Abstract
Background
Cyclo-oxygenase-2 (COX-2) is up-regulated in malignant tumours rendering it an attractive target for cancer therapeutics. However, whether long-term antagonism maintains its initial efficacy on established tumours is unclear.
Methods
4T1 cells were injected into the mammary fat pad of BALB/c mice (n = 8). Once tumour deposits were established, animals were randomized into two equal groups to receive either a selective COX-2 inhibitor (SC-236) or a drug vehicle. Further animals similarly treated (n = 7) were studied in diuresis cages allowing urine capture and analysis by mass spectrometry to determine Prostaglandin F-1 levels (PGF-1). In addition, both wild-type receiving SC-236 and COX-2 knockout mice receiving either SC 236 or vehicle were subjected to the same studies to determine whether tumour-derived or host-derived (stromal) COX-2 was the critical element. Finally, BALB/c mice with 4T1 tumours (n = 7) were treated with a combination of COX-2 and lipoxygenase (LOX) inhibition to attenuate this escape phenomenon.
Results
While selective COX-2 inhibition initially retarded tumour growth, a rapid increase in tumour growth rate occurred later (day 9). This escape phenomenon correlated with an increase in urinary PGF-1 levels. An identical trend was also observed whether COX-2 knockout mice received SC-236 or not, suggesting that this effect is due to increased tumour-derived COX-2 production rather than recovery of host COX-2 functional capacity. Finally, dual inhibition of COX and LOX pathways attenuated this escape process.
Conclusion
The anti-neoplastic effects of selective COX-2 inhibition may not be sustained as tumours demonstrate an escape capacity. However, this phenomenon maybe attenuated by a combination of COX/LOX inhibitors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cyclo-oyxgenase 2 (COX-2) expression has been demonstrated to be up-regulated in many cancers, including mammary, pancreatic, gastrointestinal, endometrial and prostatic tumours [1–6]. Both the enzyme and the prostaglandin products it facilitates play important roles in tumourigenesis, from tumour development through to invasion and metastasis. COX-2 expression itself promotes cell proliferation by the activation of EGFR and inhibits apoptosis by up-regulation of Bcl-2 [7–9]. In addition, COX-2 acts to suppress the host immune response, induce angiogenesis through augmented VEGF and bFGF expression and facilitate metastatic potential by up-regulation of uPA and MMP-2 [8, 10, 11]. Numerous studies have suggested that selective inhibition of COX-2 could be of considerable therapeutic value in the treatment and, possibly, prevention of cancer.
Lipoxygenase (LOX) metabolic pathways are emerging as key regulators of cell proliferation and neo-angiogenesis. There are three known isoforms of the lipoxygenase family; 5-lipoxygenase (5-LOX), 12-lipoxygenase (12-LOX) and 15-lipoxygenase (15-LOX). In recent years, a participation of 5-LOX in the regulation of cell proliferation and apoptosis has emerged, and the biological functions of 5-LOX in cancer cells have been examined using pharmacological inhibitors and/or antisense technology. It is reported that 5-LOX products, namely, 5(S)-hydroxyeicosatetraenoic acid (HETE) and Leukotriene A4 (LTA4) but not Leukotriene B4 (LTB4), potently up-regulate vascular endothelial growth factor (VEGF) transcription in a human malignant mesothelioma model [12]. As VEGF is also a potent proangiogenic factor and is therefore crucial for tumor growth and invasion, it might be concluded that 5-LOX promotes in vivo tumour development by a dual mechanism, a direct proliferative stimulus on cancer cells and a potentiation of the proangiogenic response by the host stromal cell. A mechanism linking COX-2 inhibition to apoptosis could lead to an increase in concentration of free, unmetabolized arachidonic acid [13].
We have previously demonstrated that effective COX-2 inhibition can indeed markedly diminish the rate of both the development and growth of primary and secondary malignant deposits soon after initiation of treatment [14, 15]. However, careful scrutiny of the slope of the tumour growth curves in these studies, and others [16], suggested subtle non-linear growth patterns occurring late in the time-periods, studied raising the possibility of a diminution of efficacy occurring over time. These observations compelled us to examine the durability of COX-2 inhibitor’s effect using the same rodent mammary model of breast cancer. In doing so, tumours were found to exhibit a capacity to escape pharmaceutical COX-2 inhibition as evidenced by increases overtime in both tumour growth and urinary prostaglandin metabolites. The relevant contribution of host to tumour COX-2 was proven by utilizing COX-2 inhibitors in COX-2 knockout mice.
