:1851–1860
Greenlund LJS, Nair KS (2003) Sarcopenia-consequences, mechanisms, and potential therapies. Mechanisms of Ageing and Development 124:287–299
Doherty T (2003) Invited Review : Aging and sarcopenia. J Appl Physiol 95:1717–1727
Robinson S, Cooper C, Aihie Sayer A (2012) Nutrition and sarcopenia: a review of the evidence and implications for preventive strategies. J Aging Res 2012:510801
Robinson SM, Jameson KA, Batelaan SF, Martin HJ, Syddall HE, Dennison EM, Cooper C, Sayer AA (2008) Diet and its relationship with grip strength in community-dwelling older men and women: the Hertfordshire cohort study. J Am Geriatr Soc 56:84–90
Nielsen FH, Lukaski HC (2006) Update on the relationship between magnesium and exercise. Magnes Res 19:180–189
Nielsen FH (2010) Magnesium, inflammation, and obesity in chronic disease. Nutr Rev 68:333–340
Lukaski HC, Nielsen FH (2002) Dietary Magnesium Depletion Affects Metabolic Responses during Submaximal Exercise in Postmenopausal Women. J Nutr 132:930–935
Dominguez L, Barbagallo M, Lauretani F, Bandinelli S, Bos A, Corsi A, Simonsick E, Ferrucci L (2006) Magnesium and muscle performance in older persons : the InCHIANTI study. Am J Clin Nutr 84:419–426
Craig CL, Marshall AL, Sjostrom M, Bauman AE, Booth ML, Ainsworth BE, Pratt M, Ekelund U, Yngve A, Sallis JF, Oja P (2003) International physical activity questionnaire: 12-country reliability and validity. Med Sci Sports Exerc 35:1381–1395
Budziareck MB, Pureza Duarte RR, Barbosa-Silva MC (2008) Reference values and determinants for handgrip strength in healthy subjects. Clin Nutr 27:357–362
Boadella JM, Kuijer PP, Sluiter JK, Frings-Dresen MH (2005) Effect of self-selected handgrip position on maximal handgrip strength. Arch Phys Med Rehabil 86:328–331
Macaluso A, Young A, Gibb KS, Rowe DA, De Vito G (2003) Cycling as a novel approach to resistance training increases muscle strength, power, and selected functional abilities in healthy older women. J Appl Physiol 95:2544–2553
Rolland Y, Lauwers-Cances V, Pahor M, Fillaux J, Grandjean H, Vellas B (2004) Muscle strength in obese elderly women: effect of recreational physical activity in a cross-sectional study. Am J Clin Nutr 79:552–557
Samson MM, Meeuwsen IB, Crowe A, Dessens JA, Duursma SA, Verhaar HJ (2000) Relationships between physical performance measures, age, height and body weight in healthy adults. Age Ageing 29:235–242
Wall JC, Bell C, Campbell S, Davis J (2000) The Timed Get-up-and-Go test revisited: measurement of the component tasks. J Rehabil Res Dev 37:109–113
Luna-Heredia E, Martin-Pena G, Ruiz-Galiana J (2005) Handgrip dynamometry in healthy adults. Clin Nutr 24:250–258
Brilla LR, Haley TF (1992) Effect of magnesium supplementation on strength training in humans. J Am Coll Nutr 11:326–329
Greenhaff PL, Bodin K, Soderlund K, Hultman E (1994) Effect of oral creatine supplementation on skeletal muscle phosphocreatine resynthesis. Am J Physiol 266:E725–730
Firoz M, Graber M (2001) Bioavailability of US commercial magnesium preparations. Magnes Res 14:257–262
Walker AF, Marakis G, Christie S, Byng M (2003) Mg citrate found more bioavailable than other Mg preparations in a randomised, double-blind study. Magnes Res 16:183–191
Guerrera MP, Volpe SL, Mao JJ (2009) Therapeutic uses of magnesium. Am Fam Physician 80:157–162
Siener R, Jahnen A, Hesse A (2011) Bioavailability of magnesium from different pharmaceutical formulations. Urol Res 39:123–127
Dimai HP, Porta S, Wirnsberger G, Lindschinger M, Pamperl I, Dobnig H, Wilders-Truschnig M, Lau KH (1998) Daily oral magnesium supplementation suppresses bone turnover in young adult males. J Clin Endocrinol Metab 83:2742–2748
