Abstract
Pregnant women in developing countries are vulnerable to multiple micronutrient deficiencies. Studies assessing serum levels of the micronutrients and magnitude of their deficiencies are very scarce in African subjects. This study was aimed at determining serum levels of micronutrients in 375 pregnant (42 HIV seropositive) and 76 non-pregnant women (20 HIV seropositive) who visited the University of Gondar Hospital, Gondar, Ethiopia. Serum concentrations of zinc,\ copper, selenium, calcium, and magnesium were determined using an inductively coupled plasma mass spectrometer. Irrespective of HIV serostatus, pregnant women had significantly higher serum concentrations of copper and copper/zinc ratio and significantly lower magnesium compared to those in non-pregnant women (P < 0.05). Except for selenium, which was significantly lower in HIV-seropositive pregnant women (P < 0.05), the mean serum concentrations of zinc, copper, calcium, and magnesium were not significantly different between pregnant women by HIV serostatus. The prevalence of deficiency in zinc, magnesium, selenium, and calcium in the pregnant women, irrespective of their HIV serostatus, was 66.7%, 25.6%, 21.9%, and 9.3%, respectively. The magnitude of deficiency in zinc, magnesium, and selenium was significantly higher in HIV seropositive pregnant women (76.2%, 52.4%, and 45.2%) than that in HIV-seronegative pregnant women (65.5%, 22.2%, and 18.9%) and in HIV-seronegative non-pregnant women (42.9%, 8.1%, and 30.4%; P < 0.05). Deficiency in one, two, three, or four mineral elements was observed in 44.8%, 14.4%, 9.9%, and 5.1% of the pregnant women, respectively. Only 25.9% of the pregnant women and 44.7% of the non-pregnant women were not deficient in any of the micronutrients. The high prevalence of micronutrient deficiencies in pregnant and non-pregnant women in Gondar, Ethiopia warrants the need for strategies on prevention and control of the deficiencies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Minerals such as zinc, copper, selenium, calcium, and magnesium are essential for normal human development and functioning of the body [1]. They have been implicated to play important roles in immuno-physiologic functions [1, 2]. For example, zinc is an integral part of more than 200 enzymes and has significant task in nucleic acid metabolism, cell replication, tissue repair, and growth. Its deprivation leads to profound alteration of thymic function with resultant loss of T-cell-mediated responses and increased susceptibility to infectious diseases [1, 2]. The antioxidation functions of selenium in glutathione peroxidase is essential in protecting the biological system from oxidation caused by peroxides. Superoxide dismutases, which usually contain copper and/or zinc, act as antioxidants against superoxides. Calcium plays an important role in muscle contraction and regulation of water balance in cells. Modification of plasma calcium concentration leads to the alteration of blood pressure. Magnesium has been known as an essential co-factor for many enzyme systems. It also plays an important role in neurochemical transmission and peripheral vasodilation [1].
Deficiency of the minerals in women of reproductive age is recognized as a major public health problem in many developing countries [3–7]. Pregnant women are particularly vulnerable to nutritional deficiencies because of the increased metabolic demands imposed by pregnancy involving a growing placenta, fetus, and maternal tissues, coupled with associated dietary risks [8, 9]. In turn, the maternal undernutrition can have adverse effects such as anemia, pregnancy-induced hypertension, complications of labor, and even death. Furthermore, the fetus can be affected, resulting in stillbirth, pre-term delivery, intrauterine growth retardation, congenital malformations, reduced immunocompetence, and abnormal organ development [9, 10].
The essential nature of minerals has been recognized through the identification of clinical conditions associated with severe deficiencies of particular minerals and through subsequent animal experiments. Although the importance in pregnancy of a few mineral deficiencies, such as iron and iodine, has been long recognized, the role of many others is only recently becoming appreciated [3]. Gaining this understanding is complicated by the finding that mineral deficiencies often co-exist and that deficiencies of specific minerals vary by stage of life, economic status, health status, etc. This variability is due to consumption of diets with differing content and bioavailability of micronutrients and differing losses of and requirements for micronutrients [3].
