Abstract
Purpose
Shoulder dysfunction is one of the most bothersome questions for breast cancer survivors. Studies show that mirror therapy can improve shoulder function in patients with a limited shoulder range of motion and shoulder pain. Here, this article reports the results of a randomized controlled trial investigating the effects of the mirror therapy on shoulder function in patients with breast cancer following surgical treatments.
Methods
Totally, 79 participants were divided to two groups receiving active range-of-motion upper limb exercise based on the mirror therapy or active range-of-motion upper limb exercise respectively for 8 weeks. Shoulder range of motion, Constant-Murley Score, Disabilities of Arm, Shoulder, and Hand Questionnaire, Tampa Scale of Kinesiophobia, Visual analog scale, and grip strength were measured at baseline (T0), 2 weeks (T1), 4 weeks (T2), and 8 weeks (T3). The effects of the intervention on shoulder function were analyzed in generalized estimation equation, from group, time, and the interactions between group and time based on the data from participants who completed at least one post-baseline observation
Results
At least one post-baseline observation was performed by 69 participants (n=34 mirror group, n=35 control group). 28(82.35%) participants in the mirror group adhered to the exercise compared to 30(85.71%) in the control group. Generalized estimation equation model showed group had main effects on forward flexion (Waldχ²=6.476, P=0.011), with the Cohen’s d=0.54. The effects of the group on abduction, Constant-Murley Score, and Disabilities of Arm, Shoulder, and Hand Questionnaire were significant when fix the effects of the time. At 8 weeks, participants in the mirror group showed an improvement in abduction compared to the control group (P=0.005), the Cohen’s d was 0.70. At 8 weeks, participants in the mirror group had a higher Constant-Murley Score than control group (P=0.009), with Cohen’s effect size value of d=0.64. The mirror group showed a greater improvement on the Disability of Arm, Shoulder, and Hand Questionnaire than control group at 2 weeks, 4 weeks, and 8 weeks (P≤0.032), but with a weak effect size value of all (r≤0.32). Group had main effects on Tampa Scale of Kinesiophobia (Waldχ²=6.631, P=0.010), with the Cohen’s effect size value of d=0.56.
Conclusions
Mirror therapy improved shoulder flexion, abduction, shoulder function in daily life, and arm function and symptom of the affected shoulder in patients with breast cancer following surgical treatment, while decreasing fear of movement/(re)injury. Mirror configuration needs to be improved in further research to increase its feasibility.
Implications for Cancer Survivors
Breast cancer survivors can try mirror therapy as a practical and effective method in shoulder rehabilitation for a promotion on effects.
Trial registration
ClinicalTrial.gov Identifier: ChiCTR2000033080.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Breast cancer is a globally prevalent malignancy [1]. Following the advancements in the field of oncology, the survival rates for breast cancer patients improved much, with 5-year survival rates of 85%–90% in developed countries and 80%-84% in developing countries [1, 2]. Although surgery is currently the primary therapeutic modality for breast cancer, it still has limitations. Shoulder dysfunction is a common and persistent complication of breast cancer surgery, characterized by a restricted range of motion (ROM), decreased arm strength, pain and numbness in the shoulder, and a decline in function, significantly impacting the physical health and quality of life of breast cancer survivors [3, 4]. Mastectomy and lymph node dissection are surgical procedures that involve removal of significant amounts of soft tissue and pectoral fascia that are instrumental in supporting free shoulder movement due to their extensibility and elasticity. Consequently, muscle excision and myofascial involvement result in a wide area of tissue tightness and gliding restrictions in the neck, upper arm, axilla, chest wall, and abdomen [5], as well as a subsequent reduction in ROM, particularly in abduction and forward flexion, with an average reduction of 41º and 32º, respectively, 33 months after surgery [6]. Even less invasive therapies like sentinel lymph node biopsy (SLNB) have been shown to cause a reduction of more than 10º in abduction and forward flexion in patients two years after the surgery [7]. Moreover, patients treated with breast conserving surgery exhibited a decrease in forward flexion and abduction by 19º and 28º after 9 months, respectively, while their pectoralis minor muscle shrank by 1 cm [8]. Shoulder function deteriorates in patients undergoing radiation therapy, with local pain, edema, and shoulder stiffness [9], thereby increasing the risk of rotator cuff disease and shoulder adhesive capsulitis [10]. Patients with shoulder dysfunction report difficulty returning to work and performing daily activities, as well as emotional issues and a low quality of life [11-13]. Exercise is critical in rehabilitating shoulder function of breast cancer survivors. Typically, in the early postoperative period, exercise involve active or passive movement of fingers and arms, beginning on the first to third postoperative day. The effects of exercise have been well documented in the literatures, including improvements in shoulder ROM and activity of daily living [14, 15].
Despite the potential benefits of early ROM exercise, many patients reported shoulder dysfunction even years after breast cancer surgery [16]. Literatures reported that breast cancer survivors may be hesitant to move their arms due to concerns about surgical complications and postoperative pain [17, 18]. However, systematic reviews have indicated that early exercise did not exacerbate surgical side effects, such pain, seroma, or slower wound healing, when compared to delayed exercise [15]. It appears that the fear of movement/(re)injury among breast cancer survivors is rooted in psychological factors. The fear is characterized as an excessive, irrational, and debilitating fear of physical movement and activity caused by an excessive fear of painful injury or reinjury, with the most severe form of fear known as “kinesiophobia” [19]. Literatures demonstrated that fear of movement/(re)injury is strongly associated with disability, including decreased joint ROM, poor participation in daily activities and task performance, work loss, and other negative outcomes [20-22]. Recent research by Van der Gucht E et.al revealed that kinesiophobia, a related factor that often outweighs pain intensity and pain catastrophizing, is a major contributor to pain-related disability in breast cancer survivors after treatment [23]. Similarly, Gencay Can A et al. discovered that breast cancer survivors with kinesiophobia have higher morbidity of lymphedema, arm function disability, and lower quality of life than patients without kinesiophobia [24]. These findings suggest that various breast cancer sequelae is associated with the fear of movement/(re)injury. Furthermore, the fear of movement/(re)injury often results in patients adopting a posture of bent arm and adducent shoulder, which may aggravate the adaptive shrinkage of chest tissue [10].
