Abstract
Soil respiration (Rs) is important for transporting or fixing carbon dioxide from the atmosphere, and even diminutive variations can profoundly influence the carbon cycle. However, the Rs dynamics in a loess alpine hilly region with representative sensitivity to climate change and fragile ecology remains poorly understood. This study investigated the correlation and degree of control between Rs and its photosynthetic and environmental factors in five subalpine forest cover types. We examined the correlations between Rs and variables temperature (T10) and soil moisture content at 10 cm depth (W10), net photosynthetic rate (Pn) and soil properties to establish multiple models, and the variables were measured for diurnal and monthly variations from September 2018 to August 2019. The results showed that soil physical factors are not the main drivers of Rs dynamics at the diel scale; however, the trend in the monthly variation in Rs was consistent with that of T10 and Pn. Further, Rs was significantly affected by pH, providing further evidence that coniferous forest leaves contribute to soil acidification, thus reducing Rs. Significant exponential and linear correlations were established between Rs and T10 and W10, respectively, and Rs was positively correlated with Pn. Accordingly, we established a two-factor model and a three-factor model, and the correlation coefficients (R2) was improved to different degrees compared with models based only on T10 and W10. Moreover, temperature sensitivity (Q10) was the highest in the secondary forest and lowest in the Larix principis-rupprechtii forest. Our findings suggest that the control of Rs by the environment (moisture and temperature) and photosynthesis, which are interactive or complementary effects, may influence spatial and temporal homeostasis in the region and showed that the models appropriately described the dynamic variation in Rs and the carbon cycle in different forest covers. In addition, total phosphorus (TP) and total potassium (TK) significantly affected the dynamic changes in Rs. In summary, interannual and seasonal variations in forest Rs at multiple scales and the response forces of related ecophysiological factors, especially the interactive driving effects of soil temperature, soil moisture and photosynthesis, were clarified, thus representing an important step in predicting the impact of climate change and formulating forest carbon management policies.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Climate warming from 1880 to 2012 has led to an 0.85 °C increase in the global mean temperatures on land and ocean surfaces (Zhou et al. 2018). Indeed, climate change has huge potential to damage plant growth, water storage, agricultural production and economic activities (Chesney et al. 2017) and exacerbate drought and ecological imbalances. CO2 also can affect global temperature (Friedlingstein et al. 2014). Soil respiration (Rs) is important for transporting or fixing CO2 in the atmosphere, which is one of the most important carbon fluxes (Wu et al. 2020; Raich et al. 2002). The global soil organic carbon (SOC) pool is approximately twice as large as the OC pool in atmospheric and terrestrial organisms and more than tenfold larger than that caused by human activity (e.g., fuels combustions) (Lal 2008; Hashimoto 2012; Chen et al. 2020). Small variations in Rs may profoundly influence the carbon cycle and thus affect regional and even global climates through a variety of feedback processes. A potential positive feedback between warmer temperatures and intensive Rs could eventually accelerate global warming (Vargas et al. 2010a, b).
Therefore, information about Rs and its controlling environmental factors is of great significance in estimating the concentration of CO2 in the atmosphere and its impact on global climate change, especially in response to the continuing trend in global warming trend. Monitoring soil temperatures in the field is an effective way to study the response of Rs to changes in soil temperature (Wertin et al. 2018) and provide a theoretical reference for probing the effects of long-term warming on soil C dynamics (Crowther et al. 2013). The Rs rates are dominated by soil temperatures (Ts) and soil moisture (SWC), which directly affect soil microorganisms and plant root activity and have indirect effects by changing substrate supply and plant growth (Hanson et al. 2000). A moderate temperature increase can accelerate soil humus decomposition and promote autotrophic respiration, thereby facilitating Rs activity. SWC, the main driver of net primary productivity, can intensely influence the input of C from litter and decompositions of soil organic matter, thereby affecting the output heterotrophic respiration and C output (Moyano et al. 2013). To date, the temperature sensitivity (Q10) of Rs has been shown to decrease with increasing temperature and decreasing SWC (Flanagan and Johnson 2005). Other studies have shown that different forest types differ in the major factors that affect temporal and spatial variation in Rs, including soil temperature and moisture, root biomass, litter inputs, microbial populations, plant metabolism, other annually or seasonally fluctuating conditions and processes, and even plant phonological patterns (Curiel Yuste et al. 2010; Sheng et al. 2010; Zeng et al. 2014). Rs is also influenced by soil properties that can change soil microbial community composition (Landesman et al. 2014), including soil pH, total N, available P, and litter C (Gao et al. 2018; Feng et al. 2014).