Maternal iron deficiency and the consequent anemia comprise a major problem in developing countries, affecting >50% of women during pregnancy [5, 6]. Other micronutrient deficiencies are likely to be widely prevalent, especially those of iodine, zinc, and vitamin A [1, 8, 9]. Although several studies have documented the status of one or two micronutrients among pregnant women [11–14], little information is available on the extent of multiple micronutrient deficiencies during pregnancy [15, 16]. Therefore, the present study was aimed to determine serum levels of zinc, copper, selenium, calcium, and magnesium in Ethiopian pregnant women and to compare these to those in apparently healthy non-pregnant women.
Materials and Methods
Design, Area, and Period of Study
Pregnant women attending routine antenatal care from March to June 2005 at the University of Gondar Hospital in Gondar, Ethiopia were enrolled at their first visit. As controls, apparently healthy volunteer non-pregnant women, who were living in the same geographic locality with the pregnant women, were recruited. The University of Gondar Hospital is a tertiary-level teaching- and service-rendering institution that provides health service to over 4 million people in Northwest Ethiopia. Informed consent was obtained from the participants, and the study was ethically approved by the Research Ethics Committee of the University of Gondar and The University of Tokushima, Japan.
Anthropometry
Body weight was determined to the nearest 0.1 kg on an electronic digital scale, and height was measured to the nearest 0.1 cm. Body mass index (BMI), defined as the weight in kilogram of the individual divided by the square of the height in meter, was used to determine the nutritional status of the participants into severe malnutrition (BMI < 15.9 kg/m2), moderate malnutrition (BMI = 16–16.9 kg/m2), mild malnutrition (BMI = 17–18.4 kg/m2), normal (BMI = 18.5–24.9 kg/m2), overweight (BMI = 25–29.9 kg/m2), and obese (BMI > 30).
Blood Collection, Serum Separation, and HIV Serology
About 5 ml of venous blood was collected from the participants. After clot was retracted, the blood samples were centrifuged, and sera were separated from the cells following standard procedures. The sera were tested for the presence of HIV-1 antibodies using rapid HIV-1 diagnostic test kits following the manufacturers’ instructions. The results were interpreted following the current national algorithm for screening of sera for HIV-1 infection [17]. The remaining sera were kept frozen at −20oC until used.
Determination of Trace Elements in Serum
The concentration of minerals in serum was determined using an inductively coupled plasma mass spectrometer (ICP-MS; model 8500, Schimadzu, Tokyo, Japan) following previously published procedures [18]. In brief, serum sample (200 μl) was aliquoted into Teflon tube and covered with a Teflon ball. After adding 1 ml of concentrated HNO3 (Wako Pure Chemicals, Japan), the tube was heated at 120°C for 5 h on an aluminum heating block (IWAKI, Asahi Techno Glass, Japan). The sample was further heated almost to dryness at 200°C after removing the Teflon ball. Finally, the residue was dissolved with 2 ml of 0.1 M HNO3, which contained 10 ng/ml internal standard elements (In, Re, and Tl). The diluted serum solution was used for analysis of the elements in ICP-MS. Commercially available single-element standard solutions (1,000 μg/ml) were purchased from Wako Pure Chemicals (Osaka, Japan) and used for standardization of calibration curves.
Statistical Analysis
Data were analyzed using Statistical Package for the Social Sciences (SPSS) version 10 statistical package (SPSS, Chicago, IL, USA). A one-sample Kolmogorov–Smirnov test was used to assess whether the data were normally distributed. Comparisons of serum values of the trace elements among pregnant women with and without HIV co-infection and control groups were made using a one-way analysis of variance. Post hoc Tukey test was used to determine which pairs of means differ significantly. Deficiency of zinc, selenium, copper, magnesium, and calcium was defined at their serum levels less than 75, 7, and 75 μg/dl and 1.8 and 8.4 mg/dl, respectively [19]. P values less than 0.05 were considered statistically significant.