The blockade of arachidonic metabolism may increase the intracellular levels of unesterified arachidonate, which itself induces a concentration-dependent apoptotic response. If this is the case, when multiple metabolic pathways of arachidonic acid are present within the same cell, as in cancer cells coexpressing COX-2 and 5-LOX, both enzymes should be blocked to achieve a substantial elevation in free arachidonic acid levels [17]. We therefore decided to inhibit both lipoxygenase and cyclooygenase in an orthotopic model of breast cancer to determine whether we could attenuate the escape phenomenon.
Materials and methods
The COX-2 inhibitor
SC-236 (4-[5-(4-chlorophenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl] benzene-sulfonamide) is a potent selective COX-2 inhibitor and a trivial inhibitor of COX-1 activity. The selected dose was based on previously published data [18].
The LOX inhibitor
5-LOX inhibitor [2(12-hydroxydodeca-5-10-dinyl)-3,5,6-trimethyl-1-4-benzoquinone] was administered at 10 mg/Kg in 1% DMSO as an intraperitoneal injection [19]. This was purchased from Alpha Technologies.
Animals
All animals were housed in a licensed biomedical facility (RCSI Department of Surgery, Beaumont Hospital) and every procedure was conducted under license from the Department of Health, Ireland, and in accordance with the guidelines of United Kingdom Co-ordinating Committee on Cancer Research (UKCCR). Animals were allowed to acclimatize after arrival for 1 week being housed in an air-conditioned room at a temperature of 21°C and 50% humidity under a 12-h light–dark cycle. All animals had free access to water and food (WM Connolly & Sons).
Balb-c mice were obtained from Charles River Institute, Margate, Kent, UK They were purchased in cages in groups of five prior to being used in the study.
COX-2 Knockout mice
The COX-2 knockout mice were kind gifts from Dr. Les Ballou’s group (Department of Veterans Affairs Medical Centre, Memphis, Tennessee, USA). These animals were developed on a C57BL6/OLA-129 background, which was backcrossed with DBA-1 mice for six generations, followed by extensive interbreeding.
Tumour cell culture preparation
The 4T1 tumour cell line is a spontaneously metastasizing mammary adenocarcinoma that was provided as a gift by Dr. F. Miller of Duke University, USA. Cells were stored in monolayer cultures in Dulbecco’s Modified Eagle Medium, which also contains foetal bovine serum, sodium pyruvate, non-essential amino acids, l-glutamine and vitamins (Life Technologies, Inc., GIBCO BRL, Paisley, UK). All cells were maintained in ventilated flasks in an atmosphere of 5% CO2 in air at 37°C. The tumour cells were harvested from subconfluent cultures using 0.25% trypsin–0.02% EDTA, then washed three times with PBS without magnesium/calcium and centrifuged at 1100 rpm for 5 min. Cellular viability was proven using trypan blue and single cell suspensions with greater than 90% viability were used for mammary fat pad injections.
Tumour model
The 4T1 mammary pad/pulmonary metastases model in BALB/c mice is a validated model for early metastatic disease [20]. This tumour shares many of the characteristics with human mammary cancers with respect to its immunogenicity, metastatic properties and growth characteristics making it an excellent surrogate model. In brief, this model involves each mouse undergoing anaesthesia using inhalational halothane and having 50,000 4T1 cells injected into the mammary fat pad adjacent to the right forefoot. The size of this developing primary tumour was measured daily using a Vernier caliper to assess its area (calculated as the square root of the product of two perpendicular diameters as per Pulaski et al. [21].
Study groups
Balb-c mice that had tumours induced as mentioned above were randomized into two groups on the 10th day after injection, as this time-period represents a tumour incubation phase sufficient for the tumour diameter to reliably and consistently reach approximately 8.0 ± 0.4 mm as well as for micrometastases to become established in the lungs. The first group (n = 8) then subsequently received intraperitoneal injections of 200 μl of the selective COX-2 inhibitor SC-236 at 6 mg/kg in 1% v/v dimethylsulphoxide (DMSO), while the second group (n = 8) was administered a similar volume of vehicle solution alone at the same intervals.
