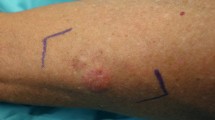
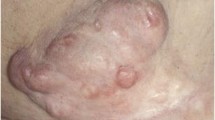
Opinion statement
Cutaneous sarcoma is a group of malignant mesenchymal tumors primarily involving the dermis, and it is characterized by extreme clinicopathological heterogeneity. Although its occurrence rate is rare, dermatofibrosarcoma protuberans (DFSP) is one of the most common types of dermal sarcoma. DFSP grows slowly and tends to relapse locally after inadequate resection. There are various histological variants of DFSP tumors and it often mimics benign lesions such as dermatofibroma and scar, which make accurate diagnosis difficult and delayed, and some cases progress to the stage where the tumor is unresectable. Recent advancements in cancer genetics and molecular biology methods have elucidated the COL1A1-PDGFB fusion gene, some novel fusion gene variants and pathways related to DFSP pathogenesis that have resulted in the evolution of cutaneous sarcoma diagnosis and treatment. For example, some clinical studies have confirmed the efficacy of imatinib methylate, an αPDGFR-targeted therapy for unresectable or metastatic DFSP. The present review summarizes recent updates in DFSP research, diagnostics, and treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cutaneous sarcoma is a group of malignant mesenchymal tumor primarily involving the dermis. These tumors are rare, but demonstrate extreme clinicopathologic and genetic heterogeneity. Although cutaneous sarcoma is thought to arise from dermal mesenchymal cells, the exact histogenesis is not yet well established. Therefore, cutaneous and soft tissue sarcomas are classified according to the line of differentiation in a given neoplasm. In other words, sarcomas show a broad range of differentiation and are histologically classified according to the non-neoplastic counterpart of mature tissue they resemble, such as fibroblast (dermatofibrosarcoma protuberans [DFSP]), smooth muscle (leiomyosarcoma), and endothelium (angiosarcoma). The most common primary cutaneous sarcoma is Kaposi sarcoma (KS) (71.1%), followed by DFSP (18.4%), leiomyosarcoma (2.2%), and angiosarcoma (1.6%) in the USA [1]. Although the most common type of KS in the USA is AIDS-associated KS, KS is extremely rare in Japan. Therefore, there is a great regional difference in the prevalence of the dermal sarcoma subtype. Since cutaneous sarcomas show similar morphological features and are composed of several subtypes and histological variants within a single entity, it is often difficult to make an accurate histological diagnosis. An accurate clinical diagnosis is often delayed, making curative treatment sometimes impossible.
Recent advancements in molecular pathology have made it possible to classify sarcomas based on underlying molecular alterations. In turn, this has enabled the development and application of new molecular targeted therapies to treat sarcomas. In the present review, we discuss recent updates regarding DFSP research, differential diagnostics, and treatment.
Clinicopathological features of DFSP
DFSP is a low-grade and locally invasive soft tissue sarcoma that originates from the dermis or superficial subcutis of young to middle-aged adults. These tumors were initially characterized as keloid-like sarcomas and were named DFSP by Hoffman in 1925 [2]. It accounts for approximately 5% of all soft tissue sarcomas and 18% of all cutaneous soft tissue sarcomas, which are most common in Japan and second common in the USA. The tumor location is usually the trunk, the head and neck being the other common locations. In the earliest growth phase, DFSP shows as polypoid protuberance or an indurated plaque on the skin.
Histologically, DFSPs are composed of mildly atypical but uniform spindle cells arranged in a prominent storiform whorled pattern, invading the surrounding subcutaneous fat (Fig. 1a–c). Immunohistochemically, DFSP tumor cells are strongly positive for cluster of differentiation (CD) 34 (Fig. 1d) and vimentin and negative for CD44, S100 protein, and factor XIIIa. Differential diagnoses of conventional DFSP include cellular dermatofibroma (factor XIIIa-positive; CD34-negative), fibrosarcoma undifferentiated pleomorphic sarcoma (UPS), atypical fibroxanthoma, desmoplastic melanoma, KS, and solitary fibrous tumor. However, it should be noted that between 6 and 10% of DFSPs are CD34-negative and approximately 15–25% are factor XIIIa-positive [3,4,5]. In addition, CD34 expression can also be observed in other sarcomas, such as myofibrosarcoma, epithelioid sarcoma, and angiosarcoma, as well as some benign fibrohistiocytic lesions. Therefore, many studies have attempted to find more specific diagnostic makers able to distinguish DFSP from other spindle cell tumors. For example, apolipoprotein D is reportedly highly expressed in the cytoplasm of DFSP (9/10 cases) and DFSP variants (12/16 cases), including Bednar tumor and giant cell fibroblastoma, but not in UPS [6].
Histological appearance of classic dermatofibrosarcoma protuberans (DFSP). a, b Panoramic (a) and lower magnification (b) view of DFSP infiltrating the surrounding adipose tissue (hematoxylin and eosin staining). c Classical DFSP is comprised of interwoven, dense, atypical spindle cells forming a storiform pattern. d Classical DFSP is generally reactive with anti-CD34 immunostaining. a–c Hematoxylin and eosin stain.
The 10-year survival rate for patients with DFSP is 99.1%; factors related to adverse treatment outcomes include advanced age, male sex, African heritage, and anatomic location of the tumor, including limbs and head [7]. Fibrosarcomatous transformation worsens the prognosis (discussed later). While the cellular origin of DFSP is unknown, principal theories of its histogenesis include precursor histiocytes, fibroblasts, and dendritic, perineural, or endoneural cells.
Histological variants of DFSP
Unusual DFSP variants are comprised of at least 10 subtypes, most commonly including pigmented (Bednar tumor), myxoid, myoid, granular cell, sclerotic, atrophic and fibrosarcomatous DFSPs, and giant cell fibroblastoma (Fig. 2) [3, 8].
Histological appearance of variants of dermatifibrosarcoma protuberans (DFSP). a Giant cell fibroblastoma is a DFSP variant characterized by bizarre, multinucleated giant cells with cellular pleomorphism. b Giant cell fibroblastoma often shows pseudo vascular pattern. c Bednar tumor is a pigmented variant of DFSP characterized by DFSP with admixed melanin-bearing dendritic cells. d Myxoid DFSP is characterized by moderately cellular areas with abundant pale to clear myxoid stroma. e Macroscopic feature of plaque-like DFSP (atrophic DFSP) lesion (chest wall). It shows depressed plaque with reduction in dermal thickness. f, g The tumor areas in the subcutis showing non-protuberance feature (f) and reactive with anti-CD34 immunostaining (g). a–d and f Hematoxylin and eosin stain.