Results
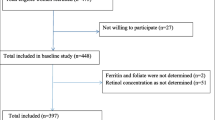
A total of 375 pregnant women and 76 apparently healthy non-pregnant women (controls) were included in this study. About 42 of the pregnant women and 20 of the controls were found co-infected with HIV. Table 1 shows the age distribution and anthropometric features of the participants. Majority of the study subjects were in the age group 20–39 years. The mean BMI of the pregnant women (23.87 ± 9.36) was significantly higher than that in the non-pregnant women (20.54 ± 2.25). Only 2.1% and 10.5% of the pregnant and non-pregnant women, respectively, had BMI less than 18.5 kg/m2.
Table 2 shows the concentrations of serum magnesium, calcium, copper, zinc, and selenium in pregnant women and controls. The mean serum level of selenium was significantly lower in pregnant women with HIV co-infection compared to that in HIV-seronegative pregnant women (P < 0.05). The mean concentrations of magnesium, calcium, copper, and zinc were not significantly different between pregnant women with and without HIV co-infection. However, the ratio of copper to zinc concentration in serum was significantly higher in HIV-seropositive pregnant women than that in HIV-uninfected pregnant women (P < 0.05). Irrespective of HIV serostatus, pregnant women had significantly higher serum concentrations of copper and copper-to-zinc ratio and significantly lower serum levels of magnesium compared to those in healthy controls (P < 0.05). Pregnant women without HIV co-infection had significantly lower serum magnesium concentration compared to controls without HIV infection (P < 0.05). On the contrary, the pregnant women without HIV co-infection had a significantly higher concentration of copper (P < 0.05) and a remarkably lower serum zinc level when compared to that in HIV-seronegative pregnant women, although not statistically significant. As a result, the ratio of copper to zinc concentration was significantly higher in serum of pregnant women without HIV infection as compared to controls without HIV co-infection. On the other hand, except for serum concentrations of copper and copper-to-zinc ratio, the levels of calcium, zinc, and selenium in HIV-seropositive pregnant women were not significantly different when compared to their corresponding levels in controls with HIV co-infection. Non-pregnant women did not have significant difference in concentrations of copper, zinc, selenium, and copper-to-zinc ratio by HIV serostatus. However, only serum calcium was significantly higher in sera of those without HIV co-infection compared to its level in HIV-seropositive non-pregnant women (P < 0.05).
Table 3 shows the distribution of pregnant women and healthy controls by serum status of minerals and HIV infection. The prevalence of deficiency in zinc, magnesium, selenium, and calcium in the pregnant women, irrespective of their HIV serostatus, was 66.7%, 25.6%, 21.9%, and 9.3%, respectively. The magnitude of deficiency in zinc, magnesium, and selenium was significantly higher in HIV-seropositive pregnant women (76.2%, 52.4%, and 45.2%) than that in HIV-seronegative pregnant women (65.5%, 22.2%, and 18.9%; P < 0.05). Deficiency in one, two, three, or four micronutrients was observed in 44.8%, 14.4%, 9.9%, and 5.1% of the pregnant women, respectively. Only 25.9% of the pregnant women and 44.7% of non-pregnant women were not deficient in any of the micronutrients (Table 4).
Discussion
This study demonstrated that 74.1% of pregnant women and 55% of controls in Gondar, Ethiopia were deficient in serum levels of one or more micronutrients. The deficiencies were more pronounced in the women with HIV co-infection. In fact, infection with HIV is a major health problem in the country in general and among pregnant women in particular [20]. Antenatal-based sentinel surveillance data from 44 rural and 38 urban sites indicated an HIV prevalence of 2.2% and 9.5%, respectively, in pregnant women in the year 2005 [20]. Infection with HIV has long been shown to affect nutritional status.
