Abstract
Purpose of Review
Spontaneous coronary artery dissection (SCAD) is an increasingly recognized entity leading to myocardial infarction especially in women. Multiple observational studies have been published in the recent years. This review summarizes the current body of knowledge and recent developments in SCAD epidemiology, presentation, diagnosis, and management.
Recent Findings
Contemporary series have estimated the incidence of SCAD to be up to ~ 4% of all patients presenting with acute coronary syndrome (ACS). Fibromuscular dysplasia, hormonal therapy, multiparity, connective tissue disorders, and systemic inflammatory diseases have been identified as predisposing factors for SCAD. Although SCAD typically presents as ACS, emerging evidence shows that cardiogenic shock, ventricular arrhythmia, and cardiac arrest are not uncommon, with higher incidence of complication among peripartum women. Advances in SCAD diagnosis include a novel angiographic classification of SCAD and the use of optical coherence tomography and intravascular ultrasound to aid in diagnosis. Management depends on clinical stability and the presence of high-risk features. Beta-blockers are the mainstay of medical therapy long term and was shown to reduce recurrent SCAD. The largest study to date including 750 patients across 22 North American centers showed that the majority of patients was managed conservatively and that the technical success rates of percutaneous coronary intervention were low. Following discharge, SCAD-specific cardiac rehabilitation had been shown to have long-term cardiovascular benefits.
Summary
The recent years have seen a surge in SCAD awareness and publication. Centralized patient networks as well as core lab interpretation of diagnostic data have contributed significantly to the growing body of knowledge. Current efforts to delineate the pathogenesis of SCAD; improve medical and revascularization therapy; and understand complications such as cardiogenic shock and cardiac arrest will serve as foundational building blocks for future research and innovation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Spontaneous coronary artery dissection (SCAD) is an increasingly recognized condition that can lead to myocardial infarction (MI), cardiac arrest, and death in young women who do not have traditional cardiovascular risk factors [1]. Since the first reported case in 1931 by Pretty, SCAD remained underdiagnosed for many decades due to the low index of suspicion for acute coronary syndrome in this unique population [2]. Over the past decade, the awareness of SCAD has dramatically increased. This is largely attributed to the improved diagnosis by coronary angiography as well as the establishment of SCAD patient networks and centralized registries [3,4,5,6]. Recently, both the American Heart Association and European Society of Cardiology published position statements on the diagnosis and management of SCAD [7••, 8]. In this review, we summarize the current body of knowledge as well as recent developments around SCAD presentation, diagnosis, and management.
Definition and Pathogenesis
SCAD is defined as a non-traumatic, non-iatrogenic separation of the coronary arterial wall, which is unrelated to atherosclerosis [1]. Myocardial ischemia or infarction occurs primarily due to the formation of an intramural hematoma and subsequent arterial luminal stenosis [7••]. Two mechanisms of SCAD pathogenesis have been proposed. The first one involves tearing of the intima, hemorrhage into the media, and formation of a false lumen with compression of the true lumen [9, 10] (Fig. 1). The second mechanism is less well delineated and involves spontaneous rupture of the vaso vasorum and bleeding into the arterial wall [11].
Coronary artery schematic showing SCAD due to intimal rupture with intramural hematoma in the false lumen compressing the true lumen. Reprinted with permission from reference [1]. Copyright Elsevier Inc. 2016
The most common predisposing condition in those presenting with SCAD is fibromuscular dysplasia (FMD), a non-inflammatory arteriopathy which has been shown to affect coronary arteries [6, 12, 13]. The incidence of FMD in SCAD is reported to be between 31 and 72% [6, 14••]. This highlights the importance of routine screening of other vascular beds for FMD, such as cerebrovascular, renal, and iliac arteries. Recently, the Canadian SCAD study (large multicenter observational study of 750 patients) was published and reported lower incidence of other predisposing factors for SCAD, including active hormonal therapy (10.0%), grand multigravida (defined as 5 or more pregnancies (8.9%)), and multiparity (defined as 4 or more births (8.5%)) [14••]. Systemic inflammatory diseases such as rheumatoid arthritis and systemic lupus erythematosus have also been shown to be predisposing factors, with prevalence between 1.5 and 4.7% [14••, 15]. Inherited connective tissue disorders such as Marfan and Ehlers-Danlos syndromes were reported in up to 3.6% of SCAD patients [14••, 15]. In one study where 59 SCAD patients underwent genetic testing for a heritable connective tissue disorder, three (5.1%) received such a diagnosis [16]. The genetic predisposition to SCAD has not been well studied. An analysis of the Mayo Clinic Registry showed a low prevalence of familial cases of 1.2% [17]. Ongoing studies are underway to ascertain genes associated with SCAD and other types of disease including inherited cardiac conditions.