Giant cell fibroblastoma
Giant cell fibroblastoma (GCF) is a morphological variant of DFSP. Previously, GCF was considered as a juvenile form or precursor lesion of DFSP, because this tumor is often observed in infants and children. However, recent morphological, immunohistochemical, and molecular studies suggest that GCF is on a same spectrum with DFSP [9]. These tumors are composed of loosely arranged, spindle to pleomorphic cells that are often, multinucleated, and infiltrate into the dermis and subcutaneous tissue (Fig. 2a). Pseudovascular tissue spaces lined by multinucleated giant cells are often observed (Fig. 2b) [9, 10]. Some studies have reported the occurrence of a GCF in recurrent lesions following DFSP or Bednar tumors. Furthermore, although local recurrence has been observed, metastasis has not been reported yet.
Pigmented DFSP (Bednar tumor)
Bednar tumor is a pigmented DFSP characterized by melanin-containing dendritic cells. It was first reported by Bednar in 1957 [11], occurs predominantly in persons of African heritage, and represents 1–5% of all DFSP cases [12]. The incidence of local recurrence from Bednar tumor is 11–13%, which is lower than that from conventional DFSP (20–50%). Metastasis of Bednar tumors is extremely rare and in later disease stages, and the principle sites of metastasis include the lungs, bones, liver, pancreas, stomach, intestine, thyroid, and brain [13]. Histologically, these tumors are characterized by spindle cells arranged in a tight storiform pattern and admixed melanin-containing dendritic cells (Fig. 2c). Ultrastructural studies have also confirmed the pigmented cells mostly contain mature or stage IV melanosomes [14, 15].
While the origin of the Bednar tumor remains unclear and controversial, two main theories propose its histogenesis via neuroectodermal differentiation or melanocytic colonization. The differential diagnosis of Bednar tumor includes fibrous histiocytoma, neurofibroma, malignant melanoma, and cellular blue nevus. Immunohistochemically, the vast majority of Bednar tumors are CD34-positive, but their positivity for melanocytic markers (e.g., human melanoma black-45), and tyrosinase are controversial. While the first reported case by Bednar was negative for S-100, some have reported cases where melanin-containing dendritic cells were positive for usual melanocytic makers, including S-100 [11].
The fact that conventional DFSP sometimes recurs as pigmented DFSP supports the idea that these subtypes have different morphologic expression of the same neoplasm.
Plaque-like DFSP (atrophic DFSP)
The very early clinical stage of DFSP is considered to be that of a non-protuberant flat lesion, which eventually develops into the typical protuberant form [16]. But in some cases, the lesion retains the non-protuberant feature. This is called atrophic or plaque-like DFSP, which is characterized by the presence of a depressed plaque with more than 50% reduction in dermal thickness. The dermal thickness is measured by comparing the subcutis with the surrounding dermis and fat (Fig. 2e–g) [3]. The first case was reported by Lambert in 1985 [17]. Atrophic DFSP tumors often occur in young adults (median age, 24 years old), but may also occur in infants and children [18, 19]. Clinically, these tumors often mimic benign lesions, such as morphea, atrophoderma, atrophic scars, anetoderma, lipoatrophy, or medallion-like dermal dendrocyte hamartoma [3].
Sclerosing DFSP (sclerotic DFSP)
Sclerosing or sclerotic DFSP is characterized by the existence of paucicellular or hypocellular collagen accounting for at least 50% of the tumor [20,21,22]. This tumor is often misdiagnosed as other benign sclerosing lesions and mesenchymal neoplasms, such as sclerosing epithelioid fibrosarcoma, sclerotic or desmoplastic LMS, and desmoplastic melanoma [20]. This variant is often admixed with classical DFSP, and histological transition from classical DFSP to sclerosing DFSP has also been observed. Although its pathogenesis remains unknown, some studies suggest its morphological features have resulted due to the production of large amounts of collagen by neoplastic cells [22].
Myxoid DFSP
Myxoid DFSP is a DFSP subtype composed of stellate or spindle-shaped tumor cells that are embedded in an abundant pale to clear, myxoid stroma (comprising at least 50% of the tumor) (Fig. 2d); prominent but delicate thin-walled blood vessels are also admixed. Clinically, myxoid DFSPs are similar to conventional DFSP, and it is important to distinguish myxoid DFSPs from benign tumors with myxoid stroma, such as spindle cell lipoma [23]. Immunohistochemically, the majority of myxoid tumors are CD34-positive and S-100-negative, similar to conventional DFSP. Furthermore, these tumors sometimes coexist with focal areas of fibrosarcomatous and giant cell fibroblastoma-like component [24,25,26].
Myoid DFSP
In 1996, Calonje and Fletcher first reported five cases of DFSP with myoid differentiation [27]. This feature is more often associated with fibrosarcomatous DFSP than with conventional DFSP [27,28,29]. The origin of myoid features is thought to be myofibroblastic differentiated tumor cells or reactive hyperproliferated pericytes or vessel wall smooth muscle [30]. Immunohistochemically, myoid tumor areas are CD34-negative, desmin-negative, and smooth muscle actin-positive.
Granular cell DFSP
Previously, three cases of granular cell variant of DFSP were described as tumors characterized by an intimate mixture of spindle cells with a significant population of cells having abundant lysosomal granules around eccentric nuclei and prominent nucleoli [31]; these granules are diastase-resistant and periodic acid-Schiff-positive [31].
Immunohistochemically, the granular cells are strongly positive for natural killer cell inhibitory factor 1C3 but negative for S-100, neurospecific enolase, and macrophage marker 387 (MAC387) [31].