COX-2 knockout (−/−) mice and their wild-type equivalent were also injected with 50,000 4T1 cells. Again, once tumour diameter reached approximately 8.0 ± 0.5 mm, these animals were randomized (n = 5/group) to receive either a selective COX-2 inhibitor [200 μl of the selective COX-2 inhibitor (SC 236 at 6 mg/Kg in 1% DMSO)] or a control drug vehicle (daily intra-peritoneal injections of 200 μl vehicle (1% v/v DMSO). Lastly, three groups (n = 7) of Balb-c mice were again incubated with 4T1 tumours, and once tumours reached 8.0 ± 0.5 mm, they were randomized to receive drug vehicle, selective COX-2 inhibitor or a combination of COX-2 and LOX inhibitors.
Analysis of urinary PGF-1
Further, balb-c mice underwent the same treatment as above in the same groups, but on this occasion they were housed in a diuresis cage to allow 24-h collection of pooled urine throughout the study period.
Preparation of mouse urine samples
Daily, approximately 4–5 ml of pooled urine was collected from the diuresis cages of mice in both groups. Samples were filtered and then centrifuged at 5,000 rpm for 5 min before being subdivided into two separate aliquots. The first was used for estimation of urinary creatinine levels, while the second was stored in a −80°C freezer for later analysis using mass spectrometry.
Urinary prostacyclin metabolite analysis by mass spectrometry
Using mass spectrometry, levels of 6-keto PGF-1α, a stable compound derived from PGI-2, were measured in the collected urine as level of this substance accurately reflects overall COX-2 activity. Prostaglandin production (PGF-1) was by this means determined at selected time points during the experiment (namely, day 3, 6, 7, 9, 10, and 12).
Mass spectrometry analysis
High performance liquid chromatography (HPLC) conditions
The HPLC conditions employed a binary reverse phase gradient system and totaled 25 min of active run preceded by a 5-min pre-equilibration step before each run. The column used was Omnisphere C18 (Varian), dimensions 2.1 mm diameter and 100 mm length, and packed with 3 μm particles. The total combined binary flow into the mass spectrometer was 200 μl/min. Total run time—including pre-equilibration step—was 30 min per sample. Typical retention time for 2,3 dinor-6-ketoPGF1α was 12 min. Typical peak width was 12 s ensuring 12 scans per mass transition for the dwell of 500 ms per mass transition.
Mass spectrometry conditions
Mass spectrometry used electrospray ionization (Turbo-ionspray) on a triple quadrupole type instrument (API 3000-Applied Biosystems), and total HPLC effluent was fed directly into the source. Ancillary gas supplies were provided by an NM20LA generator (Peak Scientific). Nitrogen was used for curtain; collision, auxiliary and nebulizer gas, while air was used as the source exhaust gas. The mass spectrometer was operated in negative ion mode using a selected reaction monitoring (SRM) program specific for 2,3 dinor-6-ketoPGF1a and tetradeuterated 2,3 dinor-6-ketoPGF1a and an optimised collision energy of 28 volts for the following parent/daughter ion transitions.
Statistical analysis
Data are expressed as mean ± standard error mean (SEM). Differences between treatment groups were determined by unpaired t test using Instat for Windows statistics package (Graphpad Software Inc.). Data were considered as significant when P < 0.05.
Results
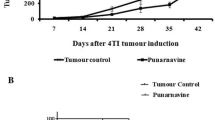
As demonstrated in Fig. (1), a significant initial decrease in tumour growth rate occurred in balb-c mice administered daily COX-2 inhibitor therapy that persisted until approximately the 9th day after treatment commenced. After this time-point, however, the trend reversed with an incremental growth in primary tumour size again becoming apparent. Prior to day 9, the mean growth rate of the COX-2 inhibited group was 0.34 mm/day, however, after that the growth rate accelerated to 0.66 mm/day. Indeed, by day 14, the average tumour diameter of the COX-2 inhibition group equaled that of the control group. Whether this observation was related to loss of effect of COX-2 inhibition (i.e., tumour escape) was verified by the levels of PGF-1α measured in mice receiving SC-236 as compared with that of vehicle alone (Fig. 2). Initially levels decreased markedly with commencement of SC-296 daily injection. However, as is evident from analysis of this data (Fig. 2), there was a significant rise in 6-keto PGF-1α after day 6. This implies an enhancement of COX activity in the presence of ongoing administration of a selective COX-2 inhibitor.
