Abstract
Creativity is the ability to produce something novel and useful. Various tasks have been used to explore the neural bases of creativity. However, studies exploring the relationship between the brain regions during divergent thinking are still rare. Given that the brain works in networks, exploring the functional connectivity (FC) patterns during divergent thinking is important. The present study explored the FC patterns during alternative uses task and its relationship with openness to experience. Psychophysiological interaction results corroborated that the inferior parietal lobule was positively connected to the precuneus and middle temporal gyrus. Middle frontal gyrus/superior frontal gyrus was positively connected to the precuneus and supramarginal gyrus. Individual difference analysis revealed that openness to experience was positively related to the strength of FCs between some key regions of default mode, cognitive control and salience networks. Findings confirmed the network-based mechanisms underlying creativity and the neural basis of individual differences of openness to experience.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Creativity is the ability to produce something novel and useful (Stein 1953; Sternberg and Lubart 1996; Runco and Jaeger 2012). It is linked not only to social development but also to almost all the areas of our everyday life (Mumford 2002; Dietrich and Kanso 2010). In the past years, various tasks have been used to explore the neural bases of creativity (Jung-Beeman et al. 2004; Qiu et al. 2010; Takeuchi et al. 2010a, b; Abraham et al. 2012; Aziz-Zadeh et al. 2013; Kleibeuker et al. 2013; Zhu et al. 2013; Park et al. 2015; Saggar et al. 2015). These studies confirmed that creativity may be related to widespread brain areas, such as the frontal areas, cingulate cortex, and temporoparietal areas (Dietrich and Kanso 2010; Jung et al. 2013; Fink and Benedek 2014). A recent meta-analysis in our laboratory used activation likelihood estimation to ascertain the role of these regions in divergent thinking tasks (Wu et al. 2015).
However, these investigations of creativity focus on which area is activated during creativity tasks. Studies investigating the relationship among the brain regions during the creativity tasks are still rare. Brain functions are supported by coordinated activities between separated brain regions (Catani et al. 2013; Smith et al. 2013; Sporns 2013), that is, the brain works in networks. Recent viewpoint highlights the role of the default mode network (DMN) and the cognitive control network (CCN) in the neural basis of creativity (Jung et al. 2013; Beaty et al. 2016a). The DMN is active during rest and deactivates during most externally-oriented tasks (Raichle et al. 2001; Greicius et al. 2003). This network associates with various cognitive processes such as mind-wandering (Mason et al. 2007), future thinking (Schacter et al. 2012), and perspective taking (Buckner and Carroll 2007). The CCN is engaged in attention-demanding cognitive tasks that associate with the top-down modulation of attention, response inhibition (Aron 2007), task-set switching (Dreher and Berman 2002), and working memory (Curtis and D'Esposito 2003). During creative tasks, these networks are likely to correspond to blind variation and selective retention components of creative cognition, respectively (Jung et al. 2013). The DMN related to the blind variation mechanism that is related to creative thinking generation and CCN related to selective retention which is related to the top-down cognitive control (Jung et al. 2013; Abraham 2014; Mok 2014). Creative idea involves the dynamic interactions of these brain networks (Beaty et al. 2016a). A recent study corroborated that functional connectivity (FC) patterns can predict individual creative ability (Beaty et al. 2018). Therefore, exploring the FC patterns during divergent thinking task is important.
Studies exploring the relationship between creativity and brain connectivity (structural and resting sate functional) give us insight into creativity-related connectivity patterns. For instance, Takeuchi et al. (2012) investigated the relationship between the creativity measured by divergent thinking and resting state FC (rsFC) and affirmed that high creativity is associated with increased rsFC between the medial prefrontal cortex and the posterior cingulate. Jung et al. (2010) used diffusion tensor imaging to explore the relationship between creativity and white matter integrity (structural connectivity). They corroborated that the composite creativity index was significantly inversely related to fractional anisotropy within the left inferior frontal white matter. These studies validated that several important connectivities were closely related to creativity. Nevertheless, limited to the neuroimaging methods, rsFC and structural connectivity were off-line and not dependent on tasks. Although rsFC or structural connectivity reflect the patterns of interaction between brain networks (Greicius et al. 2009; Honey et al. 2009), studies exploring task-based FC may give us specific functional networks during purposeful creativity tasks.
