Abstract
Purpose
This study aimed to evaluate the prevalence and risk factors of secondary osteoporosis (OP) in patients with systemic lupus erythematosus (SLE) and provide a theoretical basis for clinical prevention and treatment of SLE.
Methods
Take systematic review and meta-analysis of relevant studies. Data sources are CINAHL databases, PubMed, Embase, Wan Fang, Weipu, and CNKI databases. Eligibility criteria are cross-sectional or case–control studies which analyzed the prevalence and risk factors of OP in SLE. Two authors independently screened all studies; a third author verified and identify controversial studies. The quality of the included articles was evaluated. Stata 11 and Rev-Man 5.2 software were used for data processing.
Results
Thirty-one articles were included, with a total sample size of 3089 SLE, including 529 OP cases and 2560 non-OP cases. Meta-analysis showed that the prevalence of OP among SLE was 16% (95% CI (0.12, 0.19)). The risk of OP in SLE cases compared with controls was significantly greater with OR of 2.03 (95% CI 1.33–3.10, P = 0.001). Age, disease duration, cumulative glucocorticoid dose, duration of glucocorticoid therapy, SLICC, and menopause had significant differences between two groups. No statistical differences of daily glucocorticoid dose, SLEDAI, and BMI were found between OP and non-OP cases.
Conclusions
Our study found a statistically significant increased risk of OP in SLE patients compared with controls. SLE patients should be actively screened for OP and its consequences. Larger longitudinal studies are needed to confirm this possible association.
The prevalence of OP in SLE was 16%. Compared with controls, the risk of OP in SLE was 2.03. There were significant differences of age, disease duration, cumulative glucocorticoid dose, time of glucocorticoid, SLICC, and menopause, while daily glucocorticoid dose, SLEDAI, and BMI had no statistical differences between OP and non-OP cases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Systemic lupus erythematosus (SLE) is an autoimmune disease involving multiple organ systems and the presence of anti-nuclear antibodies [1, 2], predominantly affecting women, particularly those of childbearing age [3]. Glucocorticoids (GCs) are potent anti-inflammatory and immunosuppressive agents, which are indispensable in the treatment of SLE [4]. However, long-term use and even abuse of GCs in SLE do great harms. Ruiz-Irastorza et al. [4] reported that GCs were associated with the development of hyperglycemia, coronary heart disease, osteoporosis, osteonecrosis, cataracts, and others.
Osteoporosis (OP), as one of the most serious complications of SLE, is characterized by a reduction of bone mass per unit volume and destruction of bone microstructure, leading to increased bone fragility and fracture risk in patients with SLE [5]. OP is common in SLE. Bultink [6] indicated that the incidence rate of osteopenia in SLE was 25–74%, OP was 1.4–60.5%. In China, Ji et al. [7] found that osteopenia was 41.7%, OP was 39%, which corresponds to the increased risk of fracture. Studies have reported that the incidence of fracture in SLE was 34.6%, seriously decreasing the quality of life of patients and upregulating mortality rates [8].
In decades, OP have recently received more attention for their damage in SLE. Di Munno et al. [9] reported that risk factors for OP in female patients with SLE were disease-specific (reduced physical activity, cytokines, renal impairment, endocrine factors) and non-disease-specific mechanisms (sunshine avoidance, GCs, immunosuppressants). Bultink indicated that the etiology of bone loss in SLE is multifactorial, including inflammation, serological factors, hormonal factors, metabolic factors, possibly genetic factors, and medication-induced adverse effects [10]. Studies have different opinions. The purpose of this study is to evaluate the risk factors of secondary OP in SLE patients and prevent the occurrence of secondary OP.
Methods
This review was conducted according to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines for systematic reviews and meta-analyses [11]. Key words and study eligibility criteria were determined. The protocol for the review was registered with PROSPERO (Registration number: CRD42018117580).
Literature search
Two investigators took systematic literature search in PubMed, Embase, CINAHL databases, Wan Fang, Weipu, and CNKI databases till December 2018. The medical subject heading (MeSH) search headings were all combinations of index terms: “Systemic lupus erythematosus” or “SLE” or “lupus” and “osteoporosis” or “OP” and “risk factors”. Eligibility criteria include observational studies which analyzes the prevalence and risk factors of OP in SLE. At the same time, the reference lists of identified manuscripts were hand-searched and scrutinized to identify other relevant publications. We took the search again before the final analyses on 06/01/2019.
