Abstract
Gemfibrozil is a lipid-modifying agent that belongs to the fibric acid derivative class. Fibric acid derivatives activate peroxisome proliferator activated receptor α (PPAR-α). The primary role of these agents in clinical practice is for the management of hypertriglyceridemia. Triglycerides may be reduced by as much as 74 % in some patients. In addition to lowering triglycerides, these agents can also decrease very low-density lipoprotein cholesterol (VLDL-C) and low-density lipoprotein cholesterol (LDL-C) as well as raise high-density lipoprotein cholesterol (HDL-C). Based on the 2013 ACC/AHA Guideline on the Treatment of Blood Cholesterol to Reduce Atherosclerotic Cardiovascular Risk in Adults and the National Lipid Association, pharmacologic therapy to reduce triglycerides should be considered when triglyceride levels are ≥500 mg/dL. While the use of gemfibrozil has decreased over the years for a variety of reasons, muscle-associated adverse effects is the predominant reason and the one that is most clinically relevant. However, despite these concerns, there are situations in which the use of gemfibrozil in combination with a statin may be necessary. Understanding the metabolism of gemfibrozil and the degree of interaction with the various statins will assist health-care providers to optimize safety when this combination is clinically indicated.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gemfibrozil belongs to a class of agents known as fibric acid derivatives. This is a well-utilized class of lipid-modifying agents used primarily to treat hypertriglyceridemia. Fibric acid derivatives not only decrease triglycerides, but also increase high-density lipoprotein cholesterol (HDL-C) and decrease both very low-density lipoprotein cholesterol (VLDL-C) and low-density lipoprotein cholesterol (LDL-C). Treatment with fibric acid derivatives should be considered when triglycerides are above a given threshold. Guidelines vary on when pharmacologic therapy should be prescribed. The 2013 ACC/AHA Guideline on the Treatment of Blood Cholesterol to Reduce Atherosclerotic Cardiovascular Risk in Adults and the National Lipid Association recommends therapy initiation when triglycerides are ≥500 mg/dL to prevent pancreatitis [1, 2]. Gemfibrozil has been available for years. However, its use has fallen out of favor due to concerns for adverse effects as monotherapy, drug interactions in combination therapy with statins, as well as limited data in reducing CV morbidity and mortality. This article will review the clinical efficacy, adverse effects, drug interactions with statins, and the role in clinical practice of gemfibrozil and will provide recommendations for using gemfibrozil safely in combination with statins in patients who may warrant further reduction in lipoproteins.
Mechanism of Action
Fibric acid derivatives activate peroxisome proliferator-activated receptor α (PPAR-α). PPAR-α or NR1C1 (nuclear receptor protein subfamily 1, group 1, member 1) is a nuclear protein that is encoded by the PPARA gene in humans and is a member of the steroid hormone receptor superfamily [3]. Activation of PPAR-α promotes uptake, utilization, and catabolism of fatty acids and helps regulate several aspects of lipid metabolism [4, 5]. In hepatocytes, gemfibrozil and other fibric acid derivatives decrease triglycerides and VLDL levels by increasing VLDL catabolism and triglyceride clearance. Triglyceride metabolism is regulated by lipoprotein lipase (LPL), a water-soluble enzyme that hydrolyzes triglycerides in lipoproteins, such as those found in chylomicrons and very low-density lipoproteins (VLDL). APOC3 is an inhibitor of lipoprotein lipase and is thought to inhibit the hepatic uptake of triglyceride-rich particles. Production of Apo C-III is decreased and LPL activity is increased by fibrate therapy. This results in reduced circulating levels of TG and VLDL. The reduction in serum triglyceride levels results in a beneficial alteration in the size and composition of LDL particles. Particles are transformed from small, dense, atherogenic LDL particles to large, bouyant, non-atherogenic LDL particles. This alteration in particle size is important as small, dense LDL-C particles are more highly atherogenic due to increased susceptibility to oxidation [4]. In general, gemfibrozil will slightly lower LDL-C but only in individuals with normal triglyceride levels.