Furthermore, as a complex cognitive ability, creativity is closely related to personal characteristics, especially openness to experience. Considerable literature affirmed the consistently positive correlation between openness to experience and creativity (Feist 1998; Feist and Barron 2003; Silvia et al. 2009). However, studies addressing the potential relationship between openness to experience and creativity from the perspective of neuroscience are still rare. Li et al. (2014) explored the relationship among brain structure, openness to experience, and trait creativity and verified that openness to experience mediated the association between the gray matter volume of the middle temporal gyrus and trait creativity. The role of openness in creativity was affirmed by this study, but this study only explored this question at the regional brain level. The relationship between openness to experience and dynamic interaction of brain networks is still unknown.
The present study aims to explore the FC pattern during creative tasks and its relationship to openness to experience. To this end, the most widely used task, alternative uses task (AUT), was used. First, we hypothesize that the prefrontal cortex, temporal lobe and inferior parietal lobule will be activated in creative tasks. Second, these regions would connect to other key regions of the DMN and the CCN. Third, individuals’ personality trait (openness to experience) would be associated with the FC of these regions during tasks.
Materials and methods
Participants
Twenty-nine right-handed university students (17 women and 12 men) from Southwest University, China, participated in the experiment. The age of the participants ranged from 19 to 21 years (19.48 ± 0.74). None of them had a history of neurological or psychiatric illnesses. In accordance with the Declaration of Helsinki (1991), written informed consent was obtained from all the participants. The experimental procedure was approved by the local ethics committee.
fMRI task
The AUT and control task (object characteristics task, OCT) were used. Each task had 12 items, and each item was presented in separate blocks. In the AUT, the participants were asked to think of the original uses of everyday objects as many as possible in 20 s. In the OCT, the participants were required to think of the typical characteristics of everyday objects in 20 s. After each item, they had 4 s to press the buttons according to the number of ideas they generated. Four choices were provided: 0–1 ideas, 2–3 ideas, 4–5 ideas, and more than 5 ideas. Four buttons that represent each choice were available. The participants would press one of the buttons according to the number of their ideas. A fixation lasting for 20 s between the items were present. Tasks were presented in fixed sequence (AUT1 – Fixation – OCT1 – Fixation – AUT 2 – Fixation – OCT2...). After scanning, the participants were asked to write down the ideas they generated in a scanner.
All the generated ideas were rated by three trained raters with regard to their originality on a five-point rating scale ranging from 1 (“not original”) to 5 (“highly original”). Fluency and originality scores were obtained by averaging the raters’ scores. The raters displayed high internal consistency in their ratings (mean Cronbach’s alphas = 0.90).
Measure of openness to experience
Openness to experience was assessed using the revised version of the NEO Personality Inventory (Costa and McCrae 1992) that is based on a 5-factor model of personality. Questions were answered on a 5-point scale and personality were measured in five domains: neuroticism, extraversion, openness to experience, agreeableness and conscientiousness. In the present study, we mainly focused on the openness to experience dimension based on the a priori hypothesis predicting the relationship between openness and creativity (Feist 1998; Feist and Barron 2003; Silvia et al. 2008).
fMRI image acquisition
Images were acquired using a Siemens TRIO 3-Tesla scanner. The participants lay supine with their heads snugly fixed with foam pads to minimize head movements. They were instructed to keep still. A total of 535 BOLD images were obtained using Echo Planar Imaging (EPI) sequence with the following parameters: slices = 32; repetition time (TR)/echo time (TE) = 2000/30 ms; flip angle = 90 deg. FOV = 200× 200 mm2; voxel size = 3.4× 3.4× 4 mm3; thickness = 3 mm; and slice gap = 1 mm.
fMRI data preprocessing
Functional imaging data analysis was performed with SPM8 software (Wellcome Department of Imaging Neuroscience, London, U.K.). First, the functional data of each participant were motion-corrected. The participants who exhibited head motion of 3 mm maximum translation or 3.0° rotation were excluded. Thereafter, each participant’s functional image was normalized to EPI templates based on the Montreal Neurological Institute space (resampling voxel size = 3 × 3 × 3 mm3). Spatial smoothing (8 mm FWHM Gaussian kernel) was conducted to decrease spatial noise.
