Abstract
The diversity of bottom substrates is a primary driver of taxonomic richness and species abundance patterns of freshwater benthic insects in space and time. Here, we examine the influence of substrate composition on the seasonal patterns of benthic insect communities in streams of arid regions. Benthic insects were sampled monthly over a year at three sampling sites distributed along the Bouilef stream within the Belezma biosphere reserve (Algeria). Different substrate types (sand, gravel, pebbles, boulders, and emergent macrophytes) were sampled in the rainy and dry seasons. During the two sampling seasons, a total of 8599 insects belonging to six orders, 26 families, and 39 genera/species were sampled and identified. Student’s t-test analysis showed that season influences significantly the mean abundance of benthic insects, which increases in the rainy season. However, the season does not affect taxa richness. Substrate types influence taxa composition and variation of benthic insect communities. The results showed that pebbles and boulders are the most populated by gathering and filtering collectors in terms of taxa abundance. Emergent macrophytes are more favorable for herbivores shredders. Principal Coordinates Analysis ‘’PCoA’’ also showed that pebbles and boulders differed significantly from other substrates, whereas sand and gravel substrates exhibited similar taxa mean abundance. Such assessments can help propose conservation measures needed to successfully safeguard these fragile ecosystems.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Owing to their high diversity and variable tolerance to habitat variation, benthic macroinvertebrates are widely used for the biomonitoring of freshwaters across ecosystem types (e.g., lakes and rivers) and bioclimatic regions (Moisan and Pelletier 2013; Laini et al. 2022). Several physicochemical and biological factors drive the composition and variability of benthic invertebrate communities, either independently or combined (Benzina and Si Bachir 2018; Benzina et al. 2021; Buffagni 2020). Among those important physical factors, the nature and diversity of the bottom substrate are of utmost importance in shaping a benthic community. The characteristics of benthic substrates can vary across geographical and bioclimatic regions, and according to stream size and location within a drainage basin. Both local and regional settings can therefore affect the relationships between substrates and the seasonal dynamics of benthic communities. In most of the Mediterranean region, the substrate composition is closely related to the orography of the area crossed by streams, and to seasonal events of flooding and drying over an annual cycle (Gasith and Resh 1999). Precipitation regimes also have a major influence on aquatic ecosystems (Dodds et al. 2019; Carey et al. 2021), because they directly influence substrate size through river competence and the flow seasonality that determines the variation and composition of benthic insects (Patrick et al. 2019; Crabot et al. 2021; Carvallo et al. 2022). We know for instance that some species prefer large rocky substrates; others are typically found under pebbles or borrowed in sandy areas, while siltation tends to clog up the bottom and eliminate species (Burd et al. 2008). Some studies even provided evidence for ontogenetic habitat shifts in benthic invertebrates (Hanquet et al. 2004), thus highlighting the underlying role of substrate diversity in community seasonality. Yet, most of our current knowledge of species-substrate relationships in freshwaters has come from studies of temperate and tropical streams.
Arid-land streams of North Africa show much higher hydrological variability than their temperate and tropical counterparts (Dakki 1986, 1987; Gasith and Resh 1999; Arab et al. 2004; Benzina et al. 2019). Although the diversity of benthic macroinvertebrates communities decreases in arid regions, as they are submitted to highly selective environments. In addition, in such arid regions, stream substrate tends to be finer, and enriched of sediments such as sand, silt, and clay. Soil particles can be more compact due to aridity and low vegetation cover, which can affect permeability and water retention in the substrate (Naorem et al. 2022). This seasonal contrast is due to the fact that the seasonal patterns in these regions can vary with generally two seasons. The dry season sometimes extends up to 9 months with punctual periods of floods, which often results in increased erosion of shorelines and surrounding land. This can lead to greater sediment transport in streams, altering their substrate and morphology (Mueller and Pitlick 2013). The rainy season is generally short and characterized by low and recurrent rainfall that leads to increases in the diversity and richness of benthic macroinvertebrate communities (Tonkin et al. 2017; Patrick et al. 2019; Carvallo et al. 2022). These variations affect the structure and distribution patterns of these benthic macroinvertebrates’ communities (Benzina 2019).