Epidemiology
The incidence of SCAD has been historically underestimated, due to the limitations of coronary angiography for SCAD diagnosis and the low index of suspicion for acute coronary syndrome (ACS) in relatively young women without conventional cardiovascular risk factors. Contemporary series have reported that in all patients presenting with ACS; the prevalence of SCAD ranges between 0.2 and 4.9% [5, 7••, 18,19,20,21,22]. Within subgroups of young women, the prevalence of SCAD has been reported to be much higher. Vanzetto et al. showed that in women aged < 50 presenting with ACS, the prevalence of SCAD is 8.7% and 10.8% in those presenting with ST segment elevation myocardial infarction (STEMI) [5]. Similarly, Saw et al. found that SCAD was the cause of ACS in 24% of cases in women aged 50 or younger [6]. In this single-center series that examined coronary angiograms performed in women younger than 50 in British Columbia over a 2-year period, SCAD was diagnosed in 9% of women and 24.4% of those who presented with MI [23]. The authors attributed this higher prevalence largely to the improved detection rate of type 2 SCAD on angiography at this center [10]. Recently, the largest population-based analysis to date of SCAD in the setting of acute MI was conducted on the National Inpatient Sample database between 2009 and 2014 [24]. Among women with a discharge diagnosis of acute MI, 0.98% of the women had a co-diagnosis of SCAD. Of the total number of women with SCAD and acute MI, 44.9% of these had the diagnosis of STEMI.
Clinical Presentation
Patients with SCAD typically present with ACS. This manifests as non-ST elevation ACS (NSTEACS) in between 13 and 67% of cases and STEMI in 33 to 87% of cases [6, 14••, 21, 25,26,27]. In the Canadian SCAD (CanSCAD) study, the largest multicenter, prospective observational study to date, precipitating stressors and conditions were frequently found. Precipitating stressors were reported in 66.4% of patients. Of these, emotional stressors were reported in 50.3% of cases, physical stressors in 28.9%, and heavy isometric activities (lifting more than 50 pounds) in 9.8% of cases [14••].
Cardiogenic shock and ventricular arrhythmias may occur with SCAD. The incidence of shock has been reported to range between 1.2 and 15.9% [5, 6, 14••, 20, 25, 27, 28]. Ventricular tachycardia or fibrillation was reported in 3.6 to 11.8% of patients [5, 6, 14••, 29, 30], with 1.6 to 2.8% suffering a cardiac arrest [25, 27]. Among the series of peripartum SCAD, the incidence of shock is much higher at between 20.3 and 24.2% [29, 30], with one series reporting cardiac arrest incidence of 11.4%. Data (reported but unpublished) from our center showed that cardiogenic shock SCAD patients was more likely to be peripartum, presented with STEMI, developed ventricular arrhythmia, and required revascularization.
SCAD is the most common cause of pregnancy-associated MI. In one study examining acute MI among pregnant patients, SCAD was found to be the culprit in 43% of cases [31]. It is thought that hormonal changes during pregnancy, mainly progesterone-mediated effects, impair collagen synthesis and weakens the tunica media [32]. In a large registry of 323 female SCAD patients, the prevalence of SCAD in pregnancy was 16.7% [33]. The overall incidence of SCAD during pregnancy has been reported to be 1.81 per 100,000 pregnancies [30]. Multiple studies have shown that SCAD tends to occur in the peripartum period and less frequently in the antenatal period (most commonly during the third trimester) [29, 30, 33]. Importantly, SCAD during pregnancy is associated with more high-risk features: lower mean left ventricular ejection fraction at the time of SCAD diagnosis; left main or multi-vessel dissection; and requiring coronary artery bypass grafting [33].