Activation analysis
In the first-level analysis of each participant, each task type was modeled separately with a general linear model. Movement correction parameters were added as covariates of no interest. Blocks in which participants did not generate any answer were not included in further analysis. In light of the research questions and hypotheses, we performed a comparison to produce a ‘contrast image’ for each participant: AUT versus OCT (AUT > OCT). In the group level, a one-sample t test for contrast image was used to obtain the activity pattern. Cluster-level family-wise error (FWE) was used for multiple comparisons. The significance threshold for FWE was set at p < 0.01 (cluster-forming threshold at voxel-level: p < 0.001).
Psychophysiological interaction analysis
On the basis of the regional activation results in the present study and previous literature, we defined the bilateral inferior parietal lobule (IPL), right middle frontal gyrus/superior frontal gyrus (MFG/SFG), right MFG, inferior frontal gyrus (IFG)/Insula, and cuneus as seed regions (VOI, Volume of Interest). The psychophysiological interaction (PPI) analysis consists of a design matrix with three regressors: (i) psychological variable: the cognitive process of interest (here AUT vs. OCT), (ii) physiological variable: the neural response of VOIs, and (iii) interaction term of (i) and (ii). VOIs’ time courses were extracted from the left IPL (x = −51, y = −54, z = 39), right IPL (x = 48, y = −54, z = 48), right MFG/SFG (x = 33, y = 54, z = 6), right MFG (x = 39, y = 33, z = 39), IFG/insula (x = 63, y = 12, z = 3), and right cuneus (x = 12, y = −87, z = 12) (6 mm radius sphere at the local peak). The psychological variable used was a vector coding for the specific task (1 for AUT, −1 for OCT) convolved with the HRF. Subsequently, the psychophysiological interaction term was created with the time course and psychological variable.
PPI analysis was then carried out for each VOI in each subject, and a group-level one-sample t-test was conducted to examine the significant FC. Whole brain cluster-level FWE was used for multiple comparisons. The significance threshold for FWE was set at p < 0.01 (cluster-forming threshold at voxel-level: p < 0.001).
Brain behavior correlation
Multiple regressions were used to explore the relationship between individual differences in openness and FC patterns. Personality score (openness to experience) was used as the variable of interest. Whole brain cluster-level FWE was used for multiple comparisons. The significance threshold for FWE was set at p < 0.01 (cluster-forming threshold at voxel-level: p < 0.001).
Results
Behavior data
During the task, the mean originality was 2.95 (SD = 0.25), and the mean fluency was 2.64 (SD = 1.24). Openness scores range from 133 to 202 (M = 161.59, SD = 18.38).
Activation analysis
In the contrast of AUT > OCT, one sample t test of data revealed stronger activation in posterior brain areas such as cuneus (Fig. 1, Table 1). The opposite contrast (AUT < OCT) found in widespread activation included the bilateral temporoparietal areas and the right lateral prefrontal cortex (Table 1, Fig. 1).
Psychophysiological interaction analysis
For all the participants, one-sample t-test showed that the left IPL was positively connected to the bilateral precuneus (Prec), the right middle temporal gyrus (MTG) (see Table 2, Fig. 2). Similar results were found when using right IPL as seed region. Right MFG/SFG was positively connected to bilateral Prec, MTG, and supramarginal gyrus (SMG). The right MFG/SFG was negatively connected to bilateral MOG and left SFG (see Table 2, Fig. 2). Right MFG was negatively connected to bilateral MOG. Right cuneus has adverse FC patterns compared with IPL (see Table 2). No significant result exists when using right IFG/insula as a seed region.