Fibric acid derivatives also increase HDL production by increasing the synthesis of Apo A-I and Apo A-II [6, 7]. The degree of reduction in triglycerides as well as the increase in HDL-C observed with fibric acid therapy will vary depending on baseline values. On average, triglycerides are reduced by 25 to 50 %, but may be reduced by as much as 74 % in patients with severe hypertriglyceridemia, while HDL-C levels are increased on average by 10–20 % but may be increased by as much as 35 % [8]. Gemfibrozil is indicated for the treatment of patients with hypertriglyceridemia (types IV and V hyperlipidemia) who are at risk for pancreatitis and for reducing the risk of developing CAD in type IIb patients.
Efficacy in Reducing Cardiovascular Disease Events
Three major trials have evaluated the effects of fibrate monotherapy on cardiovascular events. The Helsinki Heart Study (HHS) was a primary prevention trial evaluating the efficacy of increasing HDL-C and lowering non-HDL-C with gemfibrozil in reducing the risk of CHD events [9]. Middle-aged men (n = 4081) aged 40–55 years with primary dyslipidemia (non-HDL-C ≥200 mg/day) were randomized to gemfibrozil 600 mg twice daily or to placebo. Results demonstrated an increase in HDL-C from 47 mg/dL at baseline to 51 mg/dL (8 % increase) and decreased non-HDL-C from 196 to 174 mg/dL (12 % decrease). In patients randomized to gemfibrozil, there was a 34 % reduction in the relative risk of fatal and non-fatal MI (p < 0.02). The patients who demonstrated the highest reduction in event rates were those with higher baseline triglycerides levels (>200 mg/dL) and high LDL-C/HDL-C ratios (>5). While there was a reduction in fatal and non-fatal MI, there was no difference in total mortality between the two groups. The study was underpowered to adequately assess the mortality endpoint.
However, based on the results of this trial, gemfibrozil received an indication for primary prevention cardiovascular disease in patients with the lipid triad of increased triglycerides, increased LDL-C, and decreased HDL-C.
The Veterans Affairs High-Density Lipoprotein Intervention (VA-HIT) trial was a secondary prevention trial examining the effect of gemfibrozil monotherapy compared to placebo on major CHD events in patients with dyslipidemia [10]. The study included 2531 men with a history of CHD who had low HDL-C (mean 32 mg/dL) and low LDL-C (mean 111 mg/dL). Patients were randomized to receive either 600 mg twice daily of gemfibrozil or placebo. Treatment with gemfibrozil resulted in 7 % increase in HDL-C and 31 % reduction in triglycerides. The concentration of HDL-C was inversely related to CHD events. Multivariate analysis showed an 11 % reduction in CHD events with gemfibrozil for every 5 mg/dL increase in HDL-C (p = 0.02). Neither triglyceride nor LDL-C levels predicted events. Overall, there was a significant 22 % relative risk reduction in death from CHD in patients assigned to gemfibrozil (p < 0.006). There was also a 24 % reduction in the combined endpoint of death from coronary revascularization, hospitalization due to angina, non-fatal MI, and stroke (p < 0.001). This study suggests that the use of gemfibrozil may provide significant additional risk reduction in patients with low LDL-C (range 100–129 mg/dL). A follow-up study from the VA-HIT trial demonstrated that the patients that derived the most benefit from gemfibrozil were those with diabetes or who were insulin resistant [11]. Gemfibrozil is also useful in treating hypertriglyceridemia associated with protease inhibitors in patients with human immunodeficiency virus (HIV) or acquired immune deficiency syndrome (AIDS) [12].
