Abstract
Background
Although Legg-Calvé-Perthes’ disease (LCPD) is frequently associated with varying degrees of femoral head deformity and leg length discrepancy (LLD), no factors that predict residual shortening have been clearly identified.
Questions/purposes
We attempted to determine whether (1) the extent of femoral head involvement; (2) varus osteotomy; and (3) patient demographic characteristics are associated with LLD at skeletal maturity in patients with LCPD.
Methods
We retrospectively reviewed the records of 168 skeletally mature patients with unilateral LCPD. The mean age at diagnosis was 7 years (range, 2–14 years). The extent of femoral head involvement was determined from the initial radiographs using the Herring lateral pillar and Catterall classifications. LLD was defined as shortening by ≥ 1.0 cm as measured from scanograms. The patient’s sex and the treatment modalities used were also recorded.
Results
LLD ranging from 10 to 38 mm (mean, 19 mm) occurred in 93 (55%) patients and was associated with the extent of femoral head involvement. Varus osteotomy was not associated with residual shortening. The patient’s age at diagnosis did not affect the LLD at skeletal maturity. The strongest predictor of LLD was a lateral pillar classification of B/C or C (odds ratio, 3.5; 95% confidence interval, 1.39–8.79).
Conclusions
The extent of femoral head involvement, but not the patient’s age at diagnosis or sex or the treatment modality, can predict the LLD at skeletal maturity in patients with unilateral LCPD.
Level of Evidence
Level II, prognostic study. See Guidelines for Authors for a complete description of levels of evidence.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Legg-Calvé-Perthes’ disease (LCPD) frequently results in varying degrees of femoral head deformity and leg length discrepancy (LLD) despite attempts to preserve the anatomy of the femoral head and proximal femoral physis [9, 14, 35]. The consequences of LCPD such as residual deformity of the femoral head are related to the restriction of joint mobility [36], femoroacetabular impingement [10, 33], pain [13], and premature osteoarthritis [2]. LLD can also lead to abductor weakness and limping.
Several parameters reportedly predict the shape of the femoral head after healing. These include the age at diagnosis [17, 32], head-at-risk sign [29], and the extent of femoral head involvement [6]. However, most studies have focused on the deformation of the spherical shape of the affected femoral head as evaluated radiographically.
Several analyses of the relationship between LCPD and LLD have documented shortening of the affected limb ranging in length from 1 mm to 7 cm [9, 14, 28, 31, 35, 39]. However, the incidence and predictors of LLD in patients with LCPD remain unclear; furthermore, it is not known whether parameters such as age at diagnosis, sex, type of treatment, and the extent of femoral head involvement can predict the occurrence of LLD.
Our goal was to determine whether (1) the extent of femoral head involvement; (2) varus osteotomy; and (3) patient demographic characteristics are associated with LLD at skeletal maturity in patients with LCPD.
Patients and Methods
We retrospectively reviewed the medical records of 168 patients who had been diagnosed with LCPD from April 2004 through February 2008. All patients had reached skeletal maturity by the time of the last followup visit (at least 4 years after diagnosis in all patients). Our criteria for skeletal maturity were age ≥ 17 years for males or ≥ 15 years for females or the presence of radiographic physeal closure.
Two of the authors (KWP, KSJ) determined the skeletal maturity at the time of the last followup visit by examining the closure of the distal femoral and proximal tibial physes on AP and lateral radiographs of the lower extremities. The authors agreed that a physis had reached closure when the radiolucent gap between the epiphysis and metaphysis of the distal femur and proximal tibia had disappeared. If the authors disagreed on closure of the gap, skeletal maturity was considered to be the midpoint of a period during which the length of the lower limb did not change in two successive radiological measurements [21]. Scanograms were obtained to measure leg lengths at each visit to our clinic including the initial visit. Patients with bilateral LCPD, an initial LLD exceeding 1.0 cm, or other underlying diseases or risk factors related to osteonecrosis were excluded from our study. We enrolled 146 (87%) male and 22 (13%) female patients with a mean age at diagnosis of 7 years (range, 2–14 years). No patients were lost to followup. The minimum duration of followup was 4 years (mean, 9 years; range, 4–25 years). All data were obtained from medical records and radiographs, and no patient was called back for an additional visit for the purpose of the study.
