Abstract
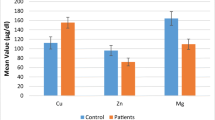
The purpose of the study was to determine the serum concentration of trace elements of panic disorder patients and to find out the relationship between trace element levels and nutritional status or socio-economic factors. The study was conducted among 54 panic disorder patients and 52 healthy volunteers. Patients were recruited from Bangabandhu Sheikh Mujib Medical University by random sampling. Serum trace element concentrations were determined by flame atomic absorption spectroscopy (for Mg, Zn, Ca, and Cu) as well as graphite furnace (for Mn). Data were analyzed by independent t test, Pearson’s correlation analysis, regression analysis, and ANOVA. The serum concentration of Mn, Zn, Ca, Cu, and Mg in panic disorder patients were 0.37 ± 0.30, 0.67 ± 0.20, 99.91 ± 15.15, 0.83 ± 0.23, and 21.14 ± 3.72 mg/L, while those were 0.4163 ± 0.2527, 0.86 ± 0.3, 106.6073 ± 18.6531, 0.8514 ± 0.3646, and 21.37 ± 2.03 mg/L in control subjects, respectively. The serum concentration of Zn decreased significantly (p = 0.001) in patient group. But the differences of the concentration of Mn, Ca, Cu, and Mg between patient and control group were not significant (p = 0.522, p = 0.065, p = 0.800, and p = 0.712, respectively). Socio-economic data reveal that most of the patients were very poor and middle aged. Mean BMIs of the control group (23.74 ± 2.71 kg/m2) and the patient group (22.62 ± 3.74 kg/m2) were within the normal range (18.5–25.0 kg/m2). There was no significant relationship between serum zinc level and BMI of patients (r = 0.038; p = 0.809). So the decreased level of serum zinc in panic disorder patients was not because of other reasons, but rather it may provide a prognostic tool for the diagnosis and treatment of this disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Panic disorder, as described by the Diagnostic and Statistical Manual of Mental Disorders Fourth Edition (DSM-IV), is a psychological and psychiatric condition characterized by recurring panic attacks in combination with significant behavioral change or at least a month of ongoing worry about the implications or concern about having other attacks. Panic disorder often begins in late adolescence or early adulthood and is twice as common in women as men [1]. Panic disorder is associated with several psychiatric conditions such as depression and other anxiety disorders [2]. Symptoms of panic disorder as described by DSM-IV include palpitations, sweating, shaking, shortness of breath, chest pain or discomfort, depersonalization, losing control, and numbness or tingling sensations [3]. Panic disorder is often accompanied by other serious problems such as depression, drug abuse, or alcoholism [4, 5]. Panic disorder may result from an abnormally sensitive fear network, which includes the prefrontal cortex, insula, thalamus, amygdala, and projections from the amygdala to the locus ceruleus, hypothalamus, periaqueductal gray substance, and parabrachial nucleus [6]. But the exact etiology of panic disorder (PD) remains a mystery. In panic disorder, the body’s normal “alarm system” tends to be triggered unnecessarily and the reason is not well understood [7]. However, distinguishing panic attacks from medical disorders is difficult [8]. Alternatively, the pathophysiology of panic disorder remains unknown and the diagnosis of panic disorder also remains difficult [9, 10], and currently there is no medical or laboratory diagnostic test to confirm panic disorder.
Trace elements play a dual role in the biological system through their interaction with biomolecules. They regulate a number of cellular metabolic reactions while a few of them act as etiological agents in many environmentally induced neurological disorders [11]. In addition, they are involved in the function of sub-cellular systems such as mitochondria as well as in membrane transport, nerve conduction, and muscle contraction. Some of them (Zn, Cu, Mn, and Se) act as antioxidants. The very complex balance of trace elements is crucial for all areas of maintaining human health, preventing as well as overcoming health problems [12]. Therefore, altered homeostasis of trace elements may adversely affect biological processes, and are associated with many diseases [13]. Copper is an important component of several metalloenzymes, including tyrosinase and dopamine hydroxylase [14]. It also plays a vital role in neurological disorders [15]. Zinc plays an essential role in many aspects of metabolism including the activity of more than 300 enzymes, the structure of many proteins, and control of genetic expression [16, 17]. Zinc is an important cofactor in many biochemical reactions essential for normal neurological function and a cofactor for several neurotransmitters. Zinc status affects basic processes of cell division, growth, differentiation, development, performance, and aging through its requirement not only for synthesis and repair of DNA, RNA, and protein but also for many other aspects of metabolism [18, 19]. Zinc is unevenly distributed within the brain particularly at high concentrations in the hippocampal mossy fiber system where it acts as a neuromodulator [20]. Interactions with inhibitory [21] and excitatory amino acid neurotransmitter are well known with Zn [22] and high concentration of zinc is neurotoxic [23]. Paul et al. stated that panic attacks are highly associated with copper toxicity. Zinc deficiency due to copper excess may contribute to this problem. Brain is a major target tissue for higher level of manganese. Manganese has been shown to accumulate in vivo in the mitochondria of brain areas associated with neurological symptoms [24]. Lower level of Mn plays a significant role in the development of schizophrenia possibly by interfering with the membrane stability [25]. Magnesium is a natural calcium channel blocker and significant magnesium deficiency has been reported in depression [26].