The study of benthic macroinvertebrates and their relationship to the substrate in arid regions can contribute to our understanding of the ecology of arid rivers and the assessment of water quality in these fragile ecosystems (Qian et al. 2021; Webster et al. 2022). It is, however, difficult to isolate substrate and seasonal effects on benthic macroinvertebrate communities from other factors such as water chemistry, temperature, or human-induced degradation of the habitats. Here we investigate how do substrate types affect the seasonal composition and functional feeding groups variation of benthic insects in the Bouilef stream located in an arid region (northeastern Algeria).
Materials and methods
Study region and sites selection
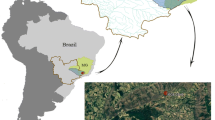
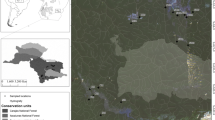
The study sites are located in the Bouilef stream within the Belezma biosphere reserve, which ranges from 35°32′40” and 35°37′46′′ N, to 5°55′10” and 6°10′45′′ E. This protected area is part of a mountain range located in the semi-arid part of northeastern Algeria, near Batna city. The climate of the study region is influenced by its location and suffers a long period of summer drought. The climate of the reserve is globally semi-arid with cold winters and dry-hot summers at low elevations (900–1200 m a.s.l) (Boukerker and Si Bachir 2015). Over the last 10 years, mean temperatures (± SD) were 17.46 ± 0.27 ◦C, with maxima of 25.86 ± 0.49 ◦C and minima of 8.94 ± 0.38 ◦C at an elevation of 1052 m a.s.l. Annual precipitations varied between 160.8 and 362.2 mm with a mean value of 310.2 ± 74.7 mm (Fig. 1). Nevertheless, in recent years, the reserve has undergone significant changes in climate and land use causing, among other adverse effects, the decline of Atlas Cedar Cedrus atlantica, an endemic tree species of North Africa (Cherak et al. 2021).
The Gaussen ombrothermic diagram for the study period (from September 2021 to August 2022), indicated the presence of a rainy season, spanning from February to April, and a dry season encompassing the remaining months. Precipitations and air temperature in the rainy season and dry season were 115.4 and 105.3 mm and 10.6 and.
18.2 ◦C, respectively. This rainy season is characterized by hydrological stability, with no occurrences of flooding before or during the insect sampling. While the dry season is marked by occasional moments of flooding.
The Bouilef stream was selected for its permanent flow compared to other streams of the protected area. It belongs to the hydrographic basin of the Constantine high plateaus (Watershed No. 07, area: 9,578 km2), and stretches over a length of 2.5 km with an average slope of 10% (ANRH, 2001). The forested stream is characterized by the presence of dense riparian vegetation on both river banks, composed mainly of trees of Atlas cedar (Cedrus atlantica), Aleppo pine (Pinus halepensis), Holm oak (Quercus ilex), elm leaf blackberry (Rubus ulmifolius), mastic (Pistacia lentiscus) and herbaceous vegetation. The presence of this riparian vegetation still implies higher shading and allochthonous material input into the stream (Boukerker and Si Bachir 2015).
Over the study period, three sites A, B, and C very close to each other (140 m between A and B and 180 m between B and C), and belonged to the same Bouilef stream (A: 35°37’03.0"N, 6°11’13.9"E, B: 35°37’01.0"N, 6°11’18.0"E, C: 35°36’55.8"N, 6°11’14.6"E) were sampled monthly. Each site (5 m2 of surface area approximately) is located at elevations of 1065–1078 m (a.s.l) and was selected based on the existence of different substrate types, including sand (< 2 mm), gravel (2–16 mm), pebbles (16–64 mm), boulders (64–256 mm), and emergent macrophytes (Tachet et al. 2010).
Physicochemical parameters of water
At the field (sites), seven parameters were measured: pH, temperature (T °C), total dissolved solids (TDS ppm), and electrical conductivity (EC, µS cm− 1), were measured using a multi-parameter waterproof meter HI991300® (HANNA instruments). Turbidity (NTU: Nephrometric Turbidity Unit) was measured with a turbidimeter (HACH instruments), and water depth (cm) was measured using a graduated wooden board. Additionally, water flow velocity is measured by a flow scale: (1) very slow speed; (2) slow speed; (3) average speed, and (4) fast speed. In the laboratory, three parameters were analyzed: Salinity (mg L− 1), dissolved organic matter (DOM mg L− 1), and dissolved oxygen (DO mg L− 1) from 500 ml samples taken on each sampling occasion, using the standardized water quality monitoring protocols (AFNOR 2005).
