Abstract
Exposure to heavy metals has been associated to a higher incidence of multiple sclerosis. In this work, we present a possible relationship between serum mercury levels and development of multiple sclerosis in Isfahan, the third largest city in Iran. Seventy-four patients affected by multiple sclerosis were retrieved from multiple sclerosis (MS) clinic in Isfahan, Iran. By matching sex and age, 74 healthy volunteers were chosen as control group. Blood samples were collected and serum mercury content was determined. Serum mercury level in MS patients was significantly higher than controls (9.6 ± 10.17 vs. 5.7 ± 8.6, P = 0.037). Concerning all MS patients, serum mercury value was significantly higher than the mercury concentration founded in control subjects {odd ratio: 2.39 (CI, 1.96–2.94), P = 0.00}. Serum mercury level is higher in MS patients with odd ratio equal to 2.39 compared with healthy individuals. It may reveal that high mercury levels in serum might help MS development in susceptible individuals. More studies with larger sample size are needed to confirm this hypothesis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Multiple sclerosis (MS) is an inflammatory demyelinating disorder of the central nervous system (CNS) [1]. Although genetic and immunological factors have been strongly mentioned to be associated with MS pathogenesis, the ultimate etiology of MS remains uncertain [2, 3]. Apparently, both genetics and environmental factors are important in pathogenesis of multiple sclerosis [4]. Recently, environmental factors such as pathogens and chemicals (toxic elements) have been suggested to play a significant role in MS pathogenesis. Moreover, exposure to heavy metals has been associated to a higher incidence of multiple sclerosis [5–8]. Among all, mercury is assumed to be one of the most toxic within nonradioactive elements. Dietary use of predatory fish is considered to be the most frequent resource of organic mercury exposure [9]. Moreover, mercury and its derivatives have been widely used in different products including antiparasitics, antiseptics, dental amalgams, medical equipment, and beauty creams. Subsequent to growing industrial applications of mercury, its environmental level is estimated to increase more than threefold in the past century [10, 11]. Casualties who exposed to mass exposure in Japan during 1950–1960 along with victims in Iraq through 1971–1972, were related to mercury-contaminated fish and fungicide in seed grains, respectively. This highlights the role of mercury as an important neurotoxicant [12, 13]. Meanwhile, there are many unsubstantiated claims that chronic mercury poisoning causes neurological diseases and behavioral problems. MS is also reported to be due to chronic mercury intoxication. Review of recent literature claims that mercury used in dental amalgams may lead to nephrotoxicity, neurobehavioral changes, autoimmunity, and oxidative stress [14–16]. Dental amalgam is an alloy which contains about 50% mercury and is frequently used in dentistry as a tooth-filling material [11, 17]. There are many studies which have shown that dental amalgams may contribute in pathogenesis of various tissue burdens, particularly within CNS, kidneys, and certain glands [12]. Meanwhile, in 1986, a correlation between multiple sclerosis and chronic exposure to mercury via dental amalgam fillings was reported [18].
Due to high incidence of multiple sclerosis and also industrial pollutions in Isfahan, we studied the possible relation between serum mercury level and development of MS in Isfahan that is the third largest city of Iran.
Methods
Patients
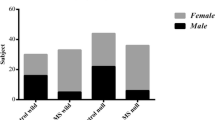
Seventy-four patients (60 females and 14 males) affected by multiple sclerosis were retrieved from MS clinic in Isfahan, Iran. Patients mean age was 30.97 ± 9.07 years and only individuals with expanded disability status scale less than 3 were enrolled in our study. All patients were definite MS diagnosed by a neurologist according to McDonald criteria [19]. Indeed, they were all in relapsing remitting status and were clinically stable for the past 3 months. Meanwhile, by matching sex and age and the place for residence for the patient group, 74 healthy volunteers (58 female and 16 male; p value, 0.275) were chosen as control group. The control group was chosen from volunteers, who were students of Isfahan University of Medical Sciences, School of Pharmacy. Mean age of control subjects was 29.50 ± 10.58 and the difference between patients and controls was not significant (p value, 0.395). In both groups, individuals with cardiological, kidney, respiratory or liver diseases and also vegetarians, were excluded. The project was approved by the ethics committee of Isfahan University of Medical Sciences and both control and patient groups filled out the inform consents accepted by the committee. Neurological disability was rated based on the Kurtzke scale and all patients were assessed during a stable period of their illness [20].
