Abstract
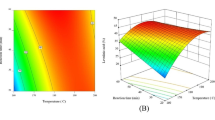
This study is focused on the possibility of using crustacean waste shells for sustainable biofuels and chemical production. We investigated the synthesis of ethyl levulinate (EL) from glucosamine by the methanesulfonic acidcatalyzed hydrothermal reaction using Box-Behnken design. In the ethyl levulinate synthesis, higher water content highly inhibited the formation of EL. Among the reaction factors, reaction temperature, catalyst concentration, and reaction time positively affected the outcome more than substrate concentration. The optimized reaction conditions were 200 °C reaction temperature, 60 g/L substrate concentration, 0.75 M catalyst concentration, and 44.9 min. Under these conditions, a 22.76 mol% EL yield was achieved. These results suggest that crustacean waste shells can be used for renewable feedstocks to produce valuable chemicals and biofuels.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
S. K Hoekman, Renew. Energy, 34, 14 (2009).
D. J. Hayes, S. Fitzpatrick, M. H. B. Hayes, J. R. H. Ross, Biorefineries — industrial processes and products, B. Kamm, P. R. Gruber and M. Kamm (Eds.), WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim, 139 (2006).
T. Werpy and G. Petersen, Top value added chemicals from biomass, volume I — Results of screening for potential candidates from sugars and synthesis gas, National Renewable Energy Lab., Golden, CO (2004).
J. N. Chheda, Y. Roman-Leshkov and J. A. Dumesic, Green Chem., 9, 342 (2007).
C. H. Ra, P. Sirisuk, J. H. Jung, G. T. Jeong and S. K. Kim, Bioprocess Biosyst. Eng., 41, 457 (2018).
M. R. Park, S. K. Kim and G. T. Jeong, Biotechnol. Bioprocess Eng., 23, 302 (2018).
M. D. N. Meinita, B. Marhaeni, G. T. Jeong and Y. K. Hong, J. Appl. Phycol., 31(4), 2507 (2019).
K. Inokuma, M. Takano and K. Hoshino, Biochem. Eng. J., 72, 24 (2013).
M. Rinaudo, Prog. Polym. Sci., 31, 603 (2006).
Y. Wang, C. M. Pederson, T. Deng, Y. Qiao and X. Hou, Bioresour. Technol., 143, 384 (2013).
H. S. Kim, M. R. Park, S. K. Kim and G. T. Jeong, Korean J. Chem. Eng., 35, 1290 (2018).
H. S. Kim, S. K. Kim and G. T. Jeong, RSC Adv., 8, 3198 (2018).
B. Y. Coh, J. W. Lee, E. S. Kim and Y. S. Park, J. Chitin Chitosan, 8(3), 127 (2003).
M. J. Hulsey, Green Energy Environ., 3, 318 (2018).
Y. W. Tiong, C. L. Yap, S. Gan and W. S. P. Yap, Ind. Crop. Prod., 128, 221 (2019).
S. Quereshi, E. Ahmad, K. K. Pant and S. Dutta., Catal. Today, 291, 187 (2017).
C. Chang, G. Xu and X. Jiang, Bioresour. Technol., 121, 93 (2012).
L. Peng, L. Lin, J. Zhang, J. Shi and S. Liu, Appl. Catal. A: Gen., 397, 259 (2011).
E. Ahmad, Md. I. Alama, K. K. Pant and M. A. Haider, Green Chem., 18, 4804 (2016).
Q. Guan, T. Lei, Z. Wang, H. Xu, L. Lin, G. Chen, X. Li and Z. Li, Ind. Crop. Prod., 113, 150 (2018).
J. J. Bozell and G. R. Petersen, Green Chem., 12(4), 539 (2010).
M. Popova, P. Shestakova, H. Lazarova, M. Dimitrov, D. Kovacheva, A. Szegedi, G. Mali, V. Dasireddy, B. Likozar, N. Wilde and R. Glaser, Appl. Catal. A Gen., 560, 119 (2018).
G. Pasquale, P. Vázquez, G. Romanelli and G. Baronetti, Catal. Commun., 18, 115 (2012).
Z. Zhang, K. Dong and Z. K. Zhao, ChemSusChem., 4, 112 (2011).
M. R. Park, H. S. Kim, S. K. Kim and G. T. Jeong, Fuel Process. Technol., 172, 115 (2018).
G. L. Miller, Anal. Chem., 31, 426 (1959).
G. Xu, C. Chang, S. Fang and X. Ma, Renew. Energy, 78, 583 (2015).
F. Rataboul and N. Essayem, Ind. Eng. Chem. Res., 50, 799 (2011).
M. Mascal and E. B. Nikitin, ChemSusChem., 3, 1349 (2010).
S. De, S. Dutta and B. Saha, Green Chem., 13, 2859 (2011).
G. Zhao, M. Liu, X. Xia, L. Li and B. Xu, Molecules, 24, 1881 (2019).
K W. Omari, J. E. Besaw and F. M. Kerton, Green Chem., 14, 1480 (2012).
Acknowledgements
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (NRF-2015R1D1A3A01015882, NRF-2018R1D1A3B07048973).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jeong, GT., Kim, SK. Efficient conversion of glucosamine to ethyl levulinate catalyzed by methanesulfonic acid. Korean J. Chem. Eng. 37, 1743–1750 (2020). https://doi.org/10.1007/s11814-020-0594-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-020-0594-2