Diagnosis
Patients with SCAD are typically women, young, and present with a paucity of traditional cardiovascular risk factors. The management of ACS secondary to SCAD is significantly different from that of atherosclerotic coronary artery disease and plaque rupture. Therefore, accurate diagnosis is of paramount importance. Evaluation should begin with a detailed history and physical examination, along with a high index of suspicion in the appropriate patient population.
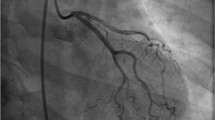
Early invasive coronary angiography is the gold standard test for diagnosis of SCAD. The classic appearance is multiple radiolucent lumens on angiography; this is absent, however, in > 70% of SCAD cases [9]. Three angiographic patterns of SCAD have been recently characterized (Fig. 2). Type 1 is the pathognomonic appearance with contrast staining of arterial wall with multiple lumens. This may occur from a tear in the intima, with hemorrhage into the media, and formation of a false lumen with compression of the true lumen. Alternatively, intramural hematoma from rupture of vaso vasorum can rupture into the true lumen, causing intimal dissection appearance. Type 2 is the most common type of SCAD and is often missed on angiography. It appears as long, diffuse narrowing of the artery, ranging from mild stenosis to complete occlusion [9, 10]. This is due to intramural hematoma in the false lumen compressing the true lumen. Type 3 SCAD mimics atherosclerosis with focal or tubular stenosis and can be challenging to differentiate from atherosclerosis without additional intracoronary imaging.
The pathognomonic appearance of multiple radiolucent lumens is absent in the majority of cases [6, 34]. In these situations, optical coherence tomography (OCT) and intravascular ultrasound (IVUS) can be employed to assess for SCAD. OCT allows visualization of the coronary artery wall with a resolution of 10 μm [34]. The diagnosis of SCAD on OCT is made upon visualization of an intimomedial flap or an intramural hematoma. OCT has been used in multiple studies and has been shown to facilitate an early and accurate diagnosis of SCAD, particularly among patients with concomitant atherosclerotic coronary artery disease, as well as among patients with type 3 angiographic SCAD, which may mimic atherosclerosis [9, 34, 35]. The CanSCAD study reported the mean length of dissection to be 33.2 mm. The majority of patients had one artery (86.9%) and one segment (74.8%) affected. A quarter of patients (25.2%) had two or more segments affected. The left anterior descending artery (LAD) was the most commonly affected artery (52.1%), followed by the left circumflex artery (37.7%) and the right coronary artery (23.2%) [14••]. Angiographically, type 2 SCAD was the most common presentation (60.2%). The median segment diameter stenosis was 79.0% [14••]. Forty-one patients (5.5%) had OCT-confirmed SCAD, and sixteen patients (2.1%) had IVUS-confirmed SCAD.
Cardiac computed tomography angiography (CCTA) is increasingly employed as an efficient means to evaluate low- and intermediate-risk patients presenting with ACS. However, the role of CCTA in SCAD has not been studied. Currently, CCTA is not recommended as the first-line imaging modality for suspected acute SCAD and does not have sufficient sensitivity to exclude SCAD [7••, 36, 37]. This is because of the lower spatial and temporal resolution of CCTA for SCAD compared with invasive coronary angiography [38]. Intramural hematoma can mimic the appearance of cardiac motion artifact and further limits the utility of this modality. Potential advantages of CCTA include its accessibility and non-invasive nature [39]. Several series have used CCTA as a means to non-invasively follow patients to monitor for resolution of SCAD, particularly as the risk of recurrence is significant in this group. One study followed 24 patients post-SCAD using CCTA. At a mean follow-up of 121 days, twenty had complete resolution of SCAD, while the rest had persistent dissection, aneurysm reaction, and one case where angioplasty was performed in the false lumen [40].
