Abstract
Background
Infected acute necrotic collections (ANC) and walled-off necrosis (WON) of the pancreas are associated with high mortality. The difference in mortality between open necrosectomy and minimally invasive therapies in these patients remains unclear.
Methods
This retrospective multicenter cohort study was conducted among 44 institutions in Japan from 2009 to 2013. Patients who had undergone invasive treatment for suspected infected ANC/WON were enrolled and classified into open necrosectomy and minimally invasive treatment (laparoscopic, percutaneous, and endoscopic) groups. The association of each treatment with mortality was evaluated and compared.
Results
Of 1159 patients with severe acute pancreatitis, 122 with suspected infected ANC or WON underwent the following treatments: open necrosectomy (33) and minimally invasive treatment (89), (laparoscopic three, percutaneous 49, endoscopic 37). Although the open necrosectomy group had a significantly higher mortality on univariate analysis (p = 0.047), multivariate analysis showed no significant associations between open necrosectomy or Charlson index and mortality (p = 0.29, p = 0.19, respectively). However, age (for each additional 10 years, p = 0.012, odds ratio [OR] 1.50, 95% confidence interval [CI] 1.09–2.06) and revised Atlanta criteria-severe (p = 0.001, OR 7.84, 95% CI 2.40–25.6) were significantly associated with mortality.
Conclusions
In patients with acute pancreatitis and infected ANC/WON, age and revised Atlanta criteria-severe classification are significantly associated with mortality whereas open necrosectomy is not. The mortality risk for patients undergoing open necrosectomy and minimally invasive treatment does not differ significantly. Although minimally invasive surgery is generally preferred for patients with infected ANC/WON, open necrosectomy may be considered if clinically indicated.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acute pancreatitis is a relatively common disease with a frequency of 13–45 patients per 100,000 population per year,1 5–20% of whom develop necrotizing pancreatitis and require intensive care.2,3,4 Acute necrotic collections (ANC) develop in almost all patients with necrotizing pancreatitis, and approximately 50% of these patients proceed to develop walled-off necrosis (WON).2,5 Approximately one-third of patients with pancreatic necrosis, including those with ANC and WON, develop severe infections6 and these patients have a mortality rate of 30–39%.7 Intensive care, including drainage and/or necrosectomy of the infected necrotizing tissue, is mandatory to maximize survival.
A step-up approach, where drainage is performed before necrosectomy, is currently preferred over primary open necrosectomy before drainage in patients with infected ANC and WON.8 However, no randomized control trials (RCT) or meta-analyses have shown a significant difference in mortality rate between primary open necrosectomy and minimally invasive treatment, possibly because of small sample sizes.8,9,10 We therefore hypothesized that open necrosectomy may be associated with greater mortality than treatment with minimally invasive therapies in patients with infected ANC/WON and drew on a large multicenter database of patients with acute pancreatitis to investigate this possibility. Moreover, no evaluation of secondary open necrosectomy after drainage has yet been reported. We therefore also compared the effect of primary open necrosectomy before drainage with secondary open necrosectomy after drainage on mortality, our hypothesis being that secondary open necrosectomy is associated with lower mortality than primary open necrosectomy.
Materials and Methods
Study Design and Patients
This study is a post hoc analysis of findings from a retrospective multicenter cohort study11 that investigated the efficacy of continuous regional arterial infusion (CRAI) of protease inhibitors for patients with severe acute pancreatitis (SAP). The original study was registered with the University Hospital Medical Information Network (UMIN:000012220). Patients with SAP aged 18 years or older treated in 44 institutions in Japan between 2009 and 2013 were enrolled. In this study, SAP was diagnosed using the criteria of the Japanese Ministry of Health, Labour and Welfare study group for the severity of acute pancreatitis. The efficacy of these criteria has been confirmed by multiple studies.12,13,14 The criteria include various prognostic factors and computed tomography (CT) grade (Supplementary Table 1).12 The required measures were obtained at diagnosis and the patient evaluated repeatedly from the onset to 72 h. SAP was diagnosed when the total prognostic factor score was three points or more, or computed tomography (CT) grade was two or more. For the present study, anonymized data from the database with no link to individual patient information was used. The Institutional Review Boards waived the need for a review of the present study because the data was anonymous.
