Abstract
Purpose of Review
Asthma is one of the most common chronic respiratory diseases worldwide, yet only a small percentage of patients are categorized as having severe disease. Severe asthmatics, however, are responsible for the largest burden of healthcare costs and lost productivity. Several recent guidelines have addressed disease pathogenesis and treatment modalities for these complex patients. Herein, we review the severe asthma guidelines, compare the existing guidelines, address key areas that are yet to be addressed in the guidelines, and discuss future directions for severe asthma research.
Recent Findings
This is a narrative review of the 2019 European Respiratory Society/American Thoracic Society (ERS/ATS) and Global Initiative for Asthma (GINA) guidelines that specifically address the diagnosis and management of severe asthma. The pathophysiological mechanisms that underlie severe asthma are reviewed, and novel therapies that target specific pathophysiological pathways in severe asthma are discussed in detail. Although the guidelines address the use of novel biological therapies for patients with T2-mediated disease, data comparing these agents remain sparse. This review addresses several areas that are topics beyond the guidelines and highlight key areas where future research is warranted.
Summary
This review provides a comprehensive overview of the current state of severe asthma treatment and discusses potential avenues for future research for this patient population.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Asthma is estimated to effect 334 million people worldwide [1]. Approximately 4–10% of patients with asthma are categorized as having severe disease [2,3,4,5]. Despite this group of patients making up a minority of asthma patients, the largest burden of healthcare costs associated with asthma are dedicated to treatment of patients with severe disease [6, 7].
Patients with severe asthma are notoriously difficult to treat given refractory disease to standard asthma therapies and frequent dependence on oral corticosteroids. Until the last decade, there has been a paucity of information for clinicians on how to identify these patients and approach disease management. In 2009 and 2010, the first attempts to define severe asthma were made by the European Respiratory Society (ERS) [8] and American Thoracic Society (ATS) [9]. The initial guidelines allowed for the appropriate epidemiologic investigation of severe asthma and created standard definitions for use in clinical trials involving this subgroup of patients.
Over the past decade, there has been a growing body of literature around the optimal approach for management of patients with severe asthma. Further, the development of new biologic agents targeted towards specific inflammatory pathways has revolutionized the way these patients are treated. As a result, guidelines specifically providing an evidence-based approach to management of severe asthma have been developed with the most recent renditions being published in 2019 by ERS/ATS [10••] and by the Global Initiative for Asthma (GINA) [11••]. The purpose of this article is to discuss the diagnosis and management of adolescent and adult severe asthma in context of the most recent updates to the severe asthma guidelines. Limitations of the current guidelines as well as opportunities for future research around treatment of severe asthma will also be addressed.
Current Asthma Guidelines
There are multiple guidelines for the diagnosis and management of asthma currently available. The majority of these concentrate on the approach to treatment of mild to moderate asthma. Only a subset addresses the management of severe disease. Table 1 provides an overview of current asthma guidelines including target population and date of most recent publication. The remainder of this article will discuss the 2019 ERS/ATS and GINA guidelines that specifically address the diagnosis and management of severe asthma.
Of important note, GINA releases a yearly report on asthma management. Reference to these general guidelines will be notated as GINA Main Report along with the year published. The notation GINA SA will refer specifically to the severe asthma guidelines.
Approach to the Diagnosis of Severe Asthma
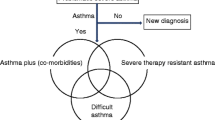
The ERS/ATS and GINA SA guidelines provide specific definitions for the diagnosis of severe asthma. Both sets of guidelines highlight that severe asthma must be differentiated from difficult-to-treat disease, which is asthma that exhibits improved control with optimal adherence to asthma treatment or after diagnosis and management of potential co-morbidities or confounders. Finally, determination of a patient’s asthma inflammatory phenotype is crucial to determining the optimal treatment strategy for severe asthma patients.
Diagnosing Severe Asthma
The ERS/ATS guidelines define severe asthma as asthma requiring treatment with high-dose inhaled corticosteroids plus a second controller medication and/or systemic corticosteroids to maintain asthma control or that remains uncontrolled despite the aforementioned medications [5]. GINA SA similarly defines severe asthma as a subset of difficult-to-treat asthma or asthma that is uncontrolled despite adherence with maximal optimized therapy (GINA step 4–5 treatment including medium- or high-dose ICS with a second controller or maintenance oral corticosteroid) and treatment of contributory factors or that worsens when high-dose treatment is decreased [11••].
The need to rule-out confounding diagnoses is vital to making an accurate diagnosis of severe asthma. When appropriate, this includes evaluation for diseases that may mimic or be associated with asthma, but may not respond to standard asthma therapies. Specific diagnoses to consider include allergic bronchopulmonary aspergillosis (ABPA), eosinophilic granulomatosis with polyangiitis (EGPA), bronchiectasis, tracheobronchomalacia, endobronchial lesions (e.g., foreign bodies, tumors, strictures), interstitial lung diseases, vocal cord dysfunction, and/or congestive heart failure [5, 11••].
Determining Asthma Phenotype
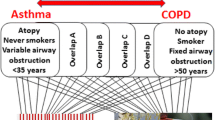
Severe asthma can be further subdivided based on the underlying inflammatory pathways driving asthma pathogenesis. Specifically, differentiating between a type 2 (T2) versus non-type 2 (non-T2) inflammatory phenotype is crucial for determining which asthma therapies a patient may ultimately be eligible for.
Approximately 55% of patients with severe asthma exhibit T2 inflammation [12, 13]. The T2 phenotype has been shown to be associated with increased asthma severity, increased frequency of exacerbations, and reduced lung function [14,15,16]. The T2 phenotype is classically described as allergic eosinophilic asthma and involves cytokines such as interleukin-4 (IL-4), interleukin-5 (IL-5), and interleukin-13 (IL-13), which drive eosinophil recruitment to the airway. T2 asthma is identified by clinical biomarkers including increased blood and sputum eosinophil counts, elevated immunoglobulin E (IgE) level, and elevated fractional exhaled nitric oxide (FeNO) [17].
If a patient does not exhibit evidence of T2 inflammation, they are considered to have a non-T2 phenotype. This subset of patients tends to include those with adult-onset disease, obese subjects, or individuals who have a history of exposure to cigarette smoke [18•]. Additionally, these individuals often respond poorly to corticosteroid therapy [19, 20]. While patients with non-T2 asthma often exhibit neutrophilic airway inflammation, there is a subgroup of patients that exhibit little airway inflammation and are classified as having smooth muscle-mediated paucigranulocytic asthma [18•]. Identification of other clinical biomarkers that can be used to identify this subgroup of patients remains an area of active research.