Management
Conservative management is the mainstay of treatment for SCAD in the absence of high-risk features. Treatment goals are to alleviate symptoms and prevent recurrence. There is a paucity of evidence regarding the optimal medical treatment of SCAD due to the lack of randomized data. Currently, long-term medical therapy includes aspirin and beta-blockers. Beta-blockers have been shown to be associated with diminished recurrence of SCAD (hazard ratio 0.36, p = 0.004) [41]. Patients who undergo coronary revascularization should continue on dual antiplatelet therapy as per current American Heart Association/American College of Cardiology and Canadian Cardiovascular Society ACS guidelines [42, 43]. In those patients who are medically managed without revascularization, the role for the addition of a P2Y12 inhibitor is less clear. Some experts recommend treatment as per usual ACS guidelines with 1 year of dual antiplatelet therapy. Others recommend limited DAPT for one up to 12 months after the event. At our center, we generally use aspirin and clopidogrel for 3 months after SCAD until further clinical reassessment.
A proposed schema of the management of SCAD patients, depending on clinical stability and the presence of high-risk features, is presented in Fig. 3. High-risk features include left main dissection, ongoing or recurrent ischemic or chest pain, ventricular tachycardia or fibrillation, and cardiogenic shock [1]. PCI for SCAD has been consistently shown to be associated with high technical failure rates of up to 54% [6, 25, 26], as well as with complications and the need for emergent CABG [20, 21, 27]. Coronary arteries in SCAD are more susceptible to iatrogenic dissection; hence, manipulation during PCI can lead to extension of intramural hematoma, thereby worsening luminal occlusion and ischemia.
Management algorithm for SCAD patients. “Ostial LAD or ≥ 2 proximal SCAD” refers to 2 or more proximal vessel segments with SCAD”. Reprinted with permission from reference [1]. Copyright Elsevier Inc. 2016
The CanSCAD study enrolled 750 patients with SCAD from 22 North American centers and evaluated the natural history of SCAD as well as outcomes. It is the largest multicenter observational study of SCAD to date. In this study, the majority of patients (86.4%) was managed initially with a conservative strategy. Of the remaining 14.7% of patients who underwent revascularization, almost all received PCI, whereas < 2% of patients underwent CABG or fibrinolysis. The most common reasons for revascularization as an initial strategy were ongoing ischemic symptoms (39.1%), ongoing ischemic changes on electrocardiogram (34.5%), and severe luminal stenosis (31.8%). Of the patients who were managed conservatively, 2% required subsequent PCI and 0.3% underwent CABG. For all patients who underwent PCI, the technical success rate was low, consistent with previous experience, with 30.1% deemed technically unsuccessful and 29.1% deemed successful [14••]. Patients managed conservatively should remain inpatient and be monitored for signs of extension of dissection, which can result in recurrent MI or unplanned revascularization. In the CanSCAD 750-patient cohort, among those treated conservatively, 4% had recurrent in-hospital MI with 2.5% needing revascularization [14••]. Expert consensus statements recommend in-patient monitoring for 3–5 days for SCAD patients treated conservatively [7••, 8].
The incidence of shock in SCAD has been reported to range between 1.2 and 15.9% [5, 6, 14••, 20, 25, 27, 28]. The literature describes limited experience with the management of cardiogenic shock. Intra-aortic balloon pump (IABP) is the most commonly used mechanical circulatory support [29, 44,45,46,47]. There is little data on improvement of the overall outcomes with IABP. There are few case reports of extra-corporeal membrane oxygenation (ECMO) used in SCAD, with most cases using ECMO as a bridge to recovery [45, 47,48,49]. The advantages of ECMO include its ability to provide biventricular support as well as oxygenation. In the CanSCAD study, 15 patients (2.0%) developed hemodynamic instability. Of these, 9 required inotropes and 6 required IABP. There were two cases managed with left ventricular assist device (LVAD) and ECMO.