Mirror therapy (MT) has been shown in to enhance shoulder function in patients with shoulder problems immediately after the intervention. Louw A et al. found that a 3-min MT improved forward flexion by 14.5° in patients with shoulder pain and limited shoulder ROM [25]. Başkaya MÇ et al. administered mirror-based shoulder exercises to the experimental group, while the control group received the same exercise with a placebo mirror. The experimental group exhibited a 24.1° increase in forward flexion immediately after the intervention [26]. MT is a conventional rehabilitation therapy utilized to treat strokes and phantom pain. The technique involves placing a mirror in front of participants with the reflective side facing the healthy limb. The patients are then prepared for MT by performing functional tasks or exercises with the healthy limb in front of the mirror and the affected limb behind it. While patients look at the image of the healthy limb in the mirror, they imagine that the affected limb can move similarly to the healthy one. A number of studies have found that MT can improve motor function and relieve unilateral pain [27-29]. The visual feedback inherent in MT underlies its effects. The appearance of the healthy limb in the mirror creates the impression that the injured limb is equally healthy, directly activating the affected limb’s proprioception, somatosensory cortex, and motor cortex through this optical illusion, thus restoring the proper sensorimotor closed loop [30-32]. Given the efficacy of MT in improving shoulder and other motor function, it is reasonable to incorporate MT into shoulder rehabilitation after breast surgery procedures. This two-arm prospective randomized controlled trial aims to evaluate the effects of active ROM upper limb exercise based on the MT on ROM, shoulder function of daily living, shoulder pain, handgrip strength, and fear of movement/(re)injury in patients after breast cancer surgery.
Methods
Design, setting, and participants
Between July 2020 and June 2021, a total of 89 participants were recruited from two hospitals in Shanghai and underwent screening before being randomly assigned to receive the active ROM upper limb exercise based on the MT or receive the active ROM upper limb exercise. The 8-week intervention commenced on the first postoperative day, and outcome measurements were taken at the baseline (pre-surgery), 2 weeks, 4 weeks, and 8 weeks. Eligible participants were women aged 18 years or older who had been newly diagnosed with breast cancer and were scheduled to undergo surgical treatment. Breast surgery was limited to modified radical mastectomy (MRM), mastectomy (Mas), or conservation surgery (Con), and axillary surgery was limited to axillary lymph node dissection (ALND) or SLNB. Additionally, participants were required to have the ability to use WeChat, a practical social software in China with phonetic and video function. Patients with bilateral breast cancer, cancer metastasis, impaired upper limbs, physical and mental illnesses, or visual defects were excluded from the study. Patients who underwent immediate breast reconstruction during the trial period were excluded. Dropouts included breast reconstruction, withdrawal, death, and loss of contact.
Ethical statements
The study protocol adheres to the Helsinki Declaration, and has been published [33]. It is registered in the Chinese Clinical Trial Registry (ChiCTR2000033080), and has been approved by the Longhua Hospital Shanghai University of Traditional Chinese Medicine’s Ethical Committee (2020LCSY016). Prior to the intervention, all participants signed an informed consent form.
Procedure
Participants were recruited every Monday and every Thursday from two hospitals. A rater blinded to the study completed the subsequent baseline measurement after the primary researcher introduced the study to eligible participants and obtained informed consent. Participants were then randomly assigned 1:1 to either the mirror group or control group using a computer-generated random sequence. To ensure allocation blindness, allocation information was written on carbonless copy paper and placed in sealed, opaque, sequentially numbered envelopes. After the assignment, both groups received training in active ROM exercises to become acquainted with the intervention, and the mirror group received additional mirror treatment. Treatment began on the first postoperative day. Study nurses monitored both groups via exercise logs, phone calls, and WeChat. Except for the rehabilitation therapist and study nurses, the rater and statistician were blinded to the study and were not permitted to ask participants about group allocation or intervention information.
Intervention
Both the mirror group and control group received active ROM upper limb exercise, with the mirror group undergoing exercise based on the mirror therapy while the control group underwent active ROM upper limb exercise alone. The Chinese Anti-Cancer Association, Committee of Breast Cancer Society, recommends active ROM upper limb exercise as part of the guidelines and specifications for breast cancer diagnosis and treatment (2019 edition). Prior to surgical treatment, training was utilized to familiarize participants in both groups with the intervention. Intervention was administered on the first day following surgery, usually one or two days after admission. Participants received written booklets and videos specifically created for the intervention upon discharge. Furthermore, the research team provided a mirror configuration for the mirror group participants. Participants continued to exercise at home after discharge and were supervised by study nurses every Tuesday or Thursday via WeChat or phone of exercise logs, including time and frequency, as well as adverse events. Those who completed the exercise were regularly rewarded with small items such as a small juicer, vacuum cup, towel, or toothpaste. Biweekly instructions on diet, self-care, and sexual life were also provided to both groups. Participants who reported morning stiffness on their shoulders for more than 30 min could take some stretch exercise or massage at morning to relieve the stiffness. The measurements are aim to protect participants and reduce the damage. During the intervention period, neither group was instructed to do exercises such as arm stretching, yoga, or Pilates, and use of elastic bandages. The intervention in both groups is described in detail below.
Control group
Appendix shows the active ROM upper limb exercise given to participants in the control group.
Mirror group
The mirror group received the active ROM upper limb exercise based on mirror therapy. The active ROM upper limb exercise is the same as the intervention used in the control group. The trial’s standing mirror consisted of a plane mirror, an adjustable bracket, and two wheels, measuring 70 cm in length and 50 cm in width, with the mirror’s height above the ground adjustable up to 1.9 m. (Fig. 1). The mirror group’s program is described below.
-
(1)
Preparation: The MT was performed in a separate, quiet room, with participants required to remove any accessories (watches, bracelets, rings, hairbands, etc.) from both upper limbs before beginning the intervention. Throughout the intervention, participants were free to sit or stand. The standing mirror was placed in front of the participants along their midsagittal plane, with their unaffected limb placed in front of the reflective side, allowing the affected limb to be hidden behind the mirror. Participants could lean slightly forward to view the entire image of the unaffected limb in the mirror (Fig. 2).