Benthic insect sampling
At the field (sites), sampling of macroinvertebrates is carried out on the five types of substrates at each site using a Surber net with a 25 cm aperture and 500 μm mesh size. The percentage coverage of the different substrates was visually assessed (Touron-Poncet et al. 2014). We collected samples from the sand, gravel, and emergent macrophyte substrates by scraping them onto the Surber net surface. The same surface area was used to sample pebbles and boulders. All collected specimens were manually separated from the substrates and placed in a 4% formaldehyde solution for preservation. In the laboratory, the benthic insects were sorted, counted, and identified to the lowest possible taxonomic level (usually genus) using appropriate keys (Tachet et al. 2010; Thorp and Rogers 2016; Ochieng et al. 2019). Finally, the specimens were preserved in 70% ethanol.
Data analysis
The benthic insects were assigned to five functional feeding groups (FFGs): gathering collectors (Gc), filtering collectors (Fc), herbivores shredders (Hs), scrapers (Sc), and predators (Pr). The FFGs assignment was at the family level according to Ramírez et al. (2014) and Cummins (2016).
The mean abundance was determined by dividing the total number of individuals by the number of surveys conducted, whether it be for seasons or substrate types. The mean taxa richness was calculated by dividing the total number of taxa by the number of surveys conducted, whether it be for seasons or substrate types.
The values of physicochemical water parameters and the abundance (Taxa and FFGs) were logx + 1 transformed. Student t-tests were performed to assess differences in the mean richness and abundance of benthic insects by season, and substrate type. T-tests were also used to assess differences in physicochemical parameters. To evaluate the seasonal variation of mean taxa richness and mean abundance of FFGs according to substrate types, a one-way non-parametric ANOVA followed by Kruskal Wallis posthoc tests were performed (p ˂ 0.05). The distribution of substrates according to their abundance of FFGs was summarized using Principal Coordinates Analysis (PCoA) with Bray-Curtis distance (abundance transformed in log (x + 1)). Substrates lacking benthic insects were removed from the PCoA analysis. The analyses were performed with SPSS version 20 and PAST 4.03 software.
Results
Physicochemical parameters of water in the dry and rainy seasons
The physicochemical parameters of water showed only small variations during the rainy season and the dry seasons, and there was no significant difference between the two seasons, except flow velocity (Table 1).
Comparison between seasonal richness and abundances of benthic communities
Overall, we collected 8599 specimens belonging to six orders, 26 families, and 39 genera/species during the two studied seasons. Coleoptera was the most diversified order with 15 taxa (Table 2). Trichoptera was the most abundant taxa with 4159 individuals, followed by Ephemeroptera, Diptera, and Coleoptera with 3009, 935, and 391 individuals, respectively. Heteroptera and Odonata were only found with 95 and 10 individuals, respectively. The most abundant families were: Sericostomatidae (2148 individuals), Hydropsychidae (2004), Baetidae (1921), and Caenidae (1053). In terms of individual numbers, the dominant FFGs were: gathering collectors with (3371 individuals), followed by filtering collectors (2943); herbivore shredders (1753); predators (102), and scrapers (150).
The student’s t-test showed a significant difference in the mean abundance of benthic insects between seasons. Benthic insect abundance was higher in the rainy season (p = 0.001) than in the dry season. However, the mean richness did not differ significantly between seasons (p = 0.22) (Fig. 2).
Comparison between seasonal richness and abundances of benthic communities according to substrate types
Our analyses showed significant differences in the mean abundance of benthic insects by substrate between the dry and rainy seasons. In the rainy season, pebbles (p = 0.001) and boulders (p = 0.03) hosted significantly more benthic insects than during the dry season (Fig. 3). There were no significant differences in the mean abundance of insects between seasons for the other substrates. There were no significant differences in substrate mean taxa richness of benthic insects between seasons: sand (p = 0.81), gravel (p = 0.85), pebbles (p = 0.41), boulders (p = 0.57), and emergent macrophytes (p = 0.06) (Fig. 3).
Comparison between seasonal richness and abundances of FFGs according to substrate types
During both the rainy and dry seasons, the non-parametric ANOVA revealed significant differences in the mean abundance of gathering collectors, filtering collectors, and herbivore shredders across different substrates. However, there were no differences in mean abundance for scrapers (p = 0.08; p = 0.09) and predators (p = 0.22; p = 0.29), neither in the rainy nor in the dry season.