Following discharge, lifestyle changes and medications are important considerations to lower the risk of recurrent SCAD. Patients are advised to avoid situations that may increase intracoronary shear stress in the same way that beta-blockers are recommended to decrease the shear stress of the coronary arteries. These include activities that increase the thoracoabdominal pressures or catecholamines. Patients are asked to avoid Valsalva maneuvers, heavy weightlifting (maximum 30 pounds for women, 50 pounds for men). Cardiac rehabilitation programs are recommended for all SCAD patients. These programs can be SCAD-specific cardiac rehabilitation (SCAD-CR) in large referring centers, such as in our institution where we have a centralized and concentrated cohort of SCAD patients [50]. These programs include exercise programs, psychological counseling, and peer support. SCAD-CR has been shown to be safe and beneficial and associated with improvement in exercise capacity, psychological measures, and reduction in chest pain. There were long-term cardiovascular benefits observed in those patients who attended our SCAD-CR compared with those who did not. Although the rate of beta-blocker uptake in the SCAD-CR was 85%, cardiac rehabilitation is thought to have additive effect from beta-blockers and perhaps even synergistic [50].
Conclusion/Future Directions
SCAD is a unique and challenging entity which is increasingly recognized as a cause of morbidity and mortality in both men and women presenting with ACS. Despite increased awareness and improved detection, much remains to be known about the pathogenesis and management of SCAD. Future research efforts should focus on identifying new predisposing factors and consolidating established connections between underlying conditions and SCAD presentations; optimizing imaging modalities to facilitate early diagnosis of SCAD; further investigating the role of medical therapy; identifying strategies to improve success of PCI; evaluating the role of mechanical circulatory support in cardiogenic shock complicating SCAD; and delineating optimal exercise and cardiac rehabilitation regimen for patients post-SCAD. Lastly, support should continue to be directed toward patient networks and referral hubs as to facilitate the establishment of specialized centers for SCAD management and research.
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance
Saw J, Mancini GBJ, Humphries KH. Contemporary review on spontaneous coronary artery dissection. J Am Coll Cardiol. 2016;68:297–312.
Pretty H. Dissecting aneurysm of coronary artery in a woman aged 42: rupture. BMJ. 1931;1:667.
Tweet MS, Gulati R, Aase LA, Hayes SN. Spontaneous coronary artery dissection: a disease-specific, social networking community-initiated study. Mayo Clin Proc. 2011;86:845–50.
Fontanelli A, Olivari Z, La Vecchia L, Basso C, Pagliani L, Marzocchi A, et al. Spontaneous dissections of coronary arteries and acute coronary syndromes: rationale and design of the DISCOVERY, a multicenter prospective registry with a case-control group. J Cardiovasc Med Hagerstown Md. 2009;10:94–9.
Vanzetto G, Berger-Coz E, Barone-Rochette G, Chavanon O, Bouvaist H, Hacini R, et al. Prevalence, therapeutic management and medium-term prognosis of spontaneous coronary artery dissection: results from a database of 11,605 patients. Eur J Cardio-Thorac Surg Off J Eur Assoc Cardio Thorac Surg. 2009;35:250–4.
Saw J, Aymong E, Sedlak T, Buller CE, Starovoytov A, Ricci D, et al. Spontaneous coronary artery dissection: association with predisposing arteriopathies and precipitating stressors and cardiovascular outcomes. Circ Cardiovasc Interv. 2014;7:645–55.
•• Hayes SN, Kim ESH, Saw J, et al. Spontaneous coronary artery dissection: current state of the science: a scientific statement from the American Heart Association. Circulation. 2018. https://doi.org/10.1161/CIR.0000000000000564First North American position statement on management of SCAD. Summarizes recent findings and identifies knowledge gaps in current research.
Adlam D, Alfonso F, Maas A, Vrints C, Writing Committee, al-Hussaini A, et al. European Society of Cardiology, acute cardiovascular care association, SCAD study group: a position paper on spontaneous coronary artery dissection. Eur Heart J. 2018;39:3353–68.
Saw J, Mancini GBJ, Humphries K, Fung A, Boone R, Starovoytov A, et al. Angiographic appearance of spontaneous coronary artery dissection with intramural hematoma proven on intracoronary imaging. Catheter Cardiovasc Interv Off J Soc Card Angiogr Interv. 2016;87:E54–61.