Fibrosarcomatous DFSP
DFSPs occasionally exhibit high-grade histological transformation, namely fibrosarcomatous DFSP (FS-DFSP). This phenomenon is observed in 10–20% of DFSP cases and increases the risk of recurrence (55%) and metastasis (57%) typically resulting in adverse outcomes [32,33,34,35]. Histologically, the fibrosarcomatous area is characterized by atypical spindle or pleomorphic cells with numerous mitoses arranged in a herringbone appearance (Fig. 3a–e). Immunohistochemically, the fibrosarcomatous component is distinguished by conversion to CD34 negativity (Fig. 3b) and an elevated Ki-67 index (DFSP 8.9% ± 6.3% vs FS-DFSP 21.5% ± 5.1%) (Fig. 3c) [36], but the detailed oncogenic mechanism of FS-DFSP is not clear. In our previous study, we showed that microsatellite instability and p53 mutation status may be associated with fibrosarcomatous changes in DFSP [37]. Another report showed p53 and mouse double minute 2 homolog (MDM2) overexpressions in the fibrosarcomatous area and suggested that those factors are involved in fibrosarcomatous transformation [38]. Our recent study also showed fibrosarcomatous transformation was accompanied by an activation of the protein kinase B (Akt)-mechanistic target of rapamycin (mTOR) pathway [39]. FS-DFSP occasionally mimics other spindle cell sarcomas such as malignant peripheral nerve sheath tumor (MPNST), synovial sarcoma, and leiomyosarcoma. MPNST can be associated with neurofibromatosis type 1 and is characterized by frequent loss of histone H3 tri-methylated at K27 (H3K27me3) modification. However, surprisingly, loss of H3K27me3 modification was reported in 10–38% of FS-DFSP cases, so this aberrant modification is not specific to MPNST [40, 41]. Synovial sarcoma has specific fusion gene such as SS18-SSX1 or SS18-SSX2. Leiomyosarcomas show consistent expression of smooth muscle markers such as desmin and h-caldesmon.
Histological and macroscopic appearance of DFSP with fibrosarcomatous component [FS-DFSP]. a, b The fibrosarcomatous component (*) showed greater cell density than the conventional DFSP component (†) (a) as well as conversion to negative anti-CD34 immunoreactivity (b). c Ki-67 index is higher in fibrosarcomatous component (*) than in conventional DFSP component (†). d, e Fibrosarcomatous component often proliferates in fascicular (d) or herringbone (e) pattern. a, d, and e Hematoxylin and eosin stain.
Fusion genes and genetic findings
DFSP is characterized by t(17;22)(q22;q13) translocation of supernumerary ring chromosome or unbalanced linear translocation der(22) that results in the fusion of exon 2 of β-type platelet-derived growth factor (PDGFB) to various exons (exons 6-49) of the collagen (COL) type 1 α1 (COL1A1) gene. PDGFB breakpoints are constantly found in exon 2, whereas COL1A1 breakpoints vary from exons 6-49. This fusion product has been identified in 85–96% of cases. COL1A1-PDGFB fusions lead to upregulation of PDGFB receptor (PDGFRB) signaling through an autocrine activation loop [42, 43]. In routine practice, diagnostic assays for detecting the PDGFB rearrangement in DFSP involve reverse transcriptase-polymerase chain reaction (RT-PCR) and fluorescence in situ hybridization (FISH) with dual fusion or break-apart probes. A COL1A1-PDGFB fusion gene or PDGFB rearrangement is considered to be present if found in at least 10% of tumor cells by FISH.
Previous cytogenetic studies have reported the possibility of other translocation events, including t(2;17)(q33;q25), t(17;22)(q22;q13), t(9;22)(q32;q12.2), t(5;8)(q13–14;p21), and t(X;7)(q21.2;q11;2). Recently, three novel fusion gene variants (COL1A2-PDGFB, COL6A3-PDGFD, and elastin microfibril interface 2 [EMILIN2]-PDGFD) were identified from DFSP using RNA-sequencing [44•, 45•, 46•] (Table 1). A COL6A3 breakpoint occurred in exon 42 in most of cases (n = 8/9), whereas COL1A1 breakpoints varied. COL6A3 overexpression has been observed in various carcinomas, including gastric, ovarian, colorectal, and pancreatic cancers. Clinicopathological, morphological, and immunohistochemical features of DFSPs with COL6A3-PDGFD fusion gene are identical to those of conventional DFSP with PDGFR-COL1A1 [45•, 46•].
PDGFD is a newly identified isoform of PDGFs. The PDGF family comprises four different polypeptide chains encoded by different genes: classical PDGFA and PDGFB and the novel PDGFC and PDGFD. PDGFD specifically binds to and activates beta-receptors [10]. The main pathological effect of PDGFR activation is cellular proliferation and migration.
Previously, two cases of FS-DFSP containing EMILIN2-PDGFD were reported. In addition, both cases showed homozygous cyclin-dependent kinase inhibitor (CDK) 2A (CDKN2A) deletion, which is often observed in hypercellular areas of conventional DFSP and FS-DFSP [45•, 47]. It remains controversial whether fibrosarcomatous transformation is related to EMILIN2-PDGFD fusion transcripts or not.
Recently, Saab et al. reported a case of DFSP-like tumors with COL1A1 copy number gain in the absence of t(17;22) [48]. This case showed similar morphology to DFSP, as well as CD34 expression, and responded to imatinib therapy. Therefore, this case might be a novel molecular variant of DFSP. An awareness of PDGFB-COL1A1-negative DFSP is important for pathologists because it will not be detected using conventional RT-PCR- or FISH-based diagnostic assays for DFSP. These fusion genes described above are summarized in Table 1.
Treatment of DFSP
DFSP can be treated by local excision with wide margins (2.8–4 cm). Due to the difficulty in obtaining clear surgical margins, Mohs micrographic surgery is typically used and leads to a significantly lower recurrence rate [49]. Nonetheless, DFSP has a high recurrence rate as approximately 17–90% cases recur locally and approximately 1–6% distantly metastasize, predominantly to the lung [34, 50]. In addition, DFSP responds poorly to conventional chemotherapy. Recent advances in DFSP genomic and molecular studies led to the identification of various candidates of molecular targeted therapies. Various molecular targeted therapies, (described below), have been utilized to treat the unresectable DFSP. However, to date, only a tyrosine-kinase inhibitor, imatinib mesylate, has proven to be a highly promising therapeutic option.
Mohs micrographic surgery
Conventional resection with predetermined margins has only evaluated the limited histological margin because of using representative vertical section. Mohs micrographic surgery is a horizontal sectioning technique that can evaluate 100% of the surgical margin. The lesion is excised with a narrow margin of normal tissue 45 degrees from the standard vertical cut. Then, the tissue is pressed flat so that the peripheral margins are flattened into the same plane for sectioning. Finally, the tissue is rapidly frozen and sectioned in a microtome. For more information on this technique, see the article of Acosta and Vélez [51].
Conventional chemotherapy
To date, no conventional chemotherapy has been reported to be effective against DFSP, with one exception, a pediatric case showing favorable outcome after administration of vinblastine (6 mg/m2) and methotrexate (20 mg/m2) [52]. Before applying vinblastine and methotrexate, the patient had been given vincristine, actinomycin, and cyclophosphamide therapy; however, there was no response.