Selection criteria
Inclusion criteria include (1) met SLE classification criteria revised by the American Society of Rheumatology (ACR); (2) included secondary OP and non-secondary OP groups; (3) OP diagnostic criteria which met World Health Organization (WHO) diagnostic criteria—Dual Energy X-ray Absorption Density (DXA). Patients were determined to be either normal (T score > − 1.0), osteogenic (T score < − 1.0), and osteoporotic (T score < − 2.5) [12].
Exclusion criteria include (1) history of taking estrogen, androgen, anticoagulant, and drugs affecting bone metabolism; (2) severe liver and kidney dysfunction, Cushing syndrome, history of thyroid and parathyroid diseases, ovariectomy, and other diseases affecting bone metabolism.
Study selection
Two authors independently screened the title and abstract of all included studies. We excluded unrelated studies. We took full-text review for the rest of the studies. Reviewers also took discussion to solve disagreement over study eligibility at both the title/abstract and full-text review stages. A third reviewer was identified in case of disagreement or uncertainty. Any disagreements were resolved by discussion.
Data extraction
The following information was extracted from each included study: first author, publication year, country, risk factors (age, disease duration, cumulative oral GCs doses, duration of GCs therapy, daily GCs dose, SLEDAI, SLE damage index (SLICC/ACR), menopause, and BMI), numbers of OP cases, numbers of SLE cases, method of ascertainment of OP, and study quality. Extracted data were extracted into a standardized Excel (Microsoft Corporation, Redmond, WA, USA) file. Missing information were obtained by email. The data were independently extracted by two authors.
Assessment of study quality
Two reviewers used the Newcastle-Ottawa Scale (NOS) and Agency for Healthcare Research and Quality (AHRQ) to assess methodological quality independently. NOS uses a “star” rating system (range from 0 star to 9 stars) to judge quality. Seven to 9 points were considered to be high quality, < 7 points were considered to be low quality [13]. AHRQ, 11-item checklist, an item would be scored “0” if it was answered “No” or “Unclear”, if it was answered “Yes”, then the item scored “1” [14]. Article quality was assessed as follows: low quality, 0–3; moderate quality, 4–7; and high quality, 8–11. Finally, to ensure the quality of research, we choose studies with a score of more than 7 points.
Data analysis
Meta-analysis was performed using Stata 11 and RevMan 5.2 software recommended by Cochrane Collaboration Network. Bi-categorized variables were analyzed by odds ratio (OR), and continuous variables were analyzed by mean difference (MD). Age, disease duration, cumulative oral GC dose, duration of GC therapy, daily GC dose, SLEDAI, SLICC, menopause, and BMI as research statistics were analyzed. Each effect was expressed by 95% confidence interval (CI), P < 0.05 was the significant difference. Heterogeneity was assessed visually with forest plots and quantified numerically using the I2 index. Non-heterogeneity (I2 < 50%) data used a fixed effect model, heterogeneity (I2 > 50%) data used sensitivity analysis to find out the causes of heterogeneity, and if there was no clinical heterogeneity, a random effect model was used. If there is clinical heterogeneity, subgroup analysis can be done according to the origin of heterogeneity.
Results
Study selection
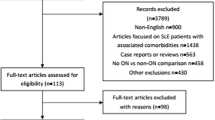
\The details of the study selection process are shown in Fig. 1. One thousand twelve potentially relevant articles were identified using the search strategy. After removal of duplicates, 742 remained. Following title/abstract review, animal model studies, case reports, and those with unavailable, 665 articles were excluded. The 77 remaining full-length articles were screened. Finally, 31 were included in the qualitative and quantitative analysis [14,15,16,17,18,19,20,21, 12, 22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44].
Study characteristics
The summary of the included study characteristics is shown in Table 1. Thirty-one identified studies matched the inclusion criteria, reporting on 3650 cases, healthy control group 561 cases and SLE 3089 cases. The included articles were published between 1985 and 2017. The sample size of the studies is different and the population is female dominated. This meta-analysis included 13 case–control studies and 18 cross-sectional studies. Most studies (16/31) were published in China. The diagnostic criteria for OP was mainly through DXA. The quality of studies evaluated by the NOS and AHRQ is shown in Table 1. The correlations between OP and risk factors in SLE patients are also shown in Table 2. “+” mainly refers to the development of osteoporosis related to those risk factors. “−” mainly refers to the development of osteoporosis not related to those risk factors. The blank indicates that the risk factor was not discussed in the article.