Guideline-Directed Treatment of Severe Asthma
The ERS/ATS and GINA SA guidelines published in 2019 provide an updated framework for managing severe asthma patients. The following sections will discuss severe asthma management in the context of the new guidelines, and key differences between the guidelines will be highlighted.
Biologic Therapies
Treatment of severe asthma has evolved over the past decade with the development of targeted biologic therapies aimed at downregulating the T2 inflammatory cascade. Currently, there are five US Federal Drug Administration (FDA)-approved biologic therapies for the treatment of severe T2 asthma, with several other agents currently in development. Clinically, these therapies have been shown to reduce exacerbations [21, 22••, 23,24,25,26,27,28,29], decrease corticosteroid use [27, 30, 31••], improve lung function [22••, 23, 29, 32], and subjectively improve symptoms in patients with severe T2 asthma [23, 33, 34]. The 2019 severe asthma guidelines recommend consideration of add-on biologic therapy in adolescents and adults with uncontrolled severe asthma who exhibit a T2 inflammatory phenotype and/or with severe corticosteroid–dependent asthma [10••, 11••].
The first biologic agent used for treatment of severe asthma was the humanized recombinant monoclonal anti-IgE antibody, Omalizumab. Initially approved in 2003, Omalizumab functions via binding directly to IgE. These Omalizumab–IgE complexes prevent IgE from interacting with the IgE receptors on the surface of mast cells and basophils leading to decreased release of allergic response mediators [35]. Selective use of Omalizumab as add-on therapy in patients classified as having severe allergic asthma (proven via sensitivity testing) with elevated IgE levels has been guideline recommended dating back to 2010 [36]. The current 2019 ERS/ATS and GINA SA guidelines give additional specifications for whom Omalizumab therapy should be considered. They recommend add-on anti-IgE therapy in adolescents and adults with severe allergic asthma whom have blood eosinophil levels ≥ 260/μl and elevated FeNO ≥ 20 [10••, 11••].
Three agents aimed at downregulating the IL-5 inflammatory pathway have also been developed. Mepolizumab and Reslizumab are human monoclonal antibodies to IL-5 (anti-IL5). Through inactivation of IL-5, they lead to reduced levels of eosinophils and eosinophil precursors in the bone marrow and bronchial mucosa [37,38,39]. Benralizumab is a monoclonal antibody against the alpha chain of the IL-5 receptor (anti-IL5Rα) leading to antibody-dependent cell-mediated cytotoxicity of eosinophils and reduced levels of eosinophils in the bone marrow, blood, and peripheral tissues [37, 40]. Both the 2019 ERS/ATS and GINA SA guidelines recommend consideration of anti-IL5/IL5Rα therapies for adults with uncontrolled severe eosinophilic asthma [10••, 11••]; however, the guidelines differ in what biomarker cutoffs should be used when identifying patients likely to benefit from these therapies. The ERS/ATS guidelines recommend using a blood eosinophil cut-point of ≥ 150/μl to guide anti-IL5/IL5Rα initiation [10••], whereas the GINA SA guidelines recommend a blood eosinophil cutoff ≥ 300/μl [11••].
Most recently, the human monoclonal antibody, Dupilumab, was FDA approved for the management of moderate-to-severe asthma in October 2018. In addition to severe asthma, Dupilumab carries additional FDA approvals for the management of atopic dermatitis and chronic rhinosinusitis with nasal polyposis. Dupilumab binds with the alpha subunit of the IL-4 receptor, decreasing signal transduction of IL-4 and IL-13 leading to a decreased T2-mediated inflammation [41]. While just recently approved for severe asthma treatment, both the 2019 ERS/ATS and GINA SA guidelines recommend its consideration for add-on therapy for patients with severe eosinophilic asthma [10••, 11••]. The guidelines differ in which subset of severe asthmatic patients Dupilumab should be considered in. The ERS/ATS guidelines recommend use of Dupilumab in adults with severe eosinophilic asthma or those with severe corticosteroid-dependent asthma regardless of blood eosinophil levels [10••]. GINA SA recommends Dupilumab for adult patients with severe eosinophilic asthma and ongoing exacerbations who have blood eosinophils ≥ 150/μl, FeNO ≥ 25 ppb, need for maintenance oral corticosteroids, or in patients with concurrent moderate-to-severe atopic dermatitis and/or nasal polyposis [11••].
Long-Acting Muscarinic Agents
Long-acting muscarinic antagonists (LAMAs) have been shown to have beneficial effects on lung function and improved asthma control in adults and adolescents with severe asthma [42, 43•, 44]. Use of the LAMA, tiotropium, for add-on therapy in adolescents and adults with severe uncontrolled asthma despite GINA step 4–5 or NAEPP step 5 therapies is recommended by the ERS/ATS and GINA SA 2019 guidelines [10••, 11••]. GINA SA also recommends consideration of a trial of tiotropium in patients with severe asthma and a non-T2 inflammatory phenotype [11••]. Neither set of guidelines comments on if or how other LAMAs, such as umeclidinium and glycopyrroniumm, should be utilized citing the lack of data on use of these agents as adjunctive therapy in severe asthma.
Macrolide Antibiotics
Macrolide antibiotics have been shown to significantly reduce rates of asthma exacerbations and improve quality of life in adult patients with moderate-to-severe persistent asthma [45, 46]. While prior versions of the severe asthma guidelines had recommended against their use, the 2019 guidelines suggest a trial of chronic macrolide treatment with azithromycin or clarithromycin to reduce asthma exacerbations in severe adult asthmatics. However, its use in children and adolescents is not advised [10••, 11••]. GINA SA further specifies that macrolide use should be considered in non-T2 asthmatics or T2 asthmatics without access to biologic therapies. They note that use of chronic macrolide therapy for asthma is off-label, and there is the potential for the development of antibiotic resistance with use [11••]. Thus, the risks and benefits must be weighed and discussed with the patient when this therapy is considered.
Other Therapies
Bronchial thermoplasty has been shown to improve quality of life and decrease the rate of severe exacerbations in patients with severe asthma [47, 48]. Use of bronchial thermoplasty for management of severe asthma was addressed in the 2014 ERS/ATS severe asthma guidelines [5], but not in the most recent 2019 guidelines. GINA SA recommends consideration of bronchial thermoplasty in severe asthmatics with non-T2 inflammation and in patients with T2 inflammation that do not respond adequately to targeted biologic therapy [11••].
Finally, the use of anti-fungal agents are recommended for use in severe adult asthmatics with recurrent exacerbations of allergic bronchopulmonary aspergillosis. Use of anti-fungal agents in asthmatics without confirmed allergic bronchopulmonary aspergillosis is not recommended [5, 11••].