Traditionally, the diurnal and seasonal variations in Rs have been expressed as empirical functions of Ts or SWC for various ecosystems (Heinemeyer et al. 2012). However, plant photosynthetic activity plays a significant role in regulating and forecasting Rs, especially in drylands (semiarid and arid areas), which have low levels of microbial activity and organic C in the soil; moreover, litter can lead to rhizosphere respiration becoming the dominant factor in Rs (Jia et al. 2018) because the source of C for the rhizosphere respiration of plants is provided by photosynthesis. Therefore, Ts and SWC may not fully represent Rs across multiple spatial and temporal scales. In addition, diurnal and weekly variation in Rs may be closely related to canopy photosynthesis, while the correlation between Rs and canopy photosynthesis (Hölttä 2010)under seasonal and interannual variation needs to be assessed by estimating productivity (Vargas et al. 2010a, b). However, due to the limitations of complex local geographical conditions and other problems, it is difficult to evaluate productivity using an eddy correlation system, a method to explore the annual variation law of Rs via single leaf photosynthesis (Wertin et al. 2018). In addition, after a large area of near-natural stand transformation occurs, ecosystems with the same stand type will appear as "fragmented" (large-scale pure forest planting area disappears and is replaced by other tree species), which adds to the difficulties and uncertainties in macroscopic analysis of the photosynthetic characteristics of different stand types. Consequently, it may be more accurate to assess the correlation by measuring the combination of single leaf photosynthesis with Rs.
The study site in the eastern part of the Qinghai-Tibet Plateau is representative of areas that are sensitive to climate change and of fragile, complex ecological environments with unique geographical features (Yao et al. 2005; Fu et al. 2006; Liu and Chen 2000). In recent years, arid and semiarid areas have been identified as potential C sinks (Ardö and Olsson 2003). However, as a major component of the semiarid and arid region, the response to the carbon cycle in various ecosystems under climate change in the Qinghai Tibet Plateau is poorly understood, especially in subalpine forest ecosystems (in contrast to the relatively well-studied Rs component in alpine, subalpine meadow and permafrost ecosystems) (Ren et al. 2017; Li et al. 2017; Peng et al. 2015). Forest Rs mainly emerges from the root system, microbial community and soil organic matter metabolism. The habitat provided by different forest stands determines which ecological and biophysiological factors lead to differences in Rs. Therefore, the annual and seasonal changes in remote sensing for specific types of subalpine forest ecosystems on the Qinghai-Tibet Plateau, the response to relevant ecological and physiological factors, and the sensitivity to temperature need to be further explored and studied on a multispatial scale.
Hence, we established a fitting model between the Rs and the SWC, the Ts and combination of SWC and Ts to illustrate the interaction of temperature and moisture on Rs. Moreover, we introduce Pn as the main evaluation factor to establish a three-factor model to more comprehensively assess Rs regulation and response to various factors in this research area to provide direction and theoretical support for the study of Rs in typical stands of subalpine forest ecosystems in alpine hilly areas. The objectives were to (1) reveal any multitemporal scale changes in Rs in different forest covers; (2) test for a correlation between Rs and Ts, SWC and combination of Ts and SWC; (3) evaluate the effect of single leaf photosynthesis on Rs; (4) compare the adaptability of different forest covers to warming trends based on Q10; and (5) investigate the correlation between Rs and other ecological environmental factors.
Materials and methods
Study site
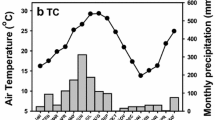
The research was carried out at the several forest reserves in Datong County (37°23′N, 101°51′E) in eastern Qinghai Province, including Tal Gou, Yang Jia Zhai, and An Men Tan. This region is the transition zone between the Qinghai-Tibet Plateau and the Loess Plateau, with an altitude of 2280–4622 m. Typical continental climate predominates in this area, with an annual mean temperature and relative humidity of 4.9 ℃ and 56%, respectively. The annual mean precipitation and evapotranspiration are 523.3 mm and 1762.8 mm, respectively. April to September (the plant growing season) is the rainy season in which 87% of the rainfall occurs, and the dry season is usually from November to March (Fig. 1). Major afforestation species include Picea crassifolia Kom., Juniperus przewalskii Kom., Populus cathayana Rehd., Larix principis-rupprechtii Mayr, Betula platyphylla Suk., and Caragana korshinskii Kom.
Experimental plots
Five representative forest covers containing secondary forest (S1), L. principis-rupprechtii (S2), P. crassifolia (S3), P. cathayana (S4) and B. platyphylla (S5) plantations were chosen as experimental plots during the spring through early fall of 2018. All slope directions were shady, and slopes did not exceed 20°. The total number of plots (the fixed size was 20 × 20 m) was 15, which included three replicates (a similar plot was selected according to afforestation data from the local forestry bureau) per forest cover, and each plot was separated by a buffer of at least 20 m. Elevation, slope, tree height, diameter at breast height (DBH), forest age, stand density, canopy density and physical and chemical properties in the topsoil (0–20 cm) were measured for each plot (Tables 1, 2). The main vegetation types of secondary forest were B. platyphylla., L. principis-rupprechtii, P. cathayana, and P. crassifolia (not long after replanting, the forest age was 10 years) present at a ratio of 3:3:2:2.
Experimental design and measurements
Soil physicochemical properties
Measurements (total two) of all variables were completed in August 2019 according to government standards (Tables 1, 2). Five replicates per plot of undisturbed soil and the soil profile at 0–20 cm depth were collected with cutting rings, placed in separate sealed bags and brought to the laboratory to determine the physical properties of soil. Soil pH can be directly determined with a pH meter (Gao et al. 2018). Soil bulk density was determined using the method of White (1988). Soil samples were dried, ground, and sievied to determine the soil organic carbon (SOC), total nitrogen (TN), total phosphorus (TP), and total potassium (TK) contents as previously described (Clark et al. 1998; Mitchell et al. 1999; Udelhoven et al. 2003; West et al. 1989).