Radiotherapy
DFSPs are moderately radio-responsive. Several previous reports have shown that adjuvant radiotherapy decreased the risk of local recurrence, especially in patients with close or positive margins [53,54,55]. Meta-analysis of two retrospective cohort studies revealed that there was no significant difference in pooled odds ratios between adjuvant radiation therapy and surgery alone (odd’s ratio 0.31; P = 0.07). However, there was a trend of lower recurrence toward adjuvant radiotherapy with positive or close surgical margins (33%) versus surgery alone (17.3%) [56]. The recommended dose varies from 50 to 70 Gy and depends on the residual tumor size and site [57]. Although some cases using preoperative radiotherapy have been reported, the effectiveness is largely unknown because case sizes are still small.
Imatinib
Recently, various molecular target therapies have been attempted for DFSP. Since COL1A1-PDGFB fusions lead to upregulation of PDGFRB signaling through autocrine activation loops [58], administration of the tyrosine-kinase inhibitor imatinib mesylate (specifically inhibits breakpoint cluster region/Abelson murine leukemia, KIT, PDGFRA, and PDGFRB) is currently Food and Drug Administration-approved for adults with unresectable recurrent or metastatic DFSP in the USA (though not in Japan). The combined results of two phase II studies by the European Organization for Research and Treatment of Cancer and Southwest Oncology Group show a 50% response rate with imatinib regardless of dose (400 or 800 mg/day). In 2010, a phase II multicenter trial of neoadjuvant imatinib therapy for 2 months reported nine out of 25 (36%) patients achieved a clinical response [29]. The median decrease in tumor size was 20.0%, and tumors showed reduced cellularity and hyaline changes after treatment.
In metastatic DFSP, imatinib has been shown to result in a median survival of more than 19 months but had unfavorable outcomes with FS-DFSP [59]. Unfortunately, imatinib response rates are only approximately 50% in most DFSP cases, transient in FS-DFSP cases, and are associated with nonresponse in pigmented DFSP [59]. Imatinib has also reportedly ineffective for treatment of DFSP without COL1A1-PDGFB [59, 60].
Resistance to imatinib often emerges rapidly. However, the biological mechanisms of resistance to imatinib are not well characterized in DFSP. Stacchiotti et al. reported low level of PDGFRB phosphorylation, such as of Akt and extracellular signal-regulated kinase 1/2, in the presence of high levels of pS6 and p4EBP1 are associated with imatinib resistance [61]. Recently, homozygous deletion of CDKN2A and CDKN2B was identified in DFSP and it is associated with loss of p16 expression and imatinib resistance [47]. Loss of the p16/CDKN2A/inhibitors of CDK4A locus at 9p was also reported in metastatic FS-DFSP [47]. Oh et al. proposed a clonal evolution model to explain imatinib resistance in DFSP by analyzing whole exome sequencing data of serial tumor samples obtained from a patient with a 10-year history of recurrent and metastatic DFSP [62]. They found the breakpoint of COL1A1-PDGFB was identical in all sample series. However, the somatic mutation pattern was completely different and new focal amplification which had not been observed in the primary clone before metastasis was also observed. These results imply the emergence of a new subclonal population of cells that became dominant as a result of selective pressure, such as radiotherapy, chemotherapy, or specific drugs.
Sunitinib
Sunitinib is an oral multitargeted receptor tyrosine-kinase inhibitor that inhibits PDGFR, vascular endothelial growth factor receptors 1–3, KIT, colony stimulating factor-1 receptor, and fms-like tyrosine-kinase-3. Its binding capacity for PDGFRB is approximately 10-times greater than that of imatinib [63]. Sunitinib therapy is approved by the U.S. FDA for the treatment of renal cell carcinoma (RCC) and imatinib-resistant gastrointestinal stromal tumor (GIST). Sunitinib treatment has been shown to have favorable outcomes for patients with DFSP after imatinib failure [64, 65].
Sorafenib
Sorafenib is also an oral multikinase inhibitor approved by the U.S. FDA for the treatment of unresectable hepatocellular carcinoma, RCC, and metastatic or recurrent thyroid carcinoma, which targets both RAF family of serine/threonine kinases and the tyrosine-kinase receptor VEGFR-2 (KDR), VEGFR-3 (Flt-4), Flt-3, PDGFR, and KIT [66]. Kamar et al. reported effective sorafenib (800 mg/day) treatment of recurrent DFSP after imatinib failure [67]. This case showed a dramatic response with 5 months of progression-free survival.
Pazopanib
Pazopanib is a multitargeted tyrosine-kinase inhibitor with high affinity for vascular endothelial growth factor receptor and lower affinity for PDGFR. Reports of its efficacy are controversial [68, 69]. Although PDGFR is an essential therapeutic target of DFSP, epidermal growth factor receptor, insulin receptor, and insulin-like growth factor I receptor phosphorylation [59, 61], as well as increased epidermal growth factor receptor expression with DFSP progression to high-grade sarcoma, have also been reported [70].
Everolimus
Everolimus is a mammalian target of rapamycin (mTOR) inhibitor. Upregulation of PDGFB is known to activate Ras-mitogen-associated protein kinase and phosphatidylinositol-3-kinaseK-Akt-mTOR pathways leading to cell growth and differentiation [39]. A single case of imatinib-resistant fibrosarcomatous DFSP with a beneficial response to everolimus treatment has been reported [61].
Programmed death (PD)-ligand 1 (PD-L1)
Recently, immunotherapy targeting the PD-1/PD-L1 pathway has been reported to be effective against various malignancies. PD-1/PDL1 interaction inhibits the function of T cells and tumor-infiltrating lymphocytes and blockade of the interaction by anti-PD-L1 antibodies brakes the immune tolerance, and a case of DFSP with PD-L1 expression has been reported [69]. In this case, PD-L1 expression was observed in the metastatic tumor but not in primary tumors without fibrosarcomatous changes.
Other drug candidates
As DFSP is often more aggressive during pregnancy, hormonal receptors have been investigated [71, 72]. Progesterone receptors have been found to be expressed in approximately 30% of pregnant DFSP cases, whereas estrogen receptor expression is controversial [71,72,73]. These results suggest progesterone expression might be associated with DFSP growth.
