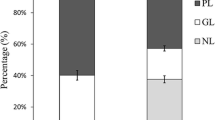
Abstract
The FA composition of glycerophospholipid (GPL) classes and subclasses was investigated in whole animals of three marine bivalve mollusks: the Japanese oyster Crassostrea gigas, the blue mussel Mytilus edulis, and the Manila clam Ruditapes philippinarum. Individual organs (gills, mantle, foot, siphon, and muscle) of the Manila clam also were examined. The PS plasmalogen (PSplsm), PE plasmalogen (PEplsm), and PC plasmalogen (PCplsm) subclasses were isolated by HPLC, and their individual FA compositions were examined using GC. Plasmalogen forms of PS and PE, when compared to their respective diacyl forms, were found to be specifically enriched with non-methylene-interrupted (NMI) FA (7,15–22∶2, 7, 13–22∶2, and their precursors) and 20∶1n−11 FA. Such a clear specific association was not found for PCplsm. Interestingly, this trend was most apparent in PSplsm, and the above FA were found to be, respectively, the predominant PUFA and monounsaturated FA in the PSplsm isolated from the three species. This specificity was maintained in all the analyzed organs of the Manila clam but varied in proportions: The highest level of plasmalogens, NMI FA, and 20∶1n−11 was measured in gills and the lowest was in muscle. These results represent the first comprehensive report on a FA composition of the PSplsm subclass isolated from mollusks. The fact that NMI FA and 20∶1n−11, which are thought to be biosynthesized FA, were mainly associated with aminophospholipid plasmalogens (PE and PS) is likely to have a functional significance in bivalve membranes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- CL:
-
cardiolipin common name of diphosphatidylglycerol
- GPL:
-
glycerophospholipid(s)
- MUFA:
-
monounsaturated FA
- NMI:
-
non-methylene-interrupted
- ΣNMI:
-
sum of all non-methylene-interrupted fatty acids
- PCplsm:
-
PC plasmalogen
- PEplsm:
-
PE plasmalogen
- PSplsm:
-
PS plasmalogen
- SFA:
-
saturated fatty acid(s)
References
Nagan, N., and Zoeller, R.A. (2001) Plasmalogens: Biosynthesis and Functions, Prog. Lipid Res. 40, 199–229.
Leray, C., Cazenave, J.-P., and Gachet, C. (2002) Platelet Phospholipids Are Differentially Protected Against Oxidative Degradation by Plasmalogens, Lipids 37, 285–290.
Maeba, R., Sawada, Y., Shimasaki, H., Takahashi, I., and Ueta, N. (2002) Ethanolamine Plasmalogens Protect Cholesterol-Rich Liposomal Membranes from Oxidation Caused by Free Radicals, Chem. Phys. Lipids 120, 145–151.
Maeba, R., and Ueta, N. (2003) Ethanolamine Plasmalogen and Cholesterol Reduce the Total Membrane Oxidizability Measured by the Oxygen Uptake Method, Biochem. Biophys. Commun. Res. 302, 265–270.
Sargent, J.R. (1987) Ether-Linked Glycerides in Marine Animals, in Marine Biogenic Lipids. Fats, and Oils (Ackman, R.G., ed.), pp. 176–193, CRC Press, Boca Raton, FL.
Joseph, J.D. (1982) Lipid Composition of Marine and Estuarine Invertebrates. Part II: Mollusca, Prog. Lipid Res. 21, 109–153.
Schrakamp, G., Schutgens, R.B.H., Schalkwijk, C.G., and Van Den Bosch, H. (1991) Comparative Studies on Plasmalogen Biosynthesis in Rat Brain Homogenates and Isolated Oligodendroglial Cells During Development, Brain Dysfunct. 4, 217–227.
Dembitsky, V.M., and Vaskovsky, V.E. (1976) Distribution of Plasmalogens in Different Classes of Phospholipids of Marine Invertebrates, Biol. Morya (Kiey) 5, 68–72.