Muscle-Related Adverse Effects of Gemfibrozil-Statin Combination Therapy
Co-administration of a statin with a fibrate may sometimes be warranted for the treatment of complex dyslipidemias with hypertriglyceridemia to reduce the risk of pancreatitis. However, concerns have emerged regarding gemfibrozil-statin combination therapy due to an observed increase in the risk of muscle-related adverse effects. Evaluating the adverse effects of gemfibrozil-statin combination therapy is complicated as both agents are associated with muscle-related adverse effects when used in monotherapy. The combined use of a statin with a fibrate is known to increase the risk of muscle-related adverse effects and use of this combination is often an exclusion criterion in randomized controlled trials of lipid-lowering drugs [13]. Therefore, evaluating statin-associated adverse events and drug interactions through either the FDA Adverse Event Reporting System (AERS) case reports or uncontrolled observational studies is common [14]. Muscle-associated adverse effects are of most concern and most clinically relevant.
The spectrum of statin-associated muscle adverse events that can increase with statin-gemfibrozil combination therapy include myalgia, myositis, and myonecrosis, with clinical rhabdomyolysis representing the most serious, life-threatening, though rare, manifestation [15]. Data from the FDA AERS have demonstrated that the risk of rhabdomyolysis with statin therapy is significantly increased when administered in combination with a fibrate [16]. Moreover, the gemfibrozil-statin combination results in a 15 times higher rate of rhabdomyolysis compared with statin-fenofibrate combination [17•]. Large-scale population-based studies have used administrative and pharmacy claims data to evaluate the safety of statin-gemfibrozil combination therapy [18–20]. These analyses demonstrate that the risk of statin-associated muscle adverse events, including clinical rhabdomyolysis, is significantly increased with statin-fibrate combination therapy and the risk is greatest with gemfibrozil-statin co-administration.
Gemfibrozil-Statin Drug-Drug Interaction Warnings
There have been several attempts to alert clinicians of the importance of the statin-gemfibrozil drug-drug interactions. These attempts started with an ACC/AHA Clinical Advisory in 2002 that reinforced the labeling dose limits for certain statins when used in combination with gemfibrozil [21]. However, clinicians continued to prescribe this combination with certain statins (especially simvastatin and lovastatin) that went against FDA labeling [22]. A 2011 revision of the FDA labeling for simvastatin addressed the gemfibrozil-statin interaction by listing that simvastatin in combination with gemfibrozil as contraindicated [23].
Gemfibrozil Pharmacokinetics
Gemfibrozil is rapidly absorbed with nearly 100 % bioavailability, achieving peak plasma concentrations within 1–2 h. It is highly protein-bound, has a short elimination T1/2 of approximately 1.5 h, and must be dosed twice daily [24]. The active drug undergoes conjugation to its acyl glucuronide, gemfibrozil 1-0-β glucuronide, which is then oxidized via the CYP450 system [25]. The majority of the gemfibrozil dose is eliminated as the glucuronide conjugate in the urine with little excreted as unchanged gemfibrozil.
Pharmacokinetic interactions, in which a drug affects the absorption, distribution, metabolism, or excretion of a co-administered drug, play an important role in the increased risk of muscle-related adverse effects with gemfibrozil-statin combination therapy. Most of the clinically significant drug-drug interactions with statin therapy are a result of cytochrome (CYP) p-450 isoenzyme system inhibition. Gemfibrozil is an inhibitor of CYP2C8, but this only affects fluvastatin. Therefore, the interaction between gemfibrozil and statin therapy is most likely explained through different mechanisms, though exact pathways and explanations of this complex interaction are not completely defined. Unlike fenofibrate or fenofibric acid, gemfibrozil, alters the pharmacokinetics of most statins and multiple mechanisms contribute to this interaction [26].
Gemfibrozil can inhibit membrane transporters, including the organic anion transporting polypeptide (OATP) 1B1. This transporter mediates transport of statins from portal circulation into the hepatocyte into the liver [27]. Inhibiting this step could result in increased plasma statin concentrations [28]. Elimination of statins and/or their metabolites into the bile and feces is also mediated by transport proteins. One of the multiple transporters involved with this elimination pathway includes OATP1B1.