Prevalence of OP in SLE
The prevalence of OP in SLE was 4 to 42.4% in individual studies (Table 1). Meta-analyses revealed the prevalence of major OP was 16% (95% CI (0.12, 0.19)), with high heterogeneity (I2 = 88.9%) (Fig. 2a). After subgroup analysis, the prevalence of major OP in premenopausal SLE was 11% (95% CI (0.06, 0.14), I2 = 59.6%) (Fig. 2b). On meta-analysis of nine studies, the risk of OP in SLE patients compared with the healthy control group was significantly greater with OR 2.03 (95% CI (1.33, 3.10), I2 = 32%) (Fig. 3).
Meta-analysis results
To explore the risk factors of OP in SLE patients, we undertook sub-analyses. We compared the risk factors between with OP and without OP SLE patients.
Age
Among the 14 studies that reported the age of patients, heterogeneity analysis of the included studies indicated that there was heterogeneity (P < 0.01, I2 = 80%), but there was no clinical heterogeneity. A random effect model was used. The results showed that there were significant differences of age between the two groups (MD = 4.73, 95% CI (2.4, 7.05), P < 0.01) (Fig. 4a).
Meta-analysis of risk factors that may place patients with systemic lupus erythematosus at a higher risk for the development of osteoporosis (OP). a Age. b Disease duration. c Subgroup of disease duration. d Cumulative corticosteroid dose. e Time of hormone. f Daily steroid dose. g SLEDAI. h SLICC. i Menopause. j BMI
Disease duration
Among the 9 studies that reported the disease duration of patients, heterogeneity analysis of the included studies indicated that there was heterogeneity (P < 0.01, I2 = 87%) (Fig. 4b). Removing untreated study, heterogeneity decreased (P = 0.21, I2 = 28%). A fixed effect model was used. The results showed that there were significant differences in disease duration between the two groups (MD = 5.09, 95% CI (3.75, 6.44), P < 0.01) (Fig. 4c).
Cumulative GC dose
Among the 13 studies that reported the cumulative GC dose of patients, heterogeneity analysis of the included literature indicated that there was no heterogeneity (P = 0.07, I2 = 41%). A fixed effect model was used. The results showed that there were significant differences in cumulative GC dose between the two groups (MD = 16.11, 95% CI (13.42,18.79), P < 0.01) (Fig. 4d).
Duration of GC therapy
Among the 10 studies that reported the duration of GC therapy of patients, heterogeneity analysis of the included literature indicated that there was heterogeneity (P < 0.01, I2 = 92%) with no clinical heterogeneity. A random effect model was conducted. The results showed that there was a significant difference in the duration of GC therapy between the two groups (MD = 9.22, 95% CI (4.78, 13.66), P < 0.01) (Fig. 4e).
Daily GC dose
Among the 6 studies that reported the daily GC dose of patients, heterogeneity analysis of the included literature indicated that there was no heterogeneity (P = 0.14, I2 = 39%). A fixed effect model was used. The results showed that there was no significant difference in daily GC dose between the two groups (MD = − 0.71, 95% CI (− 2.73, 0.91), P = 0.39) (Fig. 4f).
SLEDAI
Among the 9 studies that reported the SLEDAI of patients, heterogeneity analysis of the included literature indicated that there was no heterogeneity (P = 0.86, I2 = 0%). A fixed effect model was used. The results showed that there was no significant difference in SLEDAI between the two groups (MD = − 0.46, 95% CI (− 1.06, 0.15), P = 0.14) (Fig. 4g).
SLICC
Among the 5 studies that reported the SLICC of patients, heterogeneity analysis of the included literature indicated that there was heterogeneity (P = 0.07, I2 = 53%), but there was no clinical heterogeneity. A random effect model was used. The results showed that there was a significant difference in SLICC between the two groups (MD = 0.78, 95% CI (0.38, 1.18), P < 0.01) (Fig. 4h).