Results of mass spectrometry analysis of mouse urine for determination of Prostaglandin F-1 (PGF1). Same groups as Fig. 1
Tumour growth in wild-type mice treated with a selective COX-2 inhibitor is again shown (Fig. 3) but this time in contrast with COX-2 knock-out mice receiving either SC-236 or vehicle solution. On day 9 a visible change in the slope of the growth curve can again be ascertained that is consistent with previous escape observations. Due to the lack of host COX-2 expression, there is an attenuation of tumour growth in both knock-out mouse groups. Also, this effect is not sustainable with accelerated tumour growth occurring again at approximately about day 9. Examination of the tumour growth curves in COX-2 knock-out mice receiving the COX-2 inhibitor, however, reveals that there is loss of the gompertzian growth pattern typically associated with tumour proliferation.
Progression of primary tumours in three rodent groups with established mammary pad tumours in (filled diamond) wild-type mice (with COX-2 expressing tumours) treated with a selective COX-2 inhibitor, (filled square) COX-2 knock-out mice (cox-2 −/−) with COX-2 expressive tumours treated with control drug vehicle and lastly (filled triangle) a COX-2 knock-out group that had their COX-2 tumours treated with selective COX-2 inhibitors. Data expressed as mean ± standard error mean (SEM)
On examination of the tumour growth curves later (Fig. 4), the control tumours maintained their gompertzian growth pattern as expected. The selective COX-2 inhibited group, as previously observed, displayed the escape process again at similar time points. However, more importantly, the combined COX-2/5-LOX inhibited group failed to demonstrate any escape. This finding supports the initial hypothesis that this combined inhibition approach would retard tumour growth and attenuate the escape phenomenon.
Tumour growth curves of rodent model of breast cancer. Ten days after implantation with 4T1 cells, animals began receiving daily injections of either vehicle solution (controls), selective COX-2 inhibitor SC-236, lipoxygenase inhibitor, or a combination of selective COX-2 and 5-LOX inhibitor for a further 14 days
Discussion
It first became apparent that cyclooxygenase had a significant role in carcinogenesis when Thun et al. [22] demonstrated a significant reduction in the risk of fatal colon cancer in patients on long-term aspirin. In the early 1990s, the discovery of COX-2 inhibitors appeared to offer cancer patients and people with a genetic predisposition to developing cancer a new potential therapy without the traditional side-effect profile of non-selective non-steroidal anti-inflammatory drugs. Over the next 10 years, there was tremendous interest and activity within the scientific and academic community regarding the relationship between COX-2 and the neoplastic process. Chapple et al. [23] suggested that COX-2 expression is found in both epithelial and stromal components of sporadic human colorectal cancers. Shattuck-Brandt et al. [24] also observed high levels of COX-2 mRNA expressed in stromal fibroblasts of both human and rodent colorectal cancers. Furthermore, it was determined that COX-2 in stromal fibroblasts could act to promote tumour growth by producing bioactive prostaglandins that have paracrine effects on nearby carcinoma cells. Kinzler et al. [25] further suggested the idea that COX-2 expression in the stromal component of a solid tumour could influence either its growth or expression of proangiogenic factors.
Numerous experimental, clinical and epidemiologic studies indicate that NSAIDs, particularly the highly selective COX-2 inhibitors, show promise as anticancer drugs. Indeed, we ourselves have previously shown that a selective COX-2 inhibitor can significantly attenuate both primary and secondary cancer deposits [14, 15]. In this follow-up study, however, we have shown that the initial effects of this therapy are not sustained, thus raising important issues for the clinical use of these medications as anti-neoplastic agents. On evaluation of the current medical literature, the only previous clear suggestion of the existence of a tumour escape process from COX-2 inhibition is in a study by Williams et al. [26] who treated tumour-implanted wild-type and COX-2 (−/−) mice with selective COX-2 inhibitors. The comment offered by Williams et al. for the increase in growth rate was ‘that the absence of COX-2 in the host did not permanently halt carcinoma growth’. Interestingly, the increase in growth rate in their study occurred from day 20 until day 25 post tumour implantation which correlates roughly with our study’s observation of escape at day 9 post initiation of COX-2 inhibition or day 19 after tumour establishment. A significant difference between their study and ours, however, is that they pretreated the animals with the selective COX-2 inhibitor before tumour implantation whereas we commenced treatment with COX-2 inhibitors once tumours were well established (i.e., average diameter was 8.0 ± 0.5 mm).
