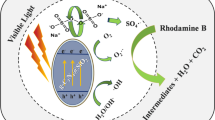
Abstract
We designed photoelectrochemical cells to achieve efficient oxidation of rhodamine B (RhB) without the need for photocatalyst or supporting electrolyte. RhB, the metal anode/cathode, and O2 formed an energy-relay structure, enabling the efficient formation of O–2 species under ultraviolet illumination. In a single-compartment cell (S cell) containing a titanium (Ti) anode, Ti cathode, and 10 mg·mL–1 RhB in water, the zero-order rate constant of the photoelectrochemical oxidation (kPEC) of RhB was 0.049 mg·L–1·min–1, while those of the photochemical and electrochemical oxidations of RhB were nearly zero. kPEC remained almost the same when 0.5 mol·L–1 Na2SO4 was included in the reactive solution, regardless of the increase in the photocurrent of the S cell. The kPEC of the illuminated anode compartment in the two-compartment cell, including a Ti anode, Ti cathode, and 10 mg·mL–1 RhB in water, was higher than that of the S cell. These results support a simple, eco-friendly, and energysaving method to realize the efficient degradation of RhB.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Chen G (2004). Electrochemical technologies in wastewater treatment. Separation and Purification Technology, 38(1): 11–41
Choi W, Termin A, Hoffmann M R (1994). Effects of metal-ion dopants on the photocatalytic reactivity of quantum-sized TiO2 particles. Angewandte Chemie, 33(10): 1091–1092
Chong M N, Jin B, Chow C W, Saint C (2010). Recent developments in photocatalytic water treatment technology: A review. Water Research, 44(10): 2997–3027
Feng X, Shang J, Zhu T (2016). Highly efficient photoelectrocatalytic reduction of hexavalent chromium based on the cascade energy transfer towards using no semiconducting photocatalysts. Electrochimica Acta, 188: 752–756
Forgacs E, Cserháti T, Oros G (2004). Removal of synthetic dyes from wastewaters: a review. Environment International, 30(7): 953–971
Fu H, Zhang S, Xu T, Zhu Y, Chen J (2008). Photocatalytic degradation of RhB by fluorinated Bi2WO6 and distributions of the intermediate products. Environmental Science & Technology, 42(6): 2085–2091
Gaya U I, Abdullah A H (2008). Heterogeneous photocatalytic degradation of organic contaminants over titanium dioxide: A review of fundamentals, progress and problems. Journal of Photochemistry and Photobiology C, Photochemistry Reviews, 9(1): 1–12
Knupfer M (2003). Exciton binding energies in organic semiconductors. Applied Physics. A, Materials Science & Processing, 77(5): 623–626
Leng W H, Zhang Z, Zhang J Q, Cao C N (2005). Investigation of the kinetics of a TiO2 photoelectrocatalytic reaction involving charge transfer and recombination through surface states by electrochemical impedance spectroscopy. Journal of Physical Chemistry B, 109(31): 15008–15023
Li Y, Sun S, Ma M, Ouyang Y, Yan W (2008). Kinetic study and model of the photocatalytic degradation of rhodamine B (RhB) by a TiO2-coated activated carbon catalyst: Effects of initial RhB content, light intensity and TiO2 content in the catalyst. Chemical Engineering Journal, 142(2): 147–155
Litter M I (1999). Heterogeneous photocatalysis: Transition metal ions in photocatalytic systems. Applied Catalysis B: Environmental, 23(2–3): 89–114
Macedo L C, Zaia D A M, Moore G J, Santana H D (2007). Degradation of leather dye on TiO2: A study of applied experimental parameters on photoelectrocatalysis. Journal of Photochemistry and Photobiology A Chemistry, 185(1): 86–93
Machado Ê L, Dambros V D S, Kist L T, Lobo E A A, Tedesco S B, Moro C C (2012). Use of ozonization for the treatment of dye wastewaters containing rhodamine B in the agate industry. Water, Air, and Soil Pollution, 223(4): 1753–1764
Markovic N M, Gasteiger H A, Ross P N Jr, Jiang X D, Villegas I, Weaver M J (1995). Electro-oxidation mechanisms of methanol and formic acid on Pt-Ru alloy surfaces. Electrochimica Acta, 40(1): 91–98
Martínez-Huitle C A, Brillas E (2009). Decontamination of wastewaters containing synthetic organic dyes by electrochemical methods: A general review. Applied Catalysis B: Environmental, 87(3–4): 105–145
Okamot K I, Yamamoto Y, Tanaka H, Itaya A (1985). Kinetics of heterogeneous photocatalytic decomposition of phenol over anatase TiO2 powder. Bulletin of the Chemical Society of Japan, 58(7): 2023–2028
Pekakis P A, Xekoukoulotakis N P, Mantzavinos D (2006). Treatment of textile dyehouse wastewater by TiO2 photocatalysis. Water Research, 40(6): 1276–1286
Shang J, Zhao F W, Zhu T, Wang Q, Song H, Zhang Y H (2010). Electric-agitation-enhanced photodegradation of rhodamine B over planar photoelectrocatalytic devices using a TiO2 nanosized layer. Applied Catalysis B: Environmental, 96(1–2): 185–189
Sun M, Liu Y, Xiang W, Zhai L F (2015). Electricity-induced catalytic oxidation of RhB by O2 at a graphite anode. Electrochimica Acta, 158: 314–320
Wei T, Li X, Zhao Q, Zhao J, Zhang D (2012). In situ capture of active species and oxidation mechanism of RhB and MB dyes over sunlight-driven Ag/Ag3PO4 plasmonic nanocatalyst. Applied Catalysis B: Environmental, 125(3): 538–545
Acknowledgements
The authors are grateful to the financial support provided by National Natural Science Foundation of China (Grant Nos. 21577003, 21277004 and 41421064), the National Key Research and Development Program of China (No. 2016YFC0202200), and Beijing Natural Science Foundation (No. 8132035).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Wang, X., Feng, X. & Shang, J. Efficient photoelectrochemical oxidation of rhodamine B on metal electrodes without photocatalyst or supporting electrolyte. Front. Environ. Sci. Eng. 12, 11 (2018). https://doi.org/10.1007/s11783-018-1061-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11783-018-1061-8