Abstract
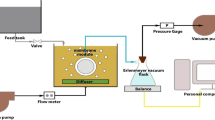
The feasibility of PES membrane with hydrophobic property, high glass transition temperature, excellent thermal and chemical stability, with good mechanical properties as porous polymeric support was studied in membrane distillation (MD) process. Several strategies were considered and investigated to decrease membrane wetting by keeping up salt rejection and also to improve the permeate flux of PES membrane in the MD process. From this point of view, hydrophobic PMHS polymer with different concentrations was used as a surface coating solution. The morphology and structure of prepared membranes were evaluated by AFM and FESEM analyses. After coating modification, the salt rejection was increased up to 98% and the contact angle was changed from 65.11° to >100.2°. To increase the water flux, the effect of feed temperature (55 °C and 75 °C) and draw solution utilization containing magnetic nanoparticle (Fe3O4) was investigated. Using draw solution provided a driving force from feed solution to permeate side and increased average permeate flux from 2.9 kg/m2·h to 6.9 kg/m2·h in the 0.06 g/l of draw solute concentration. It improved permeate flux and salt rejection simultaneously.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
D. E. Suk, T. Matsuura H. B. Park and Y. M. Lee, Desalination, 300, 312 (2010).
J. Deshpande, K. Nithyanandam and R. Pitchumani, J. Membr. Sci., 523, 301 (2017).
D. Wintera, J. Koschikowskia, F. Grossa, D. Mauchera, D. Düvera, M. Jositza and T. Mannb, J. Membr. Sci., 758, 771 (2017).
N. Shafaei, M. Jahanshahi, M. Peyravi and Q. Najafpour, Korean J. Chem. Eng., 2968, 2981 (2016).
S. Bandehali, F. Parvizian, S. M. Hosseini, T. Matsuura, E. Drioli, J. Shen, A. Moghadassi and A. S. Adeleye, Chemosphere, 283, 131207 (2021).
F. Ardeshiri, S. Salehi, M. Peyravi, M. Jahanshahi, A. Amiri and A. Shokuhi Rad, Asia-Pacific J. Chem. Eng., 13, 3 (2018).
F. Ardeshiri, A. Akbari, M. Peyravi and M. Jahanshahi, Korean J. Chem. Eng., 255, 264 (2019).
S. Salehi, M. Jahanshahi and M. Peyravi, Chem. Eng. Technol., 1994, 2004 (2019).
P. Pouresmaeel-Selkjani, M. Jahanshahi and M. Peyravi, High Performance Polymers, 759, 771 (2016).
R. R. Darabi, M. Peyravi, M. Jahanshahi and A. A. Q. Amiri, Korean J. Chem. Eng., 2311, 2324 (2017).
M. L. Lind, A. K. Ghosh, A. Jawor, X. Huang, W. Hou, Y. Yang and E. M. V. Hoek, Langmuir, 25, 10139 (2009).
Q. Ge, J. Su, T. Sh. Chung and G. Amy, Am. Chem. Soc., 382, 388 (2011).
Q. Ge, M. Ling and T. Sh. Chung, J. Membr. Sci., 225, 237 (2013).
R. R. Darabi, M. Jahanshahi and M. Peyravi, Chem. Eng. Res. Des., 11, 25 (2018).
Y. Xua, X. Peng, Ch. Y. Tangb, Q. Shiang Fud and Sh. Niea, J. Membr. Sci., 298, 309 (2010).
Y. Na, S. Yang and S. Lee, Desalination, 34, 42 (2014).
M. Khajouei, M. Peyravi and M. Jahanshahi, J. Membr. Sci. Res., 2, 12 (2017).
A. B. Rostam, M. Peyravi, M. Ghorbani and M. Jahanshahi, Appl. Surf. Sci., 17, 28 (2018).
G. Racz, S. Kerker, Z. Kovacs and G. Vatai, Chem. Eng., 81, 91 (2014).
E. Jannatduost, A. A. Babaluo, F. Abbasi, M. A. Ardestani and M. Peyravi, Desalination, 1136, 1139 (2010).
F. Ardeshiri, A. Akbari, M. Peyravi and M. Jahanshahi, J. Ind. Eng. Chem., 74, 14 (2019).
L. D. Tijing, Y.C. Woo, W.G. Shim, T. He, J.S. Choi and S.H. Kim, J. Membr. Sci., 158, 170 (2016).
H. Fan and Y. Peng, Chem. Eng. Sei., 94, 102 (2012).
R. Roshani, F. Ardeshiri, M. Peyravi and M. Jahanshahi, RSC Adv., 23499, 23515 (2018).
R. Dasa, K. Sondhib, S. Majumdara and S. Sarkara, J. Asian Ceram. Soc., 4, 243 (2016).
L. Zheng, Zh. Wu, Y. Wei, Y. Zhang and Y. Yuan, J. Membr. Sci., 71, 85 (2016).
K. J. Lu, J. Zuo and T. ShungChung, J. Membr. Sci., 65, 75 (2016).
F. Esfandian, M. Peyravi, A. A. Qoreyshi and M. Jahanshahi, J. Membr. Sci. Res., 26, 32 (2016).
M. Khajouei, M. Peyravi and M. Jahanshahi, J. Membr. Sci. Res., 2, 12 (2017).
M. Jahanshahi, A. Rahimpour and M. Peyravi, Desalination, 29, 36 (2010).
A. Rahimpour and M. Jahanshahi, Desalination and Water Treatment, 14013, 14023 (2016).
M. Manttarii, A. Pihlajamski, E. Kaipainen and M. Nystram, Desalination, 81, 86 (2002).
T. Y. Cath, V. D. Adams and A. E. Childress, J. Membr. Sci., 5, 16 (2004).
L. M. Diez and M. I. Gonzalez, J. Membr. Sci., 265, 273 (1999).
A. M. Alklaibi and N. Lior, Desalination, 111, 131 (2005).
A. Razmjou, E. Arifin, G. Dong, J. Mansouri and V. Chen, J. Membr. Sci., 850, 863 (2012).
W. TaXu, Zh. P. Zhao, M. Liu and K. Ch. Chen, J. Membr. Sci., 110, 120 (2015).
M. Gryta, Membranes, 415, 429 (2012).
Z. Shabani and A. Rahimpour, Iran. Polym. J., 25, 887 (2016).
R. Ramezani, M. Peyravi, M. Jahanshahi and S. A. A. Ghoreishi Amiri, Korean J. Chem. Eng., 2311, 2324 (2017).
Q. Ge, L. Yang, J. Cai, W. Xu, Q. Chen and M. Liu, J. Membr. Sci., 520, 550 (2016).
A. Rastegarpanah and H. R. Mortaheb, Desalination, 99, 107 (2016).
L. Eykens, K. D. Sitter, L. Stoops, C. Dotremont, L. Pinoy and B. V. Bruggen, Appl. Polym, 1, 11 (2017).
Acknowledgement
The authors acknowledge the funding support of Babol Noshirvani University of Technology through Grant program No. BNUT/389026/00.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supporting Information
Additional information as noted in the text. This information is available via the Internet at http://www.springer.com/chemistry/journal/11814.
Rights and permissions
About this article
Cite this article
Salehi, S., Jahanshahi, M. & Peyravi, M. Feasibility of hydrophobized PES membrane in hybrid MD/FO process using magnetic draw solution. Korean J. Chem. Eng. 39, 1557–1565 (2022). https://doi.org/10.1007/s11814-021-1058-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-021-1058-z