Soil respiration
From September 2018 to August 2019, in each plot, five PVC soil collars (20 cm in diameter × 10 cm in height) were randomly inserted to a 5 cm depth into the corresponding soil cores, which were extracted with a rubber hammer for Rs measurement. Rs was measured using a LI-8100 portable soil CO2 flux system (LI-Cor Inc., Lincoln, NE, USA) (Xu et al. 2018), soil temperatures at a depth of 10 cm (T10, °C) were measured with a thermocouple probe (LI-8100–201) connected to the LI-8100 system, and moisture in the 0–10 cm layer (W10, % vol) near the collar was measured simultaneously with a time-domain reflectometry (TDR) probe (model TDR300, Spectrum Technologies, Plainfield, IL, USA) (Gao et al. 2018). All indicators were observed between 09:00 and 11:00 h at least every 2 weeks because of their accuracy in estimating the daily mean value (Xu and Qi 2001a). In addition, in October 2018 and January, April, and July 2019, we measured diurnal variation every 2 h from 09:00 h to 07:00 h the next day. The final calculated values reflected the average of 5 repeated measurements. Before sampling, the plants in the collars should be carefully removed.
Photosynthetic variables
Pn was determined for plants around each collar using a LI − COR 6400 photosynthesis system (LI − COR, Lincoln, USA) using randomly selected five fully expanded and representative upper leaves for broad-leaf species P. cathayana and B. platyphylla and a sprig with numerous leaves for coniferous species L. principis-rupprechtii and P. crassifolia at the same time as Rs was measured, including measurement of diurnal variations (daytime: 09:00–19:00 h). After each measurement, the leaf area in the chamber was estimated, and the gas exchange flux was modified accordingly. The leaf area of the plants to be measured was estimated using the leaf area index (LAI), which was determined using fixed points by a canopy analyzer and analyzed by WinSCANOPY2006a. The variation in Pn in S1 was obtained by calculating the ratio of total net photosynthetic yield to the total leaf area of the canopy of different tree species in the sample plot.
Model calculation
To quantify the relationship between Rs, T10 and W10, we established correlation models to analyze and compare the temperature sensitivity in different forest covers. The linear and nonlinear regression models were as follows:
where a, b and c are undetermined parameters. Rs is soil respiration.
Q10 was calculated by the following formula:
Data analyses
Data are represented as the means ± standard errors (SE). Means for each variable were analyzed using a one-way ANOVA, and significant differences among the means were tested by Duncan's multiple range test using a significance level of α = 0.05 or 0.01 via SPSS 26.0 (IBM, Armonk, NY, USA). Means for several important parameters were tested for correlations using a Pearson correlation analysis. Simple linear regression analyses were used to examine the relationships between relative changes in Rs and Q10 values, and linear or nonlinear regression was used to evaluate the relationships between Rs and SOC, TN, TP, TK, soil C: N ratio, and Pn, which described the effects of biotic and abiotic factors on Rs. Results were plotted using Origin 2018 software (OriginLab, Massachusetts, USA).
Results
Diurnal variations in R s and P n
Maximum and minimum values for Rs were measured at 15:00 − 17:00 and 5:00 − 7:00, respectively (Fig. 2A). Rs of broad-leaf species was markedly higher than that of coniferous species during spring, summer, and autumn. Compared with the pure forest (S2–S5), the mixed secondary forest (S1) had significantly higher values, except in winter (p < 0.05). During winter, no dramatic variation was detected in any forest covers (p > 0.05).
Diurnal variations in (A) soil respiration (Rs), (B) soil temperature (T10) and (C) soil moisture content (W10) at 10 cm depth, and (D) net photosynthetic rate (Pn) for different forest covers: Secondary forest (S1), L. principis-rupprechtii (S2), P. crassifolia (S3), P. cathayana (S4), and B. platyphylla (S5) plantations in October 2018 and January, April, and July 2019. Vertical bars represented the means ± SE
The diurnal variation in T10 in each stand did not significantly fluctuate, except in April, and the temperature was higher during the day than at night. Moreover, the amplitude of variation was marginal in W10, with only a certain fluctuation in April and July (Fig. 2B, C). The variation in W10 was remarkable in S4 during summer.
As shown in Fig. 2D, P. cathayana. and L. principis-rupprechtii were not measured in the winter, because the leaves had fallen off completely. The diurnal variations in Pn in the ved species emerged as a bimodal type, which was different from the unimodal type of coniferous species. Furthermore, no remarkable diurnal variations in Pn were demonstrated with the five forest covers during the winter, and only those of S1 and S3 represented a significant change.
Monthly variations in R s, T 10, W 10 and P n
As shown in Fig. 3, the trend of T10 decreased from September to January and then rebounded until August; with the lowest values in January. The temperature range in this area is large, and reaches − 6.23 to −.94 in January and 12.07 –3.15 in August. Overall, significant differences were not observed between the forest covers, except in January, July and August.