Gemfibrozil may also inhibit statin metabolism by inhibition of hepatic glucuronidation [29, 30]. Several statins undergo glucuronidation via the UDP glucuronosyl transferase followed by lactonization as a route of elimination. In particular, the lipid-soluble statins, simvastatin, and lovastatin, are converted to water soluble salts and glucuronide conjugates that are subsequently eliminated from the body. Lactonized statins may be converted to the open acid form again by an esterase enzyme and metabolized, excreted into the bile or urine, or directly metabolized by the CYP 450 system. Inhibition of glucuronidation by gemfibrozil may inhibit subsequent metabolic steps and elimination of these statins.
Magnitude of Gemfibrozil on Statin Systemic Exposure
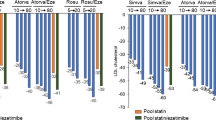
The effect of gemfibrozil on statin pharmacokinetic parameters, specifically maximum concentration (C max), has been summarized by the National Lipid Association, based on data evaluating this interaction (Table 1) [13]. In a double-blind, randomized, crossover, single-dose pharmacokinetic study of healthy volunteers, subjects were pretreated with gemfibrozil 600 mg or placebo twice daily for 2 days. On day 3, simvastatin 40 mg was co-administered with the morning dose of gemfibrozil. Gemfibrozil increased the mean AUC of simvastatin by only 35 % (p < 0.01), but the AUC of the active form, simvastatin acid, was increased by approximately 2–3-fold (p < 0.001) [31]. In a similar study of healthy volunteers, gemfibrozil increased the mean AUC of active lovastatin acid by nearly 3-fold (p < 0.001), but did not affect the plasma concentrations of the parent lovastatin [32].
The effects of multiple dose administration of gemfibrozil and fenofibrate on the single-dose pharmacokinetics of atorvastatin were compared in a study of 72 healthy volunteers. The AUC values of atorvastatin and its active metabolites (2-hydroxyatorvastatin acid, 2-hydroxyatorvastatin lactone, 4-hydroxyatorvastatin lactone) were modestly (1.3- to 1.7-fold) but significantly increased when co-administered with gemfibrozil, but there was no significant effect of gemfibrozil on atorvastatin lactone [33, 34]. It is suggested that these interactions are likely due to the inhibition of OATP2-mediated hepatic uptake, since simvastatin, lovastatin, and atorvastatin are metabolized by CYP3A4 which is not significantly affected by gemfibrozil. There was no significant pharmacokinetic interaction with fenofibrate-atorvastatin combination therapy and no increase in systemic exposure to atorvastatin.
The pharmacokinetic profile of pravastatin differs from other available statins. Pravastatin is more hydrophilic and does not easily cross cell membranes, relying upon transporters for absorption, tissue uptake, and elimination. The drug is minimally metabolized and is not significantly eliminated via CYP450 enzymes. Of the bioavailable dose, approximately 47 % undergoes renal elimination primarily in the unchanged form. However, in a single-dose pharmacokinetic study (n = 10 healthy volunteers) gemfibrozil increased plasma pravastatin concentrations by 202 % (range, 40–412 %) and significantly reduced mean renal clearance of the drug from 25 L/h to 14 L/h (p < 0.0001) in healthy volunteers without a significant effect on amount of pravastatin excreted in the urine (p = 0.42) [35•]. It is hypothesized that these findings are consistent with the inhibition of OATP1B1-mediated uptake as well as inhibition of the renal transporter OAT3.
Drug-drug interactions with rosuvastatin are thought to primarily involve drug transporters rather enzymes. Active uptake via the OATP2, OATP1B1/3, NTCP, BRCP, and NTCP transporters and active renal tubular secretion via OAT3 are important for the elimination of rosuvastatin [36•]. Though CYP2C9 plays a minor role in rosuvastatin clearance, the drug is not extensively metabolized and is primarily eliminated unchanged in the urine and feces. Gemfibrozil increased plasma concentrations of rosuvastatin by approximately 2-fold (range, 1.56–1.88) in a randomized, double-blind crossover trial of gemfibrozil or placebo in 20 healthy volunteers [37•]. In an accompanying in vitro study of the effect of gemfibrozil on the uptake of radiolabeled rosuvastatin by OATP2-transfected oocytes demonstrated a 50 % reduction in uptake, suggesting gemfibrozil inhibition of OATP2-mediated hepatic uptake may contribute to the mechanism of the drug-drug interaction. Metabolism is only a minor pathway for pitavastatin via CYP2C9 which is unaffected by gemfibrozil. Fluvastatin transport in human hepatocytes via the OATP1B1, OATP2B1, and OATP1B3 transporters is potently inhibited by gemfibrozil [38]. However, in at least one study of 17 subjects with hyperlipidemia and established ASCVD or family history of CAD, no significant difference was observed in AUC and C max when comparing the combination with fluvastatin or gemfibrozil alone [39•].