Menopause
Among the 6 studies that reported menopause of patients, heterogeneity analysis of the included literature indicated that there was heterogeneity (P = 0.03, I2 = 62%), but there was no clinical heterogeneity. A random effect model was used. The results showed that there was significant difference of menopause between the two groups (MD = 4.4, 95% CI (2.13, 9.08), P < 0.01] (Fig. 4i).
BMI
Among the 9 studies that reported the BMI of patients, heterogeneity analysis of the included literature indicated that there was heterogeneity (P = 0.008, I2 = 61%), but there was no clinical heterogeneity. A random effect model was used. The results showed that there was no significant difference in BMI between the two groups (MD = − 0.32, 95% CI (− 1.18, 0.55), P = 0.47) (Fig. 4j).
Publication bias
The analysis of 31 studies shows that funnel charts are symmetrical and considered to have no obvious publication bias.
Discussion
SLE is a chronic systemic autoimmune disease involving multiple systems. OP, as one of the complications of SLE, has received a great deal of attention due to its great harm [45]. OP is very common in SLE people. Compared with healthy control groups, the risk of OP in SLE patients was 2.03. In our study, the prevalence of OP in SLE was 16%. OP can easily lead to fracture. Bultink and Lems [46] reported that vertebral fractures often occurred asymptomatically, but were associated with a reduced quality of life, increased future fracture risk, and an increased mortality risk. Le et al. [45] also indicated that vertebral and hip fractures were significant contributors to mortality in SLE patients.
Current studies have found that the occurrence of OP in SLE patients is closely related to disease activity and treatment options [9]. Bultink and Lems [46] reported that age, disease duration, disease severity, and GC use were important risk factors in SLE patients with OP. Briot et al. [47] have reported that the inflammatory disease activity was on top of the osteoporotic risk factors. Based on the results above, age and disease duration were the risk factors of OP, which is consistent with most of studies.
GC plays an important role in SLE. Most of the patients receive GCs, which have their own deleterious effects on bone. Long-term GC use leads to increased incidence of osteoporosis in patients with SLE [48]. Balasubramanian et al. [49] reported fracture risk increased with increasing cumulative exposure, being approximately 2.5-fold higher at cumulative dose ≥ 5400 mg compared with < 675 mg. GCs can increase bone resorption and reduce bone remodeling by various actions, which affects the balance of bone metabolism and causes secondary OP [50]. From our meta-analysis, except daily GC dose, cumulative GC dose and duration of GC therapy are associated with OP. This may be related to different disease stages of lupus patients. In stable lupus patients, the dose of GCs gradually decreases, or it is not even use. Therefore, the daily dose of GCs has nothing to do with the occurrence of OP.
SLE can also have a direct or indirect negative impact on BMD. Abnormal levels of cytokines directly affect BMD. Lower levels of P1NP, the most specific bone formation marker, are predictive of BMD loss over 12 months in premenopausal SLE patients [51]. Systemic multiple organ damage can indirectly cause bone mass loss. SLEDAI scored disease activity in SLE patients and SLICC/ACR scored cumulative organ damage. Interesting, SLICC was associated with secondary OP in SLE patients, while SLEDAI has no correlation. This might be attributed that SLEDAI only assesses nine systems with 24 indicators. At the time of BMD measurements, SLEDAI serves for the model of global assessment of activity. However, SLICC/ACR is designed to assess cumulative organ damage due to SLE caused by the disease itself or by treatment since the onset of the disease. Therefore, the Systemic Lupus International Cooperation Clinical Center/American Society of Rheumatology Damage Index is better at predicting osteoporosis.
Natural menopause and aging are involved in the development of OP. Vitku et al. [52] indicated that after menopause, when estrogen levels decrease, there is room for the activity of anthropogenic substances with estrogenic properties—endocrine disruptors (EDs)—that can interfere with bone remodeling and changes in calcium-phosphate metabolism. Thus, menopause contributes to osteoporosis. Our meta-analysis is consistent with other studies. The effect of BMI on osteoporosis is still controversial. Salman-Monte et al. [53] reported that low BMI was correlated with total hip and femoral neck osteopenia. However, Carli et al. [16] indicated that BMI had no difference in two groups. Our meta-analysis showed that BMI has no correlation with the secondary OP in SLE patients.