Stacchiotti et al. reported FS-DFSP showed upregulation of enhancer of zeste homolog 2 (EZH2), a histone H3H3K27 methyltransferase [74]. Aberrant overexpression of EZH2 is also found in various malignant tumors, such as those of prostate cancer, breast cancer, ovarian cancer, and non-Hodgkin’s lymphoma and is associated with poor outcomes [75,76,77]. EZH2 functions as a transcriptional suppressor and as a transcriptional co-activator, depending on H3K27me3 or not [78]. Phase 1 clinical trial of tazemetostat, an EZH2 inhibitor, in patients with relapsed or refractory B cell non-Hodgkin’s lymphoma and those with advanced solid tumors was completed and durable objective responses were observed in 38% of patients with B cell non-Hodgkin lymphoma and 5% of patients with solid tumors [77]. These results suggest EZH2 may be a druggable target for DFSP.
Aurora kinase A (AURKA) is a G2-M kinase involved in cell division. Aberrant AURKA overexpression has been observed in many malignancies, including gastrointestinal tract cancer, pancreatic cancer, and sarcoma. A phase II clinical trial study of the AURKA inhibitor MLN8237 (alisertib) for treatment of sarcoma showed a median progression-free survival of 11.7 weeks [79]. Furthermore, a Japanese group reported AURKA overexpression in the vast majority of DFSP tumors examined, and its expression levels significantly correlated with CD34 expression [80]. Any DFSP patients participate in the phase II trial of AURKA inhibitor, but AURKA overexpression in DFSP may present the potential efficacy of alisertib therapy for the treatment of patients with DFSP.
Conclusion
In summary, DFSP demonstrates various morphological subtypes and mimics various benign or malignant lesions. Recent advances in the investigation of DFSP revealed some specific molecular diagnostic makers including novel fusion genes apart from COL1A1-PDGFB fusion genes and those underlying molecular genetic features and pathological mechanisms. These novel findings provide insight into molecular alterations being possible candidates for molecular target therapy, such as imatinib methylate therapy. Further novel insights into the biology of DFSPs will provide a comprehensive understanding of molecular target therapy and establish accurate diagnosis for patients with DFSPs.
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance
Rouhani P, Fletcher CD, Devesa SS, Toro JR. Cutaneous soft tissue sarcoma incidence patterns in the U.S. : an analysis of 12,114 cases. Cancer. 2008;113(3):616–27. https://doi.org/10.1002/cncr.23571.
Hoffmann EI. Über das knollentreibende Fibrosarkom der Haut (Dermatofibrosarkoma protuberans). Dermatology. 1925;43(1–2):1–28.
Llombart B, Serra-Guillen C, Monteagudo C, Lopez Guerrero JA, Sanmartin O. Dermatofibrosarcoma protuberans: a comprehensive review and update on diagnosis and management. Semin Diagn Pathol. 2013;30(1):13–28. https://doi.org/10.1053/j.semdp.2012.01.002.
Kim HJ, Lee JY, Kim SH, Seo YJ, Lee JH, Park JK, et al. Stromelysin-3 expression in the differential diagnosis of dermatofibroma and dermatofibrosarcoma protuberans: comparison with factor XIIIa and CD34. Br J Dermatol. 2007;157(2):319–24. https://doi.org/10.1111/j.1365-2133.2007.08033.x.
Kahn HJ, Fekete E, From L. Tenascin differentiates dermatofibroma from dermatofibrosarcoma protuberans: comparison with CD34 and factor XIIIa. Hum Pathol. 2001;32(1):50–6. https://doi.org/10.1053/hupa.2001.21137.
West RB, Harvell J, Linn SC, Liu CL, Prapong W, Hernandez-Boussard T, et al. Apo D in soft tissue tumors: a novel marker for dermatofibrosarcoma protuberans. Am J Surg Pathol. 2004;28(8):1063–9.
Criscito MC, Martires KJ, Stein JA. Prognostic factors, treatment, and survival in dermatofibrosarcoma protuberans. JAMA Dermatol. 2016;152(12):1365–71. https://doi.org/10.1001/jamadermatol.2016.1886.
Brenner W, Schaefler K, Chhabra H, Postel A. Dermatofibrosarcoma protuberans metastatic to a regional lymph node. Report of a case and review. Cancer. 1975;36(5):1897–902.
Jha P, Moosavi C, Fanburg-Smith JC. Giant cell fibroblastoma: an update and addition of 86 new cases from the armed forces Institute of Pathology, in honor of Dr. Franz M Enzinger. Ann Diagn Pathol. 2007;11(2):81–8. https://doi.org/10.1016/j.anndiagpath.2006.12.010.
Zamecnik M, Michal M, Chlumska A. Composite dermatofibrosarcoma protuberans-giant cell fibroblastoma recurring as Bednar tumor-giant cell fibroblastoma with mucoid lakes and with amputation neuroma. Cesk Patol. 2002;38(4):173–7.
Bednar B. Storiform neurofibromas of the skin, pigmented and nonpigmented. Cancer. 1957;10(2):368–76.
Dupree WB, Langloss JM, Weiss SW. Pigmented dermatofibrosarcoma protuberans (Bednar tumor). A pathologic, ultrastructural, and immunohistochemical study. Am J Surg Pathol. 1985;9(9):630–9.
Suehara Y, Yazawa Y, Hitachi K. Metastatic Bednar tumor (pigmented dermatofibrosarcoma protuberans) with fibrosarcomatous change: a case report. J Orthop Sci. 2004;9(6):662–5. https://doi.org/10.1007/s00776-004-0831-2.
Kagoura M, Toyoda M, Nagahori H, Makino T, Morohashi M. An ultrastructural and immunohistochemical study of pigmented dermatofibrosarcoma protuberans (Bednar tumor). Eur J Dermatol. 1999;9(5):366–9.
Seo IS, Goheen M, Min KW. Bednar tumor: report of a case with immunohistochemical and ultrastructural study. Ultrastruct Pathol. 2003;27(3):205–10.
Bakry O, Attia A. Atrophic dermatofibrosarcoma protuberans. J Dermatol Case Rep. 2012;6(1):14–7. https://doi.org/10.3315/jdcr.2012.1089.
Lambert WC, Abramovits W, Gonzalez-Sevra A, Souchon E, Schwartz RA, Little WP Jr. Dermatofibrosarcoma non-protuberans: description and report of five cases of a morpheaform variant of dermatofibrosarcoma. J Surg Oncol. 1985;28(1):7–11.
Martin L, Combemale P, Dupin M, Chouvet B, Kanitakis J, Bouyssou-Gauthier ML, et al. The atrophic variant of dermatofibrosarcoma protuberans in childhood: a report of six cases. Brit J Dermatol. 1998;139(4):719–25.