Role in Clinical Practice
The clinical relevance of the gemfibrozil-statin drug-drug interaction must be considered. The ACC/AHA Blood Cholesterol Guidelines state that “gemfibrozil should not be initiated in patients on statin therapy because of an increased risk for muscle symptoms and rhabdomyolysis (Classification of Recommendation, III Harm; Level of Evidence B)” [1]. The European Society of Cardiology/European Atherosclerosis Society Guidelines for the Management of Dyslipidaemias also state that “avoidance of adding gemfibrozil to a statin regimen is advised” [40]. These recommendations are derived from observational data and statin trials in which patients with serious comorbidities and those taking concomitant medications (including gemfibrozil) that may increase the risk of statin muscle related toxicity were excluded, rather than on specific pharmacokinetic data.
The FDA-approved statin product labels for statins comment on the gemfibrozil-statin drug-drug interaction (see Table 2) [41–47]. They all state that combined use of gemfibrozil with statins “should be avoided” and that the combination of gemfibrozil with simvastatin is contraindicated. The FDA-approved labeling for rosuvastatin additionally states that if used in combination with gemfibrozil, the maximum dose of rosuvastatin should not exceed 10 mg daily. However, clinicians often do not comply with FDA-labeled statin dose restrictions when prescribing combinations known to result in drug-drug interactions [22, 48•].
Pharmacokinetic data and available evidence from statin RCTs and observational studies indicate a clear increased risk of muscle-related adverse effects with gemfibrozil-statin therapy. When the combination of a fibrate and statin therapy is indicated, fenofibrate or fenofibric acid are the preferred agents in combination with a statin. However, pharmacokinetic studies indicate that the magnitudes of the interaction between gemfibrozil and atorvastatin, pitavastatin, and rosuvastatin are minor, while the magnitudes for lovastatin, pravastatin, and simvastatin are moderate.
Conclusion
Recommendations for Safe Use of Gemfibrozil
There are clinical circumstances in which fenofibrate is not an available option. These may include when fenofibrate is poorly tolerated, when the cost of fenofibrate/fenofibric acid is prohibitive, or when gemfibrozil is the only available fibrate offered by payer. Therefore, the clinician must consider if gemfibrozil is a safe option. Based on pharmacokinetic data, demonstrating a significant, greater than 2-fold increase in statin systemic exposure when co-administered with gemfibrozil, under all circumstances, the use of gemfibrozil should be avoided in combination with lovastatin, pravastatin, and simvastatin. If gemfibrozil must be used in combination with atorvastatin, pitavastatin, or rosuvastatin, consideration should be given to use a lower statin dose in order to minimize risk of muscle-related adverse effects. The use of rosuvastatin in combination with gemfibrozil is included in FDA-approved product labeling, but it is recommended that the daily dose of rosuvastatin be limited to 10 mg daily.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Stone NJ, Robinson JG, Lichtenstein AH, et al. 2013 ACC/AHA Guideline on the Treatment of Blood Cholesterol to Reduce Atherosclerotic Cardiovascular Risk in Adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014;129:S1–45.
Jacobson TA, Ito MK, Maki KC, et al. National Lipid Association recommendations for patient-centered management of dyslipidemia: part 1—executive summary. J Clin Lipidol. 2014;8:473–88.