Treatment of Severe Asthma Beyond the Guidelines
It is well-known that there is significant heterogeneity among patients with severe asthma, and this heterogeneity is present even within the classic T2 and non-T2 asthma phenotypes [17, 18•]. Thus, it not surprising that the response to biologic therapy is variable within the clinical setting, despite patients with similar levels of clinical biomarkers. While the 2019 severe asthma guidelines provide evidence-based recommendations for use of targeted biologic therapy in severe asthmatics, there are several limitations to the current guidelines. The following sections will further consider the limitations of the 2019 severe asthma guidelines, with a primary focus on the choice and use of biologic therapies in the management of severe asthma. Current knowledge as well as opportunities for ongoing research in these areas will be discussed.
Choosing Between Biologic Therapies
The 2019 ERS/ATS and GINA SA guidelines provide a general framework for clinicians to use when considering initiation of biologic therapy in patients with severe eosinophilic asthma. However, there is minimal guidance on how to choose between the various biologic agents in a patient who may meet criteria for more than one agent.
Lack of direct comparisons between currently approved biologic therapies significantly contributes to the paucity of recommendations around choosing between biologic agents. Recently, use of indirect comparisons between biologic therapies has been attempted to try and fill this existing knowledge gap. However, the results of these studies are conflicting. A 2018 systemic review by Bourdin and colleagues found Benralizumab and Mepolizumab had similar efficacy in reducing exacerbation rates and improving FEV1 when baseline patient characteristics were matched across clinical trials [49]. A 2019 network meta-analysis by Casale and colleagues suggested Reslizumab to have greater efficacy than Benralizumab in patients with moderate-to-severe eosinophilic asthma with elevated peripheral eosinophil count and two or more exacerbations in the previous year [50]. However, other meta-analyses have failed to show significant benefit of one biologic over the others [51, 52]. Future studies directly comparing biologic therapies are necessary to clarify differences in efficacy between these agents.
Use of clinical biomarkers has also been proposed as a means for predicting treatment response, but their use in clinical practice remains controversial. Measurement of airway eosinophilia through induced sputum samples has been considered the gold standard for identifying patients with eosinophilic asthma [53]. However, lack of availability in routine clinical practice has led to preferential use of alternative non-invasive biomarkers for identifying patients with a T2 phenotype. Clinical biomarkers, including peripheral eosinophil count, FeNO, serum periostin, and number of exacerbations in the past year at baseline, have been evaluated for the use of predictors of therapeutic response in severe asthma [54, 55].
Several studies have shown that elevated peripheral eosinophil counts correlate with airway eosinophilia in severe asthma [56,57,58]. From studies on anti-IL5/anti-IL5Rα therapies, baseline peripheral eosinophil count has been the primary identified determinant of who may benefit from therapy, with increased levels of peripheral eosinophils predicting improved response [21, 25, 59, 60]. However, there are several important limitations to using peripheral eosinophilia as a sole marker for predicting biologic treatment response. For one, a low blood eosinophil count does not necessarily rule-out the presence of airway eosinophilia [61, 62] as the use of OCS therapy is known to lower the peripheral eosinophil count [63]. Furthermore, eosinophil counts have also been shown to fluctuate in a circadian pattern [64]. Thus, the timing of measurement may be an important consideration when measuring eosinophil counts. More recently, a combined predictor of peripheral eosinophil count and FeNO was shown to have greater predictive value for response to Mepolizumab therapy than using either clinical predictor alone. These findings were replicated in a cohort of patients treated with Dupilumab [65]. These results suggest composite clinical biomarkers may have superior clinical predictive value than use of a single biomarker alone and deserve exploration in future studies.
Serum periostin has been shown to be a reliable, non-invasive biomarker in the research setting for identification of eosinophilic inflammation in asthma [66]. There is limited data on use of serum periostin as a predictor for biologic treatment response; however, the few studies that have been performed show promising results. A study of 30 patients on Omalizumab therapy for at least one year showed that baseline periostin levels were significantly higher in patients without asthma exacerbations during the first year of treatment [67]. A retrospective analysis of the EXTRA study, which included 850 patients with uncontrolled severe persistent allergic asthma, showed a combined predictor with high peripheral eosinophil count (≥ 260/ml), FeNO (≥ 19.5 ppb), and periostin levels (≥ 50 ng/ml) at baseline predicted decreased asthma exacerbation rates after treatment with Omalizumab [68]. Finally, a phase IIb study of the anti-IL13 antibody, Lebrikizumab, for the treatment of steroid-resistant asthma showed that high periostin levels were significantly associated with the primary endpoint of greater FEV1 improvement at 12 weeks when compared to the placebo group. No efficacy was seen for Lebrikizumab in the low periostin group when compared to the placebo [69]. There are currently no published studies evaluating serum periostin and treatment response to anti-IL5/IL5Rα therapies or Dupilumab. Despite encouraging results from research studies, use of serum periostin as a predictive biomarker has seen minimal use in routine practice given lack of clinical availability. Further, use of this biomarker may be restricted to adults as it is produced from growing bone in children and adolescents making measurements difficult to interpret [70]. Additional studies confirming efficacy are ultimately needed prior to widespread implementation of this biomarker in clinical practice.
Finally, gene expression has also been considered as a potential biomarker for predicting treatment response in severe asthma. Research has previously demonstrated that both bronchial and nasal epithelial RNA sequencing can be used as biomarkers for differentiating asthma patients from controls [71, 72•] as well as for classifying asthmatic patients into T2 and non-T2 phenotypes [73]. Studies have additionally shown airway gene expression changes following initiation of asthma treatment [74], and baseline gene expression is associated with clinical response to inhaled corticosteroids [72•]. To date, there have been no studies using airway gene expression to predict response to biologic therapies, but this remains an area of opportunity for future research efforts.
Assessment of Biologic Treatment Response
The 2019 ERS/ATS and GINA SA guidelines provide limited guidance on assessment of treatment response to targeted therapies and duration of therapy. The ERS/ATS guidelines provide no comment on assessing response to treatment with biologic therapy [10••]. For biologic therapies, GINA suggests evaluating patients after 3–6 months of therapy and taking “exacerbations, symptom control, lung function, side-effects, treatment intensity (including OCS dose), and patient satisfaction” into consideration when determining treatment response [11••].
Part of the ambiguity around these guideline recommendations stems from lack of a standard definition in the literature for defining adequate treatment response to biologic therapy. Randomized control trials on biologic therapies have used improvement in lung function [22••, 23, 29, 32], decreased exacerbation rates [21, 22••, 23, 25,26,27,28,29], reduction in corticosteroid doses [27, 30, 31••], and subjective improvement in asthma symptoms [23, 33, 34] as outcomes for indicating treatment response. Additional studies have suggested use of reduction in serum biomarkers associated with T2 inflammation for assessing treatment response [75, 76]. However, no studies to date have proposed a standard definition for treatment response. Determining specific criteria that can be used to measure biologic treatment response will allow for improved definitions for determining efficacy of these therapies and provide much needed guidance for practitioners caring for severe asthma patients in the clinical setting.