Saw J. Coronary angiogram classification of spontaneous coronary artery dissection. Catheter Cardiovasc Interv Off J Soc Card Angiogr Interv. 2014;84:1115–22.
Maehara A, Mintz GS, Castagna MT, Pichard AD, Satler LF, Waksman R, et al. Intravascular ultrasound assessment of spontaneous coronary artery dissection. Am J Cardiol. 2002;89:466–8.
Maas AHEM, Bouatia-Naji N, Persu A, Adlam D. Spontaneous coronary artery dissections and fibromuscular dysplasia: current insights on pathophysiology, sex and gender. Int J Cardiol. 2019;286:220–5.
Lee C, Starovoytov A, Saw J. Case reports of coronary fibromuscular dysplasia and spontaneous coronary artery dissections. Catheter Cardiovasc Interv Off J Soc Card Angiogr Interv. 2019;93:631–4.
•• Saw J, Starovoytov A, Humphries K, et al. Canadian spontaneous coronary artery dissection cohort study: in-hospital and 30-day outcomes. Eur Heart J. 2019;40:1188–97 This is the largest comprehensive multicenter cohort study to date of SCAD patients examining risk factors, presentation, management and outcomes.
Krittanawong C, Kumar A, Johnson KW, Luo Y, Yue B, Wang Z, et al. Conditions and factors associated with spontaneous coronary artery dissection (from a national population-based cohort study). Am J Cardiol. 2019;123:249–53.
Henkin S, Negrotto SM, Tweet MS, Kirmani S, Deyle DR, Gulati R, et al. Spontaneous coronary artery dissection and its association with heritable connective tissue disorders. Heart Br Card Soc. 2016;102:876–81.
Goel K, Tweet M, Olson TM, Maleszewski JJ, Gulati R, Hayes SN. Familial spontaneous coronary artery dissection: evidence for genetic susceptibility. JAMA Intern Med. 2015;175:821–6.
Mortensen KH, Thuesen L, Kristensen IB, Christiansen EH. Spontaneous coronary artery dissection: a Western Denmark heart registry study. Catheter Cardiovasc Interv Off J Soc Card Angiogr Interv. 2009;74:710–7.
Alfonso F, Bastante T, García-Guimaraes M, Pozo E, Cuesta J, Rivero F, et al. Spontaneous coronary artery dissection: new insights into diagnosis and treatment. Coron Artery Dis. 2016;27:696–706.
Nakashima T, Noguchi T, Haruta S, Yamamoto Y, Oshima S, Nakao K, et al. Prognostic impact of spontaneous coronary artery dissection in young female patients with acute myocardial infarction: a report from the angina pectoris-myocardial infarction multicenter Investigators in Japan. Int J Cardiol. 2016;207:341–8.
Rashid HNZ, Wong DTL, Wijesekera H, Gutman SJ, Shanmugam VB, Gulati R, et al. Incidence and characterisation of spontaneous coronary artery dissection as a cause of acute coronary syndrome--a single-centre Australian experience. Int J Cardiol. 2016;202:336–8.
Nishiguchi T, Tanaka A, Ozaki Y, Taruya A, Fukuda S, Taguchi H, et al. Prevalence of spontaneous coronary artery dissection in patients with acute coronary syndrome. Eur Heart J Acute Cardiovasc Care. 2016;5:263–70.
Saw J, Aymong E, Mancini GBJ, Sedlak T, Starovoytov A, Ricci D. Nonatherosclerotic coronary artery disease in young women. Can J Cardiol. 2014;30:814–9.
Mahmoud AN, Taduru SS, Mentias A, Mahtta D, Barakat AF, Saad M, et al. Trends of incidence, clinical presentation, and in-hospital mortality among women with acute myocardial infarction with or without spontaneous coronary artery dissection: a population-based analysis. JACC Cardiovasc Interv. 2018;11:80–90.