Sher T, Yi HF, McBride OW, Gonzalez FJ. cDNA cloning, chromosomal mapping, and functional characterization of the human peroxisome proliferator activated receptor. Biochemistry. 1993;32:5598–604.
Staels B, Dallongeville J, Auwerx J, Schoonjans K, Leitersdorf E, Fruchart JC. Mechanism of action of fibrates on lipid and lipoprotein metabolism. Circulation. 1998;98:2088–93.
Packard CJ. Overview of fenofibrate. Eur Heart J. 1998;19(Suppl A):A62–5.
Bard JM, Parra HJ, Camare R, et al. A multicenter comparison of the effects of simvastatin and fenofibrate therapy in severe primary hypercholesterolemia, with particular emphasis on lipoproteins defined by their apolipoprotein composition. Metab Clin Exp. 1992;41:498–503.
Lussier-Cacan S, Bard JM, Boulet L, et al. Lipoprotein composition changes induced by fenofibrate in dysbetalipoproteinemia type III. Atherosclerosis. 1989;78:167–82.
Leaf DA, Connor WE, Illingworth DR, Bacon SP, Sexton G. The hypolipidemic effects of gemfibrozil in type V hyperlipidemia. A double-blind, crossover study. JAMA. 1989;262:3154–60.
Frick MH, Elo O, Haapa K, et al. Helsinki Heart Study: primary-prevention trial with gemfibrozil in middle-aged men with dyslipidemia. Safety of treatment, changes in risk factors, and incidence of coronary heart disease. N Engl J Med. 1987;317:1237–45.
Rubins HB, Robins SJ, Collins D, et al. Gemfibrozil for the secondary prevention of coronary heart disease in men with low levels of high-density lipoprotein cholesterol. Veterans Affairs High-Density Lipoprotein Cholesterol Intervention Trial Study Group. N Engl J Med. 1999;341:410–8.
Rubins HB, Robins SJ, Collins D, et al. Diabetes, plasma insulin, and cardiovascular disease: subgroup analysis from the Department of Veterans Affairs High-Density Lipoprotein Intervention Trial (VA-HIT). Arch Intern Med. 2002;162:2597–604.
Henry K, Melroe H, Huebesch J, et al. Atorvastatin and gemfibrozil for protease-inhibitor-related lipid abnormalities. Lancet. 1998;52:1031–2.
Jacobson TA. NLA Task Force on Statin Safety—2014 update. J Clin Lipidol. 2014;8:S1–4.
Omar MA, Wilson JP. FDA adverse event reports on statin-associated rhabdomyolysis. Ann Pharmacother. 2002;36:288–95.
Rosenson RS, Baker SK, Jacobson TA, Kopecky SL, Parker BA. An assessment by the Statin Muscle Safety Task Force: 2014 update. J Clin Lipidol. 2014;8:S58–71.
Chang JT, Staffa JA, Parks M, Green L. Rhabdomyolysis with HMG-CoA reductase inhibitors and gemfibrozil combination therapy. Pharmacoepidemiol Drug Saf. 2004;13:417–26.
Jones PH, Davidson MH. Reporting rate of rhabdomyolysis with fenofibrate + statin versus gemfibrozil + any statin. Am J Cardiol. 2005;95:120–2. This was a search of the FDA adverse event report system to evaluate if the incidence of myotoxicity association with the statin/fibrate combination is greater with the combination of gemfibrozil/statin or fenofibrate/statin. There were fewer reports of rhabdomyolysis with fenofibrate than with gemfibrozil when used in combination with a statin.
Andrade SE, Graham DJ, Staffa JA, et al. Health plan administrative databases can efficiently identify serious myopathy and rhabdomyolysis. J Clin Epidemiol. 2005;58:171–4.
Cziraky MJ, Willey VJ, McKenney JM, et al. Statin safety: an assessment using an administrative claims database. Am J Cardiol. 2006;97:61C–8.
Graham DJ, Staffa JA, Shatin D, et al. Incidence of hospitalized rhabdomyolysis in patients treated with lipid-lowering drugs. JAMA. 2004;292:2585–90.