Brain behavior correlation
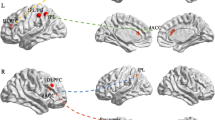
Whole brain analysis was used to explore the relationship between openness and the strength of FC. Using left IPL as a seed region, openness was positively related to the strength of FC with Prec and insula (see Table 3, Fig. 3a). Right IPL has similar results with left IPL (see Table 3, Fig. 3b). Using the right MFG as a seed region, openness was positively related to the strength of FC with the superior temporal gyrus (STG), MTG, insula and putamen (see Table 3, Fig. 3d). Right MFG/SFG has similar results with MFG (see Table 3, Fig. 3c). No significant result exists for cuneus and IFG/insula.
Relationship between behavior and functional connectivity patterns. a Using the left IPL as a seed region, openness was positively related to the strength of FC with Prec and insula. b Right IPL has similar results with left IPL. c Right MFG/SFG has similar results with MFG. d Using the right MFG as a seed region, openness was positively related to the strength of FC with STG, MTG, insula and putamen. All effects are corrected by FWE
Discussion
The present study aimed to explore the FC patterns during creative tasks and their relationship with openness to experience. We observed the activities of the bilateral MTG, bilateral IPL, MFG, and the cuneus. Results were in agreement with several findings of brain activity during the creative process compared with non-creative cognition (Fink et al. 2009; Abraham et al. 2012). PPI results corroborated that IPL was positively connected to Prec and MTG. MFG/SFG was positively connected to Prec and SMG. Brain behavior correlation analysis revealed that openness was positively related to the strength of FC between IPL/MFG and insula/Prec. The results were concordant with our hypothesis.
Brain regions are always co-activated during creativity tasks. We validated that both the left and right IPL connectivity patterns were moderated by experimental conditions. Bilateral IPL has increased connectivity with Prec and MTG during AUT compared with OCT. MRI studies have confirmed the role of these regions in the creative process (Jung et al. 2010; Wu et al. 2015). For instance, Takeuchi et al. (2010a, b) verified that a significant positive relationship existed between fractional anisotropy in the regions of the bilateral temporoparietal junction and the right IPL and individual creativity. Dandan et al. (2013) proved that the Prec is relevant to the successful storage and retrieval of heuristic knowledge that is important in creative problem solving. IPL was discussed in terms of bottom–up attention and automatically activated knowledge (Berkowitz and Ansari 2008; Fink et al. 2010). Prec was involved in the storage and retrieval of knowledge (Dandan et al. 2013), and MTG was related to the distant semantic process (Jung-Beeman et al. 2004; Whitney et al. 2011). The FC between these regions showed that the generation of original ideas needs the cooperation of different types of cognitive processes that involve in different brain regions. Furthermore, from the perspective of the network, IPL, Prec and MTG are the subregions of DMN. Given that DMN is devoted to the blind variation mechanism that is related to creative thinking generation (Jung et al. 2013; Beaty et al. 2016a), results confirmed that the cooperation of regions within the DMN related to the creative thinking process.
In the present study, MFG/SFG was positively connected to Prec and SMG. The lateral prefrontal cortex (LPFC) is related to several high-level cognitive functions such as cognitive flexibility, top-down cognitive control, sustained attention and working memory (Koechlin et al. 2003; Petrides 2005; Alvarez and Emory 2006; Dietrich and Kanso 2010). This region is widely implicated in creative thinking tasks (Abraham et al. 2012; Kleibeuker et al. 2013; Liu et al. 2015; Sun et al. 2016). Several studies have contended that LPFC connectivity with other brain regions at resting state was related to creativity (Beaty et al. 2014; Li et al. 2016). During tasks, LPFC connectivity with Prec and SMG revealed the top-down cognitive control process. From the perspective of large-scale networks, LPFC is the key region of CCN and Prec and SMG are the DMN nodes. The DMN is involved in the generation of original ideas, and the CCN is involved in the allocation of cognitive resources. Our results corroborated that the dynamic cooperation of these large-scale networks genrates original ideas.
