Abstract
Supercritical fluid extraction of flaxseed oil with carbon dioxide was performed. Effects of particle size, pressure, temperature and the flow rate of supercritical carbon dioxide (SC-CO2) were investigated. Response surface methodology was used to determine the effects of pressure (30–50 MPa), temperature (50–70 °C) and SC-CO2 flow rate (2–4 g/min) on flaxseed oil yield in SC-CO2. The oil yield was represented by a second order response surface equation (R 2 = 0.993) using the Box-Behnken design of experiments. The oil yield increased significantly with increasing pressure (p < 0.01), temperature (p < 0.05) and SC-CO2 flow rate (p < 0.01). The maximum oil yield from the response surface equation was predicted as 0.267 g/g flaxseed for 15 min extraction of 5 g flaxseed particles (particle diameter <0.850 mm) at 50 MPa pressure and 70 °C temperature, with 4 g/min solvent flow rate. Total extraction time at these conditions was predicted as 22 min.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Supercritical fluid extraction is a method for the extraction of valuable ingredients from natural substances with high yield and quality, for this reason it has been studied widely as an alternative to conventional oil extraction methods. Supercritical carbon dioxide (SC-CO2) is the most preferred supercritical solvent in food extractions because it is non-toxic, non-explosive, separated easily and completely from the extract, and it has a low critical temperature (31.1 °C) and low price. The low critical temperature of SC-CO2 enables extraction of thermo-labile compounds without degradation [1]. SC-CO2 extraction of oils from oil seeds and nuts includes rapeseed [2], peanut [3], canola [4, 5], almond [6], pistachio nut [7], sunflower [8], sesame [9], walnut [10], poppy seed [11], hazelnut [12], apricot kernel oil [13], safflower seed [14], and sacha inchi seed [15].
Flax (Linum usitatissimum) is an economically important oilseed crop and is the most prominent oilseed studied to date as a functional food [16]. Flaxseed is a leading source of an omega-3 fatty acid, α-Linolenic acid (ALA), which makes up 55–60% of total fatty acids [17, 18]. ALA reduces blood pressure in cases of hypertension and lowers serum triglycerides and cholesterol [19]. ALA is converted to eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) which have cardioprotective properties. It has been shown that flaxseed oil increases the plasma concentrations of these cardioprotective omega-3 fatty acids in humans [20]. Flaxseed oil, also, contains tocopherols and tocotrienols [17]. They are important antioxidants in stabilizing unsaturated fatty acids in foods, and together with other antioxidants like phenolics they provide effective protection against oxidative stress in the human body [21].
Many factors including pressure and temperature of the process, flow rate of solvent, particle size of seed etc. and can affect the efficiency of the SC-CO2 extraction process. To design and develop a SC-CO2 extraction process, the effects of these parameters on the total oil yield must be determined. In classical methods, process parameters are optimized by conducting experiments concentrating on one factor at a time. This method is laborious and time consuming as well as ignoring the interaction effect of parameters. Compared to the classical methods, response surface methodology is more efficient, require fewer data and provides interaction effects on the response besides factor effects. Therefore, response surface methodology has been very popular for optimization purposes in recent years [22, 23]. Response surfaces of pistachio nut oil [7], turmeric oil [22], hazelnut oil [24], apricot kernel oil [25], cotton seed oil [26], wheat germ oil [27], passion fruit seed oil [28], and coconut oil [29] yields in SC-CO2 have been reported in the literature.
Bozan and Temelli [17] studied SC-CO2 extraction of flaxseed oil. They investigated the effects of pressure (21, 35 and 55 MPa) and temperature (50 and 70 °C) on oil yield and the solubility of the oil. The maximal solubility of the oil was reported as 11.3 mg oil/g CO2 at 55 MPa and 70 °C. However, only 66% of the total oil was recovered under these conditions. They, also, indicated that the ALA content of the oil extracted by SC-CO2 was higher than that of the oil extracted by solvent extraction. Barthet and Daun [30] investigated the possibility of supercritical fluid extraction as an analytical method for determining the fat content of flax and some other soft oil seeds and they reported that the seed matrix is important for oil recovery. Although, studies of SC-CO2 extraction of flax seed oil exist, there is a need for detailed analysis of the effects of process parameters on extraction yield.
Therefore, the aims of this study were to determine the effects of pressure, temperature SC-CO2 flow rate and particle size of seeds on flaxseed oil yield in SC-CO2 and develop a response surface equation for oil yield showing the effects of pressure, temperature SC-CO2 flow rate by using response surface methodology.