If the escape process was independent of COX-2, then the levels of PGF-1α would have remained at consistently suppressed levels throughout the experiment. Moreover, if the observed phenomenon was due to tumour resistance to selective COX-2 inhibition, then PGF-1 levels should continue to decrease despite an increase in tumour size. If this is in fact a tumour escape strategy, then PGF-1 levels should initially decrease and then increase as tumours become larger. This suggests that tumours have the potential to overcome, adapt and escape selective COX-2 inhibition despite the initial antitumour effect. To determine whether this was a function of host-recovery of COX-2 capacity or was tumour-related, the final part of the study utilized COX-2 knock-out mice (−/−) given tumours which express COX-2 being treated with a COX-2 inhibitor. Although these animals are incapable of producing host COX-2 themselves, tumour escape from COX-2 inhibition was still apparent although somewhat delayed than previously observed.
These findings suggest that this phenomenon is due to up-regulation of tumour derived COX-2 expression. Interestingly, the overall daily tumour growth rate for the wild-type (treated with COX-2 inhibitor) and the knock-out mice (treated with control vehicle) was the same, i.e., 0.883 mm/day. This would imply that the effect of a selective COX-2 inhibitor is similar if not identical to the effect of genetic disruption of host COX-2 expression. However, it is also possible that this increase in activity could be due to increased COX-2 activity from sites of metastases. Studies have shown that COX-2 is expressed in liver metastatic deposits and, therefore, it is possible that as the primary neoplasm becomes metastatic, treatment with the same dose of selective COX-2 inhibitors becomes less effective [27].
There is of course already a vast array of rodent mammary carcinogenesis models in the literature that used COX-2 inhibitors and clearly demonstrated a statistically significant benefit. However, when we evaluated these models more closely, we identified several potential reasons why an escape process was not observed. Firstly, a large number of studies commenced COX-2 inhibition either prior to or at the same time as tumour cell injection [28–32]. NSAIDs and selective COX-2 inhibitors suppress tumor growth to a greater extent when treatment is begun before or coincident with exposure to the carcinogen than when it is delayed until the tumor promotion/progression phase [33]. This also explains why in rodent models of Familial Adenomatous Polyposis Coli (FAP), blockade of COX-2 either by gene deletion or by pharmaceutical inhibition of enzyme activity leads to suppression of intestinal polyp formation [34]. In contrast in our study, selective COX-2 inhibition was used to treat established primary tumours with metastatic potential. Therefore, we believe that this model is more representative and more accurate in reflecting the clinical situation where appropriate treatment commences only after the patients present with a primary tumour. Some similar studies, however, did not observe any evidence of an escape process which may be due to different tumour biology or chronology [14], although several studies have used markedly higher doses of COX-2 inhibitors to achieve their tumour growth retardation suggesting dose-dependent effects [32].
COX-2 and 5-LOX display similarities in expression and function in human cancer. Firstly, COX-2 and 5-LOX are coexpressed and up-regulated in quite a large number of cancer cells and human tumours, including lung, colon, prostate, breast and mesothelioma [17, 35–38]. Secondly, both 5-LOX and COX-2 are proangiogenic with a convergent targeting on VEGF expression and release [12, 26, 39, 40]. Thirdly, COX-2, as well as 5-LOX inhibitors, arrest cell cycle progression and induce apoptotic cell death in a number of cancer cells [12, 41, 42]. It is therefore not surprising that a combination of these agents is more effective than either agent alone and that this escape process can be attenuated by dual COX/LOX inhibition.
Our findings therefore support a future role for selective COX-2 inhibitors as neoadjuvant agents only rather in prolonged courses. It is also possible that a short course of COX-2 inhibition preoperatively may attenuate the inflammatory response associated with surgery which is known to be prometastatic and in addition act as radiosensitizers for post-operative radiotherapy [11, 14]. Such focused therapy would also avoid the cardiac side-effects associated with long-term use of COX-2 inhibitors as has been demonstrated to be of concern in both the Adenomatous Polyp Prevention on Vioxx (APPROVe) and the Adenoma Prevention with Celecoxib (APC) study [34, 43]. Neoadjuvant treatment over a short time period with low dose COX-2 inhibitors alone or in combination with other agents (e.g., LOX inhibitors) in selected patients (i.e., good cardiac function) may be utilized to downstage tumours prior to resection. However, the clinical application of these drugs as antineoplastic agents remains limited by a lack of randomized evidence of their efficacy in populations other than those with FAP and against endpoints other than adenomatous colorectal polyps. Unresolved questions concerning the mechanisms by which these drugs act, the optimal dose, treatment regimen and the balance of risks and benefits in specific populations must still be addressed.