When conducting pairwise comparisons using Kruskal-Wallis tests (Table 3)., there were no significant variations in the mean rank abundance for gathering collectors and filtering collectors between pebbles and boulders in both seasons (Group A). Similarly, no significant differences were observed between emergent macrophytes, gravel, and sand (Group B).
Nonetheless, it’s important to note that gathering collectors and filtering collectors displayed greater abundance on pebbles and boulders compared to the other substrates. Additionally, in the case of emergent macrophytes (Group A), there was a higher abundance of herbivore shredders compared to other substrates, including pebbles and boulders (Group B), gravel, and sand (Group C) (Table 3).
No significant variations were observed in the mean rank of FFGs taxa richness between substrates during both seasons (Table 3). Pairwise analysis was not conducted for the group of predators and scrapers since no significant differences were identified between substrates.
The results of the PCoA showed a distinct separation between FFG abundances according to substrate types. Substrates ranging from pebbles to boulders, which are of intermediate to large sizes, were situated on the left side of axis 1 and were characterized by a high and homogeneous abundance of benthic insects, particularly for gathering and filtering collectors. On the other hand, substrates ranging from sand to gravel, which are of smaller sizes, were grouped together on the right side of axis 1 and exhibited a low abundance of predators and scrabers. Emergent macrophytes were distributed throughout the ordination and presented intermediate abundance for herbivore shredders (Fig. 4).
Discussion
In the present study, except for the flow velocity, no measured physicochemical parameters were significantly different from each other between seasons. This low contrast may have been caused by the permanent flow of the Bouilef stream and the riparian vegetation which provides good shade for the water, meaning that we could reasonably focus on the importance of substrates to benthic communities while minimizing the potential effect of combining physicochemical factors. The mean taxa richness was not influenced by seasons or substrate types. This means either that most insects have non-seasonal life histories extending over one year or more, or that temporal turnover (multivoltine character) occurs within the identified taxonomic units (genus). On the other hand, the mean abundance of benthic insects was higher in the rainy season, especially on pebbles and boulders. Moreover, the average flow velocity in the rainy season ensures the availability of food resources (more water, hence more fresh organic matter, algae, and detritus available) likely supports higher population densities (Theodoropoulos et al. 2018; Buffagni 2020). Also, these larger substrates (pebbles and boulders) exhibit greater stability when faced with hydrological variations (Van der Meer et al. 2021), so their distribution over river beds is less sensitive to seasonal fluctuations in water flow. Similar observations were reported by Vagenas et al. (2022) in a study carried out in the largest Moroccan river (Oum Errabia) under the same type of climate. These authors found that pebbles and boulders are the most suitable for benthic macroinvertebrates. Theodoropoulos et al. (2018) also found similar results in temperate Mediterranean areas of Greece. In tropical regions, Pereira et al. (2017) found that pebbles are the most attractive substrates for benthic macroinvertebrates compared to sand and gravel (Jones et al. 2012; Schriever et al. 2015; Theodoropoulos et al. 2018; Thomas and Thomas 2022). The low level of mean abundance observed in both seasons on sand and gravel in our study can be explained by the fact that these substrates are more vulnerable to hydrodynamic degradation and are poor in food resources (Fidelis et al. 2008; Baptista et al. 2001).
Furthermore, the water flow during the rainy season removes accumulated fine sediments from the larger substrate such as boulders and pebbles. This creates suitable habitats and opportunities for the settlement of benthic insects, especially for gathering collectors and filtering collectors. These FFG groups aggregate smaller particulate organic matter (allochthonous material input from the riparian zone) deposited on the substrate and filter organic particles directly from the water column respectively. Although autochthonous food sources may be a more important part of the diets of benthic macroinvertebrates especially for gathering collectors, filtering collectors, and shredders in forested streams (Torres-Ruiz et al. 2007; Vannote et al. 1980).
The increased water flow during this period creates also microhabitats such as riffles and pools, expanding the range of niches available for benthic insects to inhabit and exploit (Fenoglio et al. 2020; McCaffrey 2021).
During the dry season, the occurrence of floods erodes the sediments from the streambed, resulting in an increased concentration of fine particles such as sand (< 2 mm) and gravel (2–16 mm) that are easily displaced by the water flow (Holomuzki and Biggs 1999). This sand substrate, when suspended in the water column can, on one hand, affect the filtering collectors by adhering and damaging the filtration structures and decreasing the food amount to be ingested (Vasconcelos and Melo 2008), and, on the other hand, obstruct crevices and interstitial spaces of substrates for attachment and shelter that gathering organisms rely on.