Pregnancy is associated with continuous physiologic adjustments that affect the metabolism of all nutrients [8]. About 40 days after conception, the placenta, a complex organ of internal secretions, releases numerous hormones and enzymes into the maternal circulation, which affect the metabolism of all nutrients. These adjustments in nutrient metabolism in addition to changes in the anatomy and physiology of the mother support fetal growth and development while maintaining maternal homeostasis and preparing for lactation [8]. These processes account for the raised metabolic rate in the pregnant state.
We observed zinc deficiency in 66.7% of the pregnant women and in 46.1% of the non-pregnant women irrespective of their HIV serostatus. This finding is comparable to studies on pregnant women from elsewhere where prevalences ranging from 22% to as high as 73.5% were reported [12, 15, 16, 21]. The higher prevalence of zinc deficiency found in the present study could be due to an inadequate zinc intake and poor bio-absorption. It is worthy to note that several dietary factors are known to affect zinc absorption as a result of physico-chemical interactions in the intestine. Phytate, a component in plants with the highest concentration found in seeds (cereal grains/legumes/nuts), inhibits zinc absorption [22]. This might hold true in the subjects of the present study, as the staple foods in Gondar and its surrounding areas are cereal based.
The high prevalence of zinc deficiency has a far-reaching implication, as zinc is an important element performing a range of functions in the body. Zinc is a co-factor for the synthesis of a number of enzymes, DNA, and RNA [1]. Zinc deficiency has been associated with complications of pregnancy and delivery, as well as with growth retardation and congenital abnormalities in the fetus [3]. During pregnancy, there is a decline in circulating zinc and a decline also occurs as pregnancy progresses, possibly due to a decrease in zinc binding and increased transfer of zinc from the mother to the fetus [14]. Several studies globally have documented relationships between lowered zinc concentrations during pregnancy and low birth weight, suggesting that there is a threshold for serum zinc concentration below which adverse pregnancy outcome increases significantly [3, 23].
In this study, unlike zinc, no pregnant or non-pregnant women were found deficient in serum copper levels. Studies have reported that the increase in serum copper during pregnancy is mainly due to an increase in its carrier proteins, ceruloplasmin, which increase in response to stimulation by elevated levels of maternal estrogens [13, 24]. In line with our observation, a very low prevalence (2.7%) of copper deficiency was reported from pregnant women in India [16] and China [15]. Copper is an important trace element involved in the function of several cuproenzymes that are essential for life [25]. Copper deficiency during embryonic and fetal development can result in numerous gross structural and biochemical abnormalities, as the fetus is fully dependent on the maternal copper supply [26].
Deficiency in magnesium was also high in the pregnant women of the present study, as 25.6% of them had its serum levels <1.80 mg/dl. However, this prevalence is lower than the 43.6% [16] and 51.5% [15] deficiency reported in Indian and Chinese pregnant women. The low serum magnesium status during pregnancy could be due to an active transport of the element across the placenta into the fetus [27]. Low dietary intake coupled with increase in demand for growth and accelerated metabolism during pregnancy could be the other contributing factors [28]. The mineral is essential for proper bone formation and in various intracellular enzymatic processes [29]. Deficiency of magnesium may possibly be associated with pre-eclampsia, pre-term delivery, and low birth weight [30]. It has been documented that magnesium deficiency during gestation significantly increases neonatal mortality and morbidity [31].
We observed selenium deficiency in a large number of pregnant women, especially in those with HIV co-infection. Selenium is an integral part of the enzyme glutathione peroxidase, which forms a major cellular defense system against oxidative injury [32]. Selenium deficiency has been incriminated in the causation of several diseases including malignancies. The selenium requirement of pregnant and lactating women increases as a result of selenium transport via the placenta and via breast milk [33]. Blood and serum selenium levels therefore decrease during pregnancy and are lower than those in non-pregnant women [33].