Monthly variations in soil temperature (T10) and (C) soil moisture content (W10) at 10 cm depth, soil respiration (Rs), and net photosynthetic rate (Pn) in different forest covers (Secondary forest (S1), L. principis-rupprechtii (S2), P. crassifolia (S3), P. cathayana (S4), and B. platyphylla (S5) plantations) from September 2018 to August 2019. Vertical bars represented the means ± SE
The W10 values were highest in April, fluctuated between May and August. During the spring and summer, the W10 values of forest covers represented extremely strong significance (p < 0.01). The seasonal coefficients of variation (CV) were 40.59%, 40.09%, 39.23%, 34.41%, and 36.54%, respectively, which demonstrated severe fluctuations in the water changes in the plantations of the five forest stands. In addition, the W10 of S4 was apparently lower than that of other forests during the growing season.
The trend of Rs changes with month was similar to that of T10. The Rs readings of S1 were markedly higher than those of the other forest stands, while no significant difference was observed between S2 and S3. During the nongrowing season (from October to March in the study site), the Rs values of P. cathayana. plantations were dramatically lower than the others. The CV was 26.86%, 15.59%, 13.94%, 26.51%, and 21.94% for S1 − S5.
The variation regulation of Pn was similar to that of Rs throughout the year, except for the values in S3, which were significantly higher than those of S1 and S5 during the winter. The Pn of S4 was significantly lower than that of the others, and no conspicuous difference was exhibited between S1 and S5.
Fitting relationship between the R s and the T 10, W 10 and combination of T 10 and W 10
A significant exponential correlation was observed between Rs and T10 for the five forest covers (p < 0.05). The highest R2 was observed in S1 (0.716), but it did not exceed 0.6 in the three other areas (Fig. 4). Q10 was highest in S1 and lowest in S2. The Q10 value of broad-leaf forest was remarkably higher than that of coniferous forest (Table 3).
Exponential and linear fitting relationship between Rs and T10 and W10, respectively. R2 represents the goodness of model fit after adjustment, the same as below. S1, S2 − S5 refer to five representative forest covers containing secondary forest (S1), L. principis-rupprechtii (S2), P. crassifolia (S3), P. cathayana (S4) and B. platyphylla (S5) plantations
The goodness of fit for W10 was weaker, which was 35% less than the correlation between Rs and T10. Furthermore, the fitted Rs model revealed remarkable effects of W10 on forest stands.
For the two-factor model with respect to T10 and W10, R2 was improved to different degrees in all forest covers, which explained 51.8% − 76.1% of the temporal variation in Rs.
Response of R s to other ecological factors
Rs had a positive correlation with all ecological factors (Fig. 5). However, significant correlations were not observed between Rs and the SOC, TK and soil C:N ratio (p > 0.05). The correlation between Rs and Pn is shown in Fig. 6. Research has found that Rs increases with increasing Pn in various forest stands. Moreover, the slope of the relationship was greater for measurements for S1 and S5, while the lowest was revealed for S2. Additionally, R2 value was lowest in S4 and highest in S1.
The relationship between R s and the combination of T10, W10 and P n
T10, W10, Pn had significant effects on Rs (Figs. 4–6), and photosynthesis can provide the main supply of soil organic substrate for Rs. Pn was an important part of the assessment of Rs via the principal component analysis (mean variance contribution rate was 14.2%, not shown); thus, it was necessary to explore the combined effects of the three factors on Rs. Based on the model between Rs and the combination of T10 and W10 validated by predecessors, we further introduced Pn as a variable to establish the equation as follows: Rs = aebTWcPnd, where a, b, c and d are undermined parameters. The model (Table 4) shows that the model fitting effect improved by introducing the Pn value was compared to the relationship between Rs and combination of the T10 and W10 (Table 3).
Discussion
Ts and soil water content (SWC) are the main environmental factors controlling Rs, and their changes will affect Rs in different stand types to different degrees. Limited diurnal variations occurred T10 and W10 in different forest covers because of the influence of closed canopies, and the result was different from the fluctuation of Rs, indicating that soil physical factors are not the main drivers of Rs dynamics at the diel scale. These phenomena were in line with findings in arid and semiarid regions(Jia et al. 2018). Furthermore, the Rs of the broad-leaved species was much higher than that of coniferous species during spring, summer, and autumn, which may be due to the increased alduronic acid secreted by the leaves of S2 and S3, which caused soil acidification (refer to the pH value in Table 1) and slow decomposition of litter, thereby inhibiting the activity of microorganisms to reduce heterotrophic respiration. The Rs values in the daytime (photosynthesis was carried out synchronously) were greater than those at night, indicating that photosynthesis was coupled with respiratory mechanisms on a daily time scale. The seasonal variation trend of Rs was conspicuously regulated by T10 and W10, in accordance with Chen’s research on different subalpine ecosystems (Chen et al. 2014), and demonstrated the higher sensitivity of Rs to T10 and W10 on a seasonal scale compared to that on the daily scale. Moreover, photosynthetic productivity was an important driver of soil CO2 flux, not only on the daily time scale, but also on the seasonal and annual scales (Michael et al. 2008). In our study, the monthly variations in Rs and Pn maintained a high degree of consistency in different forest covers. Even in the winter when S2 and S4 lacked photosynthetic products, their Rs values simultaneously decreased to the minimum values. Our research also found that the Rs of S1 was significantly higher than that of pure forests throughout the year. By comparing the soil characteristics of each forest (Table 1), we found that the soil bulk density and the SOC, TN, TP and TK were significantly lower and higher than those of other forests, respectively. This result showed that the secondary forest structure (multilayer forest) was the most reasonable and could effectively alleviate intraspecies competition and promote plant growth and metabolism, thereby providing material conditions for carbon cycling.