Patients with acute pancreatitis who had undergone treatment for suspected infected ANC/WON were included. Physicians at the relevant institutions reviewed the patents’ medical records for the following data: age, gender, cause of pancreatitis, acute physiology and chronic health evaluation (APACHE II, highest score within 24 h after diagnosis of SAP), prognostic factor score, Charlson index, CT severity index, revised Atlanta classification, treatment (CRAI, enteral feeding within the first 48 h, volume of fluid administered within the first 24 h, treatment for ANC/WON, mechanical ventilation, hemodiafiltration, preventive use of antibacterial drugs), and outcome (hospital mortality, proved infection, length of hospital stay). Pancreatitis with persistent single or multiple organ failure over 48 h was regarded as SAP in accordance with the revised Atlanta criteria. Organ failure was defined as a score of 2 or more for one of three organ systems (respiratory, cardiovascular, and renal) according to the modified Marshall scoring system.2 Patients in this study were followed from hospitalization to discharge. All data were collected at a central research secretariat and analyzed.
Infected ANC/WON
ANC/WON were diagnosed according to the revised Atlanta classification.2 Suspected infected ANC/WON was defined as a condition in which a patient with ANC or WON had evidence of possible infection (e.g., fever, pain, or inflammatory response on blood tests). The diagnosis of infected ANC/WON was established when bacteria were identified in blood cultures, ANC/WON tissue, or aspirates.
Treatments and Outcomes
We divided patients into two groups: those who had undergone open necrosectomy and those who had undergone minimally invasive treatment. The open necrosectomy group included both patients who had undergone primary open necrosectomy before drainage and secondary open necrosectomy after drainage. Typically, open necrosectomy was indicated for patients with extensive retroperitoneal necrosis. Minimally invasive treatment was defined as laparoscopic, percutaneous, or endoscopic approaches and included drainage only, necrosectomy only, and a combination of both. The endoscopic ultrasonography (EUS)-guided approach was classified as an endoscopic treatment and video-assisted retroperitoneal debridement (VARD) as a laparoscopic treatment. Criteria for selection of the intervention were not standardized, the decision being at the discretion of the physician in charge of the patient’s care.
Statistical Analysis
Selected characteristics were compared between the open necrosectomy and minimally invasive treatment groups. The χ2 test or Fisher’s exact test was used for comparison of categorical data, and Student’s t test or the Mann-Whitney U test was used to compare continuous data. Univariate analyses were performed with independent variables to evaluate the risk factors for mortality. Multivariate logistic regression was performed to evaluate the association of open necrosectomy with mortality. The covariates, including age, revised SAP according to Atlanta criteria, and the Charlson index, were selected because they are associated with mortality according to previous studies.15,16 We selected the revised Atlanta classification as the most established means of classifying the severity of patients with acute pancreatitis, and did not use other prognostic factors or the APACHE II scores to avoid multiple collinearities. The odds ratio and 95% confidence intervals (CI) for secondary open necrosectomy and primary open necrosectomy versus minimally invasive treatment were determined. A p value of less than 0.05 was considered to denote statistical significance, and all tests were two-tailed. All statistical analyses were performed using SPSS version 23 for Windows (IBM Corp., Armonk, NY, USA). Subgroup analyses were conducted on patients with proved infected ANC/WON and patients with more than 50% pancreatic necrosis in an identical manner. Furthermore, the three groups (primary open necrosectomy, secondary open necrosectomy, and minimally invasive treatment) were compared statistically. Multivariate analysis was also performed using age, Charlson index, and SAP according to the revised Atlanta criteria. The study complied with the Strengthening Reporting of Observational Studies in Epidemiology guidelines.17
Results
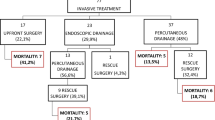
Of 1159 patients with SAP, 425 had pancreatic necrosis, 122 of whom had suspected infected ANC/WON and underwent invasive treatment (Fig. 1). The mean follow-up was 102.2 (SD, 80.0) days. Their mean age was 62.3 years, 70% were men, 83 (68%) were diagnosed with SAP according to the revised Atlanta criteria, and 42 (34%) died. Of 122 patients with suspected infected ANC/WON, 95 (78%) were shown to have an infection based on culture results (Table 1).