Lettieri C, Zavalloni D, Rossini R, Morici N, Ettori F, Leonzi O, et al. Management and long-term prognosis of spontaneous coronary artery dissection. Am J Cardiol. 2015;116:66–73.
Tweet MS, Eleid MF, Best PJM, Lennon RJ, Lerman A, Rihal CS, et al. Spontaneous coronary artery dissection: revascularization versus conservative therapy. Circ Cardiovasc Interv. 2014;7:777–86.
Rogowski S, Maeder MT, Weilenmann D, Haager PK, Ammann P, Rohner F, et al. Spontaneous coronary artery dissection: angiographic follow-up and long-term clinical outcome in a predominantly medically treated population. Catheter Cardiovasc Interv Off J Soc Card Angiogr Interv. 2017;89:59–68.
Motreff P, Malcles G, Combaret N, et al. How and when to suspect spontaneous coronary artery dissection: novel insights from a single-centre series on prevalence and angiographic appearance. EuroIntervention J Eur Collab Work Group Interv Cardiol Eur Soc Cardiol. 2017;12:e2236–43.
Havakuk O, Goland S, Mehra A, Elkayam U. Pregnancy and the risk of spontaneous coronary artery dissection: an analysis of 120 contemporary cases. Circ Cardiovasc Interv. 2017;10. https://doi.org/10.1161/CIRCINTERVENTIONS.117.004941.
Faden MS, Bottega N, Benjamin A, Brown RN. A nationwide evaluation of spontaneous coronary artery dissection in pregnancy and the puerperium. Heart Br Card Soc. 2016;102:1974–9.
Elkayam U, Jalnapurkar S, Barakkat MN, Khatri N, Kealey AJ, Mehra A, et al. Pregnancy-associated acute myocardial infarction: a review of contemporary experience in 150 cases between 2006 and 2011. Circulation. 2014;129:1695–702.
Yip A, Saw J. Spontaneous coronary artery dissection-a review. Cardiovasc Diagn Ther. 2015;5:37–48.
Tweet MS, Hayes SN, Codsi E, Gulati R, Rose CH, Best PJM. Spontaneous coronary artery dissection associated with pregnancy. J Am Coll Cardiol. 2017;70:426–35.
Alfonso F, Paulo M, Gonzalo N, Dutary J, Jimenez-Quevedo P, Lennie V, et al. Diagnosis of spontaneous coronary artery dissection by optical coherence tomography. J Am Coll Cardiol. 2012;59:1073–9.
Saw J, Poulter R, Fung A. Intracoronary imaging of coronary fibromuscular dysplasia with OCT and IVUS. Catheter Cardiovasc Interv Off J Soc Card Angiogr Interv. 2013;82:E879–83.
Emergency Department Patients With Chest Pain Writing Panel, Rybicki FJ, Udelson JE, et al. 2015 ACR/ACC/AHA/AATS/ACEP/ASNC/NASCI/SAEM/SCCT/SCMR/SCPC/SNMMI/STR/STS appropriate utilization of cardiovascular imaging in emergency department patients with chest pain: a joint document of the American College of Radiology Appropriateness Criteria Committee and the American College of Cardiology Appropriate Use Criteria Task Force. J Am Coll Radiol JACR. 2016;13:e1–e29.
American College of Cardiology Foundation Task Force on Expert Consensus Documents, Mark DB, Berman DS, et al. ACCF/ACR/AHA/NASCI/SAIP/SCAI/SCCT 2010 expert consensus document on coronary computed tomographic angiography: a report of the American College of Cardiology Foundation task force on expert consensus documents. Circulation. 2010;121:2509–43.
Stefanini GG, Windecker S. Can coronary computed tomography angiography replace invasive angiography? Coronary computed tomography angiography cannot replace invasive angiography. Circulation. 2015;131:418–25 discussion 426.
Tweet MS, Gulati R, Williamson EE, Vrtiska TJ, Hayes SN. Multimodality imaging for spontaneous coronary artery dissection in women. JACC Cardiovasc Imaging. 2016;9:436–50.