Marini M, Saponaro A, Magarinos G, de Baldrich A, Lynch P, Remorino L. Congenital atrophic dermatofibrosarcoma protuberans. Int J Dermatol. 2001;40(7):448–50.
Sabater-Marco V, Perez-Valles A, Berzal-Cantalejo F, Rodriguez-Serna M, Martinez-Diaz F, Martorell-Cebollada M. Sclerosing dermatofibrosarcoma protuberans (DFSP): an unusual variant with focus on the histopathologic differential diagnosis. Int J Dermatol. 2006;45(1):59–62. https://doi.org/10.1111/j.1365-4632.2004.02340.x.
Hattori H. Nodular sclerotic change in dermatofibrosarcoma protuberans: a potential diagnostic problem. Br J Dermatol. 2003;148(2):357–60.
Diaz-Cascajo C, Weyers W, Borghi S. Sclerosing dermatofibrosarcoma protuberans. J Cutan Pathol. 1998;25(8):440–4.
Tran P, Henderson GP, McLemore M. An unusual clinical presentation of myxoid dermatofibrosarcoma protuberans with a prominent vasculature: a potential pitfall in the diagnosis of myxoid soft tissue tumors. J Cutan Pathol. 2018;45(6):419–22. https://doi.org/10.1111/cup.13130.
Sato N, Kimura K, Tomita Y. Recurrent dermatofibrosarcoma protuberans with myxoid and fibrosarcomatous changes paralleled by loss of CD34 expression. J Dermatol. 1995;22(9):665–72.
Zamecnik M, Michal M. Myxoid variant of dermatofibrosarcoma protuberans with fibrosarcomatous areas. Zentralbl Pathol. 1993;139(4–5):373–6.
Mentzel T, Scharer L, Kazakov DV, Michal M. Myxoid dermatofibrosarcoma protuberans: clinicopathologic, immunohistochemical, and molecular analysis of eight cases. Am J Dermatopathol. 2007;29(5):443–8. https://doi.org/10.1097/DAD.0b013e318145413c.
Calonje E, Fletcher CD. Myoid differentiation in dermatofibrosarcoma protuberans and its fibrosarcomatous variant: clinicopathologic analysis of 5 cases. J Cutan Pathol. 1996;23(1):30–6.
Al-Zaid T, Khoja H. Acral dermatofibrosarcoma protuberans with myoid differentiation: a report of 2 cases. J Cutan Pathol. 2017;44(9):794–7. https://doi.org/10.1111/cup.12982.
Kerob D, Porcher R, Verola O, Dalle S, Maubec E, Aubin F, et al. Imatinib mesylate as a preoperative therapy in dermatofibrosarcoma: results of a multicenter phase II study on 25 patients. Clin Cancer Res. 2010;16(12):3288–95. https://doi.org/10.1158/1078-0432.CCR-09-3401.
Sanz-Trelles A, Ayala-Carbonero A, Rodrigo-Fernandez I, Weil-Lara B. Leiomyomatous nodules and bundles of vascular origin in the fibrosarcomatous variant of dermatofibrosarcoma protuberans. J Cutan Pathol. 1998;25(1):44–9.
Banerjee SS, Harris M, Eyden BP, Hamid BN. Granular cell variant of dermatofibrosarcoma protuberans. Histopathology. 1990;17(4):375–8.
Wrotnowski U, Cooper PH, Shmookler BM. Fibrosarcomatous change in dermatofibrosarcoma protuberans. Am J Surg Pathol. 1988;12(4):287–93.
Llombart B, Monteagudo C, Sanmartin O, Lopez-Guerrero JA, Serra-Guillen C, Poveda A, et al. Dermatofibrosarcoma protuberans: a clinicopathological, immunohistochemical, genetic (COL1A1-PDGFB), and therapeutic study of low-grade versus high-grade (fibrosarcomatous) tumors. J Am Acad Dermatol. 2011;65(3):564–75. https://doi.org/10.1016/j.jaad.2010.06.020.
Lyu A, Wang Q. Dermatofibrosarcoma protuberans: a clinical analysis. Oncol Lett. 2018;16(2):1855–62. https://doi.org/10.3892/ol.2018.8802.
Hayakawa K, Matsumoto S, Ae K, Tanizawa T, Gokita T, Funauchi Y, et al. Risk factors for distant metastasis of dermatofibrosarcoma protuberans. J Orthop Traumatol. 2016;17(3):261–6. https://doi.org/10.1007/s10195-016-0415-x.
Sasaki M, Ishida T, Horiuchi H, MacHinami R. Dermatofibrosarcoma protuberans: an analysis of proliferative activity, DNA flow cytometry and p53 overexpression with emphasis on its progression. Pathol Int. 1999;49(9):799–806.
Takahira T, Oda Y, Tamiya S, Yamamoto H, Kawaguchi K, Kobayashi C, et al. Microsatellite instability and p53 mutation associated with tumor progression in dermatofibrosarcoma protuberans. Hum Pathol. 2004;35(2):240–5.
Hisaoka M, Okamoto S, Morimitsu Y, Tsuji S, Hashimoto H. Dermatofibrosarcoma protuberans with fibrosarcomatous areas. Molecular abnormalities of the p53 pathway in fibrosarcomatous transformation of dermatofibrosarcoma protuberans. Virchows Arch. 1998;433(4):323–9.
Hiraki-Hotokebuchi Y, Yamada Y, Kohashi K, Yamamoto H, Endo M, Setsu N, et al. Alteration of PDGFRbeta-Akt-mTOR pathway signaling in fibrosarcomatous transformation of dermatofibrosarcoma protuberans. Hum Pathol. 2017;67:60–8. https://doi.org/10.1016/j.humpath.2017.07.001.
Cleven AH, Al Sannaa GA, Briaire-de Bruijn I, Ingram DR, van de Rijn M, Rubin BP, et al. Loss of H3K27 tri-methylation is a diagnostic marker for malignant peripheral nerve sheath tumors and an indicator for an inferior survival. Mod Pathol. 2016;29(9):1113. https://doi.org/10.1038/modpathol.2016.103.
Pekmezci M, Cuevas-Ocampo AK, Perry A, Horvai AE. Significance of H3K27me3 loss in the diagnosis of malignant peripheral nerve sheath tumors. Mod Pathol. 2017;30(12):1710–9. https://doi.org/10.1038/modpathol.2017.97.