Experimental Procedures
Materials
Flaxseeds were obtained from a local market and stored at +4 °C in sealed glass jars. The moisture and oil contents were 3.4 and 32.7%, respectively. Without any pre-treatment, samples were ground into small sizes by using a kitchen grinder (Braun MultiQuick 6550), sieved and fractionated according to particle size by certified test sieves (Endecotts Ltd., London, England). Sieving was performed by a shaker (Octagon 200, Endecotts Ltd., London, England). CO2 was purchased from Habaş (Turkey).
Methods
SC-CO2 Extraction
Extractions of flaxseed oil with SC-CO2 were performed by using a Supercritical Fluid Extraction System (SFX System 5100, Isco Inc., Lincoln, NE, USA), which consists of an extractor (SFX 3560) and two syringe pumps (Model 100DX) enabling co-solvent addition. The extractor is a 10-ml aluminum sample cartridge where SC-CO2 flows downward.
5 g flaxseed particles were extracted with SC-CO2. The extract was passed through the coaxially heated adjustable restrictor, which was set at 110 °C, and the oil was precipitated in test tubes containing glass wool. The yield was determined gravimetrically. Extractions of response surface analysis were done for 15 min.
Analytical Methods
Moisture content of the samples was determined using AOAC Method 926.12 (AOAC, 1995). For total fat determination, a 5-g flaxseed sample was extracted with n-hexane (Merck) using a Soxhlet extractor [31]. The analyses were performed in triplicates.
Experimental Design
Response surface methodology was used to determine the effects of pressure, temperature and solvent flow rate on flaxseed oil yield in SC-CO2. The three level Box-Behnken design with three independent variables was applied for response function fitting. Independent variables of the design were pressure, temperature and SC-CO2 flow rate. They were coded as −1, 0 and 1 and coded levels were found from equations
Experimental points used according to the Box-Behnken design and their coded values are given in Table 1.
A second order polynomial equation was used to express the yield as a function of independent variables,
where, Y is oil yield in SC-CO2 as g oil/g flaxseed and a ij are coefficients of the function. Multiple regression analysis was performed to find the coefficients of the response function (Minitab Inc., Minitab release 12.1, 1998) by using experimental oil yield values obtained after 15 min of extraction (Table 1).
Results and Discussion
Effects of pressure, temperature and solvent flow rate on flaxseed oil yield in SC-CO2 were investigated by response surface methodology. Before extractions used in response surface regression, preliminary experiments were performed to analyze effects of parameters (including particle size) to determine the extraction mechanism and consequently, the extraction conditions to be used in the experimental design. The levels of independent parameters (Table 1) were determined based on these preliminary experiments.
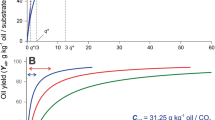
Extraction curves of the preliminary experiments are shown in Fig. 1. Conditions and results of these extractions are summarized in Table 2. Extraction of flaxseed oil, similar to oil seeds, can be divided into two periods as fast (initial linear period plus transition period) and slow extraction periods (second linear period). Extraction of the flaxseed oil occurred mainly in the fast extraction period and, as particle size decreased, oil yield increased. For extraction 1, oil yield and time of the fast extraction period were about 0.24 g oil/g flaxseed (73% of the initial oil content) and 25 min, however, oil yield and time of the slow extraction period were about 0.25 g/g flaxseed (76% of the initial oil content) and 25 min. Only 0.01 g/g flaxseed oil (3% of the initial oil content) was extracted during the slow extraction period. For extraction 4, oil yield and time of the fast extraction period were about 0.29 g oil/g flaxseed (89% of the initial oil content) and 50 min, however, oil yield and time of the slow extraction period were about 0.31 g/g flaxseed (95% of the initial oil content) and 70 min. Only 0.02 g/g flaxseed oil (6% of the initial oil content) was extracted during the slow extraction period. Similar trend exists for other extractions (Fig. 1 and Table 2). Oil seeds were ground to break oil cells releasing oil out of the cells for easy recovery. As particle size decreases, the amount of the released oil increases [13, 32]. In the fast extraction period, the oil released from the oil cells is extracted from the surface of the particles, however, in slow extraction period the unreleased oil from the intact cells is extracted. In the fast extraction period, the mass transfer rate is determined by the solubility of the oil in SC-CO2, while in the slow extraction period, it is controlled by diffusion of the oil in the particles. The mass transfer rate was low and the oil yield was insignificant in the slow extraction period compared to those in the fast extraction period [12, 13, 24]. Therefore, continuation of extraction in the slow extraction period was not feasible and extraction was ended after the fast extraction period and the total oil yields obtained at the end of the extractions were considered as the extractable oil amounts for specific particle sizes [24].