Serum calcium is composed of protein-bound calcium, diffusible calcium complexes, and ionized calcium. Its serum concentration is so exquisitely regulated with the normal serum concentrations ranging from 8.5 to 10.5 mg/dl in adults. However, we observed relatively higher mean serum calcium in the Ethiopian pregnant women in the present study. In line with our finding, Hamlin [34] ascribed the lowest rates of edema, proteinuria, and hypertension-gestosis in Ethiopian pregnant woman to diets high in calcium. A study involving Swedish pregnant women indicated a decrease in serum calcium as pregnancy advances, where its mean level declined from 2.235 ± 0.097 mM/l at week 10 to 2.353 ± 0.097 mM/l at week 32 [35]. The decreased serum calcium levels were thought to be due to consequences of increased blood volume and transfer of calcium to the fetus [35].
This study also showed that deficiencies of minerals occur concurrently in pregnant women (29.4%) and controls (19.7%). This indicates the coexistence of micronutrient deficiencies probably due to common etiology and underlying mechanisms. For instance, a diet rich in phytate and low in animal proteins, as is common in most developing countries including Ethiopia, predisposes one to insufficient intake and absorption of zinc [22, 36]. In addition, direct interactions between micronutrients were described previously, such as the antagonistic effect of iron supplementation on zinc uptake, although the mechanisms underlying these interactions are not yet fully understood [22]. Apart from the dietary factors, the pregnant women living in developing countries are exposed to recurrent respiratory and diarrheal infections, prolonged lactation, and recurrent pregnancy, all of which can influence micro-nutrition [4].
In summary, the findings of the present study reveal a high prevalence of micronutrient deficiencies, individually as well as concomitantly, among the pregnant and non-pregnant women in Gondar. These calls for the need to undertake multicentric studies in various parts of the country to substantiate the data obtained in the present study so that any intervention measures, if required, can be initiated.
References
WHO (1996) Trace elements in human nutrition and health. WHO, Geneva, Switzerland
Failla ML (2003) Trace elements and host defense: recent advances and continuing challenges. J Nutr 133:1443S–1447S
Black RE (2001) Micronutrients in pregnancy. Br J Nutr 85:S193–S197
Abdulla M, Khan AH, Reis MF (1996) Trace element nutrition in developing countries. Asia Pacific J Clin Nutr 5:186–190
Ramakrishnan U (2002) Prevalence of micronutrient malnutrition worldwide. Nutr Rev 60:S46–S52
Seshadri S (2001) Prevalence of micronutrient deficiency particularly of iron, zinc and folic acid in pregnant women in South East Asia. Br J Nutr 85:S87–S92
Christian P (2003) Micronutrients and reproductive health issues: an international perspective. J Nutr 133:1969S–1973S
King JC (2000) Physiology of pregnancy and nutrient metabolism. Amer J Clin Nutr 71:1218–1225
Kontic-Vucinic O, Sulovic N, Radunovic N (2006) Micronutrients in women's reproductive health: II. Minerals and trace elements. Int J Fertil Womens Med 51:116–124
Ramakrishnan U, Manjrekar R, Rivera J, Gonzales-Cossio T, Martorell R (1999) Micronutrients and pregnancy outcome: a review of the literature. Nutr Res 19:103–159
Seydoux J, Girardin E, Paunier L, Beguin F (1992) Serum and intracellular magnesium during normal pregnancy and in patients with pre-eclampsia. Br J Obstet Gynecol 99:207–211
Yasodhara P, Ramaraju LA, Raman L (1994) Trace minerals in pregnancy 1. Copper and zinc. Nutr Res 11:15–21