Two of the authors (KWP, KSJ) classified the extent of femoral head involvement of the affected hips from AP and lateral plain radiographs obtained at the time of presentation according to Catterall’s four-category classification system [6]. The estimated kappa value for the interobserver reliability of Catterall’s classification system was 0.51. These were recategorized into a simpler two-group classification [38] by combining Catterall Group 1 with Group 2 and Groups 3 with Group 4. We also used the lateral pillar classification developed by Herring et al. [16, 18], which classifies hips into Groups A, B, B/C border, and C. In this study, the hips were classified according to the simplified two-group classification, in which Groups A and B together represent mild involvement and Groups B/C border and C severe involvement of the femoral head. The kappa value for the interobserver reliability of Herring’s lateral pillar classification system was 0.79.
The treatment modalities used in these cases included physiotherapy alone, abduction bracing, adductor tenotomy, varus osteotomy of the proximal femur, innominate osteotomy of the pelvis, and shelf acetabuloplasty. The indications for physiotherapy were (1) age ≤ 6 years; (2) a lateral pillar classification of A; and (3) full ROM of the hip. The indications for abduction bracing were (1) age ≤ 8 years; (2) a lateral pillar classification of A or B; and (3) full ROM of the hip. In cases in which the motion of the hip was limited, with adductor tightness after 2 weeks of traction, the joint status was evaluated by arthrogram obtained under general anesthesia. Adductor tenotomy was considered for patients with a spherical femoral head, full ROM, and no hinged abduction. The indications for varus osteotomy of the proximal femur were (1) age ≥ 8 years; (2) a lateral pillar classification of B, B/C border, or C; and (3) no hinged abduction. A single patient, who was 14 years old at the time of the initial assessment, was treated with Salter innominate osteotomy and shelf acetabuloplasty. The contraindications for surgery were (1) age ≤ 8 years; and (2) a lateral pillar classification of A. We considered all of these approaches to be containment methods. To determine the effect of the treatment modality on LLD, we divided the patients into two groups: a nonoperative group consisting of patients who were treated with physiotherapy alone, abduction bracing, or adductor tenotomy and an operative group consisting of patients who underwent varus osteotomy, innominate osteotomy, or shelf acetabuloplasty. One hundred four patients (62%) were treated with physiotherapy alone, abduction bracing, or adductor tenotomy, whereas 64 patients (38%) were treated surgically (63 of 64 underwent varus osteotomy of the proximal femur).
Chi square tests were used to determine if the dichotomized age, sex, and treatment modality used differed between the femoral involvement groups as classified by each system: Groups I and II versus III and IV and Groups A and B versus B/C and C. The age at diagnosis, sex, and treatment modality used did not differ between the two groups for either classification system (Table 1).
Two of the authors (KWP, KSJ) measured the residual LLD from scanograms taken at the final followup visit. A difference in length between the legs of < 1.0 cm was considered normal, a difference ≥ 1.0 cm to < 2.5 cm was considered mild residual LLD, and a difference ≥ 2.5 cm was considered moderate-to-severe residual LLD. Patients with an LLD ≤ 2.5 cm usually required no active intervention and at most needed only a shoe lift [12]. We estimated the intraclass correlation coefficient values and corresponding 95% confidence intervals (CIs) for the interobserver reliability of the measured residual LLD. These were calculated with a two-way random effect model assuming a single measurement and absolute agreement. The estimate of interobserver reliability of residual LLD was 0.96 (95% CI, 0.91–0.99).
Chi square tests were used to determine whether potential predictive factors (sex, age at diagnosis, treatment modality, Catterall classification, and lateral pillar classification) differed among the three LLD groups (Table 2). We used multinomial logistic regression analysis to determine whether varus osteotomy was independently associated with the LLD at skeletal maturity. As described, rather than analyzing the length of the LLD as a continuous variable, we divided the patients into three groups according to their LLD measurements. The group with an LLD < 1.0 cm, which was considered normal, was used as the reference group. Separate odds ratios (ORs) were determined for the treatment modality (nonoperative versus operative) for each LLD group with the exception of the reference category (no LLD group), which was omitted from the analysis. We used multiple logistic regression analysis to determine whether any of the potential predictors were associated with LLD. The group with LLD < 1.0 cm was again used as the reference category, and separate ORs were determined for the mildly and moderately to severely affected groups.