Calcium and magnesium are necessary for relaxation and proper nerve function. Calcium excites nerves; magnesium calms them down. Ca is also capable of inducing structural and cognitive deficits in schizophrenia and it has been proposed that altered Ca signaling may constitute the central unifying molecular pathology in schizophrenia [27]. A lack of calcium or magnesium may increase susceptibility to anxiety and panic. Low Mg/Ca ratio may trigger the stress response [28].
These observations suggest that alterations in essential trace elements like Cu, Zn, Mn, Ca, and Mg may play a role in the pathogenesis of panic disorder. As no work has been reported regarding serum trace element concentration in panic disorder patients, therefore attempts were made to investigate the correlation between trace element level and the degree of disorder, if any.
Materials and Methods
Fifty four panic disorder patients (44 males and 10 females of mean age 29 ± 7 years) were randomly recruited from Bangabandhu Sheikh Mujib Medical University (BSMMU) by a specialist psychiatrist who was trained in Diagnostic and Statistical Manual of Psychiatric Disorders (Fourth Edition) and 52 sex-, education-, and age-matched healthy volunteers (42 males and 10 females mean age 30 ± 6 years) were recruited purposively as control. The study subjects were briefed about the purpose of the study and written consent was obtained from each of them. All subjects had to go through clinical examinations to find out the existence of other diseases that might alter trace element level. Subjects also had to undergo a routine physical checkup including their organ activity, weight, nutritional condition, blood pressure, chest X-ray, and ECG. Hematocrit, blood urea, nitrogen, creatinine, glucose, and liver enzyme tests were also done for all subjects to find out their actual pathological condition. These subjects did not have diabetes, kidney failure, or other disease nor had they been treated with drugs, which can interfere with concentration of the elements (diuretics, antihypertensive drugs or mineral supplements, etc.). Patients who were mentally retarded and suffered from co-morbid psychiatric disorders were also excluded from the study. Socio-demographic data were also collected in a questionnaire form which contains important information including socio-economic data, history of illness, family history, etc. Ethical approval was obtained from the BSMMU authority.
Five milliliters of venous blood was drawn from each patient and controls after overnight fasting using a plastic syringe fitted with a stainless steel needle. The blood sample was collected into a metal-free plastic tube and allowed to clot at room temperature for 1 h, so that clotting factors could be removed from the serum. Then the blood sample was centrifuged at 3,000 rpm for 15 min at room temperature to separate the serum. The serum was aliquoted into an Eppendorf tube and was left to stand at −80°C until analysis. Blood collection and serum separation were carried out in a dust-free environment.
Analysis of the trace element was carried out by using flame atomic absorption spectrometry (Varian SpectraAA 220) as well as graphite furnace [29]. Serum samples were diluted by deionized water by a factor of 10. Different concentrations (0.5, 1.0, 2.0, 5.0, and 10.0 mg/L) of trace elements were used for calibration of standard graphs. Absorbances were read at 324.8, 213.9, 279.5, 285.2, and 422.7 nm for copper, zinc, manganese, magnesium, and calcium, respectively, in the atomic absorption spectrometer. To verify the assay accuracy and to maintain quality, the standard solutions were run for every 10-test sample. A software package (SpectrAA software) was used to calculate concentration of copper, zinc, manganese, magnesium, and calcium. The precaution for both collection and subsequent handling of serum were taken in order to avoid or minimize trace elements contamination. The results were expressed as mean ± SD. Differences between groups’ means were determined by independent sample t test. The significance level was set at p < 0.05. Pearson’s correlation analysis was performed to find out the correlation of BMI and socio-economic factors with the trace element concentration of the patients. A multiple regression analysis and one-way analysis of variance (ANOVA) were also performed to determine the extent of involvement of socio-economic factors and BMI that affects the level of trace elements.