Martin-Lagos F, Navarro-Alarcon M, Terres-Martos C, Lopez-Garcia de la Serrana H, Perez-Valero V, Lopez-Martinez MC (1998) Zinc and copper concentrations in serum from Spanish women during pregnancy. Biol Trace Elem Res 61:61–70
Tamura T, Goldenberg RL, Johnston KE, DuBard M (2000) Maternal plasma zinc concentrations and pregnancy outcome. Am J Clin Nutr 71:109–113
Ma AG, Chen XC, Xu RX, Zheng MC, Wang Y, Li JS (2004) Comparison of serum levels of iron, zinc and copper in anaemic and non-anemic pregnant women in China. Asia Pac J Clin Nutr 13:348–352
Pathak P, Kapil U, Kapoor SK et al (2004) Prevalence of multiple micronutrient deficiencies amongst pregnant women in a rural area of Haryana. Indian J Pediatr 71:1007–1014
Ethiopian Health and Nutrition Research Institute (2002) Manual for HIV-1 diagnosis. Ethiopian Health and Nutrition Research Institute, Addis Ababa, Ethiopia
Kassu A, Yabutani T, Mahmud ZH et al (2006) Alterations in serum levels of trace elements in tuberculosis HIV infection. Eur J Clin Nutr 60:580–586
Sauberlich HE (1999) Laboratory tests for the assessment of nutritional status, 2nd edn. CRC, Boca Raton
Ethiopian Ministry of Health (2006) AIDS in Ethiopia, 6th edn. Ethiopian Ministry of Health, Addis Ababa, Ethiopia
Kapil U, Pathak P, Singh P, Singh C (2002) Zinc and magnesium nutriture amongst pregnant mothers of urban slum communities in Delhi: a pilot study. Indian Pediatr 39:365–368
Gibson RS (1994) Zinc nutrition in developing countries. Nutr Res Rev 7:151–173
Krebs NF (2000) Overview of zinc absorption and excretion in the human gastrointestinal tract. J Nutr 130(5S Suppl):1374S–1377S
Kalra R, Kalra VB, Sareen PM, Khandelwal R (1989) Serum copper and ceruloplasmin in pregnancy with anaemia. Indian J Pathol Microbiol 32:28–32
Olivares M, Uauy R (1996) Copper as an essential nutrient. Am J Clin Nutr 63:791–796
Creizel AE (1995) Nutritional supplementation and prevention of congenial abnormalities. Curr Opin Obstet Gynecol 2:88–94
Gupta AP, Bhandari B, Gupta A (1984) Serum copper, zinc, magnesium and calcium in neonates. Indian Pediatr 21:469–473
Hall DG (1957) Serum magnesium during pregnancy. Obstet Gynecol 9:158–162
Grollman A, Grollman EF (1965) Pharmacology and therapeutics, 6th edn. Lea, Philadelphia
Chien PFW, Khan KS, Arnott N (1996) Magnesium sulphate in the treatment of eclampsia and pre-eclampsia: an overview of the evidence from randomized trials. Brit J Obstetr Gynecol 103:1085–1091
Almonte RA, Heath DL, Whitehall J, Russell MJ, Pathole S, Vink R (1999) Gestational magnesium deficiency is deleterious to fetal outcome. Biol Neonate 76:26–32
McKenzie RC, Rafferty TS, Beckett GJ (1998) Selenium: an essential element for immune function. Immunol Today 19:342–345
Zachara BA, Wardak C, Didkowski W, Maciag A, Marchaluk E (1993) Changes in blood selenium and glutathione concentrations and glutathione peroxidase activity in human pregnancy. Gynekol Obstet Invest 35:12–17
Hamlin RJH (1962) Prevention of preeclampsia. Lancet 1:864–865
Lagerkvist BJ, Ekesrydh S, Englyst V, Nordberg GF, Söderberg HA, Wiklund DE (1996) Increased blood lead and decreased calcium levels during pregnancy: a prospective study of Swedish women living near a smelter. Am J Public Health 86:1247–1252
Lonnerdal B (2000) Dietary factors influencing zinc absorption. J Nutr 130:1378S–1383S
Acknowledgments
Financial support was provided by University of Gondar, Ethiopia and partly by Sasakawa Scientific Research Grant from the Japan Science Society (no. 17-241).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kassu, A., Yabutani, T., Mulu, A. et al. Serum Zinc, Copper, Selenium, Calcium, and Magnesium Levels in Pregnant and Non-Pregnant Women in Gondar, Northwest Ethiopia. Biol Trace Elem Res 122, 97–106 (2008). https://doi.org/10.1007/s12011-007-8067-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-007-8067-6