The mechanism of osteoporosis is complex. In addition to uncontrollable factors such as age and menopause, we should control disease activities and use GCs reasonably and standardly. GC is a double-edged sword. GCs, the basic drugs for SLE, have strong anti-inflammatory and immunosuppressive effects and can reverse the disease. However, GCs also cause diabetes, osteoporosis, ulcers, bacterial and fungal infections, and so on. Given the potential dangers, we should not refuse to use GCs during treatment. SLE disease activity may have direct effects on bone formation. Timely use of hormones can quickly control inflammation, thereby inhibiting cytokine-induced osteoclast activity. Therefore, a fine balance exists between disease control and the use of GCs with regard to bone health is the focus of our research. Studies have shown that adverse effects of daily doses of < 7.5 mg prednisone were least [54]. Combined treatment with HCQ and the judicious use of immunosuppressive drugs help to keep prednisone therapy within those limits [4]. In addition, all guidelines recommend supplementation with calcium and vitamin D in SLE. Bisphosphonates are considered the first choice therapy for the prevention and treatment of GIOP [55].
The study has some limitations. Firstly, the use of calcium tablets can affect the occurrence and development of OP, but we did not check medications, such as bisphosphonates, calcium, vitamin D, and Denosumab, which affected bone turnover, so the heterogeneity of study was high. Besides, there are various factors affecting osteoporosis, such as serum cytokines, illumination, immunosuppressant medications, and smoking, which increased or reduced the risk of both SLE and OP [55]. However, due to the lack of relevant studies, we did not discuss these factors. Larger sample studies will be needed to elucidate in the future. Additionally, longitudinal studies also needed to delineate a temporal association and causality of OP in the development of SLE.
Conclusion
Our report is the first systematic review to examine the association between OP and SLE. From analysis, OP significantly increased among SLE patients as compared with controls. The age, disease duration, cumulative GC dose, duration of GC therapy, SLICC, and menopause in SLE patients play an important role in OP.
References
Wang H, Wang J, Xia Y (2017) Defective suppressor of cytokine signaling 1 signaling contributes to the pathogenesis of systemic lupus erythematosus. Front Immunol 8:1292. https://doi.org/10.3389/fimmu.2017.01292
Wu H, Zhao M, Chang C, Lu Q (2015) The real culprit in systemic lupus erythematosus: abnormal epigenetic regulation. Int J Mol Sci 16(5):11013–11033. https://doi.org/10.3390/ijms160511013
Khaniukov O, Yehudina Y, Kalashnykova O, Sapozhnychenko L (2018) Management of patients with systemic lupus erythematosus in the prepregnancy and antenatal periods: challenges and solutions (review). Georgian Med News 280–281:54–61
Ruiz-Irastorza G, Danza A, Khamashta M (2012) Glucocorticoid use and abuse in SLE. Rheumatology (Oxford) 51(7):1145–1153. https://doi.org/10.1093/rheumatology/ker410
Xiao Y, Li K, Wang Z, Fu F, Shao S, Song F, Zhao J, Chen W, Liu Q, Xu J (2019) Pectolinarigenin prevents bone loss in ovariectomized mice and inhibits RANKL-induced osteoclastogenesis via blocking activation of MAPK and NFATc1 signaling. J Cell Physiol. https://doi.org/10.1002/jcp.28079
Bultink IE (2012) Osteoporosis and fractures in systemic lupus erythematosus. Arthritis Care Res 64(1):2–8. https://doi.org/10.1002/acr.20568
Ji Z, Zhang Z, Bao C, Zhao D, Lin J, Wu H, He D, Du H, Jiang L (2014) The epidemiological investigation on glucocorticoid-induced osteoporosis. Chin J Rheumatol 8:520–524. https://doi.org/10.3760/cma.j.issn.0578-1426.2017.03.006