Roura G, Ariza-Solé A, Rodriguez-Caballero IF, Gomez-Lara J, Ferreiro JL, Romaguera R, et al. Noninvasive follow-up of patients with spontaneous coronary artery dissection with CT angiography. JACC Cardiovasc Imaging. 2016;9:896–7.
Saw J, Humphries K, Aymong E, Sedlak T, Prakash R, Starovoytov A, et al. Spontaneous coronary artery dissection: clinical outcomes and risk of recurrence. J Am Coll Cardiol. 2017;70:1148–58.
Amsterdam EA, Wenger NK, Brindis RG, Casey DE Jr, Ganiats TG, Holmes DR Jr, et al. 2014 AHA/ACC guideline for the management of patients with non-ST-elevation acute coronary syndromes: a report of the American College of Cardiology/American Heart Association task force on practice guidelines. J Am Coll Cardiol. 2014;64:e139–228.
Focused 2012 Update of the Canadian Cardiovascular Society Guidelines for the use of antiplatelet therapy - PubMed. https://pubmed.ncbi.nlm.nih.gov/23978596-focused-2012-update-of-the-canadian-cardiovascular-society-guidelines-for-the-use-of-antiplatelet-therapy/?from_single_result=ccs+guideline+non-st-elevation. Accessed 15 Mar 2020.
Ferrari E, Tozzi P, von Segesser LK. Spontaneous coronary artery dissection in a young woman: from emergency coronary artery bypass grafting to heart transplantation. Eur J Cardio-Thorac Surg Off J Eur Assoc Cardio-Thorac Surg. 2005;28:349–51.
Patanè F, Boffini M, Sansone F, Campanella A, Rinaldi M. ECMO as a bridge to transplantation in biventricular dysfunction due to primary spontaneous coronary artery dissection. Transpl Int Off J Eur Soc Organ Transplant. 2009;22:500–2.
Sanghera P, Ravipati V, Kalapura T, Test V. Improving mortality in cardiogenic shock from spontaneous coronary artery dissection with percutaneous external heart assist: a case report. CHEST. 2013;144:140A.
Padukone A, Sayeed AK, Marczin N, García Sáez D, Zych B, Mohite PN, et al. Salvage myocardial revascularisation in spontaneous left main coronary artery dissection with cardiogenic shock - the role of mechanical circulatory support. Perfusion. 2017;32:171–3.
McKechnie RS, Patel D, Eitzman DT, Rajagopalan S, Murthy TH. Spontaneous coronary artery dissection in a pregnant woman. Obstet Gynecol. 2001;98:899–902.
Knapp KE, Weis RA, Cubillo EI, Chapital AB, Ramakrishna H. Spontaneous, postpartum coronary artery dissection and cardiogenic shock with extracorporeal membrane oxygenation assisted recovery in a 30-year-old patient. Case Rep Cardiol. 2016;2016:1048708.
Chou AY, Prakash R, Rajala J, Birnie T, Isserow S, Taylor CM, et al. The first dedicated cardiac rehabilitation program for patients with spontaneous coronary artery dissection: description and initial results. Can J Cardiol. 2016;32:554–60.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Dr. Saw reports grants from CIHR, grants from Heart & Stroke Foundation of Canada, other from Michael Smith Foundation of Health Research, grants and personal fees from AbbottVascular, grants and personal fees from Boston Scientific, grants from AstraZeneca, outside the submitted work. Dr. Yang and Dr. Alfadhel have nothing to disclose.
Human and Animal Rights and Informed Consent
All reported studies/experiments with human or animal subjects performed by the authors have been previously published and complied with all applicable ethical standards (including the Helsinki declaration and its amendments, institutional/national research committee standards, and international/national/institutional guidelines).
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Women and Ischemic Heart Disease
Rights and permissions
About this article
Cite this article
Yang, C., Alfadhel, M. & Saw, J. Spontaneous Coronary Artery Dissection: Latest Developments and New Frontiers. Curr Atheroscler Rep 22, 49 (2020). https://doi.org/10.1007/s11883-020-00866-4
Published:
DOI: https://doi.org/10.1007/s11883-020-00866-4