Open necrosectomy was performed on 33 patients and the remaining 89 underwent minimally invasive treatment. There were significant differences between the two groups in rate of alcoholic pancreatitis (p = 0.029), APACHE II score (p = 0.009), prognostic factor score (p = 0.003), SAP according to the revised Atlanta criteria (p = 0.015), use of mechanical ventilation (p = 0.004) and hemodiafiltration (p = 0.033), rate of proved infection (p = 0.035), and mortality (p = 0.047) (Table 1).
Risk factors for mortality by univariate analysis were open necrosectomy (OR 2.28, 95% CI 1.00–5.19, p = 0.047), age (with each 10 years increment, OR 1.48, 95% CI 1.11–1.96, p = 0.005), APACHE II score (OR 1.11, 95% CI 1.04–1.17, p < 0.001), prognostic factor score (OR 1.43, 95% CI 1.17–1.74, p < 0.001), Charlson index (OR 1.61, 95% CI 1.12–2.31, p = 0.012), SAP according to the revised Atlanta criteria (OR 7.39, 95% CI 2.41–22.7, p < 0.001), mechanical ventilation (OR 10.6, 95% CI 3.04–37.3, p < 0.001), and hemodiafiltration (OR 7.89, 95% CI 3.22–19.4, p < 0.001) (Table 2). According to multivariate analysis, open necrosectomy and Charlson index were not associated with mortality, but age (with each 10 years increment, OR 1.50, 95% CI 1.09–2.06, p = 0.012) and SAP according to the revised Atlanta criteria (OR 7.84, 95% CI 2.40–25.6, p = 0.001) were significantly associated with mortality (Table 3).
Twenty of 33 patients underwent open necrosectomy as a secondary procedure, nine as a primary open necrosectomy, and four were unknown. The three groups (minimally invasive treatment, primary, and secondary open necrosectomy) were compared in sub-analyses. The mortality odds-ratio for secondary open necrosectomy versus minimally invasive treatment was 1.25 (95% CI 0.40–3.84, p = 0.69) and for primary open necrosectomy versus minimally invasive treatment was 3.13 (95% CI 0.66–17.5, p = 0.15) when minimally invasive treatment 1 was assumed by multivariate analysis with age, Charlson index, and SAP according to the revised Atlanta criteria. Analyses were performed on the subgroup of patients with proved infection (n = 95) in an identical fashion (Table 3 and Supplementary Table 2). Neither univariate nor multivariate analyses showed a significant association between mortality and open necrosectomy (p = 0.050, 0.23, respectively). There was also no association between undergoing open necrosectomy and mortality in the subgroup with proved infected ANC/WON, but age and SAP according to the revised Atlanta criteria were significantly associated with mortality (Table 3). In univariate analyses performed on the subgroup of patients with more than 50% pancreatic necrosis (n = 31), there was no association between undergoing open necrosectomy and mortality (OR 2.55, 95% CI 0.39–16.6, p = 0.39). Multivariate analysis was not performed because this subgroup was too small.
Discussion
In this retrospective cohort study, patients suspected of having infected ANC/WON who had undergone open necrosectomy had a higher mortality than those who had undergone minimally invasive treatment according to univariate analysis. According to multivariate analysis adjusted for age, severity of underlying pancreatitis, and comorbidities, the treatment approach (open vs. minimally invasive) was not associated with mortality, although age and SAP according to the revised Atlanta criteria were significantly associated with mortality. These data suggest that age and a diagnosis of SAP should have a more significant impact on the mortality of patients with acute pancreatitis and suspected of having infected ANC/WON than the invasiveness of treatment.