Ts contributes to regulating Rs by its effects on plant root growth, microbial activity, and litter decomposition; therefore, an exponential regression model was established for both (Song et al. 2013). Based on previous studies, we adjusted the parameters of the model to apply to the study area, then established a significant exponential model to demonstrate the regulatory effect of T10 on Rs, thereby providing theoretical support for the prediction of CO2 flux variations in alpine hilly areas under global warming. The correlation coefficient between Rs and T10 ranged from 49.4% in S3 to 71.6% in S1. The reason for the difference may be due to the dissimilarity of the root systems and microbial activity (Chen et al. 2010), and even the regulation of photosynthesis by Ts (R2 = 0.73, p < 0.05) in plants may affect Rs. The correlation model of Rs and T can explain most of the seasonal and diel changes in soil C emissions, although the effect is not identical. Other factors such as soil moisture content affect Rs for a certain period of time. Therefore, we established a linear regression model of Rs and W10. The correlation between Rs and SWC was significantly different in five forest types, which might be because the variation in W10 affected the input of carbon and the decomposition of litter and soil organic matter, thus indirectly affecting heterotrophic respiration and carbon output (Moyano et al. 2013). The correlation coefficients of the model were significantly lower than those based on Rs and T10, which may have been caused by factors such as rainfall and severe evapotranspiration, and the related models established by SWC were not stable enough, as shown by the CV values (Fig. 3) for the forest types. In addition, sufficient precipitation occurred during study period, indicating that SWC was not a limiting factor for Rs.
Wang et al. (2006) believed that Ts is a great indicator for estimating Rs in a specific ecosystem, and verified the Q10 function to estimate the reliability of Rs, which is also confirmed by most ecosystems. The response of soil CO2 emission to Ts can be described by Q10 which is the Ts coefficient of the reaction (Ma et al. 2019). In this study, the Q10 value of broad-leaf forest was remarkably higher than that of coniferous forest, which was not in line with Zheng (Zheng et al. 2009), who found that the Q10 in deciduous forests was significantly higher than that in evergreen forests, and the Q10 in coniferous forests was significantly higher than that in evergreen broad-leaf forests. This difference may be because Q10 is not a long-term reflection of temperature sensitivity, but rather a comprehensive response to temperature fluctuations, root biomass and activity, humidity conditions, and other unknown variables (Janssens and Pilegaard 2003), thus resulting in indistinctness. Scholars have proposed that W10 thresholds exist in different forests. When W10 is higher than the threshold, the response of Rs to T10 may be confined by limiting aeration and CO2 diffusion, and the decrease in soil microbial activity in a low oxygen environment may also lead to the reduction of soil CO2 emissions, thereby restraining the sensitivity of Rs to T10 (Xu and Qi 2001b; Rey et al. 2002; Chen et al. 2010). Although T10 was not significantly different between the different forest stands, other ecological biological factors were significantly different, including SOC, TN, TP, TK, pH, soil bulk density and Pn. Moreover, Q10 was highest in S1 because it belonged to the enclosed forest protected from human activities, and S1 was characterized by a sufficient supply of soil organic substrate, higher root biomass, and microbial activity, and more complex and stable species composition compared to other forests. However, this result also implied that the carbon cycle might experience a greater disturbance under global warming. Therefore, the process of global warming and carbon cycle promote and influence each other and are complementary.
The combination of Ts and SWC on Rs is critical to understanding the mechanisms of climate control on Rs and its components (Ma et al. 2019). The response of Rs to T10 and W10 in this study area has been proven, and it is worth exploring whether there is an interactive or complementary effect of T10 and W10 on Rs. We established a two-factor model combining W10 and T10 compared to the model-based only on T10 or W10, which improved R2 to different degrees, this circumstance has been ascertained by others research (Zhang et al. 2010; Gao et al. 2018; Ma et al. 2019). According to our results, the variation trend of Rs increased with the escalation of W10 and T10. From May to August, when W10 remained basically stable, the increase in T10 promoted the increase in Rs by boosting the root system and microbial activity.
When exploring the response of Rs to other ecological environmental factors, we observed that the variation in Rs was not in conformity with that of SOC, TK and the soil C:N ratio, which was also confirmed by Gao et al. (2018) in a study of the correlation between Rs and environmental factors. In addition, Rs was weakly consistently correlated with TN and TP (Nielsen and Becky 2015; Wertin et al. 2018). The findings indicated that the combined indicators rather than a single factor regulate the changes of Rs, which complicates the evaluation of Rs (Okin et al. 2016). Soil properties can be influenced by forest types and act on soil microbial and root activity (Lu et al. 2014). Our research found that Rs can be significantly affected by pH, which is further evidence that coniferous forest leaves contribute to soil acidification, thus reducing Rs.