Dey M, Bukhari M (2018) Predictors of fracture risk in patients with systemic lupus erythematosus. Lupus 27(9):1547–1551. https://doi.org/10.1177/0961203318768886
Di Munno O, Mazzantini M, Delle Sedie A, Mosca M, Bombardieri S (2004) Risk factors for osteoporosis in female patients with systemic lupus erythematosus. Lupus 13(9):724–730. https://doi.org/10.1191/0961203303lu1097oa
Bultink IEM (2018) Bone disease in connective tissue disease/systemic lupus erythematosus. Calcif Tissue Int 102(5):575–591. https://doi.org/10.1007/s00223-017-0322-z
Moher D, Liberati A, Tetzlaff J, Altman DG, Group P (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. J Clin Epidemiol 62(10):1006–1012. https://doi.org/10.1016/j.jclinepi.2009.06.005
Cramarossa G, Urowitz MB, Su J, Gladman D, Touma Z (2017) Prevalence and associated factors of low bone mass in adults with systemic lupus erythematosus. Lupus 26(4):365–372. https://doi.org/10.1177/0961203316664597
Bundhun PK, Kumari A, Huang F (2017) Differences in clinical features observed between childhood-onset versus adult-onset systemic lupus erythematosus: a systematic review and meta-analysis. Medicine (Baltimore) 96(37):e8086. https://doi.org/10.1097/MD.0000000000008086
Gibofsky A (2012) Comparative effectiveness of current treatments for rheumatoid arthritis. Am J Manag Care 18(13 Suppl):S303–S314
Bultink IE, Lems WF, Kostense PJ, Dijkmans BA, Voskuyl AE (2005) Prevalence of and risk factors for low bone mineral density and vertebral fractures in patients with systemic lupus erythematosus. Arthritis Rheum 52(7):2044–2050. https://doi.org/10.1002/art.21110
Carli L, Tani C, Spera V, Vagelli R, Vagnani S, Mazzantini M, Di Munno O, Mosca M (2016) Risk factors for osteoporosis and fragility fractures in patients with systemic lupus erythematosus. Lupus Sci Med 3(1):e000098. https://doi.org/10.1136/lupus-2015-000098
Chong HC, Chee SS, Goh EM, Chow SK, Yeap SS (2007) Dietary calcium and bone mineral density in premenopausal women with systemic lupus erythematosus. Clin Rheumatol 26(2):182–185. https://doi.org/10.1007/s10067-006-0258-6
Compeyrot-Lacassagne S, Tyrrell PN, Atenafu E, Doria AS, Stephens D, Gilday D, Silverman ED (2007) Prevalence and etiology of low bone mineral density in juvenile systemic lupus erythematosus. Arthritis Rheum 56(6):1966–1973. https://doi.org/10.1002/art.22691
Ding C, Sun L, Wang H, Lin H, Zhang X (2002) Determinants of bone mineral density in systemic lupus erythematosus. Chin J Rheumatol 6(3):192–194. https://doi.org/10.3760/j:issn:1007-7480.2002.03.012
Pons F, Peris P, Guanabens N, Font J, Huguet M, Espinosa G, Ingelmo M, Munoz-Gomez J, Setoain J (1995) The effect of systemic lupus erythematosus and long-term steroid therapy on bone mass in pre-menopausal women. Br J Rheumatol 34(8):742–746
Furukawa M, Kiyohara C, Tsukamoto H, Mitoma H, Kimoto Y, Uchino A, Nakagawa M, Oryoji K, Shimoda T, Akashi K, Harada M, Horiuchi T (2011) Prevalence of and risk factors for low bone mineral density in Japanese female patients with systemic lupus erythematosus. Rheumatol Int 31(3):365–376. https://doi.org/10.1007/s00296-009-1244-5
Wang G, Li X, Li X, Wang G, Tao J, Ma Y (2017) The effect of long-term low dose prednisone on bone mineral density in patients with systemic lupus erythematosus. Chin J Intern Med 56(3):179–183. https://doi.org/10.3760/cma.j.issn.0578-1426.2017.03.006
Bhattoa HP, Bettembuk P, Balogh A, Szegedi G, Kiss E (2002) Bone mineral density in women with systemic lupus erythematosus. Clin Rheumatol 21(2):135–141
Liang J, Jiang N, Xu Z, Lu X, Liu X, Chen S (2006) The influence of long-term glucocorticoid use on bone mineral density in premenopausal women with SLE. Chin J Osteoporos 12(1):57–58,61. https://doi.org/10.3969/j.issn.1006-7108.2006.01.017
Shen L, Liu Q, Yu L, Huang A (2004) Multivariable statistical analysis of osteoporosis in patients with systemic lupus erythematosus. Chin J Rheumatol 8(2):92–96. https://doi.org/10.3760/j:issn:1007-7480.2004.02.008