Surgeons generally prefer minimally invasive surgery for patients with infected ANC/WON. However, the current study suggests that there is no association between open necrosectomy and mortality in patients with extensive pancreatic necrosis, and that secondary open necrosectomy after drainage (OR 1.25) may be better than primary open necrosectomy (OR 3.13) with regard to mortality. Therefore, open necrosectomy, especially secondary open necrosectomy after drainage, is a reasonable option for patients with infected ANC/WON if indicated, such as patients with extensive retroperitoneal necrosis extending to the small bowel mesentery. Open necrosectomy may not be a good option for patients without extensive retroperitoneal necrosis extending far from the lesser sac, i.e., patients with typical retro-gastric collections around the body and proximal tail of the pancreas.
The presence of solid necrotizing tissue or perfused tissue within ANC/WON lesions in addition to extensive pancreatic necrosis may be an indication for open necrosectomy of an infected fluid collection as opposed to further minimally invasive treatments. Although open necrosectomy is associated with a high morbidity (34–95%) and mortality (6–25%), the success rate is close to 100%, probably because the procedure enables a direct approach to an infected solid lesion.7 In the PANTER RCT cohort study, necrosectomy was eventually required for ANC/WON lesions that had a solid necrotized tissue component in 60% of patients who underwent endoscopic or percutaneous drainage.8 Several studies have shown that a percutaneous approach has only a 60–80% success rate and most patients will then require a subsequent open necrosectomy.18,19 Bleeding and other complications caused by iatrogenic damage to blood vessels and pseudoaneurysms developing in and around the lesion or unaffected visceral tissue can also be minimized by adopting an open surgical approach.19 In a recent multicenter study, various complications (e.g., bleeding, perforation, air embolism) were occurred in as many as 33% of patients treated with an EUS approach.21 Most complications can be avoided or controlled during open necrosectomy. Nevertheless, the optimal treatment depends on the patient’s condition and the expertise of the institution. In the cohort in the present study, the appropriate selection of open surgery may have affected the findings that there was no significant difference in mortality rate adjusted for severity.
Three RCTs have evaluated the association between type of intervention and mortality. The PANTER RCT reported that a step-up approach was associated with a significantly lower incidence of new-onset multiple organ failure and a lower rate of complications than primary open necrosectomy (12 vs. 42%, p = 0.002), whereas the difference in mortality between these two treatment approaches was not significant (19 vs. 16%, p = 0.70).8 The PENGUIN RCT compared surgical necrosectomy by video-assisted retroperitoneal debridement and endoscopic transgastric necrosectomy. Surgical necrosectomy was associated with higher IL-6 concentrations than endoscopic necrosectomy,9 but the effect of the type of surgical procedure used on mortality was not significant (40 vs. 10%, p = 0.30).9 Another RCT that compared endoscopic necrosectomy and minimally invasive surgical necrosectomy as part of a step-up approach also showed no significant difference in mortality based on the intervention (mortality 18% in endoscopy group vs. 13% minimally invasive surgery group, p = 0.50).20 A meta-analysis comparing endoscopic/retroperitoneal necrosectomy with open necrosectomy also found no significant difference between these procedures in the incidence of complications or mortality rate.10 The finding that the type of intervention used to treat infected ANC/WON has no significant impact on mortality in the present study is in agreement with these previous findings.