Next Steps When Initial Biologic Treatment Fails
GINA SA provides guidance on the next steps for responders to biologic therapy, including guidance on tapering asthma therapy and when to consider stopping biologic therapy. However, questions remain on what to do if a patient is deemed to have treatment failure.
As per GINA SA, if a patient fails initial biologic therapy switching to a different biologic agent can be considered. Current research does suggest that switching between biologic therapies after initial failure may be efficacious for some patients. A 24-week prospective, multicenter, open-label study of 29 patients with severe eosinophilic asthma who had poor response to Omalizumab showed switching to Reslizumab led to a significant improvement in asthma symptoms with 60% of patients achieving control of their asthma [77]. Another study showed clinical benefit when patients with uncontrolled severe eosinophilic asthma were switched to Mepolizumab after failing Omalizumab [78]. Clinical improvement when switching between anti-IL5 therapies was also described [79]. Despite the potential clinical benefit of switching between biologic therapies after initial treatment failure, there remains a lack of guidance on how to approach switching between biologic therapies or which biologic to switch to.
Combination biologic therapy can also be considered in patients who have failed to achieve adequate response to biologic monotherapy. However, there is minimal information around use of more than one biologic for management of severe eosinophilic asthma, with current data limited to a case report of 3 patients [80]. Thus, combination biologic therapy remains an area for ongoing investigation and with a need for proof of efficacy and safety prior to this approach being widely recommended.
Treatment of Non-T2 Asthma
Medication strategies and targets for therapy in non-T2 asthma remains an area of ongoing research and clinical need. As such, consensus guidelines around treatment of non-T2 asthma are currently lacking. GINA SA recommends considering trial of LAMA, leukotriene modifier, macrolide, low-dose oral corticosteroid, and bronchial thermoplasty as potential therapeutic options [11••]. Which treatment strategy to choose and what to do if these treatment strategies fail however are not addressed. Expert opinion on treatment of non-T2 asthma additionally recommends lifestyle management, including smoking cessation, avoidance of secondhand smoker exposure, and weight loss in obese patients, given these co-morbidities are associated with non-T2 asthma [81]. Importantly, there are currently no approved biologic therapies for non-T2 asthma.
Limitation around developing non-T2 asthma guidelines partially stems from lack of a specific definition for identifying this group of patients. Currently, non-T2 asthma is the default phenotype when a patient is considered to have absent markers (e.g., peripheral eosinophilia, low FeNO, normal IgE) of T2 inflammation. However, patients can be easily mischaracterized into the non-T2 phenotype if they are on treatments (e.g., chronic oral corticosteroids) that suppress T2 inflammatory biomarkers. Further, this definition may overly generalize a heterogenous group of patients. For example, a study using gene expression profiling to categorize asthma patients identified three distinct transcriptional phenotypes of asthma. One subgroup appeared most similar to T2 asthma. The other two subgroups would both be classified under the non-T2 asthma phenotype despite having distinct gene expression profiles suggesting important differences between the groups [82]. Another study using sputum cell transcriptomics to classify patients similarly showed distinct non-T2 asthma subgroups [83]. These studies provide further evidence that the non-T2 asthma classification may be an oversimplification of complex inflammatory pathways. Thus, more specific definitions and identification of biomarkers to better classify patients within this non-T2 asthma population remain areas in need of further investigation.
Adolescents and Children with Severe Asthma
Currently, there are no dedicated guidelines to the management of severe asthma in the pediatric population. The 2019 ERS/ATS and GINA SA guidelines primarily focus on the management of adolescents and adults. Management of children with asthma is addressed in the NAEPP [84], GINA Main Report [85], BTS/SIGN [86], NICE [87], and ICON [88] guidelines via a stepwise approach beginning with initiation of inhaled corticosteroids, followed by consideration of long-acting bronchodilator, leukotriene inhibitor, and/or maintenance of OCS if symptoms remain uncontrolled. However, there is limited guidance on the management of difficult-to-control severe persistent asthma in this population when the aforementioned therapies fail. This deficiency stems from few advanced therapies approved for this age group as well as lack of large randomized studies on advanced therapies that include pediatric patients.
Use of muscarinic agents in the management of pediatric asthma has been shown to be safe and effective [89,90,91]. The LAMA, tiotropium, has been FDA approved for maintenance therapy in children ≥ 6 years old, and use in this age group as add-on therapy is recommended by ERS/ATS and GINA [10••, 85]. Recently in 2018, a small randomized control trial of tiotropium demonstrated safety and the potential to reduce asthma exacerbation risk in children 1–5 years old with persistent asthma [91]. However, rigorous, well-powered studies are needed to confirm these findings prior to extending the recommendation for use of tiotropium to this age group.
Until recently, the only asthma biologic agent currently approved for use in the pediatric population was Omalizumab, which is approved for add-on treatment in children ≥ 6 years old with moderate-to-severe asthma. In September 2019, Mepolizumab also received approval for use in children ≥ 6 years old and is currently the only therapy with an anti-IL5 mechanism approved for use in the pediatric population. Randomized controlled studies on efficacy of Reslizumab, Benralizumab, and Dupilumab in severe eosinophilic asthma included adolescents [23,24,25, 29, 31••, 32], suggesting a potential role for these agents in this population. However, these agents as well as Mepolizumab have not been rigorously studied in the pediatric population. Thus, large studies specifically evaluating safety and efficacy in children and adolescents continue to be needed for future development of evidence-based guidelines around use of these agents in the pediatric population.
Asthma-Associated Sinus Disease
Asthma is well-known to be associated with rhinosinusitis and nasal polyposis [92, 93]. Severity of asthma has been shown to correlate with severity of sinus disease [94, 95], and comorbid chronic rhinosinusitis is a negative predictor of quality of life in asthma patients [96]. Finally, it has been shown that medical or surgical treatment of chronic rhinosinusitis may lead to improvement in asthma control [97, 98].
There is currently limited guideline recommendations for the management of asthma-associated sinus disease. The 2015 American Academy of Otolaryngology–Head and Neck Surgery Foundation update to the clinical practice guidelines for management of adult sinusitis added asthma and nasal polyps as conditions that modify management of rhinosinusitis, and recommended that patients with uncontrolled asthma symptoms should be evaluated for concurrent sinus disease [99]. The 2019 GINA SA guidelines recommend consideration of Dupilumab initiation for patients with severe asthma and concurrent sinus disease [11••], as this is the only biologic currently approved for chronic rhinosinusitis with nasal polyposis. Further guideline recommendations on evaluation and treatment options for chronic rhinosinusitis and nasal polyposis may benefit patients whose uncontrolled severe asthma may be confounded by untreated asthma-associated sinus disease.