Pasternak RC, Smith Jr SC, Bairey-Merz CN, Grundy SM, Cleeman JI, Lenfant C. ACC/AHA/NHLBI clinical advisory on the use and safety of statins. J Am Coll Cardiol. 2002;40:567–72.
Alford JC, Saseen JJ, Allen RR, Nair KV. Persistent use of against-label statin-fibrate combinations from 2003–2009 despite United States Food and Drug Administration dose restrictions. Pharmacotherapy. 2012;32:623–30.
U.S. Food and Drug Administration. FDA drug safety communication: new restrictions, contraindications, and dose limitations for Zocor (simvastatin) to reduce the risk of muscle injury. Published 6/8/11, Available at: http://www.fda.gov/Drugs/DrugSafety/ucm256581.htm.
Gemfibrozil (Lopid) prescribing information. New York, NY: Parke-Davis; 2012.
VandenBrink BM, Isoherranen N. The role of metabolites in predicting drug-drug interactions: focus on irreversible cytochrome P450 inhibition. Curr Opin Drug Discov Dev. 2010;13:66–77.
Bellosta S, Paoletti R, Corsini A. Safety of statins: focus on clinical pharmacokinetics and drug interactions. Circulation. 2004;109:III50–7.
Kalliokoski A, Niemi M. Impact of OATP transporters on pharmacokinetics. Br J Pharmacol. 2009;158:693–705.
Shitara Y, Hirano M, Sato H, Sugiyama Y. Gemfibrozil and its glucuronide inhibit the organic anion transporting polypeptide 2 (OATP2/OATP1B1:SLC21A6)-mediated hepatic uptake and CYP2C8-mediated metabolism of cerivastatin: analysis of the mechanism of the clinically relevant drug-drug interaction between cerivastatin and gemfibrozil. J Pharmacol Exp Ther. 2004;311:228–36.
Prueksaritanont T, Tang C, Qiu Y, Mu L, Subramanian R, Lin JH. Effects of fibrates on metabolism of statins in human hepatocytes. Drug Metab Dispos: Biol Fate Chem. 2002;30:1280–7.
Prueksaritanont T, Zhao JJ, Ma B, et al. Mechanistic studies on metabolic interactions between gemfibrozil and statins. J Pharmacol Exp Ther. 2002;301:1042–51.
Backman JT, Kyrklund C, Kivisto KT, Wang JS, Neuvonen PJ. Plasma concentrations of active simvastatin acid are increased by gemfibrozil. Clin Pharmacol Ther. 2000;68:122–9.
Kyrklund C, Backman JT, Kivisto KT, Neuvonen M, Laitila J, Neuvonen PJ. Plasma concentrations of active lovastatin acid are markedly increased by gemfibrozil but not by bezafibrate. Clin Pharmacol Ther. 2001;69:340–5.
Whitfield LR, Porcari AR, Alvey C, Abel R, Bullen W, Hartman D. Effect of gemfibrozil and fenofibrate on the pharmacokinetics of atorvastatin. J Clin Pharmacol. 2011;51:378–88.
Backman JT, Luurila H, Neuvonen M, Neuvonen PJ. Rifampin markedly decreases and gemfibrozil increases the plasma concentrations of atorvastatin and its metabolites. Clin Pharmacol Ther. 2005;78:154–67.
Kyrklund C, Backman JT, Neuvonen M, Neuvonen PJ. Gemfibrozil increases plasma pravastatin concentrations and reduces pravastatin renal clearance. Clin Pharmacol Ther. 2003;73:538–44. This was a randomized placebo-controlled, 2-phase crossover study to evaluate the effects of gemfibrozil on the pharmacokinetics of pravastatin. The results showed an increase in the plasma concentration of pravastatin when combined with gemfiborzil. The increase is in part related to a reduction in renal clearance of pravastatin.