Previous research revealed that T10 and W10 together could explain more than 75% of the variation in Rs (Keith et al. 1997). Since the components that affect changes in Rs are not completely clear, we are particularly interested in the contribution of Pn to Rs with species of contrasting functional types. We analyzed the correlation between the Pn value and Rs of each forest cover type and found that Pn was significantly positively correlated with Rs, which is consistent with studies showing the close coupling of A. hymenoides and A. confertifolia leaf-level Pn and Rs (Wertin et al. 2017, 2018). Other studies have demonstrated the close correlation between Pn and Rs in dryland ecosystems (Chen et al. 2009). This result showed that the photosynthetic C supply acted as a driving factor for Rs and Pn, and regulated Rs by stimulating root respiration and heterotrophic respiration of root deposition. Moreover, studies have also suggested a more compact coupling between photosynthetic products and Rs than between Rs and other factors (Vargas et al.; 2010a, b; Jia et al. 2018). We can also support this result by analyzing the correlation between the Pn of a single-leaf horizontal and Rs; R2 was higher than that of the model based only on W10 and T10. Furthermore, the R2 between Pn and Rs was lowest in S4 and highest in S1. We speculated that S1 was located in the fenced-in zone, which was not disturbed by human activity, and that production and behaviors would introduce more substrate and change the mechanisms underlying underground C supply, thereby reducing the effect of photosynthesis on Rs. But why is the correlation coefficient lowest in S4 if it is an artificial forest subjected to more human disturbance? The daily change in Pn is bimodal with solar radiation, temperature, and humidity in most of the observation periods, and the severe water consumption characteristics of P. cathayana reduced the biomass and microbial activity of the forest. In addition, the high incidence of black spot disease in August severely inhibited leaf photosynthesis during period of growth and metabolism, as shown in Fig. 3. Therefore, to more comprehensively evaluate the main factors controlling Rs in different forest ecosystems in subalpine hilly areas, we introduced Pn to establish a three-factor model. The results exhibited a significant improvement in the fitting correlation coefficient compared to that of the two-factor model based on the combination of T10 and W10. Our results suggest that the control of Rs by the environment (moisture and temperature) and photosynthesis may be spatially and temporally homeostatic in the region; thus, the model was feasible for describing the dynamic variation in the Rs and the carbon cycle under different forest covers.
However, the pattern of underground C supply and distribution is not yet clear. Although these findings help fill some gaps in knowledge of Rs dynamics in different forest ecosystems under current climatic conditions, further research should focuse on the driving factors of autotrophic respiration and heterotrophic respiration, and the response of microorganisms to climate change needs to be explored.
Conclusion
During spring, summer, and autumn, the diurnal variations in Rs were significantly greater in the afternoon than at other times, and the Rs was pronouncedly higher for broad-leaved species than for coniferous species in the five typical forest covers. Our study also found that Rs could be significantly affected by soil pH, which provided further evidence that coniferous forest leaves contribute to soil acidification, thus reducing Rs. Furthermore, soil physical factors are not the main drivers of Rs dynamics at the diel scale; however, the trend in the monthly variation trend of Rs was consistent with that of T10 and Pn. The Rs of S1 was significantly higher than the value found in pure forests throughout the year.
The R2 for the model based on Rs and T10 was significantly higher than for the model based on Rs and W10. Moreover, the two-factor model that combined T10 and W10 with adjusted parameters improved the R2 value at a different level compared with the model based only on T10 and W10, indicating that the Rs was influenced by the combined effect of T10 and W10, which was interactive or complementary in different forest ecosystems in the alpine hilly region. The Q10 value for the broad-leaved forest was remarkably higher than for the coniferous forest, and the highest value was discovered in the secondary forest. Our findings showed that a combination of soil chemical properties rather than a single factor may regulate the changes of Rs, which complicates the evaluation of Rs.
By evaluating the correlation between the Pn at the single-leaf level and Rs, we proved that Pn was closely coupled with Rs in all the measured forests. Furthermore, the R2 between Pn and Rs was lowest in Populus cathayana plantations and highest in the secondary forest. The results showed that the three-factor model presented significant improvements in R2 compared to the two-factor model based on the combination of T10 and W10 and thus is, feasible for describing the dynamic variation in Rs.