Conclusion
Management of severe asthma has rapidly evolved with the development of biologic agents to treat those exhibiting a T2 inflammatory phenotype. The 2019 ERS/ATS and GINA guidelines for severe asthma management provide a needed update to evidence-based recommendations around initiation of the multiple therapies currently available. However, many questions around the management of severe asthma patients remain unanswered, including how to predict and assess treatment response to biologic therapy, next steps when initial biologic therapy fails, what is the optimal approach to management of non-T2 asthma, and which therapies should be considered for children < 12 years old with severe persistent asthma. Future editions of the guidelines should address these limitations and take into further consideration the heterogeneity among severe asthma patients. This personalized approach to severe asthma management will reduce unnecessary exposure to medications and minimize healthcare costs through decreased exposure to expensive therapies.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Vos T, Flaxman AD, Naghavi M, Lozano R, Michaud C, Ezzati M, et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990-2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380(9859):2163–96.
Hekking PP, Wener RR, Amelink M, Zwinderman AH, Bouvy ML, Bel EH. The prevalence of severe refractory asthma. J Allergy Clin Immunol. 2015;135(4):896–902.
Varsano S, Segev D, Shitrit D. Severe and non-severe asthma in the community: a large electronic database analysis. Respir Med. 2017;123:131–9.
Nagase H, Adachi M, Matsunaga K, Yoshida A, Okoba T, Hayashi N, et al. Prevalence, disease burden, and treatment reality of patients with severe, uncontrolled asthma in Japan. Allergol Int. 2019;69(1):53–60.
Chung KF, Wenzel SE, Brozek JL, Bush A, Castro M, Sterk PJ, et al. International ERS/ATS guidelines on definition, evaluation and treatment of severe asthma. Eur Respir J. 2014;43(2):343–73.
Van Ganse E, Laforest L, Pietri G, Boissel JP, Gormand F, Ben-Joseph R, et al. Persistent asthma: disease control, resource utilisation and direct costs. Eur Respir J. 2002;20(2):260–7.
Accordini S, Bugiani M, Arossa W, Gerzeli S, Marinoni A, Olivieri M, et al. Poor control increases the economic cost of asthma. A multicentre population-based study. Int Arch Allergy Immunol. 2006;141(2):189–98.
Chung KF, Godard P, Adelroth E, Ayres J, Barnes N, Barnes P, et al. Difficult/therapy-resistant asthma: the need for an integrated approach to define clinical phenotypes, evaluate risk factors, understand pathophysiology and find novel therapies. ERS Task Force on Difficult/Therapy-Resistant Asthma. European Respiratory Society. Eur Respir J. 1999;13(5):1198–208.
Proceedings of the ATS workshop on refractory asthma: current understanding, recommendations, and unanswered questions. American Thoracic Society. Am J Respir Crit Care Med. 2000;162(6):2341–51.
•• Holguin F, Cardet JC, Chung KF, Diver S, Ferreira DS, Fitzpatrick A, et al. Management of severe asthma: a European Respiratory Society/American Thoracic Society guideline. Eur Respir J. 2020;55(1):1900588 ERS/ATS guideline of severe asthma in which the use of biological agents for the treatment of severe asthma is reviewed.
•• Global Initiative for Asthma (GINA). Difficult-to-treat & severe asthma in adolescent and adult patients. Diagnosis and management (updated April 2019). 2019. GINA severe asthma guideline in which biological agents, macrolides, and long-acting muscarinic agents are reviewed.
Schleich F, Brusselle G, Louis R, Vandenplas O, Michils A, Pilette C, et al. Heterogeneity of phenotypes in severe asthmatics. The Belgian Severe Asthma Registry (BSAR). Respir Med. 2014;108(12):1723–32.
Zhang JY, Wenzel SE. Tissue and BAL based biomarkers in asthma. Immunol Allergy Clin N Am. 2007;27(4):623–32 vi.
Price D, Wilson AM, Chisholm A, Rigazio A, Burden A, Thomas M, et al. Predicting frequent asthma exacerbations using blood eosinophil count and other patient data routinely available in clinical practice. J Asthma Allergy. 2016;9:1–12.
Price DB, Rigazio A, Campbell JD, Bleecker ER, Corrigan CJ, Thomas M, et al. Blood eosinophil count and prospective annual asthma disease burden: a UK cohort study. Lancet Respir Med. 2015;3(11):849–58.
Hancox RJ, Pavord ID, Sears MR. Associations between blood eosinophils and decline in lung function among adults with and without asthma. Eur Respir J. 2018;51(4):1702536.
Kuruvilla ME, Lee FE, Lee GB. Understanding asthma phenotypes, endotypes, and mechanisms of disease. Clin Rev Allergy Immunol. 2019;56(2):219–33.
• Wenzel SE. Asthma phenotypes: the evolution from clinical to molecular approaches. Nat Med. 2012;18(5):716–25 Excellent overview of asthma phenotypes and of endotype-driven treatment of asthma.
Woodruff PG, Modrek B, Choy DF, Jia G, Abbas AR, Ellwanger A, et al. T-helper type 2-driven inflammation defines major subphenotypes of asthma. Am J Respir Crit Care Med. 2009;180(5):388–95.
Green RH, Brightling CE, Woltmann G, Parker D, Wardlaw AJ, Pavord ID. Analysis of induced sputum in adults with asthma: identification of subgroup with isolated sputum neutrophilia and poor response to inhaled corticosteroids. Thorax. 2002;57(10):875–9.
Pavord ID, Korn S, Howarth P, Bleecker ER, Buhl R, Keene ON, et al. Mepolizumab for severe eosinophilic asthma (DREAM): a multicentre, double-blind, placebo-controlled trial. Lancet. 2012;380(9842):651–9.
•• Ortega HG, Liu MC, Pavord ID, Brusselle GG, FitzGerald JM, Chetta A, et al. Mepolizumab treatment in patients with severe eosinophilic asthma. N Engl J med. 2014;371(13):1198–207 Use of Mepolizumab in patients with severe eosinophilic asthma reduces asthma exacerbation rates.
Bleecker ER, FitzGerald JM, Chanez P, Papi A, Weinstein SF, Barker P, et al. Efficacy and safety of benralizumab for patients with severe asthma uncontrolled with high-dosage inhaled corticosteroids and long-acting beta2-agonists (SIROCCO): a randomised, multicentre, placebo-controlled phase 3 trial. Lancet. 2016;388(10056):2115–27.