Conclusion
The use of COX-2 inhibitors as a long-term chemopreventive agent is already considered controversial due to reports about the increased incidence of cardiovascular events. Furthermore, this study suggests that the antineoplastic effect of COX-2 inhibitors are not sustainable over long periods of time. There is, however, a window of opportunity that their employment may provide some degree of retardation early in tumour development. The use of COX-2 inhibitors in clinical practice may be most appropriate when utilized to down stage or limit further tumour progression while patients are waiting for their definitive therapy or may prove more effective in combination with other agents such as lipoxygenase inhibitors or statins.
References
Ristimaki A, Sivula A, Lundin J, Lundin M, Salminen T, Haglund C, Joensuu H, Isola J (2002) Prognostic significance of elevated cyclo-oxygenase-2 expression in breast cancer. Cancer Res 62(3):632–635
Kokawa A, Kondo H, Gotoda T, Ono H, Saito D, Nakadaira S, Kosuge T, Yosida S (2001) Increased expression of cyclooxygenase-2 in human pancreatic neoplasms and potential for chemoprevention by cyclooxygenase inhibitors. Cancer 91(2):333–338. doi:10.1002/1097-0142(20010115)91:2<333::AID-CNCR1006>3.0.CO;2-N
Sano H, Kawahito Y, Wilder RL, Hashiramoto A, Mukai S, Asai K, Kimura S, Kato H, Kondo M, Hla T (1995) Expression of cyclooxygenase-1 and 2 in human colorectal cancer. Cancer Res 55(17):3785–3789
Juo YE, Rew JS, Seo YH, Choi SK, Kim YJ, Park CS, Kim SJ (2003) Cyclooxygenase-2 overexpression correlates with vascular endothelial growth factor expression in tumour angiogenesis in gastric cancer. J Clin Gastroenterol 37(1):28–33. doi:10.1097/00004836-200307000-00009
Li W, Xu RJ, Zhang HH, Jiang LH (2006) Overexpression of cyclooxygenase-2 correlates with tumour angiogenesis in endometrial carcinoma. Int J Gynecol Cancer 16(4):1673–1678. doi:10.1111/j.1525-1438.2006.00408.x
Tjandrawinata RR, Dahiya R, Hughes-Fulford M (1997) Induction of cyclooxygenase-2 mRNA by prostaglandin E2 in human prostatic carcinoma cells. Br J Cancer 75(8):1111–1118
Pai R, Soreghan B, Szabo IL et al (2002) Prostaglandin E2 transactivates EGF receptor : a novel mechanism for promoting colon cancer growth and gastrointestinal hypertrophy. Nat Med 8:289–293. doi:10.1038/nm0302-289
Tsujii M, Kawano S, Tsuji S, Sawoaka H, Hori M, DuBois RN (1998) Cyclooxygenase regulates angiogenesis induced by colon cancer cells. Cell 93:705–716. doi:10.1016/S0092-8674(00)81433-6
Sharma S, Stolina M, Yang SC, Baratelli F, Lin JF, Atianzar K, Luo J, Zhu L, Lin Y, Huang M, Dohadwala M, Batra RK, Dubinett SM (2003) Tumour cyclooxygenase 2-dependent suppression of dendritic cell function. Clin Cancer Res 9:961–968
Li G, Yang T, Yan J (2002) Cyclooxygenase-2 increased the angiogenic and metastatic potential of tumour cells. Biochem Biophys Res Commun 299:886–890. doi:10.1016/S0006-291X(02)02707-9
Tsujii M, Kawano S, DuBois RN (1997) Cyclooxygenase-2 expression in human colon cancer cells increases metastatic potential. Proc Natl Acad Sci USA 94:3336–3340. doi:10.1073/pnas.94.7.3336
Romano M, Catalano A, Nutini M, D’Urbano E, Crescenzi C, Claria J, Libner R, Davi G, Procopio A (2001) 5-Lipoxygenase regulates malignant mesothelial cell survival: involvement of endothelial growth factor. FASEB J 15:2326–2336. doi:10.1096/fj.01-0150com