-
(2)
Warm-up: Participants were instructed to focus on the image of the unaffected limb in the mirror while performing synchronized upper limb movements for two to three minutes. This procedure was repeated three times. The mirror’s reflection allowed the affected limb to be moved normally over the image, giving the injured arm the appearance of being intact.
-
(3)
Active ROM upper limb exercise: The MT therapist instructed participants later to perform the active ROM upper limb exercise described in Appendix while using MT. Throughout the exercise, participants were instructed to look in the mirror at the image of their unaffected limb and imagine the affected shoulder moving like the healthy one.
Exercise adherence and adverse events
Adverse events, such as wound drainage volume greater than 50 mL 1 week after surgery, delayed wound healing, seroma, skin flap necrosis, and persistent shoulder pain, were monitored during the study. The nurse observed the wound and recorded daily fluid drainage before catheter withdrawal, as well as any adverse events that occurred after the participants entered the study. Participants who reported adverse events have the option to withdraw from the intervention and receive free treatment and evaluation. Participants who attended 85% or more of the planned exercise sessions were considered adherent. The exercise log records were utilized to calculate adherence.
Measures
A trained rater took measurements at the baseline (T0), 2 weeks (T1), 4 weeks (T2), and 8 weeks (T3).
Primary outcome
The main outcome was shoulder ROM on the affected limb including forward flexion, backward flexion, abduction, internal rotation, and external rotation. A 30-cm transparent goniometer usually used in the orthopedics was employed to assess the shoulder ROM with the participants seated. The goniometer is consisted with a degree marked disk and two arms. Participants were asked to remove the clothes on the affected shoulder, then attempt to attain their maximal active range of motion as much as possible at each assessment. When measured forward flexion and backward flexion, the axis of the goniometer was positioned over the acromion process, with the stationary arm of the device was set paralleled with the midaxillary line, and the moving arm was set to the central line of the upper arm. External rotation and internal rotation are measured in 90° of abducted position of the upper limb. The axis of the goniometer was set at the olecranon process, the stationary arm was set paralleled with the ground, and the moving arm was set to the ulna. For abduction measurement, the axis of the goniometer was positioned over the acromion process, the stationary arm was set paralleled with the spine, and the moving arm was set to the central line of the upper arm. The measurement protocol for the shoulder joint that follows the recommendations from Lee DK et al. [34].
Second outcome
-
The Constant-Murley Score (CMS), a dependable and effective tool for assessing shoulder function, was employed to assess shoulder function in daily living, including aspects of pain, daily activity ability, active ROM, and abduction strength. This tool allows for a more comprehensive assessment of shoulder function than the primary outcome. A higher score on the CMS denotes improved shoulder function.
-
The Disability of Arm, Shoulder, and Hand Questionnaire (DASH) was also used to assess the function and symptom of the affected arm. This widely used tool evaluates an individual’s arm function in three aspects: everyday activity (21 items), abnormal symptoms (5 items), and implications on social, occupational, sleep, and psychological functioning (4 items). A higher score on the DASH implies a severe problem with arm function and symptom. The questionnaire adapts a 5-point Likert scale in reverse scoring, and the value was determined by adding all of the points, subtracting 30, and dividing by 1.2.
-
The 13-item Tampa Scale of Kinesiophobia (TSK), which has the same reliability and validity as the original version, was used to evaluate patients’ fear of movement and (re)injury. The TSK is frequently used to evaluate the patient’s cognition of pain and attitude toward movement in order to evaluate the patients’ fear of movement/(re)injury [35]. Each item of the TSK can be graded from 1 to 4, with higher scores signifying more severe fear. The classifications of the measure are subclinical (13–22), mild (23–32), moderate (33–42), and severe (43-52) [36].
-
The Visual Analog Scale (VAS) was used to measure shoulder pain. Participants marked a number in a 10-cm straight line, with the range from 0 to 10, where 10 represented the most painful position. Participants were required to rate their level of pain during exercise and rest.
-
An electronic grip strength meter was used to assess handgrip strength. Prior to assessment, the rater adjusted the parameter (age and weight) to match each participant and adjusted the handle to suit the participants’ hand size. Participants were instructed to sit naturally before gently pulling the handle with their affected limb and exerting maximum effort. After each participant was tested twice, the highest number was selected as the final record. The participants were given a two-minute break between tests.
Statistical analysis
The GLIMMPSE program was utilized for the calculation of repeated measurement. ROM served as the study’s major endpoint and the primary effect indicator for estimating sample size in this randomized controlled experiment. The sample size was determined to be 58 after selecting a test power of 0.95 and a type 1 error rate of 0.05, with Başkaya’s findings on shoulder flexion, which had a Cohen’s effect size value of 1.3799, cited for reference [26]. Accounting for a 20% dropout rate, the final sample size was 70, with 79 cases in the study’s real samples.
Demographic data, baseline comparison, and outcomes between group comparison were reported. Continuous data that were normally distributed were displayed in means and standard deviations. Variables met test of homogeneity, examined in independent two-tailed t-test, otherwise, non-parametric Mann Whitney U-test was used. Non-normally distributed continuous variables were presented as median and interquartile ranges and examined in a non-parametric Mann Whitney U-test. Categorical variables were presented as frequencies (f) and percentages (%), and examined in chi-square test. The SPSS24.0 program was used to process the data, and the statistical significance was adjusted at P value < 0.05. All statistical tests were double-blind. All randomly assigned individuals with at least one post-baseline observation were included in the population for the statistical analysis, known as the modified intention-to-treat (mITT) population. The missing data were filled in using the last observation carried forward.
To evaluate the effects of the MT on shoulder function from group, time, and the interactions between group and time, the generalized estimation equation (GEE) was utilized on the primary and secondary outcomes, using the correlation matrix as an independent structure. To control the influence factors, weight, radiotherapy, dominant hand, type of surgery, and number of lymph nodes removed were used as covariates. Then, the group and time interaction model was tested for each outcome, if no interactions were found, the main effects were reported. When there were interaction effects, the independent two-tailed t-test or non-parametric Mann Whitney U-test were used to examine simple effects of group. Correspondingly, the results were presented as a line chart showing mean and standard error or a box plot. Cohen’s effect size and r were reported for variables analyzed in the parametric and non-parametric tests, respectively, to assess the magnitude of statistical differences.