Dembitsky, V.M. (1979) Plasmalogens in Phospholipids of Marine Invertebrates, Biol. Morya (Kiev) 5, 86–90.
Soudant, P., Marty, Y., Moal, J., and Samain, J.F. (1995) Separation of Major Polar Lipids in Pecten maximus by High-Performance Liquid Chromatography and Subsequent Determination of Their Fatty Acids Using Gas Chromatography, J. Chromatogr. B 673, 15–26.
Soudant, P., Moal, J., Marty, Y., and Samain, J.F. (1996) Impact of the Quality of Dietary Fatty Acids on Metabolism and the Composition of Polar Lipid Classes in Female Gonads of Pecten maximus (L.), J. Exp. Mar. Biol. Ecol. 205, 149–163.
Soudant, P., Moal, J., Marty, Y., and Samain, J.F. (1997) Composition of Polar Lipid Classes in Male Gonads of Pecten maximus (L.). Effect of Nutrition, J. Exp. Mar. Biol. Ecol. 215, 103–114.
Soudant, P., Marty, Y., Moal, Y., Masski, H., and Samain, J.F. (1998) Fatty Acid Composition of Polar Lipid Classes During Larval Development of Scallop Pecten maximus (L.), Comp. Biochem. Phys. A 121, 279–288.
Koizumi, C., Jeong, B.Y., and Ohshima, T. (1990) Fatty Chain Composition of Ether and Ester Glycerophospholipids in the Japanese Oyster Crassostrea gigas, Lipids 25, 363–370.
Jeong, B.Y., Ohshima, T., and Koizumi, C. (1990) Molecular Species of 1-O-Alkyl-1′-enyl-2-acyl-, 1-O-Alkyl-2-acyl-and 1,2-Diacyl Glycerophospholipids in Japanese Oyster Crassostrea gigas (Thunberg), Lipids 25, 624–632.
Ackman, R.G., and Hooper, S.N. (1973) Non-Methylene-Interrupted Fatty Acids in Lipids of Shallow-Water Marine Invertebrates: A Comparison of Two Molluses (Littorina littorea and Lunatia triserita) with the Sand Shrimp (Crangon septumspinosus), Comp. Biochem. Phys. 46B, 153–165.
Dunstan, G.A., Volkman, J.K., and Barrett, S.M. (1993) The Effect of Lyophilization on the Solvent Extraction of Lipid Classes, Fatty Acids and Sterols from the Oyster Crassostrea Gigas, Lipids 28, 937–944.
Caers, M., Coutteau, P., and Sorgeloos, P. (1999) Dietary Impact of Algal and Artificial Diets, Fed at Different Feeding Rations, on the Growth and Fatty Acid Composition of Tapes philippinarum (L.) Spat, Aquaculture 170 307–322.
Zhukova, N.V. (1986) Biosynthesis of Non-Methylene-Interrupted Fatty Acids from [14C]Acetate in Molluscs, Biochim. Biophys. Acta 878, 131–133.
Zhukova, N.V. (1991) The Pathway of the Biosynthesis of Non-Methylene-Interrupted Dienoic Fatty Acids in Molluscs, Comp. Biochem. Phys. 100B 801–804.
Chapelle, S. (1987) Plasmalogens and O-Alkylglycerophospholipids in Aquatic Animals, Comp. Biochem. Phys. 88B, 1–6.
Klingensmith, J.S. (1982) Distribution of Methylene and Non-Methylene-Interrupted Dienoic Fatty Acids in Polar Lipids and Triacylglycerols of Selected Tissues of the Hardshell Clam (Mercenaria mercenaria), Lipids 17, 976–981.
Folch, J., Lees, M., and Sloane Stanley, G.H. (1957) A Simple Method for the Isolation and Purification of Total Lipids from Animal Tissues, J. Biol. Chem. 226, 497–509.
Nelson, G.J. (1993) Isolation and Purification of Lipids from Biological Matrices, in Analyses of Fats, Oils and Derivatives (Perkins, E.G., ed.), pp. 20–59, AOCS Press, Champaign.