Results
The extent of femoral head involvement was associated with the degree of LLD. LLD was found in 93 (55%) patients and ranged in magnitude from 10 to 38 mm (mean, 19 mm). The mean amount of shortening was 12 mm (range, 0–38 mm) in skeletally mature patients and 19 mm in patients with LLD. The modified two-group Catterall classification (I and II versus III and IV) and modified two-group lateral pillar classification (A and B versus B/C border and C) were associated with the LLD at skeletal maturity (p = 0.02 for both).
Treatment with varus osteotomy was not associated with the LLD (OR for mild LLD, 0.94; 95% CI, 0.48–1.84; p = 0.85; OR for moderate to severe LLD, 1.02; 95% CI, 0.39–2.66; p = 0.97). The degree of LLD did not differ with respect to age at diagnosis, sex, or treatment modality.
The strongest association was between modified Herring Group II (lateral pillar B/C and C) and mild LLD (OR, 3.6; 95% CI, 1.41–9.14; p = 0.01) followed by that between modified Catterall Group B (III and IV) and moderate-to-severe LLD (OR, 3.32; 95% CI, 0.97–11.4; p = 0.06; Table 3). An age of onset ≥ 6 years, male sex, and operative treatment were not associated with LLD.
Discussion
The shape of the femoral head and congruency of the hip after healing stage in patients who have been treated for LCPD are determined by the extent of femoral head involvement at the time of diagnosis (Fig. 1). However, shortening of the affected limb to a variable degree is very common (Fig. 2). LLD in patients with LCPD may be caused by many different factors, including the extent of femoral head involvement [14, 22], premature closure of the capital femoral physis [1, 3, 24, 35], disuse atrophy caused by bracing or nonweightbearing [20], and varus osteotomy of the proximal femur [7, 14, 26]. However, the association between femoral head involvement as graded by different classification systems and LLD at skeletal maturity remains unclear. Furthermore, varus osteotomy of the proximal femur, one of the procedures most commonly performed for containment of LCPD, can cause iatrogenic shortening. The prognostic factors for residual shortening in patients with LCPD are also unclear. We therefore investigated whether (1) the extent of femoral head involvement; (2) varus osteotomy; and (3) patient demographic characteristics are associated with the degree of LLD at skeletal maturity.
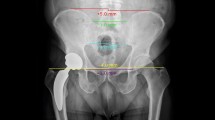
Serial radiographs of a hip with LCPD at the time of initial diagnosis show varying degrees of femoral head involvement: (A) a 7-year-old boy with lateral pillar Stage B/C, Catterall Grade 3; (B) a 6-year-old boy with lateral pillar Stage B, Catterall Grade 4; (C) a 6-year-old boy with lateral pillar Stage B/C, Catterall Grade 4; (D) a 7-year-old boy with lateral pillar Stage C, Catterall Grade 4.
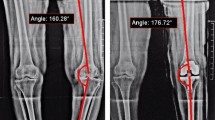
Followup standing full-length lower extremity radiographs of the same patients at skeletal maturity show (A) a spherical femoral head (Stulberg II) and no LLD; (B) a spherical femoral head (Stulberg II) and 23 mm of LLD; (C) a nonspherical, ovoid femoral head (Stulberg III) and 30 mm of LLD; (D) an ovoid femoral head (Stulberg III) and 36 mm of LLD.
Readers should be aware of the limitations of our study. First, this was by design a retrospective comparative study. However, we examined all of the data from patients who had reached skeletal maturity at the time of the final followup visit. Second, the severity of disease was classified using a simplified system. Although the Catterall classification system reportedly has poor interobserver reliability [8, 15], the modified two-group Catterall classification, with 50% involvement of the femoral head as the dividing line, may be more reliable [38]. Because lateral pillar classifications of femoral head involvement of B/C border and C have been considered to carry less favorable prognoses, we also used a modified two-group lateral pillar classification [30]. Third, because of the retrospective nature of this study, we were only able to report the measured LLD and could not determine the functional consequences of the scanographic findings. Therefore, our classification of LLD did not reflect either the patients’ symptoms or any pathological gait patterns. However, our investigation still provides valuable information for patients and their caregivers. Fourth, because of the size of the study, the patients were grouped into two general treatment categories rather than individual treatment groups. Although this was a small study using simple classification systems, our findings indicate some patients will have LLD at skeletal maturity after treatment of LCPD by certain methods. Further studies with larger numbers of participants will be required to confirm that the identified factors actually influence the LLD.