Results
Recruitment
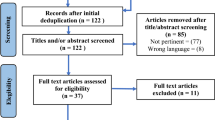
As shown in Fig. 3, a total of 89 eligible patients were screened from new case admissions between July 2020 and June 2021, and 79 patients who signed informed consents were randomly assigned to the mirror therapy group (n = 39) or the control group (n = 40). 69 cases (87.34%) had at least one post-baseline observation, and 64 cases (81.01%) had all measurements completed. Dropout rates were comparable in the mirror (n = 7) and control groups (n = 7), with reasons including breast reconstruction (n = 3), loss of contact (n = 7), withdrawal (n = 3), and death (n = 1). The GEE analyses were performed on 69 participants who provided at least one post-baseline observation. Adverse events were minimal, with only one participant in each group experiencing persistent shoulder pain, but both completing the intervention. Overall, the results suggested that mirror therapy was a safe and feasible intervention for breast cancer patients who underwent surgery.
Sample characteristics
The between group comparison of demographic characteristics were shown in Table 1. The number of cases had their dominant hand affected in the mirror group and control group are 24 and 20, the difference between groups is not significant (Z = 1.065, P = 0.302). MRM, Mas, and Con instances are 30, 4, and 5 in the mirror group, and 32, 2 and 6 in the control group, respectively. In the mirror group, 34 individuals received ALND and 5 received SLNB, while 37 participants in the control group received ALND and 3 received SLNB. In the mirror group, radiation was administered to 12, which were two more cases than control group, but statistically, there was no difference between group (χ2 = 0.327, P = 0.567). With the exception of weight, participants’ demographics were evenly distributed. In comparison to the control group of 59.19 ± 5.63 kg, the weight of the mirror group was 56.32 ± 6.20 kg, the difference was statistically significant (t = 2.009, P = 0.049). Therefore, weight was a covariate in the GEE analysis to control the influence. As shown in Table 2, there were no statistically significant differences between the groups’ baseline comparisons of all outcomes. The within timepoints comparison between group were shown in Table 3.
Shoulder ROM
According to the main effects of the group, the mirror group participants significantly increased their forward flexion compared to the control group, as shown in Table 4 (Waldχ2 = 6.476, P = 0.011), with Cohen’s d of 0.54. The results also demonstrated that time is the factor impacting forward flexion, with an increase at 8 weeks compared to 2 weeks and 4 weeks (P < 0.001). Statistic results showed that backward flexion, external rotation, and internal rotation were not affected by intervention, but rather by time (P < 0.05). Group and time interacted on abduction, according to the GEE model (Waldχ2 = 10.818, P = 0.013). In Fig. 4, abduction degree showed the difference between group at 8 weeks was substantial (P = 0.005), with Cohen’s d of 0.70, showing that the mirror group had significantly increased on abduction at this point compared to the control group.
CMS
According to Table 4, the intervention had no significant effects on pain or strength, but these variables changed significantly with time (P < 0.001). The GEE model revealed that group and time had interactions on participants’ activity of daily living (Waldχ2 = 18.055, P < 0.001). With a P value of 0.001, 0.004, and 0.004 respectively, comparison of CMS daily activity score between group in Fig. 4 revealed that the differences between group at 2, 4, and 8 weeks were significant. The effect size of r were 0.43, 0.34, and 0.35 respectively, indicating that the mirror therapy outperformed the control intervention in terms of daily activity ability. Main effects showed that shoulder ROM changed by time but not group, in addition, time and group had interactions on shoulder ROM (Waldχ2 = 17.32, P < 0.001). Comparison of shoulder ROM between group was significant only at 8 weeks (P = 0.032), with an effect size of r = 0.26 (Fig. 4), indicating that the effects of the mirror therapy on shoulder ROM were mild. Total score was affected by both time and group, and their interactions (Waldχ2 = 9.108, P < 0.028). Comparison of CMS total score between group showed that the difference at 8 weeks was significant (P = 0.009), with Cohen’s d of 0.64 (Fig. 4), indicating that the mirror group improved their shoulder function in daily life more than the control group.
Comparison outcomes that have interaction effects in the Genaralized Estimation Model. CMS The Constant-Murley Score; DASH The Disability of Arm, Shoulder, and Hand Questionnaire. Bigger of Abduction Degree indicates a better performance of abduction. Higher of CMS score indicates a better shoulder function. Lower of DASH score indicates a better function of the affected arm
DASH, TSK, handgrip strength, and VAS
The results of the GEE model in Table 4 showed that both group and time exerted effects on DASH, and these effects were found to interact (Waldχ2 = 10.557, P < 0.014). The comparison of DASH score between group revealed significant differences at 2, 4, and 8 weeks (Fig. 4), with a P value of 0.008, 0.032 and 0.030 respectively. The effect size of r were 0.032, 0.26, and 0.26, respectively, indicating a mild difference between group on DASH. A significant main effect of the group were shown in GEE model of the TSK (Waldχ2 = 6.631, P = 0.010) in Table 4, with Cohen’s d of 0.56. It indicated that the effects of mirror therapy on fear of movement were moderate when compared to the control intervention. GEE model showed that handgrip strength and VAS score were both significantly affected by time (P < 0.001), while group had no effects on either outcomes.
Adherence
In the mirror group, 28 participants completed at least the 85% of planned exercise sessions, while 30 participants in the control group attended at least the 85% of planned exercise sessions. The difference between group is not significant in statistics as showed in Table 5.
Discussion
This study aimed to investigate the effects of an 8-week active ROM upper limb exercise program based on the MT on shoulder function in women with breast cancer after the surgery. The most common side effect of breast cancer survivors is impaired shoulder function. Our trial found that participants in the mirror group exhibited significantly better shoulder flexion and abduction, CMS, and DASH than the control group, indicating that MT has a positive effect on shoulder motor function. Essentially, whatever interventions are conducted, the MT is primarily used as a mirror illusion carrier, where visual feedback from the mirror acts on the brain during bilateral limb exercises. Thus, the MT is more of a method than an intervention. Though MT is commonly used in patients with strokes and hand dysfunction, the exercise protocol is not yet stipulated, and the type of exercise patients perform depends on their disease and symptoms. Exercise types can be classified as movements that include object manipulation and movements that do not include object manipulation [37]. In this study, participants were treated with active ROM exercise, which is recommended by the Chinese Anti-Cancer Association, Committee of Breast Cancer Society as the exercise protocol.