We also asserted that personality trait was related to the FC patterns in tasks. The study of personality for creative individuals is one of the oldest areas in studies about creativity (Batey and Furnham 2006; Silvia et al. 2009). From the perspective of neuroscience, although the role of openness to experience in the creative cognition is still unknown, several studies explored the neural basis of openness to experience. For instance, Beaty et al. (2016b) used graph theory methods and affirmed that openness is related to the global efficiency of the DMN. In the present study, we affirmed a positive correlation between openness and IPL-Prec connectivity. The results are consistent with these studies and confirm that openness is related to the functional connectivities between nodes within the DMN. Open individuals are known for their cognitive flexibility and have high prefrontal activity (Sutin et al. 2009). Our study further contended that individuals with high openness scores also have high IPL-insula or MFG-insula connectivity. These regions devoted to different large-scale networks such as DMN, CCN, and salience network (SN). Among these three networks, SN modulates and interplays between the other two networks (Jung et al. 2013). The results confirmed that openness is related to the cooperation between networks. Taken together, increased FC between the key regions of the DMN, CCN and SN for open individuals may correspond to an improved ability to govern their cognitive resources to integrate and evaluate the relevant information, generating original ideas.
Certain limitations emerge in the present study. The present research extends past works by examining the interaction between brain regions during the divergent thinking task (AUT). The creative process has been explored by various creative tasks. The result is limited in the divergent thinking task that limits the generalizability of the results. Future studies should further examine connectivity patterns in the domain-general and domain-specific creative thinking tasks. Moreover, the fixed sequence of the experimental conditions may have potential effects on participants. Improvement in the design of experiment is also needed.
Conclusion
The present study confirmed that the dynamic interaction between brain regions, such as IPL, Prec, MTG, and MFG is related to creative processing. Furthermore, the task-specific pattern of FCs is related to individual differences in openness to experience. Findings affirmed the network-based mechanisms underlying creativity and its relationship with personality traits.
References
Abraham, A. (2014). Creative thinking as orchestrated by semantic processing vs. cognitive controlbrain networks. Frontiers in Human Neuroscience, 8, 95.
Abraham, A., Pieritz, K., Thybusch, K., Rutter, B., Kroger, S., Schweckendiek, J., Stark, R., Windmann, S., & Hermann, C. (2012). Creativity and the brain: uncovering the neural signature of conceptual expansion. Neuropsychologia, 50(8), 1906–1917.
Alvarez, J. A., & Emory, E. (2006). Executive function and the frontal lobes: A meta-analytic review. Neuropsychology Review, 16(1), 17–42.
Aron, A. R. (2007). The neural basis of inhibition in cognitive control. The Neuroscientist, 13(3), 214–228.
Aziz-Zadeh, L., Liew, S. L., & Dandekar, F. (2013). Exploring the neural correlates of visual creativity. Social Cognitive and Affective Neuroscience, 8(4), 475–480.
Batey, M., & Furnham, A. (2006). Creativity, intelligence, and personality: a critical review of the scattered literature. Genetic, Social, and General Psychology Monographs, 132(4), 355–429.
Beaty, R. E., Benedek, M., Wilkins, R. W., Jauk, E., Fink, A., Silvia, P. J., Hodges, D. A., Koschutnig, K., & Neubauer, A. C. (2014). Creativity and the default network: a functional connectivity analysis of the creative brain at rest. Neuropsychologia, 64, 92–98.
Beaty, R. E., Benedek, M., Silvia, P. J., & Schacter, D. L. (2016a). Creative cognition and brain network dynamics. Trends in Cognitive Sciences, 20(2), 87–95.
Beaty, R. E., Kaufman, S. B., Benedek, M., Jung, R. E., Kenett, Y. N., Jauk, E., Neubauer, A. C., & Silvia, P. J. (2016b). Personality and complex brain networks: the role of openness to experience in default network efficiency. Human Brain Mapping, 37(2), 773–779.