The increased risk associated with open necrosectomy as reported previously can be attributed to two major factors, the timing of open necrosectomy and the severity of the underlying pancreatitis. Although primary open necrosectomy without drainage was performed in the past, secondary open necrosectomy after drainage became increasingly popular between 2010 and 2013.8,21 Most open necrosectomies reported prior to 2010 were not performed after initial drainage, but as primary treatment, prior to any drainage procedure. Although one large combined cohort study demonstrated that minimally invasive surgery and endoscopic necrosectomy are associated with a lower mortality than open necrosectomy, open necrosectomy was performed as the primary treatment in 95.2% (498/523) of patients in three cohorts in which all patients underwent open necrosectomy.22 To date, no study has directly compared minimally invasive necrosectomy and secondary open necrosectomy after drainage. For most patients in the current study, open necrosectomy was conducted as a secondary procedure. The odds ratio for secondary open necrosectomy and mortality was lower than that for primary open necrosectomy, possibly explaining why open necrosectomy was not significantly associated with mortality in this study. One possible explanation for secondary open necrosectomy being safe is that it may be performed later than primary open necrosectomy. It is generally recommended that surgical necrosectomy be delayed until collections have become walled-off, typically 4 weeks after the onset of pancreatitis, because such a wall is robust and necrosis adheres strongly to the gastrointestinal tract.23 Thus, such walls may be more robust in patients who undergo secondary open necrosectomy than in those who undergo primary open necrosectomy. A future study with an appropriate design may identify that secondary open necrosectomy after drainage has a beneficial effect on mortality.
Patients who undergo open necrosectomy are likely to have more severe underlying pancreatitis,24 which may be associated with an increased rate of complications such as sepsis and multiple organ failure.25 Patients with more severe pancreatitis may need early aggressive treatment because they are unable to tolerate the multiple procedures required for a minimally invasive approach, which may take longer. In the present study, 85% of patients who underwent open necrosectomy had SAP according to the revised Atlanta classification, whereas only 62% of those who underwent minimally invasive treatment had SAP (p < 0.015). Thus, when interventions are performed in an appropriate sequence, the risk of mortality associated with open necrosectomy may be comparable to that of minimally invasive procedures.
This study has several limitations. The first limitation is that the database did not include information regarding the frequency and timing of interventions for suspected infected ANC/WON or post-interventional complications.26 We are currently collecting the relevant information in a prospective multicenter observational study on acute pancreatitis (UMIN:000025468, https://upload.umin.ac.jp/cgi-open-bin/ctr/ctr_view.cgi?recptno=R000028398). In the future, RCTs are required to confirm our findings. The second limitation is that the criteria for selecting the treatment for patients with suspected infected ANC/WON were not consistent among the participating institutions. However, the results of subgroup analyses of patients with proved infections were similar to those of the whole cohort, suggesting that this inconsistency did not have a significant impact on the results. The third limitation is that there were only three laparoscopies among the minimally invasive treatments. Percutaneous and endoscopic treatment may therefore have more strongly influenced our findings than laparoscopic treatment. The small number of laparoscopies performed is likely attributable to the fact that physicians or intensivists rather than surgeons treat pancreatitis in Japan.
Conclusions
This study showed that age and classification as “severe” by the revised Atlanta criteria have a more significant impact on mortality in patients with acute pancreatitis with infected ANC/WON than differences in the strategy for treatment (open necrosectomy vs. minimally invasive treatment). The risk of mortality after open necrosectomy itself may not be higher than after a minimally invasive approach. For patients with extensive retroperitoneal necrosis, a minimally invasive procedure is not the only surgical option, but open necrosectomy may be indicated. Among patients undergoing open necrosectomy, open necrosectomy after drainage (secondary) may be better than open necrosectomy before drainage (primary).
References
Yadav D, Lowenfels AB. The epidemiology of pancreatitis and pancreatic cancer. Gastroenterology 2013;144:1252–61.
Banks PA, Bollen TL, Dervenis C, et al. Classification of acute pancreatitis--2012: revision of the Atlanta classification and definitions by international consensus. Gut 2013;62:102–11.
Bruno MJ. Improving the Outcome of Acute Pancreatitis. Dig Dis 2016;34:540–5.
Hackert T, Buchler MW. Decision Making in Necrotizing Pancreatitis. Dig Dis 2016;34:517–24.
Sarathi Patra P, Das K, Bhattacharyya A, et al. Natural resolution or intervention for fluid collections in acute severe pancreatitis. Br J Surg 2014;101:1721–8.