Pineau CA, Urowitz MB, Fortin PJ, Ibanez D, Gladman DD (2004) Osteoporosis in systemic lupus erythematosus: factors associated with referral for bone mineral density studies, prevalence of osteoporosis and factors associated with reduced bone density. Lupus 13(6):436–441. https://doi.org/10.1191/0961203303lu1036oa
Ye Q, Xiong Y, Cao X, Du W (2013) Bone mineral density in 42 female patients with systemic lupus erythematosus. Chin J Diffic and Compl Cas 12(4):312–313. https://doi.org/10.3969/j.issn.1671-6450.2013.04.024
Sun YN, Feng XY, He L, Zeng LX, Hao ZM, Lv XH, Pu D (2015) Prevalence and possible risk factors of low bone mineral density in untreated female patients with systemic lupus erythematosus. Biomed Res Int 2015:510514. https://doi.org/10.1155/2015/510514
Zhu X, Liang J, Lin B, Zhang J, Tan G (2000) Measurement of bone mineral density in premenopausal women with systemic lupus erythematosus. Chin J Dermatol 3(1):12. https://doi.org/10.3760/j.issn:0412-4030.2000.01.004
Liu X, Zhu X, Li S (2000) Detection and analysis of bone mineral density in patients with systemic lupus erythematosus. J Nurs 15(1):14–15. https://doi.org/10.3969/j.issn.1001-4152.2000.01.005
Hou X, Zhang M, Cui L (2015) Serum immunological risk factors for osteoporosis in patients with systemic lupus erythematosus. Shandong Medical Journal 24:79–80. https://doi.org/10.3969/j.issn.1002-266X.2015.24.035
Yeap SS, Fauzi AR, Kong NC, Halim AG, Soehardy Z, Rahimah S, Chow SK, Goh EM (2009) Influences on bone mineral density in Malaysian premenopausal systemic lupus erythematosus patients on corticosteroids. Lupus 18(2):178–181. https://doi.org/10.1177/0961203308094995
Yee CS, Crabtree N, Skan J, Amft N, Bowman S, Situnayake D, Gordon C (2005) Prevalence and predictors of fragility fractures in systemic lupus erythematosus. Ann Rheum Dis 64(1):111–113. https://doi.org/10.1136/ard.2003.018127
Luo Z, Wu J, Huang L, Liao T, He Y (2014) Study on the relationship between inflammatory states of SLE and osteoporosis. J Mod Med Health 18:2730–2732. https://doi.org/10.3969/j.issn.10095519.2014.18.004
He W, Li J, Li B, Ye Z, Guo F, Tan D, Xie Y, Xu S (2009) Influencing factors of bone mineral density in premenopausal women with rheumatism. Journal of Pratical Medcine 25(3):394–396. https://doi.org/10.3969/j.issn.1006-5725.2009.03.023
Sui P, Li X (2008) Changes of bone mineral density in patients with systemic lupus erythematosus before and after hormone therapy. J Jining Med Univ 31(4):298–299. https://doi.org/10.3969/j.issn.1000-9760.2008.04.013
Yu J (2015) Effects of different doses of glucocorticoid maintenance therapy on bone mineral density and bone metabolism in patients with systemic lupus erythematosus. Bengbu Medical College
Liang H (2010) Bone mineral density changes and influencing factors in systemic lupus erythematosus. . Shantou University
Mendoza-Pinto C, García-Carrasco M, Sandoval-Cruz H, Escárcega RO, Jiménez-Hernández M, Etchegaray-Morales I, Soto-Vega E, Muñoz-Guarneros M, López-Colombo A, Delezé-Hinojosa M, Cervera R (2008) Risks factors for low bone mineral density in pre-menopausal Mexican women with systemic lupus erythematosus. Clin Rheumatol 28(1):65–70. https://doi.org/10.1007/s10067-008-0984-z
Banno S, Matsumoto Y, Naniwa T, Hayami Y, Sugiura Y, Yoshinouchi T, Ueda R (2002) Reduced bone mineral density in Japanese premenopausal women with systemic lupus erythematosus treated with glucocorticoids. Mod Rheumatol 12(4):323–328. https://doi.org/10.3109/s101650200057
Becker A, Fischer R, Scherbaum WA, Schneider M (2001) Osteoporosis screening in systemic lupus erythematosus: impact of disease duration and organ damage. Lupus 10(11):809–814. https://doi.org/10.1177/096120330101001108