In this study, only a piece of mirror was added in the mirror group to the same exercise in both groups, which resulted in an improvement in shoulder forward flexion and abduction, shoulder function in daily living, and function and symptom of the affected arm. This study’s findings are consistent with previous studies of Louw Aet al. and Başkaya MÇ et al. [25, 26], which demonstrated that MT can improve shoulder ROM. Furthermore, when compared to their findings, which only looked at the effects of a single intervention, our study found that a continuous MT intervention (8 weeks) had an effect on shoulder forward flexion and abduction. Compared to forward flexion and abduction, internal rotation, external rotation and backward flexion are less affected and improved in the study. Mirror configuration used in the study is a plane standing mirror, it probably affects the visual feedback, because the position of the shoulder image in the mirror and unaffected shoulder not overlap in those shoulder movement. Symmetrical motion is the best exercise mode for mirror therapy, but also limits the use of the mirror therapy in some body parts like facial Palsy. For the problems, Barth JM et.al adapted the mirror box as a bi-fold mirror that is opened to a right angle when use it [38]. The mirror twice reflect the unaffected half of a patient’s face, such that the patient sees a full, unaffected face. There also computer vision technology-based mirroring system is developed in studies to provide an immersive visual experience [39]. Therefore, folding mirror can be attempted in further study. Although the handgrip strength was a secondary outcome in the study, little is known about the improvement of ROM exercise alone on the strength. This study found that active ROM exercise based on the MT have not affected the handgrip strength. Aerobic exercise, which includes ROM exercise, has not been shown to significantly enhance handgrip strength in patients who had undergone breast cancer therapy, according to Yang’s meta-analysis [40]. According to McNeely’s meta-analysis [15], some trials that combined ROM training with resistance exercise revealed an increase in upper extremity strength. However, in a systematic review, Ribeiro reported that ROM exercises coupled with muscle training had no benefits on hand or shoulder strength [41]. Therefore, additional randomized controlled trials involving many training types are required to investigate the effects of ROM on strength. In short, this study found that mirror therapy can improve the shoulder joint motor function in patients undergone breast cancer surgeries at least.
The mechanism underlying the improvement of shoulder function through MT remains unclear. Some studies suggest that mirror effects may be related to cortical reorganization of the brain and pain modulation [42]. Conventional studies have shown that MT restores the congruence between motor output and sensory output in patients with unilateral pain, as well as the involvement of multisensory, visuo-proprioceptive integration [32]. As many patients with shoulder disease experience shoulder pain, Roustaee S et al. and Xie Na et al. have reported the improvement of the MT on shoulder pain in breast cancer survivors [43, 44], and Louw proposed that MT can be used to improve shoulder ROM, particularly in patients with severe pain [25]. However, in this study, there was no statistically significant difference between group in terms of shoulder pain. Shoulder pain in breast cancer survivors can be classified as musculoskeletal nociceptive pain, radio-induced and drug-induced neuropathic pain, or chronic persistent pain [45]. Shoulder pain has a direct impact on shoulder function. In this study, since participants received early exercise, few participants had musculoskeletal nociceptive type pain, only one participant in the mirror group reported moderate to severe surgery-induced neuropathic type pain and one in the control group reported moderate radio-induced type pain, while others did not complain about shoulder pain. Therefore, this study was not yet able to demonstrate the connection between the improvements on shoulder function and shoulder pain in breast cancer survivors. Mirror effects, regardless the improvement on pain or motor function, are at the heart of mirror therapy. Studies on healthy people have shown that the perceived position and judgments of kinesthetic states of their hand hidden behind a mirror can be influenced by the hand seen in the mirror [46], indicating that visual information has a dominant effect on proprioception. Solima proposed that mirror illusion can primarily affect estimates of our body’s desired and predicted states [47]. As a result, it appears that the mirrored image has an effect on motor perception of the limb behind the mirror. According to this viewpoint, when participants perform ROM exercises based on the MT, their attention, action monitoring, motor imagery, or something similar that is involved in the motor process strengthens, potentially resulting in improvements in shoulder ROM and daily function.
The TSK has been a valuable tool for assessing fear of movement (re)injury, which is defined as a fear of movement and physical activity that is (wrongly) assumed to cause reinjury. Although limited shoulder movement and arm load are appropriate for patients undergoing surgical treatments to reduce the potential risks of surgical complications, some patients avoid exercise and daily activity due to their incorrect belief that exercise will cause pain and injury. Studies have reported that fear of movement and pain is one of the barriers to full utilization of breast cancer survivors’ rehabilitation exercise, but relevant data is limited and quantitative results are lacking. Despite the absence of investigation of impact of fear of movement/(re)injury on shoulder ROM and daily function, our findings revealed that the mean TSK scores of participants in both groups showed mild fear of movement/(re)injury, indicating that breast cancer survivors are overly concerned about (re)injury within 8 weeks after the surgery. Furthermore, compared to control intervention, the MT can moderately improve the fear of movement. Fear of movement/(re)injury is usually considered as an excessive, irrational, and debilitating fear, and psychological interventions and cognitive behavioral education are effective treatments [21]. The treatments make patients believe pain is self-managed and gradually put themselves into situations and specific activity they identified as “dangerous” or “threatening” by cognition education and graded exposure, achieving desensitization to fear. Graded motor imagery, a three-stage treatment approach that combines left/right judgment, motor imagery, and MT, has been reported to improve fear of movement/(re)injury, despite not being the primary outcome in the studies. Birinci T et al. used graded motor imagery in patients with post-traumatic elbow stiffness, and reported an improvement on elbow ROM and TSK [48]. Gurudut P et al. reported that graded motor imagery added to the conventional physiotherapy was superior to conventional physiotherapy alone in terms of kinesiophobia, and improving shoulder function in patients with frozen shoulder [49]. Two studies reported that graded motor imagery can improve shoulder ROM, shoulder function, pain, and fear of movement in patients with shoulder disease. Graded motor imagery requires imagined movement of the affected arm, whereas MT provides a mirrored illusion of normal movement. Similar to cognition education and graded exposure in the therapy of fear of movement/(re)injury, the two key processes can stimulate the primary motor regions and give patients a genuine sensation of movement. Our findings, along with Louw’s, suggest that MT, a single step in the graded motor imagery sequence, can reduce patients’ anxiety of movement or (re)injury, at least with shoulder problems.