Banks PA, Freeman ML. Practice guidelines in acute pancreatitis. Am J Gastroenterol 2006;101:2379–400.
Bugiantella W, Rondelli F, Boni M, et al. Necrotizing pancreatitis: A review of the interventions. International journal of surgery (London, England) 2016;28 Suppl 1:S163–71.
van Santvoort HC, Besselink MG, Bakker OJ, et al. A step-up approach or open necrosectomy for necrotizing pancreatitis. N Engl J Med 2010;362:1491–502.
Bakker OJ, van Santvoort HC, van Brunschot S, et al. Endoscopic transgastric vs surgical necrosectomy for infected necrotizing pancreatitis: a randomized trial. Jama 2012;307:1053–61.
Isayama H, Nakai Y, Rerknimitr R, et al. Asian consensus statements on endoscopic management of walled-off necrosis. Part 2: Endoscopic management. Journal of gastroenterology and hepatology 2016;31:1555–65.
Horibe M, Sasaki M, Sanui M, et al. Continuous Regional Arterial Infusion of Protease Inhibitors Has No Efficacy in the Treatment of Severe Acute Pancreatitis: A Retrospective Multicenter Cohort Study. Pancreas 2017;46:510–17.
Ikeura T, Horibe M, Sanui M, et al. Validation of the efficacy of the prognostic factor score in the Japanese severity criteria for severe acute pancreatitis: A large multicenter study. United European gastroenterology journal 2017;5:389–97.
Yang CJ, Chen J, Phillips AR, et al. Predictors of severe and critical acute pancreatitis: a systematic review. Dig. Liver Dis. 2014; 46:446–51.
Mounzer R, Langmead CJ, Wu BU, et al. Comparison of existing clinical scoring systems to predict persistent organ failure in patients with acute pancreatitis. Gastroenterology 2012; 142: 1476–82.
Forsmark CE, Vege SS, Wilcox CM. Acute Pancreatitis. The New England journal of medicine 2016;375:1972–81.
Choi JH, Kim MH, Oh D, et al. Clinical relevance of the revised Atlanta classification focusing on severity stratification system. Pancreatology : official journal of the International Association of Pancreatology (IAP) [et al] 2014;14:324–9.
von Elm E, Altman DG, Egger M, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: Guidelines for reporting observational studies. International journal of surgery 2014;12:1495–9.
Zerem E, Imamovic G, Susic A, Haracic B. Step-up approach to infected necrotising pancreatitis: a 20-year experience of percutaneous drainage in a single centre. Digestive and liver disease : official journal of the Italian Society of Gastroenterology and the Italian Association for the Study of the Liver 2011;43:478–83.
Tong Z, Li W, Yu W, et al. Percutaneous catheter drainage for infective pancreatic necrosis: is it always the first choice for all patients? Pancreas 2012;41:302–5.
van Brunschot S, van Grinsven J, van Santvoort HC, et al. Endoscopic or surgical step-up approach for infected necrotising pancreatitis: a multicentre randomised trial. Lancet 2018;391:51–8.
IAP/APA evidence-based guidelines for the management of acute pancreatitis. Pancreatology : official journal of the International Association of Pancreatology (IAP) [et al] 2013;13:e1–15.
van Brunschot S, Hollemans RA, Bakker OJ, et al. Minimally invasive and endoscopic versus open necrosectomy for necrotising pancreatitis: a pooled analysis of individual data for 1980 patients. Gut 2018;67:697–706.
Besselink MG, Verwer TJ, Schoemaeckers EJ, et al. Timing of surgical intervention in necrotizing pancreatitis. Arch. Surg. 2007; 142: 1194–201.
Bausch D, Wellner U, Kahl S, et al. Minimally invasive operations for acute necrotizing pancreatitis: comparison of minimally invasive retroperitoneal necrosectomy with endoscopic transgastric necrosectomy. Surgery 2012;152:S128–34.