Yuan Q (2014) Bone mass and related factors in 100 female patients with diffuse connective tissue disease. Zhejiang University
Sinigaglia L, Varenna M, Binelli L, Zucchi F, Ghiringhella D, Gallazzi M, Limonta M, Zeni S, Fantini F (1999) Determinants of bone mass in systemic lupus erythematosus: a cross sectional study on premenopausal women. J Rheumatol 26(6):1280–1284
Wang Z (2017) The research of osteoporosis in patients with untreated systemic lupus erythematosus. Hebei Medical University
Le B, Waller JL, Radhakrishnan R, Oh SJ, Kheda MF, Nahman NS Jr, Carbone L (2018) Osteoporotic fractures in patients with systemic lupus erythematosus and end stage renal disease. Lupus 27(1):17–24. https://doi.org/10.1177/0961203317709953
Bultink IE, Lems WF (2016) Lupus and fractures. Curr Opin Rheumatol 28(4):426–432. https://doi.org/10.1097/BOR.0000000000000290
Briot K, Geusens P, Em Bultink I, Lems WF, Roux C (2017) Inflammatory diseases and bone fragility. Osteoporos Int 28(12):3301–3314. https://doi.org/10.1007/s00198-017-4189-7
Stojan G, Petri M (2017) The risk benefit ratio of glucocorticoids in SLE: have things changed over the past 40 years? Curr Treatm Opt Rheumatol 3(3):164–172. https://doi.org/10.1007/s40674-017-0069-8
Balasubramanian A, Wade SW, Adler RA, Saag K, Pannacciulli N, Curtis JR (2018) Glucocorticoid exposure and fracture risk in a cohort of US patients with selected conditions. J Bone Miner Res 33(10):1881–1888. https://doi.org/10.1002/jbmr.3523
Sarkissian A, Sivaraman V, Bout-Tabaku S, Ardoin SP, Moore-Clingenpeel M, Mruk V, Steigelman H, Morris K, Bowden SA (2019) Bone turnover markers in relation to vitamin D status and disease activity in adults with systemic lupus erythematosus. Lupus 28(2):156–162. https://doi.org/10.1177/0961203318815593
Seguro LP, Casella CB, Caparbo VF, Oliveira RM, Bonfa A, Bonfa E, Pereira RM (2015) Lower P1NP serum levels: a predictive marker of bone loss after 1 year follow-up in premenopausal systemic lupus erythematosus patients. Osteoporos Int 26(2):459–467. https://doi.org/10.1007/s00198-014-2860-9
Vitku J, Kolatorova L, Franekova L, Blahos J, Simkova M, Duskova M, Skodova T, Starka L (2018) Endocrine disruptors of the bisphenol and paraben families and bone metabolism. Physiol Res 67(Supplementum 3):S455–S464
Salman-Monte TC, Torrente-Segarra V, Munoz-Ortego J, Mojal S, Carbonell-Abello J (2015) Prevalence and predictors of low bone density and fragility fractures in women with systemic lupus erythematosus in a Mediterranean region. Rheumatol Int 35(3):509–515. https://doi.org/10.1007/s00296-014-3087-y
Sciascia S, Mompean E, Radin M, Roccatello D, Cuadrado MJ (2017) Rate of adverse effects of medium- to high-dose glucocorticoid therapy in systemic lupus erythematosus: a systematic review of randomized control trials. Clin Drug Investig 37(6):519–524. https://doi.org/10.1007/s40261-017-0518-z
Gracanin AG, Markovic I, Loncarevic J, Golob M, Morovic-Vergles J (2015) Bone mineral density in patients with systemic lupus erythematosus--our results. Reumatizam 62(2):16–21
Funding
This work was financially supported by the Postgraduate Research & Practice Innovation Program of Jiangsu Province (grant number KYCX19-2071 and KYCX18-2410), National Natural Science Foundation of China (grant numbers 81671616 and 81871278), and Research Center of Gerontology and Longevity, Affiliated Hospital of Nantong University, Nantong (grant number HS2018004).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Chaoyu Gu, Rui Zhao, Xiaomei Zhang are the first author
Rights and permissions
About this article
Cite this article
Gu, C., Zhao, R., Zhang, X. et al. A meta-analysis of secondary osteoporosis in systemic lupus erythematosus: prevalence and risk factors. Arch Osteoporos 15, 1 (2020). https://doi.org/10.1007/s11657-019-0667-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11657-019-0667-1