FitzGerald JM, Bleecker ER, Nair P, Korn S, Ohta K, Lommatzsch M, et al. Benralizumab, an anti-interleukin-5 receptor alpha monoclonal antibody, as add-on treatment for patients with severe, uncontrolled, eosinophilic asthma (CALIMA): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet. 2016;388(10056):2128–41.
Castro M, Zangrilli J, Wechsler ME, Bateman ED, Brusselle GG, Bardin P, et al. Reslizumab for inadequately controlled asthma with elevated blood eosinophil counts: results from two multicentre, parallel, double-blind, randomised, placebo-controlled, phase 3 trials. Lancet Respir Med. 2015;3(5):355–66.
Busse W, Corren J, Lanier BQ, McAlary M, Fowler-Taylor A, Cioppa GD, et al. Omalizumab, anti-IgE recombinant humanized monoclonal antibody, for the treatment of severe allergic asthma. J Allergy Clin Immunol. 2001;108(2):184–90.
Soler M, Matz J, Townley R, Buhl R, O'Brien J, Fox H, et al. The anti-IgE antibody omalizumab reduces exacerbations and steroid requirement in allergic asthmatics. Eur Respir J. 2001;18(2):254–61.
Wenzel S, Ford L, Pearlman D, Spector S, Sher L, Skobieranda F, et al. Dupilumab in persistent asthma with elevated eosinophil levels. N Engl J Med. 2013;368(26):2455–66.
Castro M, Corren J, Pavord ID, Maspero J, Wenzel S, Rabe KF, et al. Dupilumab efficacy and safety in moderate-to-severe uncontrolled asthma. N Engl J Med. 2018;378(26):2486–96.
Bel EH, Wenzel SE, Thompson PJ, Prazma CM, Keene ON, Yancey SW, et al. Oral glucocorticoid-sparing effect of mepolizumab in eosinophilic asthma. N Engl J Med. 2014;371(13):1189–97.
•• Rabe KF, Nair P, Brusselle G, Maspero JF, Castro M, Sher L, et al. Efficacy and safety of dupilumab in glucocorticoid-dependent severe asthma. N Engl J Med. 2018;378(26):2475–85 Randomized controlled trial demonstrating that in patients with glucoticoid-dependent severe asthma, use of Dupilumab decreased oral corticosteroid dose, reduced rates of severe exacerbations, and increased FEV1.
Bjermer L, Lemiere C, Maspero J, Weiss S, Zangrilli J, Germinaro M. Reslizumab for inadequately controlled asthma with elevated blood eosinophil levels: a randomized phase 3 study. Chest. 2016;150(4):789–98.
Chupp GL, Bradford ES, Albers FC, Bratton DJ, Wang-Jairaj J, Nelsen LM, et al. Efficacy of mepolizumab add-on therapy on health-related quality of life and markers of asthma control in severe eosinophilic asthma (MUSCA): a randomised, double-blind, placebo-controlled, parallel-group, multicentre, phase 3b trial. Lancet Respir Med. 2017;5(5):390–400.
Buhl R, Hanf G, Soler M, Bensch G, Wolfe J, Everhard F, et al. The anti-IgE antibody omalizumab improves asthma-related quality of life in patients with allergic asthma. Eur Respir J. 2002;20(5):1088–94.
Kawakami T, Blank U. From IgE to Omalizumab. J Immunol. 2016;197(11):4187–92.
Global Initiative for Asthma (GINA). Global strategy for asthma management and prevention 2010 (update). 2010.
Kolbeck R, Kozhich A, Koike M, Peng L, Andersson CK, Damschroder MM, et al. MEDI-563, a humanized anti-IL-5 receptor alpha mAb with enhanced antibody-dependent cell-mediated cytotoxicity function. J Allergy Clin Immunol. 2010;125(6):1344–53 e2.
Menzies-Gow A, Flood-Page P, Sehmi R, Burman J, Hamid Q, Robinson DS, et al. Anti-IL-5 (mepolizumab) therapy induces bone marrow eosinophil maturational arrest and decreases eosinophil progenitors in the bronchial mucosa of atopic asthmatics. J Allergy Clin Immunol. 2003;111(4):714–9.
Egan RW, Athwal D, Bodmer MW, Carter JM, Chapman RW, Chou CC, et al. Effect of Sch 55700, a humanized monoclonal antibody to human interleukin-5, on eosinophilic responses and bronchial hyperreactivity. Arzneimittelforschung. 1999;49(9):779–90.
Pham TH, Damera G, Newbold P, Ranade K. Reductions in eosinophil biomarkers by benralizumab in patients with asthma. Respir Med. 2016;111:21–9.
Harb H, Chatila TA. Mechanisms of dupilumab. Clin Exp Allergy. 2020;50(1):5–14.
Hamelmann E, Bernstein JA, Vandewalker M, Moroni-Zentgraf P, Verri D, Unseld A, et al. A randomised controlled trial of tiotropium in adolescents with severe symptomatic asthma. Eur Respir J. 2017;49(1):1601100.
• Kerstjens HA, Engel M, Dahl R, Paggiaro P, Beck E, Vandewalker M, et al. Tiotropium in asthma poorly controlled with standard combination therapy. N Engl J Med. 2012;367(13):1198–207 Randomized controlled trial demonstrating that in patients with poorly controlled asthma despite the use of inhaled glucocorticoids and long acting bronchodilators, the use of long acting muscarinic agents increased time to the first exacerbation.
Timmer W, Moroni-Zentgraf P, Cornelissen P, Unseld A, Pizzichini E, Buhl R. Once-daily tiotropium Respimat(R) 5 μg is an efficacious 24-h bronchodilator in adults with symptomatic asthma. Respir Med. 2015;109(3):329–38.
Gibson PG, Yang IA, Upham JW, Reynolds PN, Hodge S, James AL, et al. Effect of azithromycin on asthma exacerbations and quality of life in adults with persistent uncontrolled asthma (AMAZES): a randomised, double-blind, placebo-controlled trial. Lancet. 2017;390(10095):659–68.
Gibson PG, Yang IA, Upham JW, Reynolds PN, Hodge S, James AL, et al. Efficacy of azithromycin in severe asthma from the AMAZES randomised trial. ERJ Open Res. 2019;5(4):00056–2019.
Pavord ID, Cox G, Thomson NC, Rubin AS, Corris PA, Niven RM, et al. Safety and efficacy of bronchial thermoplasty in symptomatic, severe asthma. Am J Respir Crit Care Med. 2007;176(12):1185–91.