Beaty, R. E., Kenett, Y. N., Christensen, A. P., Rosenberg, M. D., Benedek, M., Chen, Q., Fink, A., Qiu, J., Kwapil, T. R., Kane, M. J., & Silvia, P. J. (2018). Robust prediction of individual creative ability from brain functional connectivity. Proceedings of the National Academy of Sciences of the United States of America, 115(5), 1087–1092.
Berkowitz, A. L., & Ansari, D. (2008). Generation of novel motor sequences: the neural correlates of musical improvisation. Neuroimage, 41(2), 535–543.
Buckner, R. L., & Carroll, D. C. (2007). Self-projection and the brain. Trends in Cognitive Sciences, 11(2), 49–57.
Catani, M., de Schotten, M. T., Slater, D., & Dell'Acqua, F. (2013). Connectomic approaches before the connectome. Neuroimage, 80, 2–13.
Costa, P. T., & McCrae, R. R. (1992). Neo PI-R professional manual. Odessa: PsychologicalAssessment Resources.
Curtis, C. E., & D'Esposito, M. (2003). Persistent activity in the prefrontal cortex during working memory. Trends in Cognitive Sciences, 7(9), 415–423.
Dandan, T., Wenfu, L., Tianen, D., Nusbaum, H. C., Jiang, Q., & Qinglin, Z. (2013). Brain mechanisms of valuable scientific problem finding inspired by heuristic knowledge. Experimental Brain Research, 228(4), 437–443.
Dietrich, A., & Kanso, R. (2010). A review of EEG, ERP, and neuroimaging studies of creativity and insight. Psychological Bulletin, 136(5), 822–848.
Dreher, J.-C., & Berman, K. F. (2002). Fractionating the neural substrate of cognitive control processes. Proceedings of the National Academy of Sciences, 99(22), 14595–14600.
Feist, G. J. (1998). A meta-analysis of personality in scientific and artistic creativity. Personality and Social Psychology Review, 2(4), 290–309.
Feist, G. J., & Barron, F. X. (2003). Predicting creativity from early to late adulthood: intellect, potential, and personality. Journal of Research in Personality, 37(2), 62–88.
Fink, A., & Benedek, M. (2014). EEG alpha power and creative ideation. Neuroscience and Biobehavioral Reviews, 44, 111–123.
Fink, A., Grabner, R. H., Benedek, M., Reishofer, G., Hauswirth, V., Fally, M., Neuper, C., Ebner, F., & Neubauer, A. C. (2009). The creative brain: investigation of brain activity during creative problem solving by means of EEG and FMRI. Human Brain Mapping, 30(3), 734–748.
Fink, A., Grabner, R. H., Gebauer, D., Reishofer, G., Koschutnig, K., & Ebner, F. (2010). Enhancing creativity by means of cognitive stimulation: evidence from an fMRI study. Neuroimage, 52(4), 1687–1695.
Greicius, M. D., Krasnow, B., Reiss, A. L., & Menon, V. (2003). Functional connectivity in the resting brain: a network analysis of the default mode hypothesis. Proceedings of the National Academy of Sciences, 100(1), 253–258.
Greicius, M. D., Supekar, K., Menon, V., & Dougherty, R. F. (2009). Resting-state functional connectivity reflects structural connectivity in the default mode network. Cerebral Cortex, 19(1), 72–78.
Honey, C., Sporns, O., Cammoun, L., Gigandet, X., Thiran, J.-P., Meuli, R., & Hagmann, P. (2009). Predicting human resting-state functional connectivity from structural connectivity. Proceedings of the National Academy of Sciences, 106(6), 2035–2040.
Jung, R. E., Grazioplene, R., Caprihan, A., Chavez, R. S., & Haier, R. J. (2010). White matter integrity, creativity, and psychopathology: disentangling constructs with diffusion tensor imaging. PLoS One, 5(3), e9818.
Jung, R. E., Mead, B. S., Carrasco, J., & Flores, R. A. (2013). The structure of creative cognition in the human brain. Frontiers in Human Neuroscience, 7, 330.