Biochemical Analysis
Blood samples were collected through venipuncture by using trace element-free evacuated tubes. After centrifugation at 1,000×g for 15 min, serum was separated and frozen at −20°C. The mercury content of serum was determined by atomic absorption spectrometry using cold-vapor atomization. The specimens were mixed with a strong oxidizing agent to convert all species of mercury to Hg. Argon was bubbled through the specimen to release Hg into the vapor phase and the transmittance was monitored, then the quantity of the total mercury in the specimen with using standard curve was measured [21].
Data Analysis
Standard statistical methods (\( \overline X \), SEM) were utilized to summarize parametric values and t test was employed to compare the control group versus group of patients affected by multiple sclerosis. Data were analyzed using SPSS software (version 15) and P value < 0.05 was regarded as significant. Moreover, up to 95th percentile of the serum mercury level in control subjects was considered as normal and higher amounts of mercury was regarded as abnormal levels, when we compared mercury amount of serum among control and patients. Then, chi-square test and risk estimation was used for categorical variables.
Results
The mean serum mercury level in MS patients and controls was 9.6 ± 10.17 vs. 5.7 ± 8.6, P = 0.037. Therefore, concerning all MS patients (n = 74), serum mercury value was significantly higher than the mercury concentration founded in control subjects. By considering more than 95th percentile of serum mercury level as abnormal, abnormal high levels of serum mercury in control subjects was estimated as 19.7 μg/L. Serum mercury level was significantly more abnormal in MS patients comparing to controls {odds ratio 2.39, (CI, 1.96–2.94), P value 0.000}. According to the subgroup of females, the odd ratio was {odds ratio 2.32, (CI 1.85–2.94), P value 0.000}. Additionally, in the male subgroup, the odd ratio was {odds ratio 2.85, (CI 1.72–4.76), P value 0.028}.
Discussion
Serum mercury level in healthy individuals and MS patients differed significantly. This difference was also observed in male and females. Additionally, abnormal high level of mercury was observed to be more in MS patients, revealing that high amounts of mercury in serum could be a factor that increases susceptibility of individuals to multiple sclerosis.
The global prevalence of MS is increasing and is expected to be approximately 3.6/100,000 person-years in women and 2.0/100 000 person-years in men, which reveals the female preponderance [22]. Nevertheless, MS course is more benign in females and younger individuals [23]. Males are older at onset and are susceptible for having more progressive disease course and a more rapid progression of the disease [24]. Here, in this study, we could not find any association between serum mercury level and different sex, so it appears that mercury alone may not contribute to the different susceptibility of sexes in developing MS. However, further studies are needed to evaluate the association between mercury exposure and different susceptibility of sexes and also varied severity in the course of MS.
Usual values for whole blood mercury are less than 6 μg/L; however, taking diets rich in fish might cause elevated mercury levels [9]. Mercury has a short half-life in the blood; therefore, if the exposure is chronic, obtaining urine levels in 24-h collections is more accurate [9]. Further researches with larger sample and different method for mercury evaluations are needed to establish the role of such toxic element in pathogenesis of MS.