Castro M, Rubin AS, Laviolette M, Fiterman J, De Andrade LM, Shah PL, et al. Effectiveness and safety of bronchial thermoplasty in the treatment of severe asthma: a multicenter, randomized, double-blind, sham-controlled clinical trial. Am J Respir Crit Care Med. 2010;181(2):116–24.
Bourdin A, Husereau D, Molinari N, Golam S, Siddiqui MK, Lindner L, et al. Matching-adjusted indirect comparison of benralizumab versus interleukin-5 inhibitors for the treatment of severe asthma: a systematic review. Eur Respir J. 2018;52(5):1801393.
Casale TB, Pacou M, Mesana L, Farge G, Sun SX, Castro M. Reslizumab compared with benralizumab in patients with eosinophilic asthma: a systematic literature review and network meta-analysis. J Allergy Clin Immunol Pract. 2019;7(1):122–30 e1.
Edris A, De Feyter S, Maes T, Joos G, Lahousse L. Monoclonal antibodies in type 2 asthma: a systematic review and network meta-analysis. Respir Res. 2019;20(1):179.
Ramonell RP, Iftikhar IH. Effect of anti-IL5, anti-IL5R, anti-IL13 therapy on asthma exacerbations: a network meta-analysis. Lung. 2020;198(1):95–103.
Walford HH, Doherty TA. Diagnosis and management of eosinophilic asthma: a US perspective. J Asthma Allergy. 2014;7:53–65.
Arron JR, Choy DF, Scheerens H, Matthews JG. Noninvasive biomarkers that predict treatment benefit from biologic therapies in asthma. Ann Am Thorac Soc. 2013;10(Suppl):S206–13.
Pavord ID, Afzalnia S, Menzies-Gow A, Heaney LG. The current and future role of biomarkers in type 2 cytokine-mediated asthma management. Clin Exp Allergy. 2017;47(2):148–60.
Fowler SJ, Tavernier G, Niven R. High blood eosinophil counts predict sputum eosinophilia in patients with severe asthma. J Allergy Clin Immunol. 2015;135(3):822–4 e2.
Gao J, Wu F. Association between fractional exhaled nitric oxide, sputum induction and peripheral blood eosinophil in uncontrolled asthma. Allergy, Asthma Clin Immunol. 2018;14:21.
Wagener AH, de Nijs SB, Lutter R, Sousa AR, Weersink EJ, Bel EH, et al. External validation of blood eosinophils, FE(NO) and serum periostin as surrogates for sputum eosinophils in asthma. Thorax. 2015;70(2):115–20.
Bleecker ER, Wechsler ME, FitzGerald JM, Menzies-Gow A, Wu Y, Hirsch I, et al. Baseline patient factors impact on the clinical efficacy of benralizumab for severe asthma. Eur Respir J. 2018;52(4):1800936.
Castro M, Wenzel SE, Bleecker ER, Pizzichini E, Kuna P, Busse WW, et al. Benralizumab, an anti-interleukin 5 receptor alpha monoclonal antibody, versus placebo for uncontrolled eosinophilic asthma: a phase 2b randomised dose-ranging study. Lancet Respir Med. 2014;2(11):879–90.
Korevaar DA, Westerhof GA, Wang J, Cohen JF, Spijker R, Sterk PJ, et al. Diagnostic accuracy of minimally invasive markers for detection of airway eosinophilia in asthma: a systematic review and meta-analysis. Lancet Respir Med. 2015;3(4):290–300.
Hastie AT, Moore WC, Li H, Rector BM, Ortega VE, Pascual RM, et al. Biomarker surrogates do not accurately predict sputum eosinophil and neutrophil percentages in asthmatic subjects. J Allergy Clin Immunol. 2013;132(1):72–80.
Prazma CM, Bel EH, Price RG, Bradford ES, Albers FC, Yancey SW. Oral corticosteroid dose changes and impact on peripheral blood eosinophil counts in patients with severe eosinophilic asthma: a post hoc analysis. Respir Res. 2019;20(1):83.
Durrington HJ, Gioan-Tavernier GO, Maidstone RJ, Krakowiak K, Loudon ASI, Blaikley JF, et al. Time of day affects eosinophil biomarkers in asthma: implications for diagnosis and treatment. Am J Respir Crit Care Med. 2018;198:1578–81.
Shrimanker R, Keene O, Hynes G, Wenzel S, Yancey S, Pavord ID. Prognostic and predictive value of blood eosinophil count, fractional exhaled nitric oxide, and their combination in severe asthma: a post hoc analysis. Am J Respir Crit Care Med. 2019;200(10):1308–12.
Jia G, Erickson RW, Choy DF, Mosesova S, Wu LC, Solberg OD, et al. Periostin is a systemic biomarker of eosinophilic airway inflammation in asthmatic patients. J Allergy Clin Immunol. 2012;130(3):647–54 e10.
Tajiri T, Matsumoto H, Gon Y, Ito R, Hashimoto S, Izuhara K, et al. Utility of serum periostin and free IgE levels in evaluating responsiveness to omalizumab in patients with severe asthma. Allergy. 2016;71(10):1472–9.
Hanania NA, Wenzel S, Rosen K, Hsieh HJ, Mosesova S, Choy DF, et al. Exploring the effects of omalizumab in allergic asthma: an analysis of biomarkers in the EXTRA study. Am J Respir Crit Care Med. 2013;187(8):804–11.
Corren J, Lemanske RF, Hanania NA, Korenblat PE, Parsey MV, Arron JR, et al. Lebrikizumab treatment in adults with asthma. N Engl J Med. 2011;365(12):1088–98.
Izuhara K, Ohta S, Ono J. Using periostin as a biomarker in the treatment of asthma. Allergy, Asthma Immunol Res. 2016;8(6):491–8.
Pandey G, Pandey OP, Rogers AJ, Ahsen ME, Hoffman GE, Raby BA, et al. A nasal brush-based classifier of asthma identified by machine learning analysis of nasal RNA sequence data. Sci Rep. 2018;8(1):8826.
• Woodruff PG, Boushey HA, Dolganov GM, Barker CS, Yang YH, Donnelly S, et al. Genome-wide profiling identifies epithelial cell genes associated with asthma and with treatment response to corticosteroids. Proc Natl Acad Sci U S A. 2007;104(40):15858–63 Gene expression study of airway epithelial cells from asthmatic subjects demonstrates that IL-13 increased expression of CLCA1, periostin, and serpinB2, an effect that was suppressed by corticosteroids.
Peters MC, Mekonnen ZK, Yuan S, Bhakta NR, Woodruff PG, Fahy JV. Measures of gene expression in sputum cells can identify TH2-high and TH2-low subtypes of asthma. J Allergy Clin Immunol. 2014;133(2):388–94.