References
Ardö J, Olsson L (2003) Assessment of soil organic carbon in semi-arid Sudan using GIS and the CENTURY model. Arid Environ 54(4):633–651
Chen SP, Lin GH, Huang JH, Jenerette GD (2009) Dependence of carbon sequestration on the differential responses of ecosystem photosynthesis and respiration to rain pulses in a semiarid steppe. Glob Change Biol 15(10):2450–2461
Chen BY, Liu SR, Ge JP, Chu JX (2010) Annual and seasonal variations of Q10 soil respiration in the sub-alpine forests of the Eastern Qinghai-Tibet Plateau. China Soil Bio Biochem 42(10):1735–1742
Chen YC, Luo J, Li W, Yu D, She J (2014) Comparison of soil respiration among three different subalpine ecosystems on eastern Tibetan Plateau. China Soil Sci Plant Nutr 60(2):231–241
ChenST WJ, Zhang TT, Hu Z (2020) Climatic, soil, and vegetation controls of the temperaturesensitivity (Q10) of soil respiration across terrestrial biomes. Glob Ecol Conserv 22:e00955
Chesney M, Lasserre P, Troja B (2017) Mitigating global warming: a real options approach. Ann Oper Res 255:465–506
Clark MS, Horwath WR, Shennan C (1998) Changes in soil chemical properties resulting from organic and low-input farming practices. Agron J 90(5):662–671
Crowther WT, Bradford MA, Johnson N (2013) Thermal acclimation in widespread heterotrophic soil microbes. Ecol Lett 16(4):469–477
Curiel Yuste J, Janssens IA, Carrar A, Ceulemans R (2010) Annual Q10 of soil respiration reflects plant phenologicalpatterns as well as temperature sensitivity[J]. Glob Change Biol 10(2):161–169. https://doi.org/10.1111/j.1529-8817.2003.00727.x
Feng YZ, Grogan P, Caporaso JG, Zhang H, Lin XG, Knight R, Chu HY (2014) pH is a good predictor of the distribution ofanoxygenic purple phototrophic bacteria in Arctic soils. Soil Biol Biochem 74(6):193–200
Flanagan LB, Johnson BG (2005) Interacting effects of temperature, soil moisture and plant biomass production on ecosystem respiration in a northern temperate grassland. Agric For Meteorol 130(3):237–253
Friedlingstein P, Andrew RM, Rogelj J, Peters GP, Canadell JG, Knutti R, Luderer G, Raupach MR, Schaeffer M, Vuuren DP (2014) Persistent growth of CO2 emissions and implications for reaching climate targets. Nat Geosci 7(10):709–715
Fu Q, Johanson CM, Wallace JM, Reichler T (2006) Enhanced mid-latitude tropospheric warming in satellite measurements. Science 312(5777):1179
Gao W, Huang ZQ, Ye GF, Yue XJ, Chen ZY (2018) Effects of forest cover types and environmental factors on soil respiration dynamics in a coastal sand dune of subtropical China. J For Res 29(6):1645–1655
Hanson PJ, Edwards NT, Garten CT, Andrews JA, Rustad LE, Huntingdon TG, Boone RD (2000) Separating root and soil microbial contributions to soil respiration: A review of methods and observations. Biogeochemistry 48(1):115–146
Hashimoto S (2012) A new estimation of global soil greenhouse gasfluxes using a simple data-oriented model. PLoS ONE 7(8):e41962
Heinemeyer A, Wilkinson M, Vargas R, Subke JA, Casella E, Morison JI, Ineson P (2012) Exploring the Boverflow tap theory: linking forest soil CO2 fluxes and individualmycorrhizosphere components to photosynthesis. Biogeosciences 9(1):79–95
Hölttä MT (2010) The significance of phloem transport for the speed with which canopy photosynthesis and belowground respiration are linked. New Phytol 185(1):189–203
Janssens IA, Pilegaard KIM (2003) Large seasonal changes in Q10 of soil respiration in a beech forest. Glob Change Biol 9(6):911–918
Jia X, Zha T, Wang S, Bourque PA, Wang B, Qin S, Zhang Y (2018) Canopy photosynthesis modulates soil respiration in a temperate semi-arid shrubland at multiple timescales. Plant Soil 432:437–450
Keith H, Jacobsen KL, Raison RJ (1997) Effects of soil phosphorus availability, temperature and moisture on soil respiration in Eucalyptus pauciflora forest. Plant Soil 190(1):127–141
Lal R (2008) Sequestration of atmospheric CO2 in global carbon pools. Energy Environ Sci 1(1):86–100
Landesman WJ, Nelson DM, Fitzpatrick MC (2014) Soil propertiesand tree species drive ß-diversity of soil bacterial communities. Soil Biol Biochem 76:201–209
Li GY, Mu JP, Liu YZ, Sun S (2017) Effect of microtopography on soil respiration in an alpine meadow of the Qinghai-Tibetan plateau. Plant Soil 421(1–2):147–155
Liu X, Chen B (2000) Climatic warming in the Tibetan Plateau during recent decades. Int J Climatol 20(14):1729–1742
Lu XQ, Toda H, Ding FJ, Fang SZ, Yang WX, Xu HG (2014) Effect of vegetation types on chemical and biological properties of soils of karst ecosystems. Eur J Soil Biol 61(3):49–57
Ma MZ, Zang ZH, Xie ZQ, Chen QS, Xu WT, Zhao CM, Shen GZ (2019) Soil respiration of four forests along elevation gradient in northern subtropical China. Ecol Evol 9(22):12846–12857
Michael B, Rodeghiero M, Anderson-Dunn M, Dore S, Gimeno S, Drösler M, Williams M, Ammann C, Berninger F, Flechard C (2008) Soil respiration in European grasslands in relation to climate and assimilate supply. Ecosystems 11(8):1352–1367
Mitchell RJ, Marrs RH, Le MGD (1999) A study of the restoration of heathland on successional sites: changes in vegetation and soil chemical properties. J Appl Ecol 36(5):770–783
Moyano FE, Stefano M, Claire C (2013) Responses of soil heterotrophic respiration to moisture availability: an exploration of processes and models. Soil Biol Biochem 59:72–85