Pascual I, Sabater L, Anon R, et al. Surgical versus nonsurgical treatment of infected pancreatic necrosis: more arguments to change the paradigm. Journal of gastrointestinal surgery : official journal of the Society for Surgery of the Alimentary Tract 2013;17:1627–33.
van Santvoort HC, Bakker OJ, Bollen TL, et al. A conservative and minimally invasive approach to necrotizing pancreatitis improves outcome. Gastroenterology 2011;141:1254–63.
Acknowledgments
We would like to acknowledge the support given by Kazuichi Okazaki, Tsuyoshi Hamada, Seiya Suzuki, Jun Kataoka, Tomohiro Adachi, Shin Namiki, Kaoru Hirose, Sakue Masuda, Tomoaki Hashida, Naoki Shinyama, Hitoshi Yamamura, Takashi Moriya, Kunihiro Shirai, Kazuo Inui, Satoshi Yamamoto, Kyoji Oe, Takashi Muraki, Tetsuya Ito, Yukiko Masuda, Junichi Sakagami, Hiroaki Yasuda, Yusuke Iizawa, Masayuki Kamochi, Nobuyuki Saito, Mizuki Sato, Kyohei Miyamoto, Mioko Kobayashi, Koji Saito, Shinjiro Saito, Junko Izai, Motohiro Sekino, Yoshimoto Seki, Tetsuya Mine, Youhei Kawashima, Naoyuki Matsuda, Masato Inaba, Mineji Hayakawa, Takuyo Misumi, and Yuki Takeda in data collection at the 44 participating institutions (Osaka Saiseikai Senri Hospital, Hiroshima City Hiroshima Citizens Hospital, Kansai Medical University Hirakata Hospital, The University of Tokyo Hospital, Iizuka Hospital, Japanese Red Cross Musashino Hospital, Tokyo Metropolitan Tama Medical Center, Japanese Redcross Maebashi Hospital, Shonan Kamakura General Hospital, Showa University Hospital, Nihon University Hospital, Saiseikai Kumamoto Hospital, Fukuyama City Hospital, Jichi Medical University Hospital, Chiba University Hospital, Osaka City University Hospital, Tohoku University Hospital, Nihon University Itabashi Hospital, Gifu University Hospital, Second Teaching Hospital, Fujita Health University, Asahi General Hospital, Shinshu University Hospital, National Hospital Organization Nagasaki Medical Center, University Hospital, Kyoto Prefectural University of Medicine, Mie University Hospital, Hospital of the University of Occupational and Environmental Health, Nippon Medical School Chiba Hokusoh Hospital, Jichi Medical University, Saitama Medical Center, Wakayama Medical University Hospital, Tokyo Metropolitan Bokutoh Hospital, Jikei University School of Medicine, Saka General Hospital, National Hospital Organization Sendai Medical Center, Nagasaki University Hospital, Keio University School of Medicine, Japanese Red Cross Akita Hospital, Ibaraki Prefectural Central Hospital, Tokai University Hospital, Nagoya University Hospital, Hokkaido University Hospital, National Cancer Center, Akita City Hospital, Kobe University Hospital, and Tokyo Rosai Hospital). We also thank the Japanese Society of Education for Physicians and Trainees in Intensive Care and the Japanese Society of Intensive Care Medicine. Additionally, we thank Dr. Trish Reynolds, MBBS, FRACP, from Edanz Group (www.edanzediting.com/ac) for editing the English text of a draft of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the collection and analysis of the data. The corresponding author takes responsibility for the accuracy and integrity of the data. All authors have read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM 1
(DOCX 34 kb)
Rights and permissions
About this article
Cite this article
Minami, K., Horibe, M., Sanui, M. et al. The Effect of an Invasive Strategy for Treating Pancreatic Necrosis on Mortality: a Retrospective Multicenter Cohort Study. J Gastrointest Surg 24, 2037–2045 (2020). https://doi.org/10.1007/s11605-019-04333-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-019-04333-7