Marty, Y., Delaunay, F., Moal, J., and Samain, J.F. (1992) Changes in the Fatty Acid Composition of the Scallop Pecten maximus (L.) During Larval Development, J. Exp. Mar. Biol. Ecol. 163, 221–234.
Soudant, P., Chu, F.L.E., and Marty, Y. (2000) Lipid Class Composition of the Protozoan Perkinsus marinus, an Oyster Parasite, and Its Metabolism of a Fluorescent Phosphatidylcholine Analog, Lipids 35, 1387–1395.
Kraffe, E., Soudant, P., Marty, Y., Kervarec, N., and Jehan, P. (2002) Evidence of a Tetradocosahexaenoic Cardiolipin in Some Marine Bivalves, Lipids 37, 507–514.
Chelomin, V.P., and Zhukova, N.V. (1981) Lipid Composition and Some Aspects of Aminophospholipid Organization in Erythrocyte Membrane of the Marine Bivalve Mollusc Scapharca broughtoni (Shrenck), Comp. Biochem. Phys. 69B, 599–604.
Zhukova, N.V., and Svetashev, V.I. (1986) Non-Methylene-Interrupted Dienoic Fatty Acids in Molluscs from the Sea of Japan, Comp. Biochem. Phys. 83B, 643–646.
Yorek, M.A. (1993) Biological Distribution, in Phospholipids Handbook (Cevc, G., ed.), pp. 745–775, Marcel Dekker, New York.
De Moreno, J.E.A., Moreno, V.J., and Brenner, R.R. (1976) Lipid Metabolism of the Yellow Clam, Mesodesma mactroïdes: 2-Polyunsaturated Fatty Acid Metabolism, Lipid 11, 561–566.
Waldock, M.J., and Holland, D.L. (1984) Fatty Acid Metabolism in Young Oysters, Crassostrea gigas: Polyunsaturated Fatty Acids, Lipids 19, 332–336.
Volkman, J.K., Jeffrey, S.W., Nichols, P.D., Rogers, G.I., and Garland, C.D. (1989) Fatty Acid and Lipid Composition of 10 Species of Microalgae Used in Mariculture, J. Exp. Mar. Biol. Ecol. 128, 219–240.
Stefanov, K., Seizova, K., Brechany, E., and Christie, W.W. (1992) An Unusual Fatty Acid Composition for a Fresh-Water Mussel, Unio tumidus, from Bulgaria, J. Nat. Prod. 55, 979–981.
Napolitano, G.E., and Ackman, R.G. (1993) Fatty Acid Dynamics in Sea Scallops Placopecten magellanicus (Gmelin, 1791) from Georges Bank, Nova Scotia, J. Shellfish Res. 12, 267–277.
Cook, H.W. (1996) Fatty Acid Desaturation and Chain Elongation in Eukaryotes, in Biochemistry of Lipids, Lipoproteins and Membranes (Vance, D.E., and Vance, J.E.), pp. 129–152, Elsevier Science, Amsterdam.
Paradis, M., and Ackman, R.G. (1977) Potential for Employing the Distribution of Anomalous NMI Dienoic Fatty Acids in Several Marine Invertebrates as Part of Food Web Studies, Lipids 12, 170–176.
Rabinovich, A.L., and Ripatti, P.O. (1991) The Flexibility of Natural Hydrocarbon Chains with Non-Methylene-Interrupted Double Bonds, Chem. Phys. Lipids 58, 185–192.
Morand, O.H., Zoeller, R.A., and Raetz, C.R.H. (1988) Disappearance of Plasmalogens from Membranes of Animal Cells Subjected to Photosensitized Oxidation, J. Biol. Chem. 263, 11597–11606.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Kraffe, E., Soudant, P. & Marty, Y. Fatty acids of serine, ethanolamine, and choline plasmalogens in some marine bivalves. Lipids 39, 59–66 (2004). https://doi.org/10.1007/s11745-004-1202-x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11745-004-1202-x