Nielsen UN, Becky AB (2015) Impacts of altered precipitation regimes on soil communities and biogeochemistry in arid and semi-arid ecosystems. Glob Change Biol 21(4):1407–1421
Okin GS, Moreno DLH, Patricia MS, Throop L, Enrique RV, Anthony JP, Wainwright J, Debra PFP (2016) Connectivity in dryland landscapes: shifting concepts of spatial interactions. Heather Ecol Environ 13(1):20–27
Peng F, XueX YQG, Tao W (2015) Warming effects on carbon release in a permafrost area of Qinghai-Tibet Plateau. Environ Earth Sci 73(1):57–66
Raich JW, Potter CS, Bhagawati D (2002) Bhagawati interannual variability in global soil respiration. Glob Change Bio 8(8):800–812
Ren F, Yang XX, Zhou HK, Chen L, Cao G, He JS (2017) Corrigendum: Contrasting effects of nitrogen and phosphorus addition on soil respiration in an alpine grassland on the Qinghai-Tibetan Plateau. Sci Rep 7(7):39895
Rey A, Pegoraro E, Tedeschi V, Parri ID, Jarvis PG, Valentini R (2002) Annual variation in soil respiration and its components in a coppice oak forest in Central Italy. Glob Change Biol 8(9):851–866
Sheng H, Yang YS, Yang ZJ, Chen GS, Xie JS, Guo JF, Zou SQ (2010) The dynamic response of soil respiration to land-use changes in subtropical China. Glob Change Biol 16(3):1107–1121
Song XZ, Yuan HY, Kimberley MO, Jiang H, Zhou GM, Wang HL (2013) Soil CO2 flux dynamics in the two main plantation forest types in subtropical China. Sci Total Environ 444:363–368
Udelhoven T, Emmerling C, Jarmer T (2003) Quantitative analysis of soil chemical properties with diffuse reflectance spectrometry and partial least-square regression: a feasibility study. Plant Soil 251(2):319–329
Vargas R, Baldocchi DD, Allen MF, Bahn M, Black TA, Collins SL, Yuste JC, Hirano T, Jassal RS, Pumpanen J (2010a) Looking deeper into the soil: biophysical controls and seasonal lags of soil CO2 production and efflux. Ecol Appl 20(6):1569–1582
Vargas R, Baldocchi DD, Allen MF, Bahn M, Black TA, CollinsSL YJC, Hirano T, Jassal RS, Pumpanen J, Tang JW (2010b) Looking deeper into the soil: biophysical controlsand seasonal lags of soil CO2 production and efflux. Ecol Appl A Pub Ecol Soc Am 20(6):1569–1582
Wang CK, Yang JY, Zhang QZ (2006) Soil respiration in six temperate forests in China. Glob Change Biol 12(11):2103–2114
Wertin TM, Belnap J, Reed SC (2017) Experimental warming in a dryland community reduced plant photosynthesis and soil CO2 efflux although the relationship between the fluxes remained unchanged. Funct Ecol 31(2):297–305
Wertin TM, Young K, Sasha CR (2018) Spatially explicit patterns in a dryland’s soil respiration and relationships with climate, whole plant photosynthesis and soil fertility. Oikos 127(9):1280–1290
West CPA, Mallarino P, Wedin WF (1989) Spatial variability of soil chemical properties in grazed pastures. Soil Sci Soc Am J 53(3):784–789
White I (1988) Measurement of soil physical properties in the field. In Steffen WL, Denmead OT (eds) Flow and transport in the natural environment: advances and applications. Springer Berlin Heidelberg, Berlin, pp 59–85
Wu X, Xu H, Tu DF, Liu GH (2020) Land use change and stand age regulate soil respiration by influencing soil substrate supply and microbial community. Geoderma 359:113991
Xu M, Qi Y (2001a) Soil-surface CO2 efflux and its spatial andtemporal variations in a young ponderosa pine plantation innorthern California. Glob Change Biol 7(6):667–677
Xu M, Qi Y (2001b) Spatial and seasonal variations of Q 10 determined by soil respiration measurements at a Sierra Nevadan forest. Global Biogeochem Cycles 15(3):687–696
Xu Z, Yin H, Zhao C (2018) Responses of soil respiration to warming vary between growing season and non-growing season in a mountain forest of southwestern China. Can J Soil Sci 98(1):70–76
Yao YZ, Wang YR, Li YH, Liu B (2005) The warming and drying of the loess plateau in China and its impact on the ecological environment. Resources Science (In Chinese) 5:67–75
Zeng X, Zhang W, Shen H, Xin Z (2014) Soilrespiration response in different vegetation types at MountTaihang China. CATENA 116(5):78–85
Zhang LH, Chen YN, Zhao RF, Li WH (2010) Significance of temperature and soil water content on soil respiration in three desert ecosystems in Northwest China. J Arid Environ 74(10):1200–1211
Zheng ZM, Yu GR, Fu YL, Wang YS, Sun XM, Wang YH (2009) Temperature sensitivity of soil respiration is affected by prevailing climatic conditions and soil organic carbon content: a trans-China based case study. Soil Biol Biochem 41(7):1531–1540
Zhou T, Ren L, Liu H, Lu J (2018) Impact of 1.5 °C and 2.0 °C global warming on aircraft takeoff performance in China. Sci Bull 63(11):700–707
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Project funding: This work was supported financially by the National Key Research and Development Plan Projects of China (2017YFC0504604).
The online version is available at http://www.springerlink.com
Corresponding editor: Zhu Hong
Rights and permissions
About this article
Cite this article
Li, Y., Lin, S., Chen, Q. et al. Response of soil respiration to environmental and photosynthetic factors in different subalpine forest-cover types in a loess alpine hilly region. J. For. Res. 33, 653–665 (2022). https://doi.org/10.1007/s11676-021-01340-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-021-01340-w