The amount of shortening of the LCPD-affected leg has been presumed to depend on the extent of femoral head involvement [1, 3, 14, 28]. Our observations suggest that more severe involvement of the femoral head is more likely to result in abnormal LLD. There could be several reasons for this association. In the pathophysiology of LCPD, disruption of the blood supply to the femoral head appears to be the critical determinant of the fate of the femoral head and proximal femoral growth plate [23]. Subsequent changes in the mechanical properties of the affected femoral head lead to deformation. Furthermore, premature closure of the proximal femoral growth plate is associated with diminished growth of the femoral head [14]. However, not all patients with LCPD develop clinically relevant LLD. Premature growth arrest is seen in fewer than 30% of patients with LCPD, suggesting that the growth plate continues to function in most patients [4, 37]. We observed a mean amount of shortening of approximately 12 mm and a maximum amount of shortening of approximately 38 mm, and we assume that this shortening of the affected limb is related to temporary or permanent growth arrest. Although we did not evaluate the status of the affected physis during growth, we also presume that bony bridge formation can exacerbate the extent of shortening.
Varus osteotomy of the proximal femur creates immediate postoperative shortening of the affected limb. However, progressive straightening of the neck-shaft angle and stimulation of the physis over a period of several years after surgery may compensate for the initial loss of length [28]. Compensatory remodeling starts after the completion of treatment and may limit the LLD [9, 35, 39]. Previous reports concerning LLD in patients with LCPD have reported various incidence rates and amounts of LLD depending on the treatment modality (Table 4). Several researchers reported that the incidence of LLD ranged from 34% to 58%, and several reports [25, 28] found a magnitude of LLD at skeletal maturity of ≥ 1 cm regardless of the treatment method. Mirovsky et al. [28] reported a 58% incidence of LLD in 55 patients who had undergone femoral osteotomy with a mean shortening of 1 cm at the time of complete or near-complete skeletal maturity. They also compared their results with the data reported by Laron et al. [25] and concluded that neither the incidence nor the magnitude of the LLD at skeletal maturity differed between patients treated nonoperatively and those treated with varus osteotomy of the proximal femur. Grzegorzewski et al. [14] stated that varus osteotomy produces more shortening than do other methods of treatment. In contrast, Rowe et al. [31] reported less residual shortening in patients treated by varus osteotomy of the proximal femur than in those treated by abduction bracing. However, our findings contrast with those of both of these studies, because we observed similar frequencies of LLD regardless of the patients’ age at diagnosis, sex, or the treatment method. Although we found an association between the extent of femoral head involvement (by the Catterall and lateral pillar classifications) and the incidence of LLD, mild LLD was most commonly observed in patients in modified Catterall Group B and modified lateral pillar Group II.
The shape of the femoral head after healing and the Stulberg classification at skeletal maturity of the patients with LCPD reportedly depend on two factors: the patient’s age at onset and the extent of femoral head involvement. Older age at onset and involvement of the entire femoral head indicate a less favorable prognosis for femoral head healing [11, 18, 19, 27]. Previous studies with mean followups of 5 to 11 years [5, 30, 38] show age at onset of < 6 years is associated with a spherical femoral head (Stulberg I and II) at skeletal maturity. However, our data suggest that the age at diagnosis does not correlate with the degree of LLD at skeletal maturity. Our findings are consistent with those of several previous studies [5, 30, 34, 38] in that the extent of femoral head involvement determined the degree of LLD at skeletal maturity. Grzegorzewski et al. [14] reviewed 261 patients with unilateral LCPD and concluded that the extent of femoral head involvement was associated with the occurrence of LLD. Mirovsky et al. [28] stated that less residual shortening can be expected in children who were aged ≤ 7 years at the time of the onset of symptoms or ≤ 8 years at the time of surgery. However, in our study, only the extent of femoral head involvement determined the final LLD at skeletal maturity.