Despite some studies suggest that MT improves adherence to upper limb exercise, this study revealed no difference in adherence between the two groups. Geller D et al. reported that adherence in home-based unimanual mirror therapy is as good as in traditional occupational therapy home-based programs in stroke patients [50]. We hypothesized that the high adherence in those studies was related to the high feasibility of MT in hand training. In this trial, since the mirror equipment limited the participants to see their shoulders in the mirror, which required them to adjust their positions and. We speculate that this process would make patients feel uncomfortable and inconvenient, affecting their experience and adherence to MT.
Limitations
This study has the potential to be further promoted in two aspects. Firstly, fear of movement/(re)injury affects motor function, and since shoulder ROM and fear of movement/(re)injury both improved in this study, fear of movement is possibly a mediating effect between shoulder ROM and MT. But we did not analyze the mediating effect of fear of movement/(re)injury. Secondly, making fear of movement/(re)injury an inclusion criteria may provide a more accurate effect of MT on fear of movement/(re)injury. However, in this study fear of movement/(re)injury occurred after the surgery, while recruitment was completed before surgery, making it infeasible to list the fear of movement/(re)injury as an inclusion criteria. We hope the two limitations can be improved in the further study.
Conclusion
The findings of this study demonstrated that 8 weeks active ROM upper limb exercise based on the MT can improve the shoulder flexion, shoulder abduction, activity ability in daily living, function and symptoms of the affected arm in patients with breast cancer following surgical treatment, while also relieving the fear of movement/(re)injury. However, since few participants reported shoulder pain in the study, the effects of the MT on pain remain unknown. This study provided a novel approach to both MT and breast cancer rehabilitation, demonstrating that MT can be a practical and feasible method in shoulder rehabilitation with a promotion effect. However, due to the limitations of study design, one single experiment cannot draw a definitive conclusion. In addition, since the MT has complex mechanisms, advanced studies are needed.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–49. https://doi.org/10.3322/caac.21660.
Allemani C, Matsuda T, Di Carlo V, Harewood R, Matz M, Nikšić M, et al. Global surveillance of trends in cancer survival 2000–14 (CONCORD-3): analysis of individual records for 37513025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries. Lancet. 2018;391(10125):1023–75. https://doi.org/10.1016/S0140-6736(17)33326-3.
Al-Hilli Z, Wilkerson A. Breast surgery: management of postoperative complications following operations for breast cancer. Surg Clin North Am. 2021;101(5):845–63. https://doi.org/10.1016/j.suc.2021.06.014.
Leonardis JM, Lulic-Kuryllo T, Lipps DB. The impact of local therapies for breast cancer on shoulder muscle health and function. Crit Rev Oncol Hematol. 2022;177:103759. https://doi.org/10.1016/j.critrevonc.2022.103759.
Fourie WJ. Considering wider myofascial involvement as a possible contributor to upper extremity dysfunction following treatment for primary breast cancer. J Bodyw Mov Ther. 2008;4:349–55. https://doi.org/10.1016/j.jbmt.2008.04.043.
Pillai US, Kayal S, Cyriac S, Nisha Y, Dharanipragada K, Kamalanathan SK, Halanaik D, Kumar N, Madasamy P, Muniswamy DK, Dubashi B. Late effects of breast cancer treatment and outcome after corrective interventions. Asian Pac J Cancer Prev. 2019;20(9):2673–9. https://doi.org/10.31557/APJCP.2019.20.9.2673.
Verbelen H, Gebruers N, Eeckhout FM, Verlinden K, Tjalma W. Shoulder and arm morbidity in sentinel node-negative breast cancer patients: a systematic review. Breast Cancer Res Treat. 2014Feb;144(1):21–31. https://doi.org/10.1007/s10549-014-2846-5.
Lee CH, Chung SY, Kim WY, Yang SN. Effect of breast cancer surgery on chest tightness and upper limb dysfunction. Medicine (Baltimore). 2019;98(19):e15524. https://doi.org/10.1097/MD.0000000000015524.
Lipps DB, Leonardis JM, Dess RT, McGinnis GJ, Marsh RB, Strauss JB, Hayman JA, Pierce LJ, Jagsi R. Mssechanical properties of the shoulder and pectoralis major in breast cancer patients undergoing breast-conserving surgery with axillary surgery and radiotherapy. Sci Rep. 2019;9(1):17737. https://doi.org/10.1038/s41598-019-54100-6.
Ebaugh D, Spinelli B, Schmitz KH. Shoulder impairments and their association with symptomatic rotator cuff disease in breast cancer survivors. Med Hypotheses. 2011;77(4):481–7. https://doi.org/10.1016/j.mehy.2011.06.015.
Vella F, Filetti V, Cirrincione L, Rapisarda V, Matera S, Skerjanc A, Cannizzaro E, Vitale E. Work ability after breast cancer: study of healthcare personnel operating in a hospital of South Italy. Int J Environ Res Public Health. 2022;19(17):10835. https://doi.org/10.3390/ijerph191710835.
Akezaki Y, Nakata E, Kikuuchi M, Tominaga R, Kurokawa H, Okamoto M, Hamada M, Aogi K, Ohsumi S, Sugihara S. Investigation of factors affecting early quality of life of patients after breast cancer surgery. Healthcare (Basel). 2021;9(2):213. https://doi.org/10.3390/healthcare9020213.