In Bouilef forest stream, the abundance of herbivores-shredders on emergent macrophytes could be attributed to a higher presence of these kinds of substrates which provided a direct food source, allowing them to consume plant tissue and utilize the resources (Waters and San Giovanni 2002; Altieri et al. 2021; Novack et al. 2021). Generally, the low numbers of shredders were partly due to less/loss of riparian vegetation resulting from human activities. Loss of riparian vegetation can cause loss of diversity and changes to structural and functional organization of macroinvertebrates in streams (Allan 2004; Jinggut et al. 2012; Ono et al. 2020).
The low abundance of scrabers and predators recorded in both seasons on sand and gravel may be attributed to the low amount of sunlight that enters the shaded stream throughout can’t provide the proliferation of algae (periphyton), which serves as the primary food source for scrabers (Grubaugh et al. 1996), and probably to the low prey and ambush shelter availability for predators (Cummins 2018). Also, this low abundance for predators may be related to their univoltine or longer generation times according to Huryn et al. 2008.
Consistently, our study validates habitat preferences for benthic insect communities, which are strongly influenced by substrate types and seasons. Distinction of pebbles and boulders from the other substrates, and the strong association between sand and gravel show that substrates are not related to specific sites. This is an important factor in the composition and variation of benthic insects (Hepp et al. 2012).
Our study provides additional proof that substrate type and season play a crucial role in determining the composition and variation of benthic insect communities (Duan et al. 2009), whether the stream is located in an arid, tropical, or temperate region (Barnes et al. 2013). Although our study showed the affinity of benthic insects for substrate types in arid area, these results remain limited to a single stream and a single year of study. In addition, it does not take into account rare species. There is, therefore, a need for further research and efforts to be conducted on a larger spatial and temporal scale. This information is crucial for effective preservation and conservation measures in arid region streams.
Data availability
Data used in the study are available upon request.
References
AFNOR (2005) Qualité De L’eau. Dosage des matières en suspension. Méthode par filtration sur filtre en fibres de verre NF EN 872. AFNOR Report
Allan JD (2004) Landscapes and riverscapes: the influence of land use on stream ecosystems. Annu Rev Ecol Evol Syst 35(1):257–284. https://doi.org/10.1146/annurev.ecolsys.35.120202.110122
Altieri P, Paz LE, Jensen RF, Donadelli J, Rodrigues Capítulo A (2021) Transplanting macrophytes as a rehabilitation technique for lowland streams and their influence on macroinvertebrate assemblages. Acad Bras Cienc 93(3):e20191029. https://doi.org/10.1590/0001-3765202120191029
ANRH (National Agency of Hydraulic Resources) (2001) Inventory of soil resources in Algeria 1963–2001
Arab A, Lek S, Lounaci A, Park YS (2004) Spatial and temporal patterns of benthic invertebrate communities in an intermittent river (North Africa). Ann Limnol Int J Lim 40(4):317–327. https://doi.org/10.1051/limn/2004029
Baptista DF, Buss DF, Dorvillé LFM, Nessimian JL (2001) Diversity and habitat preference of aquatic insects along the longitudinal gradient of the Macaé river basin, Rio De Janeiro. Brazil Rev Brasil Biol 61(2):249–258
Barnes JR, Vaughan IP, Ormerod SJ (2013) Reappraising the effect of habitat structure on river macroinvertebrates. Freshw Biol 58:2154–2167. https://doi.org/10.1111/fwb.12198
Benzina I (2019) Biodiversité des macroinvertébrés benthiques et évaluation multiparamétrique de la qualité des cours d’eau dans la réserve de biosphère du Belezma (région aride du Nord Est Algérien). Dissertation, Oum El Bouaghi University