Bergman E, Matsson EM, Hedeland M, Bondesson U, Knutson L, Lennernas H. Effect of a single gemfibrozil dose on the pharmacokinetics of rosuvastatin in bile and plasma in healthy volunteers. J Clin Pharmacol. 2010;50:1039–49. This study evaluated the plasma pharmacokinetics and biliary excretion effects of gemfibrozil on rosuvastatin. Bile and plasma levels were obtained every 20 minutes following administration for 200 minutes. The results showed that gemfibrozil increased the area under the curve by 1.56 fold, indicating that the interaction is minor and perhaps the combination can be used.
Schneck DW, Birmingham BK, Zalikowski JA, et al. The effect of gemfibrozil on the pharmacokinetics of rosuvastatin. Clin Pharmacol Ther. 2004;75:455–63. This was a randomized, double-blind trial, 2 period cross over trial evaluating the effects of gemfibrozil on the plasma concentration of rosuvastatin and its metabolites. The results showed that the area under the curve of rosuvastatin was increased by approximately 2 fold. The mechanism may be secondary to the inhibition of OATP2-mediated rosuvastatin hepatic uptake by gemfibrozil.
Noe J, Portmann R, Brun ME, Funk C. Substrate-dependent drug-drug interactions between gemfibrozil, fluvastatin and other organic anion-transporting peptide (OATP) substrates on OATP1B1, OATP2B1, and OATP1B3. Drug Metab Dispos: Biol Fate Chem. 2007;35:1308–14.
Spence JD, Munoz CE, Hendricks L, Latchinian L, Khouri HE. Pharmacokinetics of the combination of fluvastatin and gemfibrozil. Am J Cardiol. 1995;76:80A–3. This was an open-label, crossover study evaluating the impact of gemfibrozil on the pharmacokinetics of fluvastatin. The results showed no significant impact on area under the curve, time to maximum concentration, or maximum plasma concentration when fluvastatin is combined with gemfibrozil.
Reiner Z, Catapano AL, De Backer G, et al. ESC/EAS guidelines for the management of dyslipidaemias: the Task Force for the Management of Dyslipidaemias of the European Society of Cardiology (ESC) and the European Atherosclerosis Society (EAS). Eur Heart J. 2011;32:1769–818.
Atorvastatin (Lipitor) prescribing information. New York, NY: Pfizer; 2013.
Fluvastatin sodium (Lescol, Lescol XL) prescribing information. East Hanover, NJ: Novartis Pharmaceuticals Corporation; 2012.
Lovastatin (Mevacor) prescribing information. Whitehouse Station, NJ: Merck & Co., INC; 2014.
Pravastatin (Pravachol) prescribing information. Princeton, NJ: Bristol-Myers Squibb; 2013.
Rosuvastatin (Crestor) prescribing information. Wilmington, DE: AztraZeneca Pharmaceuticals; 2013.
Simvastatin (Zocor) prescribing information. Whitehouse Station, NJ: Merck & Co., INC; 2014.
Pitavastatin (Livalo) prescribing information. Montgomery, AL: Kowa Pharmaceuticals; 2013.
Tuchscherer RM, Nair K, Ghushchyan V, Saseen JJ. Simvastatin prescribing patterns before and after FDA dosing restrictions: a retrospective analysis of a large healthcare claims database. Am J Cardiovasc Drugs : Drugs Devices Other Interv. 2015;15:27–34. These references provide detail the effects of specific statins and their interaction with gemfibrozil. Given the differences in metabolism of statins the clinical impact of coadministration with gemfibrozil varies. Understanding these differences will assist with clinical decision making.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Barbara S. Wiggins and Joseph J. Saseen declare that they have no conflict of interest.
Pamela B. Morris declares serving on the advisory board for Amgen, AstraZeneca, and Sanofi Regeneron.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Statin Drugs
Rights and permissions
About this article
Cite this article
Wiggins, B.S., Saseen, J.J. & Morris, P.B. Gemfibrozil in Combination with Statins—Is It Really Contraindicated?. Curr Atheroscler Rep 18, 18 (2016). https://doi.org/10.1007/s11883-016-0571-8
Published:
DOI: https://doi.org/10.1007/s11883-016-0571-8