Cao Y, Pearmann AT, Zimmerman GA, McIntyre TM, Prescott SM (2000) Intracellular unesterified arachidonic acid signals apoptosis. Proc Natl Acad Sci USA 97:11280–11285. doi:10.1073/pnas.200367597
Connolly EM, Harmey JH, O’Grady T, Foley D, Roche-Nagle G, Kay E, Bouchier-Hayes DJ (2002) Cyclooxygenase inhibition reduces tumour growth and metastasis in an orthotopic model of breast cancer. Br J Cancer 87:231–237. doi:10.1038/sj.bjc.6600462
Roche-Nagle G, Connolly EM, Eng M, Bouchier-Hayes DJ, Harmey JH (2004) Antimetastatic activity of a cyclooxygenase-2 inhibitor. Br J Cancer 91(2):359–365
Kundu N, Fulton A (2002) Selective cyclooxygenase (cox)-1 or cox-2 inhibitors control metastatic disease in a murine model of breast cancer. Cancer Res 62:2343–2346
Hong SH, Avis I, Vos MD, Martinez A, Treston AM, Mulshine JL (1999) Relationship of arachidonic acid metabolizing enzyme expression in epithelial cancer cell lines to the growth effect of selective biochemical inhibitors. Cancer Res 59:2223–2228
Milas L, Kishi K, Hunter N, Mason K, Masferrer JL, Tofilon PJ (1999) Enhancement of tumour response to radiation by an inhibitor of cyclooxygenase-2 enzyme. J Natl Cancer Inst 91:1501–1504. doi:10.1093/jnci/91.17.1501
Tong WG, Ding XZ, Witt RC, Adrian TE (2002) Lipoxygenase inhibitors attenuate growth of human pancreatic xenografts and induce apoptosis through the mitochondrial pathway. Mol Cancer Ther 1:929–935
Pidgeon GP, Harmey JH, Kay E, Da Costa M, Redmond HP, Bouchier-Hayes DJ (1999) The role of endotoxin/lipopolysaccharide in surgically induced tumour growth in a murine model of metastatic disease. Br J Cancer 81(8):1311–1317. doi:10.1038/sj.bjc.6694369
Pulaski BA, Ostrand-Rosenberg S (2001) Mouse 4T1 breast tumour model. Curr Protoc Immunol. Chap 20: Unit 20.2
Thun MJ, Namboodiri MM, Calle EE, Flanders WD, Heath CW Jr (1993) Aspirin use and the risk of fatal cancer. Cancer Res 53:1322–1327
Chapple KS, Cartwright EJ, Howcroft G, Tisbury A, Bonifer C, Scott N, Windsor AC, Guillou PJ, Markham AF, Coletta PL, Hull MA (2000) Localization of cyclooxygenase-2 in human sporadic colorectal adenoma. Am J Pathol 156:545–553
Shattuck-Brandt RL, Varilet GW, Radhika A, Yang F, Washington MK, DuBois RN (2000) Cyclooxygenase-2 expression is increased in the subepithelial myofibroblasts of colon and caecal carcinoma from IL-10 (−/−) mice. Gastroenterology 118:337–345. doi:10.1016/S0016-5085(00)70216-2
Kinzler KW, Vogelstein B (1998) Landscaping the cancer terrain. Science 280:1036–1037. doi:10.1126/science.280.5366.1036
Williams CS, Tsujii M, Reese J, Dey SK, DuBois RN (2000) Host cyclooxygenase-2 modulates carcinoma growth. J Clin Invest 105(11):1589–1594. doi:10.1172/JCI9621
Hanahan D, Folkmann J (1996) Patterns and emerging mechanisms of the angiogenic switch during tumourigenesis. Cell 86:353–364. doi:10.1016/S0092-8674(00)80108-7
Abou-Issa HM, Alshafie GA, Seibert K, Koki AT, Masferrer JL, Harris JE (2001) Dose response effects of the COX-2 inhibitor, celecoxib, on the chemoprevention of mammary carcinogenesis. Anticancer Res 21:3425–3432
Jang TJ, Jung HG, Jung KH, O Min Ku (2002) Chemopreventive effect of celecoxib and expression of cyclooxygense-1 and cyclooygenase-2 on chemically induced rat tumour models. Int J Exp Pathol 83:173–182. doi:10.1046/j.1365-2613.2002.00228.x
Sheng H, Shao J, Kirkland SC, Isakson P, Coffey RJ, Morrow J, Beauchamp RD, DuBois RN (1997) Inhibition of human colon cancer cell growth by selective inhibition of cyclooxygenase-2. J Clin Invest 99:2254–2259. doi:10.1172/JCI119400