Benzina I, Si Bachir A (2018) Diversity of benthic macroinvertebrates and water stream quality in the national park of Belezma (Northern-East, Algeria). Int J Health Life Sci 4(1):1–18. https://doi.org/10.20319/lijhls.2018.41.0118
Benzina I, Si Bachir A, Ghazi C, Santoul F, Céréghino R (2019) How altitudinal gradient affects the diversity and composition of benthic insects in arid areas streams of northern. East Algeria? Biologia 75:567–577. https://doi.org/10.2478/s11756-019-00326-8
Benzina I, Si Bachir A, Santoul F, Céréghino R (2021) Macroinvertebrate functional trait responses to environmental gradients and anthropogenic disturbance in arid-land streams of North Africa. J Arid Environ 195:104626. https://doi.org/10.1016/j.jaridenv.2021.104626
Boukerker H, Si Bachir A (2015) Biodiversity of xylophagous insects and their role in the Cedrus Atlantica forests decline in the national park of Belezma, Batna (Algeria). Courrier Du Savoir 20:79–90
Buffagni A (2020) The lentic and lotic characteristics of habitats determine the distribution of benthic macroinvertebrates in Mediterranean rivers. Freshw Biol 66(1):13–34. https://doi.org/10.1111/fwb.13596
Burd BJ, Barnes PAG, Wright CA, Thomson RE (2008) A review of subtidal benthic habitats and invertebrate biota of the Strait of Georgia, British Columbia. Mar Environ Res 66:3–38. https://doi.org/10.1016/j.marenvres.2008.09.004
Carey N, Chester ET, Robson BJ (2021) Flow regime change alters shredder identity but not leaf litter decomposition in headwater streams affected by severe, permanent drying. Freshw Biol 66(9):1813–1830. https://doi.org/10.1111/fwb.13794
Carvallo FR, Strickland BA, Kinard SK, Reese BK, Hogan JD, Patrick CJ (2022) Structure and functional composition of macroinvertebrate communities in coastal plain streams across a precipitation gradient. Freshw Biol 67(10):1725–1738. https://doi.org/10.1111/fwb.13968
Cherak I, Si Bachir A, Cherak L, Ghazi C, Loucif L, Sellami M (2021) Diversity and distribution patterns of endophytic mycoflora of Atlas cedar, Cedrus Atlantica (Endl) G. Manetti ex Carrière, needles in Belezma biosphere reserve (Batna, Algeria). Biodivers J 12(3):573–583. https://doi.org/10.31396/Biodiv.Jour.2021.12.3.573.583
Crabot J, Polášek M, Launay B, Pařil P, Datry T (2021) Drying in newly intermittent rivers leads to higher variability of invertebrate communities. Freshw Biol 66(4):730–744. https://doi.org/10.1111/fwb.13673
Cummins KW (2018) Functional analysis of stream macroinvertebrates. In: Gökçe D (ed) Limnology: Some new aspects of Inland water ecology. IntechOpen, London, pp 64–78. https://doi.org/10.5772/intechopen.79913
Dakki M (1986) Biotypologie et gradient thermique spatiotemporels: Étude sur un cours d’eau du Moyen Atlas (Maroc). Bull Ecol 17(2):79–85
Dakki M (1987) Ecosystèmes d’eau courante du haut Sebou (Moyen Atlas): Études typologiques et analyses écologique et biogéographique des principaux peuplements entomologiques. Trav Inst Sci Rabat Série Zool 42:1–99
Dodds WK, Bruckerhoff L, Batzer D, Schechner A, Pennock C, Renner E, Tromboni F, Bigham K, Grieger S (2019) The freshwater biome gradient framework: Predicting macroscale properties based on latitude, altitude, and precipitation. Ecosphere 10(7):1–33. https://doi.org/10.1002/ecs2.2786
Fenoglio MS, Rossetti MR, Videla M (2020) Negative effects of urbanization on terrestrial arthropod communities: a meta-analysis. Glob Ecol Biogeogr 29(8):1412–1429. https://doi.org/10.1111/geb.13107
Fidelis L, Luiz Nessimian J, Hamada N (2008) Distribuição espacial de insetos aquáticos em igarapés de pequena ordem na Amazônia central spatial distribution of aquatic insects communites in small streams in central Amazonia. Acta Amaz 38(1):127–134. https://doi.org/10.1590/S0044-59672008000100014
Gasith A, Resh VH (1999) Streams in Mediterranean climate regions: abiotic influences and biotic responses to predictable seasonal events. Annu Rev Ecol Evol Syst 30:51–81. http://www.jstor.org/stable/221679