Jung-Beeman, M., Bowden, E. M., Haberman, J., Frymiare, J. L., Arambel-Liu, S., Greenblatt, R., Reber, P. J., & Kounios, J. (2004). Neural activity when people solve verbal problems with insight. PLoS Biology, 2(4), e97.
Kleibeuker, S. W., Koolschijn, P. C. M., Jolles, D. D., De Dreu, C. K., & Crone, E. A. (2013). The neural coding of creative idea generation across adolescence and early adulthood. Frontiers in Human Neuroscience, 7, 15.
Koechlin, E., Ody, C., & Kouneiher, F. (2003). The architecture of cognitive control in the human prefrontal cortex. Science, 302(5648), 1181–1185.
Li, W., Li, X., Huang, L., Kong, X., Yang, W., Wei, D., Li, J., Cheng, H., Zhang, Q., & Qiu, J. (2014). Brain structure links trait creativity to openness to experience. Social Cognitive and Affective Neuroscience, 10(2), 191–198.
Li, W., Yang, J., Zhang, Q., Li, G., & Qiu, J. (2016). The association between resting functional connectivity and visual creativity. Scientific Reports, 6, 25395.
Liu, S., Erkkinen, M. G., Healey, M. L., Xu, Y., Swett, K. E., Chow, H. M., & Braun, A. R. (2015). Brain activity and connectivity during poetry composition: toward a multidimensional model of the creative process. Human Brain Mapping, 36(9), 3351–3372.
Mason, M. F., Norton, M. I., Van Horn, J. D., Wegner, D. M., Grafton, S. T., & Macrae, C. N. (2007). Wandering minds: the default network and stimulus-independent thought. Science, 315(5810), 393–395.
Mok, L. W. (2014). The interplay between spontaneous and controlled processing in creative cognition. Frontiers in Human Neuroscience, 8, 663.
Mumford, M. D. (2002). Social innovation: ten cases from Benjamin Franklin. Creativity Research Journal, 14(2), 253–266.
Park, H. R., Kirk, I. J., & Waldie, K. E. (2015). Neural correlates of creative thinking and schizotypy. Neuropsychologia, 73, 94–107.
Petrides, M. (2005). Lateral prefrontal cortex: architectonic and functional organization. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 360(1456), 781–795.
Qiu, J., Li, H., Jou, J., Liu, J., Luo, Y., Feng, T., Wu, Z., & Zhang, Q. (2010). Neural correlates of the “Aha” experiences: evidence from an fMRI study of insight problem solving. Cortex, 46(3), 397–403.
Raichle, M. E., MacLeod, A. M., Snyder, A. Z., Powers, W. J., Gusnard, D. A., & Shulman, G. L. (2001). A default mode of brain function. Proceedings of the National Academy of Sciences of the United States of America, 98(2), 676–682.
Runco, M. A., & Jaeger, G. J. (2012). The standard definition of creativity. Creativity Research Journal, 24(1), 92–96.
Saggar, M., Quintin, E.-M., Kienitz, E., Bott, N. T., Sun, Z., Hong, W.-C., Chien, Y.-h., Liu, N., Dougherty, R. F., & Royalty, A. (2015). Pictionary-based fMRI paradigm to study the neural correlates of spontaneous improvisation and figural creativity. Scientific Reports, 5, 10894.
Schacter, D. L., Addis, D. R., Hassabis, D., Martin, V. C., Spreng, R. N., & Szpunar, K. K. (2012). The future of memory: remembering, imagining, and the brain. Neuron, 76(4), 677–694.
Silvia, P. J., Winterstein, B. P., Willse, J. T., Barona, C. M., Cram, J. T., Hess, K. I., Martinez, J. L., & Richard, C. A. (2008). Assessing creativity with divergent thinking tasks: exploring the reliability and validity of new subjective scoring methods. Psychology of Aesthetics, Creativity, and the Arts, 2(2), 68–85.