Alshafie GA, Abou-Issa HM, Seibert K, Harris RE (2000) Chemotherapeutic evaluation of celecoxib, a cyclooxygenase-2 inhibitor, in a rat mammary tumour model. Oncol Rep 7:1377–1381
Reddy BS, Maruyama H, Kelloff G (1987) Dose related inhibition of colon carcinogenesis by dietary piroxicam; a nonsteroidal anti-inflammatory drug, during different stages of rat colon tumour development. Cancer Res 47:5340–5346
Oshima M, Dinchuk JE, Kargman SL, Oshima H, Hancock B, Kwong E, Trasaskos JM, Evans JF, Taketo MM (1996) Suppression of intestinal polyposis in APC delta 716 knockout mice by inhibition of cyclooxygenase-2 (COX-2). Cell 87:803–809. doi:10.1016/S0092-8674(00)81988-1
Lagakos SW (2006) Time-to-event analyses for long-term treatments—the APPROVe trial. N Engl J Med 355(2):113–117. doi:10.1056/NEJMp068137
Kargman S, Vickers PJ, Evan JF (1992) A23187 induces translocation of 5-lipoxygenase in osteosarcoma cells. J Cell Biol 119:1701–1709. doi:10.1083/jcb.119.6.1701
Baodo RJ, Pardridge WM, Vinters HV, Black KL (1992) Differential expression of arachidonate 5-lipoxygenase transcripts in human brain tumours: evidence for the expression of a multitranscript family. Proc Natl Acad Sci USA 89:9044–9048. doi:10.1073/pnas.89.19.9044
Henning P, Ding XZ, Tong WG, Schneider MB, Standop J, Freiss H, Buchler MW, Pour PM, Adrian TE (2002) 5-Lipoxygenase and leukotrienes B(4) receptor are expressed in human pancreatic cancers but not in pancreatic ducts in normal tissue. Am J Pathol 161:421–428
Gupta S, Srivastara M, Ahmad N, Sakamoto K, Bostwick DG, Mukhtar N (2001) Lipoxygenase-5 is overexpressed in prostate adenocarcinoma. Cancer 91:737–743. doi:10.1002/1097-0142(20010215)91:4<737::AID-CNCR1059>3.0.CO;2-F
Hoper MM, Voelkel NF, Bates TO, Allard JD, Horan M, Sheppard D, Tuder RM (1997) Prostaglandins induce vascular endothelial growth factor in a human monocyte cell line and rat lungs via Camp. Am J Respir Cell Mol Biol 17:748–756
Liu XH, Kirschenbaum A, Lu M, Yao S, Dosoretz A, Holland JF, Levine AC (2002) Prostaglandin E2 induces hypoxia inducible factor-1-alpha stabilization and nuclear localization in a prostate cancer cell line. J Biol Chem 277:50081–50086. doi:10.1074/jbc.M201095200
Avis I, Hong SH, Martinez A, Moody T, Choi YH, Trepel J, Das R, Jett M, Mulshine JL (2001) Five-lipoxygenase inhibitors can mediate apoptosis in human breast cancer cell lines through complex eicosanoid interactions. FASEB J 15(11):2007–2009
Tsukada T, Nakashima K, Shirakawa S (1986) Arachidonate 5-lipoxygenase inhibitors show potent antiproliferative effects on human leukaemia cell lines. Biochem Biophys Res Commun 140:832–836. doi:10.1016/0006-291X(86)90709-6
Solomon SD, McMurray JJ, Pfeffer MA et al (2005) Cardiovascular risk associated with celecoxib in a clinical trial for colorectal adenoma prevention. N Engl J Med 352(11):1071–1080. doi:10.1056/NEJMoa050405
Acknowledgments
We thank Dr. D. Fitzgerald for the provision of the COX-2 knock-out mice. This research was supported by the Royal College of Surgeons in Ireland.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Barry, M., Cahill, R.A., Roche-Nagle, G. et al. Neoplasms escape selective COX-2 inhibition in an animal model of breast cancer. Ir J Med Sci 178, 201–208 (2009). https://doi.org/10.1007/s11845-009-0335-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-009-0335-3