In conclusion, we found the extent of femoral head involvement was the strongest predictor of the LLD at skeletal maturity in patients with unilateral LCPD, whereas the age at diagnosis, sex, and treatment modality were less important for predicting the amount of residual shortening. Clinicians should exercise caution when treating patients with mild degrees of LLD, because this disorder is influenced by the extent of femoral head involvement at the time of diagnosis rather than by the age at diagnosis or treatment method.
References
Barnes JM. Premature epiphysial closure in Perthes’ disease. J Bone Joint Surg Br. 1980;62:432–437.
Beck M, Kalhor M, Leunig M, Ganz R. Hip morphology influences the pattern of damage to the acetabular cartilage: femoroacetabular impingement as a cause of early osteoarthritis of the hip. J Bone Joint Surg Br. 2005;87:1012–1018.
Bowen JR, Foster BK, Hartzell CR. Legg-Calve-Perthes disease. Clin Orthop Relat Res. 1984;185:97–108.
Bowen JR, Schreiber FC, Foster BK, Wein BK. Premature femoral neck physeal closure in Perthes’ disease. Clin Orthop Relat Res. 1982;171:24–29.
Canavese F, Dimeglio A. Perthes’ disease: prognosis in children under six years of age. J Bone Joint Surg Br. 2008;90:940–945.
Catterall A. The natural history of Perthes’ disease. J Bone Joint Surg Br. 1971;53:37–53.
Catterall A. Legg-Calve-Perthes syndrome. Clin Orthop Relat Res. 1981;158:41–52.
Christensen F, Soballe K, Ejsted R, Luxhoj T. The Catterall classification of Perthes’ disease: an assessment of reliability. J Bone Joint Surg Br. 1986;68:614–615.
Edgren W. Coxa plana. A clinical and radiological investigation with particular reference to the importance of the metaphyseal changes for the final shape of the proximal part of the femur. Acta Orthop Scand Suppl. 1965:Suppl 84:81–129.
Eijer H, Podeszwa DA, Ganz R, Leunig M. Evaluation and treatment of young adults with femoro-acetabular impingement secondary to Perthes’ disease. Hip Int. 2006;16:273–280.
Gower WE, Johnston RC. Legg-Perthes disease. Long-term follow-up of thirty-six patients. J Bone Joint Surg Am. 1971;53:759–768.
Gross RH. Leg length discrepancy: how much is too much? Orthopedics. 1978;1:307–310.
Grossbard GD. Hip pain during adolescence after Perthes’ disease. J Bone Joint Surg Br. 1981;63:572–574.
Grzegorzewski A, Synder M, Kozlowski P, Szymczak W, Bowen RJ. Leg length discrepancy in Legg-Calve-Perthes disease. J Pediatr Orthop. 2005;25:206–209.
Hardcastle PH, Ross R, Hamalainen M, Mata A. Catterall grouping of Perthes’ disease. An assessment of observer error and prognosis using the Catterall classification. J Bone Joint Surg Br. 1980;62:428–431.
Herring JA, Kim HT, Browne R. Legg-Calve-Perthes disease. Part I: Classification of radiographs with use of the modified lateral pillar and Stulberg classifications. J Bone Joint Surg Am. 2004;86:2103–2120.
Herring JA, Kim HT, Browne R. Legg-Calve-Perthes disease. Part II: Prospective multicenter study of the effect of treatment on outcome. J Bone Joint Surg Am. 2004;86:2121–2134.
Herring JA, Neustadt JB, Williams JJ, Early JS, Browne RH. The lateral pillar classification of Legg-Calve-Perthes disease. J Pediatr Orthop. 1992;12:143–150.
Ippolito E, Tudisco C, Farsetti P. The long-term prognosis of unilateral Perthes’ disease. J Bone Joint Surg Br. 1987;69:243–250.
Karpinski MR, Newton G, Henry AP. The results and morbidity of varus osteotomy for Perthes’ disease. Clin Orthop Relat Res. 1986;209:30–40.
Kaweblum M, Aguilar MC, Blancas E, Kaweblum J, Lehman WB, Grant AD, Strongwater AM. Histological and radiographic determination of the age of physeal closure of the distal femur, proximal tibia, and proximal fibula of the New Zealand white rabbit. J Orthop Res. 1994;12:747–749.