Galiano-Castillo N, Ariza-García A, Cantarero-Villanueva I, Fernández-Lao C, Díaz-Rodríguez L, Arroyo-Morales M. Depressed mood in breast cancer survivors: associations with physical activity, cancer-related fatigue, quality of life, and fitness level. Eur J Oncol Nurs. 2014;18(2):206–10. https://doi.org/10.1016/j.ejon.2013.10.008.
Ribeiro IL, Moreira RFC, Ferrari AV, Alburquerque-Sendín F, Camargo PR, Salvini TF. Effectiveness of early rehabilitation on range of motion, muscle strength and arm function after breast cancer surgery: a systematic review of randomized controlled trials. Clin Rehabil. 2019;33(12):1876–86. https://doi.org/10.1177/0269215519873026.
McNeely ML, Campbell K, Ospina M, Rowe BH, Dabbs K, Klassen TP, Mackey J, Courneya K. Exercise interventions for upper-limb dysfunction due to breast cancer treatment. Cochrane Database Syst Rev. 2010;(6):CD005211. https://doi.org/10.1002/14651858.CD005211.pub2.
Hauerslev KR, Madsen AH, Overgaard J, Damsgaard TE, Christiansen P. Long-term follow-up on shoulder and arm morbidity in patients treated for early breast cancer. Acta Oncol. 2020;59(7):851–8. https://doi.org/10.1080/0284186X.2020.1745269.
Yeon S, Jeong A, Min J, Byeon J, Yoon YJ, Heo J, Lee C, Kim J, Park S, Kim SI, Jeon JY. Tearing down the barriers to exercise after mastectomy: a qualitative inquiry to facilitate exercise among breast cancer survivors. BMJ Open. 2022;12(7):e055157. https://doi.org/10.1136/bmjopen-2021-055157.
Brunet J, Taran S, Burke S, Sabiston CM. A qualitative exploration of barriers and motivators to physical activity participation in women treated for breast cancer. Disabil Rehabil. 2013;35(24):2038–45. https://doi.org/10.3109/09638288.2013.802378.
Kori SH, Miller RP, Todd DD. Kinesiophobia: a new view of chronic pain behavior. Pain Manag. 1990;3(1):35–43.
Luque-Suarez A, Martinez-Calderon J, Falla D. Role of kinesiophobia on pain, disability and quality of life in people suffering from chronic musculoskeletal pain: a systematic review. Br J Sports Med. 2019;53(9):554–9. https://doi.org/10.1136/bjsports-2017-098673.
Vlaeyen JW, Crombez G. Fear of movement/(re)injury, avoidance and pain disability in chronic low back pain patients. Man Ther. 1999;4(4):187–95. https://doi.org/10.1054/math.1999.0199.
Martinez-Calderon J, Flores-Cortes M, Morales-Asencio JM, Luque-Suarez A. Pain-related fear, pain intensity and function in individuals with chronic musculoskeletal pain: a systematic review and meta-analysis. J Pain. 2019;20(12):1394–415. https://doi.org/10.1016/j.jpain.2019.04.009.
Van der Gucht E, Dams L, Meeus M, Devoogdt N, Beintema A, Penen F, Hoelen W, De Vrieze T, De Groef A. Kinesiophobia contributes to pain-related disability in breast cancer survivors: a cross-sectional study. Support Care Cancer. 2020;28(9):4501–8. https://doi.org/10.1007/s00520-020-05304-4.
Gencay Can A, Can SS, Ekşioğlu E, Çakcı FA. Is kinesiophobia associated with lymphedema, upper extremity function, and psychological morbidity in breast cancer survivors? Turk J Phys Med Rehabil. 2018;65(2):139–46. https://doi.org/10.5606/tftrd.2019.2585.
Louw A, Puentedura EJ, Reese D, Parker P, Miller T, Mintken PE. Immediate effects of mirror therapy in patients with shoulder pain and decreased range of motion. Arch Phys Med Rehabil. 2017;98(10):1941–7. https://doi.org/10.1016/j.apmr.2017.03.031.
Başkaya MÇ, Erçalık C, KarataşKır Ö, Erçalık T, Tuncer T. The efficacy of mirror therapy in patients with adhesive capsulitis: A randomized, prospective, controlled study. J Back Musculoskelet Rehabil. 2018;31(6):1177–82. https://doi.org/10.3233/BMR-171050.
Breivik H, Allen SM, Stubhaug A. Mirror-therapy: an important tool in the management of complex regional pain syndrome (CRPS). Scand J Pain. 2013;4(4):190–7. https://doi.org/10.1016/j.sjpain.2013.07.019.
Deconinck FJ, Smorenburg AR, Benham A, Ledebt A, Feltham MG, Savelsbergh GJ. Reflections on mirror therapy: a systematic review of the effect of mirror visual feedback on the brain. Neurorehabil Neural Repair. 2015;29(4):349–61. https://doi.org/10.1177/1545968314546134.
Gandhi DB, Sterba A, Khatter H, Pandian JD. Mirror therapy in stroke rehabilitation: current perspectives. Ther Clin Risk Manag. 2020;7(16):75–85. https://doi.org/10.2147/TCRM.S206883.
Garry MI, Loftus A, Summers JJ. Mirror, mirror on the wall: viewing a mirror reflection of unilateral hand movements facilitates ipsilateral M1 excitability. Exp Brain Res. 2005;163(1):118–22. https://doi.org/10.1007/s00221-005-2226-9.
Fukumura K, Sugawara K, Tanabe S, Ushiba J, Tomita Y. Influence of mirror therapy on human motor cortex. Int J Neurosci. 2007;117(7):1039–48. https://doi.org/10.1080/00207450600936841.
Imaizumi S, Asai T, Koyama S. Agency over phantom limb enhanced by short-term mirror therapy. Front Hum Neurosci. 2017;4(11):483. https://doi.org/10.3389/fnhum.2017.00483.
Yuan RZ, Li KP, Wei XL, Zheng W, Ye Y, Wang MY, Jiang JT, Wu CQ. Effects of free range-of-motion upper limb exercise based on mirror therapy on shoulder function in patients after breast cancer surgery: study protocol for a randomized controlled trial. Trials. 2021;22(1):815. https://doi.org/10.1186/s13063-021-05789-2.