Shimizu A, O'Brien KP, Sjoblom T, Pietras K, Buchdunger E, Collins VP, et al. The dermatofibrosarcoma protuberans-associated collagen type I alpha 1/platelet-derived growth factor (PDGF) B-chain fusion gene generates a transforming protein that is processed to functional PDGF-BB. Cancer Res. 1999;59(15):3719–23.
McArthur G. Molecularly targeted treatment for dermatofibrosarcoma protuberans. Semin Oncol. 2004;31(2):30–6. https://doi.org/10.1053/j.seminoncol.2004.03.038.
• Nakamura I, Kariya Y, Okada E, Yasuda M, Matori S, Ishikawa O, et al. A novel chromosomal translocation associated with COL1A2-PDGFB gene fusion in dermatofibrosarcoma protuberans: PDGF expression as a new diagnostic tool. JAMA Dermatol. 2015;151(12):1330–7. https://doi.org/10.1001/jamadermatol.2015.2389. Reporting the novel fusion gene of DFSP; COL1A2-PDGFB.
• Dadone-Montaudie B, Alberti L, Duc A, Delespaul L, Lesluyes T, Perot G, et al. Alternative PDGFD rearrangements in dermatofibrosarcomas protuberans without PDGFB fusions. Mod Pathol. 2018;31(11):1683–93. https://doi.org/10.1038/s41379-018-0089-4. Next generation sequencing study reporting the novel fusion gene of DFSP; EMILIN2-PDGFD.
• Dickson BC, Hornick JL, CDM F, Demicco EG, Howarth DJ, Swanson D, et al. Dermatofibrosarcoma protuberans with a novel COL6A3-PDGFD fusion gene and apparent predilection for breast. Genes Chromosom Cancer. 2018;57(9):437–45. https://doi.org/10.1002/gcc.22663. Next generation sequencing study reporting the novel fusion gene of DFSP; COL6A3-PDGFD.
Eilers G, Czaplinski JT, Mayeda M, Bahri N, Tao D, Zhu M, et al. CDKN2A/p16 loss implicates CDK4 as a therapeutic target in imatinib-resistant dermatofibrosarcoma protuberans. Mol Cancer Ther. 2015;14(6):1346–53. https://doi.org/10.1158/1535-7163.MCT-14-0793.
Saab J, Rosenthal IM, Wang L, Busam KJ, Nehal KS, Dickson MA, et al. Dermatofibrosarcoma protuberans-like tumor with COL1A1 copy number gain in the absence of t(17;22). Am J Dermatopathol. 2017;39(4):304–9. https://doi.org/10.1097/DAD.0000000000000746.
Veronese F, Boggio P, Tiberio R, Gattoni M, Fava P, Caliendo V, et al. Wide local excision vs. Mohs Tubingen technique in the treatment of dermatofibrosarcoma protuberans: a two-centre retrospective study and literature review. J Eur Acad Dermatol Venereol. 2017;31(12):2069–76. https://doi.org/10.1111/jdv.14378.
Mendenhall WM, Zlotecki RA, Scarborough MT. Dermatofibrosarcoma protuberans. Cancer. 2004;101(11):2503–8. https://doi.org/10.1002/cncr.20678.
Acosta AE, Velez CS. Dermatofibrosarcoma Protuberans. Curr Treat Options in Oncol. 2017;18(9):56. https://doi.org/10.1007/s11864-017-0498-5.
Ng A, Nishikawa H, Lander A, Grundy R. Chemosensitivity in pediatric dermatofibrosarcoma protuberans. J Pediatr Hematol Oncol. 2005;27(2):100–2.
Suit H, Spiro I, Mankin HJ, Efird J, Rosenberg AE. Radiation in management of patients with dermatofibrosarcoma protuberans. J Clin Oncol. 1996;14(8):2365–9. https://doi.org/10.1200/JCO.1996.14.8.2365.
Sun LM, Wang CJ, Huang CC, Leung SW, Chen HC, Fang FM, et al. Dermatofibrosarcoma protuberans: treatment results of 35 cases. Radiother Oncol. 2000;57(2):175–81.
Dagan R, Morris CG, Zlotecki RA, Scarborough MT, Mendenhall WM. Radiotherapy in the treatment of dermatofibrosarcoma protuberans. Am J Clin Oncol. 2005;28(6):537–9.
Chen YT, Tu WT, Lee WR, Huang YC. The efficacy of adjuvant radiotherapy in dermatofibrosarcoma protuberans: a systemic review and meta-analysis. J Eur Acad Dermatol Venereol. 2016;30(7):1107–14. https://doi.org/10.1111/jdv.13601.
Lemm D, Mugge LO, Mentzel T, Hoffken K. Current treatment options in dermatofibrosarcoma protuberans. J Cancer Res Clin Oncol. 2009;135(5):653–65. https://doi.org/10.1007/s00432-009-0550-3.
Shimizu A, O'Brien KP, Sjoblom T, Pietras K, Buchdunger E, Collins VP, et al. The dermatofibrosarcoma protuberans-associated collagen type Ialpha1/platelet-derived growth factor (PDGF) B-chain fusion gene generates a transforming protein that is processed to functional PDGF-BB. Cancer Res. 1999;59(15):3719–23.
Ugurel S, Mentzel T, Utikal J, Helmbold P, Mohr P, Pfohler C, et al. Neoadjuvant imatinib in advanced primary or locally recurrent dermatofibrosarcoma protuberans: a multicenter phase II DeCOG trial with long-term follow-up. Clin Cancer Res. 2014;20(2):499–510. https://doi.org/10.1158/1078-0432.CCR-13-1411.
McArthur GA, Demetri GD, van Oosterom A, Heinrich MC, Debiec-Rychter M, Corless CL, et al. Molecular and clinical analysis of locally advanced dermatofibrosarcoma protuberans treated with imatinib: imatinib target exploration consortium study B2225. J Clin Oncol. 2005;23(4):866–73. https://doi.org/10.1200/JCO.2005.07.088.
Stacchiotti S, Pedeutour F, Negri T, Conca E, Marrari A, Palassini E, et al. Dermatofibrosarcoma protuberans-derived fibrosarcoma: clinical history, biological profile and sensitivity to imatinib. Int J Cancer. 2011;129(7):1761–72. https://doi.org/10.1002/ijc.25826.
Oh E, Jeong HM, Kwon MJ, Ha SY, Park HK, Song JY, et al. Unforeseen clonal evolution of tumor cell population in recurrent and metastatic dermatofibrosarcoma protuberans. PLoS One. 2017;12(10):e0185826. https://doi.org/10.1371/journal.pone.0185826.