Mercury is a potent toxin which affects the basic functions of the cell, through binding strongly with sulfhydryl and selenohydryl groups on albumin molecules in cell membranes, receptors and intracellular signal links, and by modifying the tertiary structure. One popular hypothesis that is frequently propounded in the literature suggests mercury to be toxic due to inducing production of free radicals and modifying the redox potential of the cell [25]. Otherwise, low concentrations of mercury may provoke phosphorylation and consequently intracellular signaling [26]. Mercury also inhibits the development of cytoskeleton structure in nerve cells and breaks it down [27]. Tschudi et al. studied the incidence of apoptosis within cultures of fetal rat brain and found that mercury speeds up the spontaneous apoptosis in immature cultures, especially astrocytes [28]. In another survey, Shenker et al. demonstrated that mercury is able to induce an apoptotic cascade in human T lymphocytes [29]. Duxbury et al. have also shown that mercury is toxic for oligodendroglial cells and human oligodendroglial cell line MO3.13. They choose this cell line to model the loss of oligodendrocytes occurring through episodes of MS. They show that damages are caused by specially an apoptotic manner, when exposed to low concentration of Hgcl2 [30]. On the other hand, MS is a degenerative disease of the central nervous system affecting the oligodendroglial components of the nerve. The pathological hallmark of MS is the plaque, a well-demarcated gray or pink lesion, characterized histologically by complete myelin loss, an absence of oligodendrocytes and relative sparing of axons. Oligodendrocytes are destroyed as an early event during the disease pathogenesis and are also found to be distressed in chronic lesions [31]. Although several observations were made to explain the possible role of mercury in MS etiology, the exact pathogenesis is still unclear. Circumstantial evidence includes empirical observations in which accidentally ingested mercury produces a set of symptoms similar to those observed in MS patients [32]. Dental amalgam fillings containing mercury have been suggested as a possible risk factor for MS as well [15]. Bangsi et al. [33] counted the number of amalgam restorations and found a higher amount of dental amalgams in MS patients. Yet, McGrother CW et al., found no relationship between the high levels of mercury in blood and urine as well as the raised number of amalgam fillings, and susceptibility to multiple sclerosis [16]. After all, Etminan et al., within a meta-analysis, found a slight, non-statistically significant increase of MS development while dental amalgams are used [11]. However, we showed the association between mercury blood level and MS, with odd ratio equal to 2.39 (2.94–1.94). Nevertheless, further complementary observations are needed to uncover the inter-relationship between MS and mercury as well as other heavy metals within CSF, blood, and urine samples.
In conclusion, serum mercury level is higher in MS patients with odd ratio equal to 2.39 compared with healthy individuals, revealing that high amounts of mercury in serum might be a factor that increases susceptibility to multiple sclerosis. More studies with larger sample size are needed to confirm this hypothesis.
References
Claude CA, Kerlero de Rosbo N (1992) Multiple sclerosis: an autoimmune disease of multifactorial etiology. Curr Opin Immunol 4:760–765
Ramagopalan SV, Dobson R, Meier UC, Giovannoni G (2010) Multiple sclerosis: risk factors, prodromes, and potential causal pathways. Lancet Neurol 9:727–739
Luechinetti CF, Parisi J, Bruck W (2005) The pathology of multiple sclerosis. Neurol Clin 23:77–105
Prat A, Antel J (2005) Pathogenesis of multiple sclerosis. Curr Opin Neurol 18:225–230
Yasui M, Kihira T, Ota K et al (1991) Zinc concentration in the central nervous system in a case of multiple sclerosis–comparison with other neurological diseases. No To Shinkei 43:951–955
Ingalls TH (1089) Clustering of multiple sclerosis in Galion, Ohio 1982–1985. Am J Forensic Med Pathol 10:213–215