Silvia, P. J., Nusbaum, E. C., Berg, C., Martin, C., & O’Connor, A. (2009). Openness to experience, plasticity, and creativity: exploring lower-order, high-order, and interactive effects. Journal of Research in Personality, 43(6), 1087–1090.
Smith, S. M., Vidaurre, D., Beckmann, C. F., Glasser, M. F., Jenkinson, M., Miller, K. L., Nichols, T. E., Robinson, E. C., Salimi-Khorshidi, G., & Woolrich, M. W. (2013). Functional connectomics from resting-state fMRI. Trends in Cognitive Sciences, 17(12), 666–682.
Sporns, O. (2013). The human connectome: origins and challenges. Neuroimage, 80, 53–61.
Stein, M. I. (1953). Creativity and culture. The Journal of Psychology, 36(2), 311–322.
Sternberg, R. J., & Lubart, T. I. (1996). Investing in creativity. American Psychologist, 51(7), 677–688.
Sun, J., Chen, Q., Zhang, Q., Li, Y., Li, H., Wei, D., Yang, W., & Qiu, J. (2016). Training your brain to be more creative: Brain functional and structural changes induced by divergent thinking training. Human Brain Mapping, 37, 3375–3387.
Sutin, A. R., Beason-Held, L. L., Resnick, S. M., & Costa, P. T. (2009). Sex differences in resting-state neural correlates of openness to experience among older adults. Cerebral Cortex, 19(12), 2797–2802.
Takeuchi, H., Taki, Y., Sassa, Y., Hashizume, H., Sekiguchi, A., Fukushima, A., & Kawashima, R. (2010a). Regional gray matter volume of dopaminergic system associate with creativity: evidence from voxel-based morphometry. Neuroimage, 51(2), 578–585.
Takeuchi, H., Taki, Y., Sassa, Y., Hashizume, H., Sekiguchi, A., Fukushima, A., & Kawashima, R. (2010b). White matter structures associated with creativity: evidence from diffusion tensor imaging. Neuroimage, 51(1), 11–18.
Takeuchi, H., Taki, Y., Hashizume, H., Sassa, Y., Nagase, T., Nouchi, R., & Kawashima, R. (2012). The association between resting functional connectivity and creativity. Cerebral Cortex, 22(12), 2921–2929.
Whitney, C., Jefferies, E., & Kircher, T. (2011). Heterogeneity of the left temporal lobe in semantic representation and control: priming multiple versus single meanings of ambiguous words. Cerebral Cortex, 21(4), 831–844.
Wu, X., Yang, W., Tong, D., Sun, J., Chen, Q., Wei, D., Zhang, Q., Zhang, M., & Qiu, J. (2015). A meta-analysis of neuroimaging studies on divergent thinking using activation likelihood estimation. Human Brain Mapping, 36, 2703–2718.
Zhu, F., Zhang, Q., & Qiu, J. (2013). Relating inter-individual differences in verbal creative thinking to cerebral structures: an optimal voxel-based morphometry study. PLoS One, 8(11), e79272.
Acknowledgments
This research was supported by the National Natural Science Foundation of China (31470981; 31571137; 31500885; 31600878; 31771231), Project of the National Defense Science and Technology Innovation Special Zone, Chang Jiang Scholars Program, National Outstanding Young People Plan, the Program for the Top Young Talents by Chongqing, the Fundamental Research Funds for the Central Universities (SWU1709568, SWU1609177), the Postgraduate Science Innovation Foundation of Chongqing (CYB18109), Natural Science Foundation of Chongqing (cstc2015jcyjA10106), Fok Ying Tung Education Foundation (151023), the Research Program Funds of the Collaborative Innovation Center of Assessment toward Basic Education Quality at Beijing Normal University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Brain Imaging Center Institutional Review Board of Southwest China University and with the standards of the Declaration of Helsinki (1991).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Sun, J., Shi, L., Chen, Q. et al. Openness to experience and psychophysiological interaction patterns during divergent thinking. Brain Imaging and Behavior 13, 1580–1589 (2019). https://doi.org/10.1007/s11682-018-9965-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-018-9965-2