Abstract
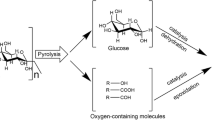
Experimental research on cellulose pyrolysis under catalysis of metallic salts was done on a thermobalance and a rapid pyrolysis system. Thermogravimetric analysis showed that K+ catalyzed the formation of active cellulose strongly and decreased the activation energy of cellulose pyrolysis. Experimental results indicated that K+ would promote the formation of char and restrain the production of bio-oil largely. Fe2+ had a similar catalysis effect on cellulose pyrolysis with K+. Fe2+ particularly catalyzed the formation of small molecule gaseous product while K+ the formation of char. The addition of K+ or Fe2+ resulted in a reduction of levoglucosan formation, and enhanced the production of hydroxyacetaldehyde and other small molecule components. Levoglucosan and hydroxyacetaldehyde were formed by the decomposition of active cellulose in a parallel mode. The secondary cracking of levoglucosan would also produce hydroxyacetaldehyde. A modified cellulose pyrolysis mechanism model was proposed based on the B-S model.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, S., Liu, Q., Liao, Y. et al. A study on the mechanism research on cellulose pyrolysis under catalysis of metallic salts. Korean J. Chem. Eng. 24, 336–340 (2007). https://doi.org/10.1007/s11814-007-5060-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-007-5060-x