It is clear that the oil yield obtained at the end of the fast extraction period was mainly dependent on particle size; however, the extraction rate and the duration of the fast extraction period were dependent on SC-CO2 flow rate, pressure and temperature (Fig. 1 and Table 2). Similar results were reported for hazelnut oil [24]. Therefore, a smaller particle size (particle diameter <0.85 mm) was used in the extractions of response surface analysis. Due to the fact that extraction of the flaxseed oil occurred mainly in the fast extraction period, the time of extractions used in response surface regression was selected as 15 min to assure extractions were in the fast extraction period. When flaxseed samples with a diameter of less than 0.850 mm were used, oil yields at the end of the fast extraction period were about 0.29–0.31 g/g flaxseed (Table 2). All of the experimental oil yields obtained for response surface analysis were less than 0.29 g/g flaxseed indicating that this was achieved (Table 1).
During the fast extraction period (including transition period) in extraction curves (first 5 points in extractions 1, 2 and 4, first 6 points in extraction 3) the relationship between extraction time and yield were linear. R 2 values obtained for the fast extraction period of the extractions 1–4 were 0.9862, 0.9837, 0.9872 and 0.9778, respectively. Released oil (more than 89% of total oil when flaxseed samples with a diameter of less than 0.850 mm were used) was extracted in this period of extraction and some of the unreleased oil (very much smaller amounts, around 4–5% of the total oil compared to oil extracted in the fast extraction period) was extracted if extraction continued in the slow extraction period. Therefore, continuation of the extraction after the fast extraction period for flaxseed is time consuming and an additional 25% more time may be enough for total extraction.
The extraction pressure was chosen between 30 and 50 MPa since the solubility of oils in SC-CO2 is low below 30 MPa [12, 33] and higher extraction pressures increase operational costs.
Experimental oil yields were used to determine the coefficients of the response surface equation (Eq. 4). Different forms of the second order regression equation were tested to give minimum standard error by removing statistically insignificant terms. Removal of the term \( a_{22} X_{2}^{2} \) (p = 0.75) decreased standard error, however, removal of other terms increased standard error. Therefore, the term \( a_{22} X_{2}^{2} \) was removed and others were left in the equation and the final form of the response surface equation having a minimum standard error was in the form,
Estimated coefficients of the reduced equation are given in Table 3 and predicted oil yields from the response surface equation are given in Table 1. A high R 2 value (0.993) indicates that the experimental data are well represented by the second-degree polynomial response surface equation. The analysis of variance also indicated that the model is highly applicable, which is evident from the high Fisher (F) ratio and the low probability value (Table 4).
Within the experimental space, effects of pressure, the flow rate and interactions between the pressure and the solvent flow rate on oil yield were found highly significant (p < 0.01); and the effects of temperature and interactions between pressure and temperature were significant (p < 0.05) (Table 3).
Response surface plots of the equation were generated to express the effect of any parameter on the yield within the experimental space under investigation. The response surface showing the effects of pressure and temperature on oil yield is given in Fig. 2, for 2 g/min (X 3 = −1) solvent flow rate. The oil yield increased both with increased pressure and temperature. In the fast extraction period, solubility behavior controls the mass transfer rate [6, 13]. Increasing the pressure increased the oil yield due to the increase in the solubility of the oil in SC-CO2 with increasing pressure [13, 17]. Although, the effect of the temperature on the oil yield was large at high pressures, as pressure decreased this effect decreased. At 50 MPa, when the temperature increased from 50 to 60 °C oil yield increased from 0.077 to 0.130 g/g flaxseed but, at 30 MPa, the same temperature difference did not increase the yield significantly from 0.034 g/g flaxseed. The solubility of oils in SC-CO2 increases with increasing pressure. However, the effect of temperature is different; increasing temperature decreases solubility below cross-over pressure but increases solubility above this pressure [33]. This shows that the cross-over pressure of flax seed oil is close to 30 MPa. This result agrees with the result of Bozan and Temelli [17], who showed that the solubility of flaxseed oil in SC-CO2, decreased at 21 MPa, however increased at 35 and 55 MPa as temperature increased from 50 to 70 °C. Increase in solubility of the oil in SC-CO2 (due to increasing temperature and pressure) increases the driving force in the fluid phase [13], therefore the extraction rate increases.