Grubaugh JW, Wallace JB, Houston ES (1996) Longitudinal changes of macroinvertebrate communities along an Appalachian stream continuum. Can J Fish Aquat Sci 53(4):896–909. https://doi.org/10.1139/f95-247
Hanquet D, Legalle M, Garbage S, Céréghino R (2004) Ontogenetic microhabitat shifts in stream invertebrates with different biological traits. Arch Hydrobiol 160(3):329–346. https://doi.org/10.1127/0003-9136/2004/0160-0329
Hepp LU, Landeiro VL, Melo AS (2012) Experimental assessment of the effects of environmental factors and longitudinal position on alpha and Beta diversities of aquatic insects in a neotropical stream. Int Rev Hydrobiol 97(2):157–167. https://doi.org/10.1002/iroh.201111405
Holomuzki JR, Biggs BJF (1999) Distributional responses to flow disturbance by a stream-dwelling snail. Oikos 87:36–47. https://doi.org/10.2307/3546994
Huryn AD, Wallace JB, Anderson NH (2008) Habitat, life history, secondary production, and behavioral adaptations of aquatic insects. An introduction to the aquatic insects of North America, 4th edn. Kendall/Hunt Publishing Company, Dubuque
Jinggut T, Yule CM, Boyero L (2012) Stream ecosystem integrity is impaired by logging and shifting agriculture in a global megadiversity center (Sarawak, Borneo). Sci Total Environ 437:83–90. https://doi.org/10.1016/j.scitotenv.2012.07.062
Jones JI, Murphy JF, Collins AL, Sear DA, Naden PS, Armitage PD (2012) The impact of fine sediment on macro-invertebrates. River Res Appl 28(8):1055–1071. https://doi.org/10.1002/rra.1516
Laini A, Burgazzi G, Chadd R, England J, Tziortzis I, Ventrucci M, Vezza P, Wood PJ, Viaroli P, Guareschi S (2022) Using invertebrate functional traits to improve flow variability assessment within European rivers. Sci Total Environ 832:155047. https://doi.org/10.1016/j.scitotenv.2022.155047
McCaffrey CM (2021) The use of mayflies, stoneflies, and caddisflies as indicators of fine sediment pollution in salmon-bearing streams of the Pacific Northwest. Dissertation, Portland State University https://doi.org/10.15760/etd.7542
Naorem A, Jayaraman S, Dalal RC, Patra A, Rao CS, Lal R (2022) Soil inorganic carbon as a potential sink in carbon storage in dryland soils - a review. Agriculture 12:1256. https://doi.org/10.3390/agriculture12081256
Novack M, Romano L, Nascimento LD, Canterle E, Barboza CN (2021) Benthic invertebrates associated with the aquatic macrophytes in a Brazilian subtropical reservoir. Eng Sanit Ambient 26(4):741–748. https://doi.org/10.1590/S1413-415220190377
Ochieng H, Okot-Okumu J, Odong R (2019) Taxonomic challenges associated with identification guides of benthic macroinvertebrates for biomonitoring freshwater bodies in East Africa: a review. Afr J Aquat Sci 44(2):113–126. https://doi.org/10.2989/16085914.2019.1612319
Ono ER, Manoel PS, Melo ALU, Uieda VS (2020) Effects of riparian vegetation removal on the functional feeding group structure of benthic macroinvertebrate assemblages. Commun Ecol 21:145–157. https://doi.org/10.1007/s42974-020-00014-7
Patrick CJ, McGarvey DJ, Larson JH, Cross WF, Allen DC, Benke AC, Brey T, Huryn AD, Jones J, Murphy CA, Ruffing C, Saffarinia P, Whiles MR, Wallace JB, Woodward G (2019) Precipitation and temperature drive continental-scale patterns in stream invertebrate production. Sci Adv 5(4):eaav2348. https://doi.org/10.1126/sciadv.aav2348
Pereira TDS, Pio JFG, Calor AR, Copatti CE (2017) Can the substrate influence the distribution and composition of benthic macroinvertebrates in streams in northeastern Brazil? Limnologica 63:27–30. https://doi.org/10.1016/j.limno.2016.12.003
Qian J, Tang S, Wang P, Lu B, Li K, Jin W, He X (2021) From source to sink: review and prospects of microplastics in wetland ecosystems. Sci Total Environ 758:143633. https://doi.org/10.1016/j.scitotenv.2020.143633