Kitagawa D, Yokota K, Gouda M, Narumi Y, Ohmoto H, Nishiwaki E, et al. Activity-based kinase profiling of approved tyrosine kinase inhibitors. Genes Cells. 2013;18(2):110–22. https://doi.org/10.1111/gtc.12022.
Fu Y, Kang H, Zhao H, Hu J, Zhang H, Li X, et al. Sunitinib for patients with locally advanced or distantly metastatic dermatofibrosarcoma protuberans but resistant to imatinib. Int J Clin Exp Med. 2015;8(5):8288–94.
Xiao W, Que Y, Peng R, Ding Y, Zhao J, Wen X, et al. A favorable outcome of advanced dermatofibrosarcoma protuberans under treatment with sunitinib after imatinib failure. Onco Targets Ther. 2018;11:2439–43. https://doi.org/10.2147/OTT.S150235.
Wilhelm SM, Carter C, Tang L, Wilkie D, McNabola A, Rong H, et al. BAY 43-9006 exhibits broad spectrum oral antitumor activity and targets the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in tumor progression and angiogenesis. Cancer Res. 2004;64(19):7099–109. https://doi.org/10.1158/0008-5472.CAN-04-1443.
Kamar FG, Kairouz VF, Sabri AN. Dermatofibrosarcoma protuberans (DFSP) successfully treated with sorafenib: case report. Clin Sarcoma Res. 2013;3(1):5. https://doi.org/10.1186/2045-3329-3-5.
Miyagawa T, Kadono T, Kimura T, Saigusa R, Yoshizaki A, Miyagaki T, et al. Pazopanib induced a partial response in a patient with metastatic fibrosarcomatous dermatofibrosarcoma protuberans without genetic translocations resistant to mesna, doxorubicin, ifosfamide and dacarbazine chemotherapy and gemcitabine-docetaxel chemotherapy. J Dermatol. 2017;44(3):e21–e2. https://doi.org/10.1111/1346-8138.13717.
Tsuchihashi K, Kusaba H, Yamada Y, Okumura Y, Shimokawa H, Komoda M, et al. Programmed death-ligand 1 expression is associated with fibrosarcomatous transformation of dermatofibrosarcoma protuberans. Mol Clin Oncol. 2017;6(5):665–8. https://doi.org/10.3892/mco.2017.1197.
Osio A, Xu S, El Bouchtaoui M, Leboeuf C, Gapihan G, Lemaignan C, et al. EGFR is involved in dermatofibrosarcoma protuberans progression to high grade sarcoma. Oncotarget. 2018;9(9):8478–88. https://doi.org/10.18632/oncotarget.23899.
Bigby SM, Oei P, Lambie NK, Symmans PJ. Dermatofibrosarcoma protuberans: report of a case with a variant ring chromosome and metastases following pregnancy. J Cutan Pathol. 2006;33(5):383–8. https://doi.org/10.1111/j.0303-6987.2006.00404.x.
Meng T, Shi XH, Wu SF, Luo YF, Wang XJ, Long X. Hormone receptors analysis in Chinese patients with dermatofibrosarcoma protuberans. J Surg Oncol. 2018;118:157–66. https://doi.org/10.1002/jso.25117.
Kreicher KL, Honda KS, Kurlander DE, Bordeaux JS. Hormone receptor expression in patients with dermatofibrosarcoma protuberans. J Am Acad Dermatol. 2016;75(6):1205–9. https://doi.org/10.1016/j.jaad.2016.07.011.
Stacchiotti S, Astolfi A, Gronchi A, Fontana A, Pantaleo MA, Negri T, et al. Evolution of dermatofibrosarcoma protuberans to DFSP-derived fibrosarcoma: an event marked by epithelial-mesenchymal transition-like process and 22q loss. Mol Cancer Res. 2016;14(9):820–9. https://doi.org/10.1158/1541-7786.MCR-16-0068.
Chase A, Cross NC. Aberrations of EZH2 in cancer. Clin Cancer Res. 2011;17(9):2613–8. https://doi.org/10.1158/1078-0432.CCR-10-2156.
Holm K, Grabau D, Lovgren K, Aradottir S, Gruvberger-Saal S, Howlin J, et al. Global H3K27 trimethylation and EZH2 abundance in breast tumor subtypes. Mol Oncol. 2012;6(5):494–506. https://doi.org/10.1016/j.molonc.2012.06.002.
Italiano A, Soria JC, Toulmonde M, Michot JM, Lucchesi C, Varga A, et al. Tazemetostat, an EZH2 inhibitor, in relapsed or refractory B-cell non-Hodgkin lymphoma and advanced solid tumours: a first-in-human, open-label, phase 1 study. Lancet Oncol. 2018;19(5):649–59. https://doi.org/10.1016/S1470-2045(18)30145-1.
Kim J, Lee Y, Lu X, Song B, Fong KW, Cao Q, et al. Polycomb- and methylation-independent roles of EZH2 as a transcription activator. Cell Rep. 2018;25(10):2808–20 e4. https://doi.org/10.1016/j.celrep.2018.11.035.
Dickson MA, Mahoney MR, Tap WD, D'Angelo SP, Keohan ML, Van Tine BA, et al. Phase II study of MLN8237 (Alisertib) in advanced/metastatic sarcoma. Ann Oncol. 2016;27(10):1855–60. https://doi.org/10.1093/annonc/mdw281.
Kanamori S, Kajihara I, Kanazawa-Yamada S, Otsuka-Maeda S, Ihn H. Expression of aurora kinase A expression in dermatofibrosarcoma protuberans. J Dermatol. 2018;45(4):507–8. https://doi.org/10.1111/1346-8138.14235.
Acknowledgements
We are very grateful to Dr. Y. Yamada, K. Kohashi, and I. Kinoshita at Kyushu University Hospital for helpful discussions. We also thank Dr. Y. Ohshiro at Matsuyama Red Cross Hospital for kindly providing the clinical samples.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Skin Cancer
Rights and permissions
About this article
Cite this article
Iwasaki, T., Yamamoto, H. & Oda, Y. Current Update on the Molecular Biology of Cutaneous Sarcoma: Dermatofibrosarcoma Protuberans. Curr. Treat. Options in Oncol. 20, 29 (2019). https://doi.org/10.1007/s11864-019-0628-3
Published:
DOI: https://doi.org/10.1007/s11864-019-0628-3