Effects of pressure and the SC-CO2 flow rate on oil yield at 70 °C are shown in Fig. 3. Increases in pressure and SC-CO2 flow rate increased oil yield and the effect of each parameter on the oil yield was high when the other parameter’s value was high. Increasing pressure increases the solubility of the oil in SC-CO2 therefore increasing the driving force in the fluid phase [13]. Increasing the SC-CO2 flow rate decreases the mass transfer resistance therefore increasing the mass transfer coefficient [13, 34, 35]. Therefore, both pressure and SC-CO2 increased the mass transfer rate and, therefore the oil yield in 15 min of extraction.
From the response surface equation, the maximum flaxseed oil yield after 15 min of extraction was found to be 0.267 g oil/g flaxseed under extraction conditions of 50 MPa, 70 °C and a 4 g/min SC-CO2 flow rate. These conditions are the highest extremes of the experimental ranges, therefore they are the best values of these ranges. The oil yield of 0.267 g oil/g flaxseed corresponds to about 82% recovery of the initial oil in 5 g flaxseed (for particle diameter <0.85 mm). Assuming 95% of the initial oil to be the maximum amount of oil that can be extracted in the fast extraction period and considering linear behaviour of the extraction curve, the time required to extract this amount of oil was determined as 17 min in this period. To calculate the total extraction time, an additional 25% time was calculated and the total extraction time was found to be 22 min. This time value indicates the time required to extract the total amount of extractable oil for 50 MPa pressure, 70 °C temperature and a 4 g/min SC-CO2 flow rate as the extraction conditions.
Expressing oil yield as a function of uncoded parameters (pressure, temperature and solvent flow rate) rather than coded parameters is practically more useful [25]. Therefore, the uncoded form of the response surface equation was developed by inserting Eqs. 1 to 3 into Eq. 5 as,
where, P is pressure (MPa), T is temperature (°C) and Q is SC-CO2 flow rate (g/min).
Conclusion
In the ranges of 30–50 MPa, 50–70 °C and 2–4 g/min pressure, SC-CO2 flow rate and interactions between pressure and solvent flow rate showed a highly significant effect on oil yield (p < 0.01); in addition, the temperature and interactions between pressure and temperature showed a significant effect on oil yield (p < 0.05). The yield increased with increasing temperature, pressure and solvent flow rate, indicating an increase in the extraction rate. Within the experimental range, the oil yield of 5 g flaxseed particles (particle diameter <0.850 mm) for 15 min of extraction time was represented by a response surface equation. The maximum oil yield from this equation was 0.267 g oil/g flaxseed under conditions of 50 MPa, 70 °C and 4 g/min SC-CO2 flow rate. Under these extraction conditions, the total extraction time to extract all the extractable oil was found to be 22 min.
References
Rizvi SSH (1996) Supercritical fluid processing of food and biomaterials. Blackie Academic and Professional, Glasgow
Eggers R (1985) High pressure extraction of oil seed. J Am Oil Chem Soc 62:1222–1230
Goodrum JW, Kilgo MB (1987) Peanut oil extraction with SC-CO2: solubility and kinetic functions. T ASAE 30:1865–1868
Fattori M, Bulley NR, Meshen A (1988) Carbon dioxide extraction of canola seed: oil solubility and effect of seed treatment. J Am Oil Chem Soc 65:968–974
Temelli F (1992) Extraction of triglycerides and phospholipids from canola with supercritical carbon dioxide and ethanol. J Food Sci 57:440–442, 457
Marrone C, Poletto M, Reverchon E, Stassi A (1998) Almond oil extraction by supercritical CO2: experiments and modeling. Chem Eng Sci 53:3711–3718
Palazoğlu TK, Balaban ME (1998) Supercritical CO2 extraction of roasted pistachio nuts. T ASAE 41:679–684
Kiriamiti HK, Rascol E, Marty A, Condoret JS (2001) Extraction rates of oil from high oleic sunflower seeds with supercritical carbon dioxide. Chem Eng Process 41:711–718
Odabaşı AZ, Balaban MO (2002) Supercritical CO2 extraction of sesame oil from raw seeds. J Food Sci Tech Mys 39:496–501
Oliveira R, Rodrigues MF, Bernardo-Gil MG (2002) Characterization and supercritical carbon dioxide extraction of walnut oil. J Am Oil Chem Soc 79:225–230