Lee DK, Kim EK. Effect of pain scrambler therapy on shoulder joint pain and range of motion in patients who had undergone arthroscopic rotator cuff repair for the first time. J Phys Ther Sci. 2016;28(7):2175–7. https://doi.org/10.1589/jpts.28.2175.
Lundberg M, Styf J, Jansson B. On what patients does the Tampa Scale for Kinesiophobia fit? Physiother Theory Pract. 2009;25(7):495–506. https://doi.org/10.3109/09593980802662160.
Neblett R, Hartzell MM, Mayer TG, Bradford EM, Gatchel RJ. Establishing clinically meaningful severity levels for the Tampa Scale for Kinesiophobia (TSK-13). Eur J Pain. 2016;20(5):701–10. https://doi.org/10.1002/ejp.795.
Morkisch N, Thieme H, Dohle C. How to perform mirror therapy after stroke? Evidence from a meta-analysis. Restor Neurol Neurosci. 2019;37(5):421–35. https://doi.org/10.3233/RNN-190935.
Barth JM, Stezar GL, Acierno GC, Kim TJ, Reilly MJ. Mirror book therapy for the treatment of idiopathic facial palsy. Ear Nose Throat J. 2020;93(9):E11–5. https://doi.org/10.1177/0145561320913211.
Sip P, Kozłowska M, Czysz D, Daroszewski P, Lisiński P. Perspectives of motor functional upper extremity recovery with the use of immersive virtual reality in stroke patients. Sensors (Basel). 2023;23(2):712. https://doi.org/10.3390/s23020712.
Yang Y, Gu D, Qian Y, Wang H, Chai X. Effectiveness of aerobic exercise on upper limb function following breast cancer treatment: a systematic review and meta-analysis. Ann Palliat Med. 2021;10(3):3396–403. https://doi.org/10.21037/apm-20-2616.
Ribeiro IL, Moreira RFC, Ferrari AV, Alburquerque-Sendín F, Camargo PR, Salvini TF. Effectiveness of early rehabilitation on range of motion, muscle strength and arm function after breast cancer surgery: a systematic review of randomized controlled trials. Clin Rehabil. 2019;33(12):1876–86. https://doi.org/10.1177/0269215519873026.
Mertens MG, Meeus M, Verborgt O, Vermeulen EHM, Schuitemaker R, Hekman KMC, van der Burg DH, Struyf F. An overview of effective and potential new conservative interventions in patients with frozen shoulder. Rheumatol Int. 2022;42(6):925–36. https://doi.org/10.1007/s00296-021-04979-0.
Roustaee S, RoudiRashtabadi O, Tirgari B, Jahani Y, Tahmasebi S. Mirror therapy effect on shoulder pain and disability and quality of life of mastectomy women: a randomized clinical trial. Disabil Rehabil. 2022;25:1–9. https://doi.org/10.1080/09638288.2022.2148296.
Na X, Xiang-mei R, Dan CH, He-wei W, Jie J. Effect of mirror therapy on persistent pain after breast cancer surgery. Chin J Rehabil Theory Pract. 2018;24(2):134–7. https://doi.org/10.3969/j.issn.1006-9771.2018.02.003.
Giacalone A, Alessandria P, Ruberti E. The physiotherapy intervention for shoulder pain in patients treated for breast cancer: systematic review. Cureus. 2019;11(12):e6416. https://doi.org/10.7759/cureus.6416.
Holmes NP, Crozier G, Spence C. When mirrors lie: “visual capture” of arm position impairs reaching performance. Cogn Affect Behav Neurosci. 2004;4(2):193–200. https://doi.org/10.3758/cabn.4.2.193.
Soliman TM, Buxbaum LJ, Jax SA. The mirror illusion’s effects on body state estimation. Cogn Neuropsychol. 2016;33(1–2):102–11. https://doi.org/10.1080/02643294.2016.1187591.
Birinci T, Kaya Mutlu E, Altun S. The efficacy of graded motor imagery in post-traumatic stiffness of elbow: a randomized controlled trial. J Shoulder Elbow Surg. 2022;31(10):2147–56. https://doi.org/10.1016/j.jse.2022.05.031.
Gurudut P, Godse AN. Effectiveness of graded motor imagery in subjects with frozen shoulder: a pilot randomized controlled trial. Korean J Pain. 2022;35(2):152–9. https://doi.org/10.3344/kjp.2022.35.2.152.
Geller D, Nilsen DM, Quinn L, Van Lew S, Bayona C, Gillen G. Home mirror therapy: a randomized controlled pilot study comparing unimanual and bimanual mirror therapy for improved arm and hand function post-stroke. Disabil Rehabil. 2022;44(22):6766–74. https://doi.org/10.1080/09638288.2021.1973121.
Acknowledgements
The authors would like to acknowledge all participants for their participation and nurses assisting with intervention.
Funding
This work was supported by the 2020 Subject Competence Enhancement Project of the School of Nursing, Shanghai University of Traditional Chinese medicine (NO. 2020HLXK06). Shanghai Nursing Association research project (NO. 2022MS-B04). The funding body is not a participant in any process including the study design, data collection, data analysis, or data interpretation of the study.
Author information
Authors and Affiliations
Contributions
R-Z Y was responsible for interpretation of the data and writing of the manuscript. X-L W and Y-Y contributed to the conduction of the study and data collection. M-Y W, J-T J, and K-P L were all involved in the material preparation, and contacts of the participants. C-Q W was responsible for the design and conception of the study, and revised the manuscript. Z. W was responsible for the questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Ethics approval was completed by Z. W. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
All procedures performed in studies were in accordance with the ethical standards of the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. This study had been approved by the Ethical Committee of the Longhua hospital affiliated with the Shanghai University of traditional Chinese medicine on 5 May 2020 (approval NO. 2020LCSY016).
Consent to participate
Written informed consent was obtained from each participant in this study.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Wei Zhu and Wei Zheng are the joint corresponding authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yuan, R., Wei, X., Ye, Y. et al. The effects of the mirror therapy on shoulder function in patients with breast cancer following surgery: a randomized controlled trial. J Cancer Surviv (2023). https://doi.org/10.1007/s11764-023-01398-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11764-023-01398-x