Melø TM, Larsen C, White LR et al (2003) Manganese, copper, and zinc in cerebrospinal fluid from patients with multiple sclerosis. Biol Trace Elem Res 93:1–8
Mutter J, Naumann J, Guethlin C (2007) Comments on the article “The Toxicology of Mercury and Its Chemical Compounds” by Clarkson and Magos (2006). Crit Rev Toxicol 37:537–549
Ibrahim D, Froberg B, Wolf A et al (2006) Heavy metal poisoning: clinical presentations and pathophysiology. Clin Lab Med 26:67–97
Golding GR, Kelly CA et al (2007) Evaluation of mercury toxicity as a predictor of mercury bioavailability. Environ Sci Technol 41:5685–5692
Aminzadeh KK, Etminan M (2007) Dental amalgam and multiple sclerosis: a systematic review and meta-analysis. J Public Health Dent 67:64–66
Ozuah PO (2000) Mercury poisoning. Curr Probl Pediatr 30:91–99
Kulig K (1998) A tragic reminder about organic mercury. N Engl J Med 338:1692–1694
Mutter J, Naumann J, Walach H et al (2005) Amalgam risk assessment with coverage of references up to 2005. Gesundheitswesen 67:204–216
Cusetta I, Invernizzi M, Granieri G (2001) Multiple sclerosis and dental amalgam: case–control study in Ferrara, Italy. Neuroepidemiology 20:134–137
McGrother CW, Dugmore C, Phillips MJ et al (1999) Multiple sclerosis, dental caries and fillings: a case–control study. Br Dent J 187:261–264
Ng DK, Chan CH, Soo MT et al (2007) Low-level chronic mercury exposure in children and adolescents: meta-analysis. Pediatr Int 49:80–87
Ingalls HT (1986) Endemic clustering of multiple sclerosis in time and place. 1934–1984. Am J Forensic Med Pathol 7:3–8
McDonald WI, Compston A, Edan G et al (2001) Recommended diagnostic criteria for multiple sclerosis: guidelines from the International Panel on the diagnosis of multiple sclerosis. Ann Neurol 50:121–127
Kurtzke JF (1983) Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS). Neurology 33:1444–1452
Barbosa F, Palmer CD, Krug FJ et al (2004) Determination of total mercury in whole blood by flow injection cold vapor atomic absorption spectrometry with room temperature digestion using tetramethylammonium hydroxide. J Anal At Spectrom 19:1000–1005
Alonso A, Hernán MA (2008) Temporal trends in the incidence of multiple sclerosis: a systematic review. Neurology 71:129–135
Yamout B, Itani S, Arabi A, Hamzeh D et al (2010) Prognostic factors of multiple sclerosis in Lebanon. Int J Neurosci 120(3):206–210
Greer JM, McCombe PA (2011) Role of gender in multiple sclerosis: clinical effects and potential molecular mechanisms. J Neuroimmunol 234(1–2):7–18
Ercal N, Gurer OH, Aykin BN (2001) Toxic metals and oxidative stress part I: mechanisms involved in metal-induced oxidative damage. Curr Top Med Chem 1:529–539
Mattingly RR, Felczak A, Chen CC et al (2001) Low concentrations of inorganic mercury inhibit Ras activation during T cell receptor mediated signal transduction. Toxicol Appl Pharmacol 176:162–168
Leong CC, Syed NL, Lorscheider FL (2001) Retrograde degeneration of neurite membrane structural integrity of nerve growth cones following in vitro exposure to mercury. Neuroreport 12:733–737
Monnet TF (1998) Induction of apoptosis by compounds depends on maturation and is not associated with microglial activation. J Neurosci Res 53:361–367
Sheuker BJ, Guo TL, Shapiro IM (2000) Mercury-induced apoptosis in human lymphoid cells: evidence that the apoptotic pathway is mercurial species dependent. Environ Res 84:89–99
Issa Y, Watts DC, Duxbury AJ et al (2003) Mercuric chloride: toxicity and apoptosis in a human oligodendroglial cell line MO3.13. Biomaterials 24:981–987
Ozawa K, Suchanek G, Breitschopf H et al (1994) Patterns of oligodendroglia pathology in multiple sclerosis. Brain 117:1311–1322
Langauer LH, Zajac NM (1997) Changes in the nervous system due to occupational metallic mercury Poisoning. Neurol Neurochir Pol 3:905–913
Bangsi D, Ghadirian P, Ducic S et al (1998) Dental amalgam and multiple sclerosis: a case–control study in Montreal, Canada. Int J Epidemiol 27:667–671
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Attar, A.M., Kharkhaneh, A., Etemadifar, M. et al. Serum Mercury Level and Multiple Sclerosis. Biol Trace Elem Res 146, 150–153 (2012). https://doi.org/10.1007/s12011-011-9239-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-011-9239-y