Ramírez A, Gutiérrez-Fonseca PE, Rio R, Juan S, Rico P (2014) Functional feeding groups of aquatic insect families in Latin America: a critical analysis and review of existing literature. Rev Biol Trop 62(S2):155–167. https://doi.org/10.15517/rbt.v62i0.15785
Schriever TA, Bogan MT, Boersma KS, Cañedo-Argüelles M, Jaeger KL, Olden JD, Lytle DA (2015) Hydrology shapes taxonomic and functional structure of desert stream invertebrate communities. Freshw Sci 34(2):399–409. https://doi.org/10.1086/680518
Tachet H, Richoux P, Bournaud M, Usseglio-Polatera P (2010) Invertébrés d’eau douce, systématique, biologie, écologie. CNRS Éditions, Paris
Theodoropoulos C, Vourka A, Skoulikidis N, Rutschmann P, Stamou A (2018) Evaluating the performance of habitat models for predicting the environmental flow requirements of benthic macroinvertebrates. J Ecohydraulics 3(1):30–44. https://doi.org/10.1080/24705357.2018.1440360
Thomas R, Thomas KR (2022) The influence of habitat structure and physico-chemical variables on entomofaunal diversity in a headwater stream of Pamba river. Kerala New Visions Biol Sci 9:150–157. https://doi.org/10.9734/bpi/nvbs/v9/1871B
Thorp J, Rogers C (2016) Thorp and Covich’s freshwater invertebrates: Key to Nearctic Fauna, 4th edn. Elsevier, Amsterdam. https://doi.org/10.1016/C2010-0-65589-1
Tonkin JD, Bogan MT, Bonada N, Rios-Touma B, Lytle DA (2017) Seasonality and predictability shape temporal species diversity. Ecology 98(5):1201–1216. https://doi.org/10.1002/ecy.1761
Torres-Ruiz M, Wehr JD, Perrone AA (2007) Trophic relations in a stream food web: importance of fatty acids for macroinvertebrate consumers. J N Am Benthol Soc 26:509–522
Touron-Poncet H, Bernadet C, Compin A, Bargier N, Céréghino R (2014) Implementing the water framework directive in overseas Europe: a multimetric macroinvertebrate index for river bioassessment in Caribbean islands. Limnologica 47:34–43. https://doi.org/10.1016/j.limno.2014.04.002
Vagenas G, Theodoropoulos C, Moutaouakil S, Benaissa H, Fendane Y, Stambouli MT, Dimitriou E, Ghamazi M, Stamo A (2022) Habitat suitability curves of macroinvertebrates from a large North African river (Oum Errabia, Morocco). Marine and Inland Waters Research Symposium, Porto Heli, Greece
Van der Meer TV, Van Der Lee GH, Verdonschot RCM, Verdonschot PFM (2021) Macroinvertebrate interactions stimulate decomposition in WWTP effluent-impacted aquatic ecosystems. Aquat Sci 83:65. https://doi.org/10.1007/s00027-021-00821-8
Vannote RL, Minshall GW, Cummins KW, Sedell JR, Cushing CE (1980) The river continuum concept. Can J Fish Aquat Sci 37:130–137. https://doi.org/10.1139/f80-017
Waters NMC, San Giovanni CR (2002) Distribution and diversity of benthic macroinvertebrates associated with aquatic macrophytes. J Freshw Ecol 17(2):223–232. https://doi.org/10.1080/02705060.2002.9663890
Webster JS, Gido KB, Hedden S, Propst DL, Whitney JE (2022) Response of arid-land macroinvertebrate communities to extremes of drought, wildfire, and monsoonal flooding. River Res Appl 38:832–845
Acknowledgements
The authors express their sincere gratitude to Pr. Bekdouche Farid, Dr. Kalbaza Ahmed Yassine, Dr. Berdja Rafik, and Dr. Arar Abdelkrim for facilitating fieldwork and technical support.
Author information
Authors and Affiliations
Contributions
MK, BR, BI, MS, and Si BA designed the research; MK, BI, and MS conducted the experiments and prepared the original draft of the manuscript; Si BA: Conceptualized and supervised the work. Si BA and CR revised the final content.
Corresponding author
Ethics declarations
Competing interests
Authors do not have any competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Meradi, K., Bounar, R., Benzina, I. et al. How do substrate types affect the seasonal richness and functional feeding groups variation of benthic insects in an arid region (northeastern Algeria)?. Biologia 79, 1749–1759 (2024). https://doi.org/10.1007/s11756-024-01657-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-024-01657-x