Bozan B, Temelli F (2003) Extraction of poppy seed oil using supercritical CO2. J Food Sci 68:422–426
Özkal SG, Salgin U, Yener ME (2005) Supercritical carbon dioxide extraction of hazelnut oil. J Food Eng 69:217–223
Özkal SG, Yener ME, Bayındırlı L (2005) Mass transfer modeling of apricot kernel oil extraction with supercritical carbon dioxide. J Supercrit Fluid 35:119–127
Han X, Cheng L, Zhang R, Bi J (2009) Extraction of safflower seed oil by supercritical CO2. J Food Eng 92:370–376
Follegatti-Romero LA, Piantino CR, Grimaldi R, Cabral FA (2009) Supercritical CO2 extraction of omega-3 rich oil from sacha inchi (Plukenetia volubilis L.) seeds. J Supercrit Fluid. doi:10.1016/j.supflu.2009.03.010
Oomah BD (2001) Flaxseed as a functional food source. J Sci Food Agric 81:889–894
Bozan B, Temelli F (2002) Supercritical CO2 extraction of flaxseed. J Am Oil Chem Soc 79:231–235
Dyerberg J (1986) Linolenate-derived polyunsaturated fatty acids and prevention of atherosclerosis. Nutr Rev 44:125–134
Cunnane SC (1995) Flaxseed ALA function in human metabolism and function of α-linolenic acid in human nutrition. In: Cunnane SC, Thompson LU (eds) Flaxseed in human nutrition. AOCS Press, Champaign, pp 99–123
Harper CR, Edwards MJ, DeFilippis AP, Jacobson TA (2006) Flaxseed oil increases the plasma concentrations of cardioprotective (n-3) fatty acids in humans. J Nutr 137:83–87
Bozan B, Temelli F (2008) Chemical composition and oxidative stability of flax, safflower and poppy seed and seed oils. Bioresour Technol 99:6354–6359
Began G, Goto M, Kodama A, Hirose T (2000) Response surfaces of total oil yield of turmeric (Curcuma longa) in supercritical carbon dioxide. Food Res Int 33:341–345
Baş D, Boyacı İH (2007) Modeling and optimization I: usability of response surface methodology. J Food Eng 78:836–845
Özkal SG, Yener ME, Salgın U, Mehmetoğlu Ü (2005) Response surfaces of hazelnut oil yield in supercritical carbon dioxide. Eur Food Res Technol 220:74–78
Özkal SG, Yener ME, Bayındırlı L (2005) Response surfaces of apricot kernel oil yield in supercritical carbon dioxide. LWT 38:611–616
Bhattacharjee P, Singhal RS, Tiwari SR (2007) Supercritical carbon dioxide extraction of cottonseed oil. J Food Eng 79:892–898
Shao P, Sun P, Ying Y (2008) Response surface optimization of wheat germ oil yield by supercritical carbon dioxide extraction. Food Bioprod Process 86:227–231
Liu S, Yang F, Zhang C, Ji H, Hong P, Deng C (2009) Optimization of process parameters for supercritical carbon dioxide extraction of Passiflora seed oil by response surface methodology. J Supercrit Fluid 48:9–14
Nik Norulaini NA, Setianto WB, Zaidul ISM, Nawi AH, Azizi CYM, Mohd Omar AK (2009) Effects of supercritical carbon dioxide extraction parameters on virgin coconut oil yield and medium-chain triglyceride content. Food chem. doi:10.1016/j.foodchem.2009.02.030
Barthet VJ, Daun JK (2002) An evaluation of supercritical fluid extraction as an analytical tool to determine fat in canola, flax, solin, and mustard. J Am Oil Chem Soc 79:245–251
Nas S, Gökalp HY, Ünsal M (1998) Bitkisel Yağ Teknolojisi. Pamukkale University Press, Denizli
Reverchon E, Marrone C (2001) Modeling and simulation of the supercritical CO2 extraction of vegetable oils. J Supercrit Fluid 19:161–175
Özkal SG, Yener ME, Bayındırlı L (2006) The solubility of apricot kernel oil in supercritical carbon dioxide. Int J Food Sci Tech 41:399–404
Subra P, Castellani S, Jestin P, Aoufi A (1998) Extraction of β-carotene with supercritical fluids: experimental and modeling. J Supercrit Fluid 12:261–269
Döker O, Salgın U, Şanal İ, Mehmetoğlu Ü, Çalımlı A (2004) Modeling of extraction of β-carotene from apricot bagasse using supercritical CO2 in packed bed extractor. J Supercrit Fluid 28:11–19
Acknowledgments
The experiments of this research were performed by using the SFE system in the Central Laboratory (R&D-Training Center) of the Middle East Technical University. The author wishes to thank Prof. Dr. M. Esra Yener and Dr. H. İbrahim Çetin for their support shown during the use of this SFE system.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Özkal, S.G. Response Surface Analysis and Modeling of Flaxseed Oil Yield in Supercritical Carbon Dioxide. J Am Oil Chem Soc 86, 1129–1135 (2009). https://doi.org/10.1007/s11746-009-1448-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11746-009-1448-6