Boudewijn IM, Lan A, Faiz A, Cox CA, Brouwer S, Schokker S, et al. Nasal gene expression changes with inhaled corticosteroid treatment in asthma. Allergy. 2019;75(1):191–4.
Hochhaus G, Brookman L, Fox H, Johnson C, Matthews J, Ren S, et al. Pharmacodynamics of omalizumab: implications for optimised dosing strategies and clinical efficacy in the treatment of allergic asthma. Curr Med Res Opin. 2003;19(6):491–8.
Nair P, Pizzichini MM, Kjarsgaard M, Inman MD, Efthimiadis A, Pizzichini E, et al. Mepolizumab for prednisone-dependent asthma with sputum eosinophilia. N Engl J Med. 2009;360(10):985–93.
Perez de Llano LA, Cosio BG, Domingo C, Urrutia I, Bobolea I, Valero A, et al. Efficacy and safety of reslizumab in patients with severe asthma with inadequate response to omalizumab: a multicenter, open-label pilot study. J Allergy Clin Immunol Pract. 2019;7(7):2277–83 e2.
Chapman KR, Albers FC, Chipps B, Munoz X, Devouassoux G, Bergna M, et al. The clinical benefit of mepolizumab replacing omalizumab in uncontrolled severe eosinophilic asthma. Allergy. 2019;74(9):1716–26.
Kurosawa M, Sutoh E. Severe uncontrolled eosinophilic asthma, which responded to benralizumab after failure to respond to mepolizumab. Ann Allergy Asthma Immunol. 2019;122(4):431–3.
Ortega G, Tongchinsub P, Carr T. Combination biologic therapy for severe persistent asthma. Ann Allergy Asthma Immunol. 2019;123(3):309–11.
Fitzpatrick AM, Chipps BE, Holguin F, Woodruff PG. T2-"low" Asthma: overview and management strategies. J Allergy Clin Immunol Pract. 2020;8(2):452–63.
Baines KJ, Simpson JL, Wood LG, Scott RJ, Gibson PG. Transcriptional phenotypes of asthma defined by gene expression profiling of induced sputum samples. J Allergy Clin Immunol. 2011;127(1):153–60 60 e1–9.
Kuo CS, Pavlidis S, Loza M, Baribaud F, Rowe A, Pandis I, et al. T-helper cell type 2 (Th2) and non-Th2 molecular phenotypes of asthma using sputum transcriptomics in U-BIOPRED. Eur Respir J. 2017;49(2):1602135.
National Asthma E, Prevention P. Expert panel report 3 (EPR-3): guidelines for the diagnosis and management of asthma-summary report 2007. J Allergy Clin Immunol. 2007;120(5 Suppl):S94–138.
Global Initiative for Asthma (GINA). Global strategy for asthma management and prevention, 2019. . 2019.
British Thoracic Society (BTS) SIGNS. British guideline on the management of asthma. 2019.
National Institute for Health Care and Excellence (NICE). Asthma: diagnosis, monitoring and chronic asthma management. 2020.
Papadopoulos NG, Arakawa H, Carlsen KH, Custovic A, Gern J, Lemanske R, et al. International consensus on (ICON) pediatric asthma. Allergy. 2012;67(8):976–97.
Szefler SJ, Murphy K, Harper T 3rd, Boner A, Laki I, Engel M, et al. A phase III randomized controlled trial of tiotropium add-on therapy in children with severe symptomatic asthma. J Allergy Clin Immunol. 2017;140(5):1277–87.
Hamelmann E, Bateman ED, Vogelberg C, Szefler SJ, Vandewalker M, Moroni-Zentgraf P, et al. Tiotropium add-on therapy in adolescents with moderate asthma: a 1-year randomized controlled trial. J Allergy Clin Immunol. 2016;138(2):441–50 e8.
Vrijlandt E, El Azzi G, Vandewalker M, Rupp N, Harper T, Graham L, et al. Safety and efficacy of tiotropium in children aged 1-5 years with persistent asthmatic symptoms: a randomised, double-blind, placebo-controlled trial. Lancet Respir Med. 2018;6(2):127–37.
Matsuno O, Ono E, Takenaka R, Okubo T, Takatani K, Ueno T, et al. Asthma and sinusitis: association and implication. Int Arch Allergy Immunol. 2008;147(1):52–8.
Jarvis D, Newson R, Lotvall J, Hastan D, Tomassen P, Keil T, et al. Asthma in adults and its association with chronic rhinosinusitis: the GA2LEN survey in Europe. Allergy. 2012;67(1):91–8.
Bresciani M, Paradis L, Des Roches A, Vernhet H, Vachier I, Godard P, et al. Rhinosinusitis in severe asthma. J Allergy Clin Immunol. 2001;107(1):73–80.
Lin DC, Chandra RK, Tan BK, Zirkle W, Conley DB, Grammer LC, et al. Association between severity of asthma and degree of chronic rhinosinusitis. Am J Rhinol Allergy. 2011;25(4):205–8.
Ek A, Middelveld RJ, Bertilsson H, Bjerg A, Ekerljung L, Malinovschi A, et al. Chronic rhinosinusitis in asthma is a negative predictor of quality of life: results from the Swedish GA(2)LEN survey. Allergy. 2013;68(10):1314–21.
Vashishta R, Soler ZM, Nguyen SA, Schlosser RJ. A systematic review and meta-analysis of asthma outcomes following endoscopic sinus surgery for chronic rhinosinusitis. Int Forum Allergy Rhinol. 2013;3(10):788–94.
Ragab S, Scadding GK, Lund VJ, Saleh H. Treatment of chronic rhinosinusitis and its effects on asthma. Eur Respir J. 2006;28(1):68–74.
Rosenfeld RM, Piccirillo JF, Chandrasekhar SS, Brook I, Ashok Kumar K, Kramper M, et al. Clinical practice guideline (update): adult sinusitis. Otolaryngol Head Neck Surg. 2015;152(2 Suppl):S1–S39.
Australia’s National Asthma Council. Australian asthma handbook. 2019.
Mensah GA, Kiley JP, Gibbons GH. Generating evidence to inform an update of asthma clinical practice guidelines: perspectives from the National Heart, Lung, and Blood Institute. J Allergy Clin Immunol. 2018;142(3):744–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflicts of interest relevant to this manuscript.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Asthma
Rights and permissions
About this article
Cite this article
Reihman, A.E., Holguin, F. & Sharma, S. Management of Severe Asthma Beyond the Guidelines. Curr Allergy Asthma Rep 20, 47 (2020). https://doi.org/10.1007/s11882-020-00940-6
Published:
DOI: https://doi.org/10.1007/s11882-020-00940-6