Results and Discussion
Socio-economic data of the schizophrenic patients and control subjects have been shown in Table 1. It was found that most of the patients were literate (94.43%), non-smokers (59%), and very poor (income 91.95 ± 80.93 US $ or less per month). Average age of patient and control group was 29 ± 7 and 30 ± 6 years, respectively, which was very similar to previous findings with other psychiatric disorder like somatization disorder, schizophrenia, mania, depression, anxiety, etc. [30–32].
Serum trace element concentrations are presented in Table 2. The concentration of Zn decreased significantly (p < 0.05) while the change of concentrations of Ca, Cu, Mn, and Mg were not statistically significant (p > 0.05). Difference between the mean BMI of patients (22.62 ± 3.74 kg/m2) and controls (23.74 ± 2.71 kg/m2) was not significant (p = 0.100). Correlative analysis was also performed using the data of serum concentration of trace elements and BMI of panic disorder patients. It was found that there is no significant correlation with the trace elements and the BMI of patient group. Correlative analysis was also performed to evaluate the effect of BMI, age, and income of patient group on serum trace element levels. No significant correlation was observed (p > 0.05). Also using serum Zn concentration of patient as dependent variable and smoking as independent variable, regression analysis was also carried out to find out the effect of smoking on serum Zn levels. Regression analysis depicted that the concentration of Zn had not changed significantly with smoking behavior (R 2 = 0.00%, t = 0.077; p = 0.939). Finally, ANOVA test was done with the same parameters that did not show a significant relationship between smoking and serum Zn concentration of the patients (F = 0.006; p = 0.939).
Other studies showed that lower magnesium level can cause depression, behavior and personality changes, apathy, irritability, and anxiety [33]. Maes et al. conducted three consecutive studies and found that serum zinc levels were significantly lower in depressed patients as compared to healthy matched controls. Studies showed that serum Cu and Mn levels were significantly higher in somatization disorder patients than in controls [34]. Yanik et al. showed that plasma Cu concentrations were significantly higher (p < 0.01) and Mn and Fe concentrations were lower (p < 0.05 and p < 0.05, respectively) in schizophrenic patients than in controls. The same study observed that alterations in essential trace elements Mn, Cu, and Fe may play a role in the pathogenesis of schizophrenia [35].
Psychiatric manifestations of zinc deficiency include behavioral disturbances, depression, and mental confusion. Within major depression populations, lower zinc levels correlate with higher depression severity [36]. Zinc is commonly found to be deficient in people with depression. Mental confusion and mental illness are strongly associated with zinc deficiency. Present finding of significant correlation of Zn level with the panic disorder patients and no significant correlation of Zn with the BMI as well as smoking and gender differences justifies that zinc may be directly linked to this disease and zinc supplement may be beneficial to treat panic disorder patients.
References
Weissman MM, Bland RC, Canino GJ, Faravelli C, Greenwald S, Hwu HG, Joyce PR, Karam EG, Lee CK, Lellouch J, Lepine JP, Newman SC, Oakley-Browne MA, Rubio-Stipec M, Wells JE, Wickramaratne PJ, Wittchen HU, Yeh EK (1997) The cross-national epidemiology of panic disorder. Arch Gen Psychiatry 54(4):305–309
Kaufman J, Charney D (2000) Comorbidity of mood and anxiety disorders. Depress Anxiety 12(1):69–76
Diagnostic and Statistical Manual of Mental Disorders (2000) 4th edn. Text rev.: DSM-IV-R. American Psychiatric Association, Washington
Regier DA, Rae DS, Narrow WE, Kaelber CT, Schatzberg AF (1998) Prevalence of anxiety disorders and their comorbidity with mood and addictive disorders. Br J Psychiatry Suppl 34:24–28
Kushner MG, Sher KJ, Beitman BD (1990) The relation between alcohol problems and the anxiety disorders. Am J Psychiatry 147(6):685–695
Gorman JM, Kent JM, Sullivan GM, Coplan JD (2000) Neuroanatomical hypothesis of panic disorder, revised. Am J Psychiatry 157:493–505