Rodriguez-Moran M, Guerrero-Romero F (2003) Oral magnesium supplementation improves insulin sensitivity and metabolic control in type 2 diabetic subjects: a randomized double-blind controlled trial. Diabetes Care 26:1147–1152
Song Y, He K, Levitan EB, Manson JE, Liu S (2006) Effects of oral magnesium supplementation on glycaemic control in Type 2 diabetes: a meta-analysis of randomized double-blind controlled trials. Diabet Med 23:1050–1056
Lee S, Park HK, Son SP, Lee CW, Kim IJ, Kim HJ (2009) Effects of oral magnesium supplementation on insulin sensitivity and blood pressure in normo-magnesemic nondiabetic overweight Korean adults. Nutr Metab Cardiovasc Dis 19:781–788
Guerrero-Romero F, Tamez-Perez HE, Gonzalez-Gonzalez G, Salinas-Martinez AM, Montes-Villarreal J, Trevino-Ortiz JH, Rodriguez-Moran M (2004) Oral magnesium supplementation improves insulin sensitivity in non-diabetic subjects with insulin resistance. A double-blind placebo-controlled randomized trial. Diabetes Metab 30:253–258
Hatzistavri LS, Sarafidis PA, Georgianos PI, Tziolas IM, Aroditis CP, Zebekakis PE, Pikilidou MI, Lasaridis AN (2009) Oral magnesium supplementation reduces ambulatory blood pressure in patients with mild hypertension. Am J Hypertens 22:1070–1075
Cesari M, Kritchevsky SB, Baumgartner RN, Atkinson HH, Penninx BW, Lenchik L, Palla SL, Ambrosius WT, Tracy RP, Pahor M (2005) Sarcopenia, obesity, and inflammation–results from the Trial of Angiotensin Converting Enzyme Inhibition and Novel Cardiovascular Risk Factors study. Am J Clin Nutr 82:428–434
Ruiz JR, Ortega FB, Warnberg J, Moreno LA, Carrero JJ, Gonzalez-Gross M, Marcos A, Gutierrez A, Sjostrom M (2008) Inflammatory proteins and muscle strength in adolescents: the Avena study. Arch Pediatr Adolesc Med 162:462–468
Maggio M, Ceda GP, Lauretani F, Cattabiani C, Avantaggiato E, Morganti S, Ablondi F, Bandinelli S, Dominguez LJ, Barbagallo M, Paolisso G, Semba RD, Ferrucci L (2011) Magnesium and anabolic hormones in older men. Int J Androl 34:e594–600
Cinar V, Polat Y, Baltaci AK, Mogulkoc R (2011) Effects of magnesium supplementation on testosterone levels of athletes and sedentary subjects at rest and after exhaustion. Biol Trace Elem Res 140:18–23
Moslehi N, Vafa M, Rahimi-Foroushani A, Golestan B (2012) Effects of oral magnesium supplementation on inflammatory markers in middle-aged overweight women. J Res Med Sci 17:607–614
Sugimoto J, Romani AM, Valentin-Torres AM, Luciano AA, Ramirez Kitchen CM, Funderburg N, Mesiano S, Bernstein HB (2012) Magnesium decreases inflammatory cytokine production: a novel innate immunomodulatory mechanism. J Immunol 188:6338–6346
Borst S (2004) Interventions for sarcopenia and muscle weakness in older people. Age and Ageing 33:548–555
Schaap LA, Pluijm SM, Deeg DJ, Harris TB, Kritchevsky SB, Newman AB, Colbert LH, Pahor M, Rubin SM, Tylavsky FA, Visser M (2009) Higher inflammatory marker levels in older persons: associations with 5-year change in muscle mass and muscle strength. J Gerontol A Biol Sci Med Sci 64:1183–1189
Acknowledgments
We would like to express our appreciation to the staff of Tehran University of Medical Sciences for their cooperation and participation in this study. This work was financially supported by Tehran University of Medical sciences
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moslehi, N., Vafa, M., Sarrafzadeh, J. et al. Does Magnesium Supplementation Improve Body Composition and Muscle Strength in Middle-Aged Overweight Women? A Double-Blind, Placebo-Controlled, Randomized Clinical Trial. Biol Trace Elem Res 153, 111–118 (2013). https://doi.org/10.1007/s12011-013-9672-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-013-9672-1