Keret D, Harrison MH, Clarke NM, Hall DJ. Coxa plana—the fate of the physis. J Bone Joint Surg Am. 1984;66:870–877.
Kim HK, Herring JA. Pathophysiology, classifications, and natural history of Perthes disease. Orthop Clin North Am. 2011;42:285–295, v.
Langenskiold A. Changes in the capital growth plate and the proximal femoral metaphysis in Legg-Calve-Perthes disease. Clin Orthop Relat Res. 1980;150:110–114.
Laron Z, Axer A, Drezner Z. Growth and development of children with Legg-Calve-Perthes syndrome in Israel. Isr J Med Sci. 1973;9:612–616.
Leitch JM, Paterson DC, Foster BK. Growth disturbance in Legg-Calve-Perthes disease and the consequences of surgical treatment. Clin Orthop Relat Res. 1991;262:178–184.
McAndrew MP, Weinstein SL. A long-term follow-up of Legg-Calve-Perthes disease. J Bone Joint Surg Am. 1984;66:860–869.
Mirovsky Y, Axer A, Hendel D. Residual shortening after osteotomy for Perthes’ disease. A comparative study. J Bone Joint Surg Br. 1984;66:184–188.
Murphy RP, Marsh HO. Incidence and natural history of ‘head at risk’ factors in Perthes’ disease. Clin Orthop Relat Res. 1978;132:102–107.
Rosenfeld SB, Herring JA, Chao JC. Legg-calve-perthes disease: a review of cases with onset before six years of age. J Bone Joint Surg Am. 2007;89:2712–2722.
Rowe SM, Moon ES, Song EK, Yoon TR, Kim MS, Cho SB. Residual shortening after Legg-Calve-Perthes disease, focusing on the response of the ipsilateral tibia. J Pediatr Orthop. 2005;25:296–299.
Salter RB, Thompson GH. Legg-Calve-Perthes disease. The prognostic significance of the subchondral fracture and a two-group classification of the femoral head involvement. J Bone Joint Surg Am. 1984;66:479–489.
Sankar WN, Flynn JM. The development of acetabular retroversion in children with Legg-Calve-Perthes disease. J Pediatr Orthop. 2008;28:440–443.
Schoenecker PL, Stone JW, Capelli AM. Legg-Perthes disease in children under 6 years old. Orthop Rev. 1993;22:201–208.
Shapiro F. Legg-Calve-Perthes disease: a study of lower extremity length discrepancies and skeletal maturation. Acta Orthop Scand. 1982;53:437–444.
Snow SW, Keret D, Scarangella S, Bowen JR. Anterior impingement of the femoral head: a late phenomenon of Legg-Calve-Perthes’ disease. J Pediatr Orthop. 1993;13:286–289.
Sponseller PD, Desai SS, Millis MB. Abnormalities of proximal femoral growth after severe Perthes’ disease. J Bone Joint Surg Br. 1989;71:610–614.
Wiig O, Terjesen T, Svenningsen S. Prognostic factors and outcome of treatment in Perthes’ disease: a prospective study of 368 patients with five-year follow-up. J Bone Joint Surg Br. 2008;90:1364–1371.
Willner S. Difference in leg length in children with coxa plana during and after treatment using unilateral unloading. Acta Orthop Scand. 1975;46:102–107.
Acknowledgments
We thank Sang-Heon Song MD, Ahmed Suparno Bahar Moni MD, and Kwang-Youn Kim for manuscript editing and technical support.
Author information
Authors and Affiliations
Corresponding author
Additional information
The institution of one or more of the authors (H-RS) has received funding from the Korea Healthcare Technology R & D Project, Ministry for Health, Welfare & Family Affairs, Republic of Korea (A110416).
All ICMJE Conflict of Interest Forms for authors and Clinical Orthopaedics and Related Research editors and board members are on file with the publication and can be viewed on request.
Each author certifies that his institution approved the human protocol for this investigation that all investigations were conducted in conformity with ethical principles of research, and that informed consent for participation in the study was obtained.
About this article
Cite this article
Park, KW., Jang, KS. & Song, HR. Can Residual Leg Shortening Be Predicted in Patients With Legg-Calvé-Perthes’ Disease?. Clin Orthop Relat Res 471, 2570–2577 (2013). https://doi.org/10.1007/s11999-013-3009-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11999-013-3009-4