Taylor (2006) Panic disorder. BMJ 332:951–955
Mary LH, Judd LL, James B, David B, Bernard B (1993) Panic Disorder Education Program ISBN 93-3508
Roy-Byrne PP, Craske MG, Stein MB (2006) Panic disorder 368(9540):1023–1032
Moore DP, Jefferson JW (2004) Handbook of Medical Psychiatry 2nd ed. Elsevier Mosby, St. Louis, pp 162–163
Sathyanarayana R, Luigi Z, Jagannatha R (2007) Trace metals, neuromelanin and neurodegeneration. An interesting area for research. Indian J Psychiatr 49(3):154–156
Charles B, Clayman (1989) American Medical Association. Am Med Assoc/Encycl Med 396:605–752
Muralidhar LH (2004) Serum trace element levels and the complexity of inter element relations in patients with Parkinson’s disease. J Trace Elem Med Biol 18:163–171
Livesay DA (2003) Conservation of electrostatic properties within enzyme families and superfamilies. Biochemistry 42(12):3464–3473
Wallwork JC (1987) Cu and central nervous system. Program Food Nutr Sci 11:203–247
Vallee B, Coleman J, Auld D (1991) Zinc fingers, zinc clusters, and zinc twists in DNA-binding protein domains. Proc Natl Acad Sci U S A 88:999–1003
Vallee B, Falchuk K (1993) The biochemical basis of zinc physiology. Physiol Rev 73:79–118
Blanchard R, Cousins R (1996) Differential display of intestinal mRNAs regulated by dietary zinc. Proc Natl Acad Sci U S A 93:6863–6868
Chesters J, Petrie L, Travis A (1990) A requirement for Zn2+ for the induction of thymidine kinase but not ornithine decarboxylase in 3T3 cells stimulated from quiescence. Biochem J 272:525–527
Xie X, Smart TG (1991) A physiological role for endogenous zinc in rat hippocampal synaptic neurotransmission. Nature 349:521–524
Smart TG, Constanti A (1990) Differential effect of zinc on the vertebrate GABA receptor complex. Br J Pharmacol 199:643–654
Yeh GC, Bonhaus DW, McNamara JO (1990) Evidence that zinc inhibits N methyl-D-aspartate receptor gated ion channel activation by noncompetitive antagonism of glycine binding. Mol Pharmacol 138:14–19
Koh JY, Choi DW (1988) Zinc alters excitatory amino acid neurotoxicity on cortical neurons. J Neurosci 8:2164–2171
Liccione JJ, Maines MD (1988) Selective vulnerability of glutathione metabolism and cellular defense mechanisms in rat striatum to manganese. ASPET 247(1):156–161
Pfeiffer CC, LaMola S (1983) Zinc and manganese in the schizophrenias. J Orthomol Psychiatry 12(3):28–48
Cox IM, Campbell MJ, Dowson D (1991) Red blood cell magnesium and chronic fatigue syndrome. Lancet 337:757–760
Lidow MS (2003) Calcium signaling dysfunction in schizophrenia: a unifying approach. Brain Res Rev 43(3):285
Seelig SM (1994) Consequences of magnesium deficiency on the enhancement of stress reactions; preventive and therapeutic implications. J Am Coll Nutr 13(5):429–446
Czupryn M, Falchuk KH, Stankiewicz A, Vallee BL (1993) A Euglena gracilis zinc endonuclease. Biochemistry 32(5):1204–1211
Karim P, Hossain I, Sadat AFMN, Nahar Z, Hossain K, Hasnat A (2006) Serum level of cadmium, calcium, lead and iron in schizophrenic patients. Dhaka Univ J Pharm Sci 5(1–2):9–13
Baker A, Sadat AFMN, Rahman L, Qusar S, Huq I, Hossain I, Hasnat A (2005) Serum trace elements in manic patients. Dhaka Univ J Pharm Sci 4(2):99–102
Bergquist J, Bergquist S, Axelsson R, Ekman R (1993) Demonstration of immunoglobulin G with affinity for dopamine in cerebrospinal fluid from psychotic patients. Clin Chim Acta 217(2):129–142
Wacker WE, Parisi AF (1968) Magnesium metabolism. NEJM 278(14):772–776
Hossain I, Islam SN, Khan MNI, Islam S, Hasnat A (2007) Serum level of copper, zinc and manganese in somatization disorder patients. Ger J Psychiatr ISSN 1433–1055
Yanik M, Kocyigit A, Tutkun H, Vural H, Herken H (2004) Plasma manganese, selenium, zinc, copper, and iron concentrations in patients with schizophrenia. Biol Trace Elem Res 98(2):109–117
Maes M, Haese PC, Scharpe S, Hondt P, Cosyns P, Broe ME (1994) Hypozincemia in depression. J Affect Disord 31(2):135–140
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nahar, Z., Azad, M.A.K., Rahman, M.A. et al. Comparative Analysis of Serum Manganese, Zinc, Calcium, Copper and Magnesium Level in Panic Disorder Patients. Biol Trace Elem Res 133, 284–290 (2010). https://doi.org/10.1007/s12011-009-8441-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-009-8441-7