Abstract
Purpose
In stage II colorectal cancer (CRC), high-risk patient selection is required, but no candidate markers have been elucidated. Our concern was whether anastomotic leakage (Lk) is a potential available clinicopathological factor for selecting high-risk stage II.
Methods
Two hundred seven patients with stage II CRC who underwent curative resection were analyzed. Clinical variables were tested for their relationship to survival.
Results
The 5-year disease-free survival rate (DFS) was 87.0%. The univariable prognostic analyses indicated that Lk (P = 0.003) was the only significant factor. The multivariable prognostic analysis revealed that Lk remained to be potently independent [hazard ratio (HR), 4.21, P = 0.021), and the DFS was 58.3% in cases with Lk, while 88.7% in the counterpart. The multivariable logistic regression analysis revealed perioperative blood transfusion (P = 0.001) was independently associated with Lk. Intriguingly, Lk was closely associated with hematogenic recurrence (P = 0.003) rather than peritoneal or local recurrence. Although sustained increase of the serum C-reactive protein at 2 weeks after operation predicted poor prognosis, the mutitivariable analysis including the C-reactive protein level revealed that Lk still indicated the prognostic potential (HR, 3.70, P = 0.075).
Conclusions
The findings concluded that Lk may be a high risk for systemic recurrence in stage II CRC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Colorectal cancer (CRC) is the second most prevalent cancer,1, and chemotherapy has dramatically improved prognostic outcome of CRC patients over the past decades.2,3 Nevertheless, CRC remains the fourth leading cause of cancer death worldwide with about 530,000 deaths every year.1 Recently, as the prognostic outcome of stage III patients has been dramatically improved due to prevalent use of adjuvant chemotherapy and improvement of chemotherapy regimens,2,4 adjuvant chemotherapy is consented as standard therapy in stage III CRC. Similarly, application of adjuvant chemotherapy is under discussion for patients with high-risk stage II disease5 although no selecting marker has been clinically identified at present. In stage II patients, approximately 20% of the patients have yet suffered from recurrence in spite of potentially curative resection.6 Therefore, pre- or postoperative prognostic markers have been anticipated for selecting high-risk patients who may benefit from adjuvant chemotherapy after curative operation of stage II CRC. Several prognostic markers or predictors of chemosensitivity for stage II patients have been reported such as allelic imbalance,7 gene expression profiling by cDNA microarray,8 or microsatellite instability,9 respectively. However, such molecular markers have been unsuitable for routine application at present because they have not been finally validated yet and are still costly and time-consuming.
Anastomotic leakage (Lk) is thought to occur at a rate ranged from 3% to 18% and has been reported to be a risk factor for local recurrences in curatively operated CRC patients.10–12 In this meaning, at least patients with Lk may be potential candidate for adjuvant chemotherapy. However, these results were based upon curatively operated patients with CRC of several stages, and the impact of Lk on long-term survival remains controversial,10–14 especially in stage II CRC. Accordingly, clinicopathological factors including Lk were prognostically analyzed within stage II patients to evaluate whether Lk could be a clinically available parameter for predicting long-term prognosis.
Patients and Methods
Characteristics of Patients with Stage II CRC
A total of 1,101 patients having electively undergone surgical resection of primary CRC at the Kitasato University Hospital from January 1, 1990 to March 31, 2000, were reviewed. Patients with colorectal multiple cancer, malignant disease of other organ, familial adenomatous polyposis, or inflammatory bowel diseases, patients who underwent resections without anastomosis, and patients undergone emergency resection for perforation or one-stage resection for obstruction were excluded. Among the remaining 946 patients of sporadic CRC, 207 patients were diagnosed (21.9%) as stage II CRC disease and were operated on with curative intent. Preoperative chemotherapy or radiation therapy had not been performed in this cohort. Patients without obstruction received mechanical bowel preparation with polyethylene glycol electrolyte solution the day before surgery, and patients with obstruction and patients with rectal cancer received bedside orthograde colorectal lavage with lukewarm water. Prophylactic intravenous antibiotics were administered at the induction of anesthesia and 3 h after the beginning of operation. Patients were followed up until the recurrence of cancer or end point (April 30 2007). All patients were followed up at least every 3 months for the first year and every 6 months thereafter. Follow-up assessment involved a medical history-taking, physical examination, biologic tests, measurement of the serum CEA and CA19-9 levels, colonoscopy, chest radiography, abdominal ultrasonography (US), and chest/abdominal computed tomography (CT). Serum CEA and CA19-9 were usually evaluated every visit, and abdominal US and CT were performed every 6 months. Chest CT and colonoscopy were examined every year. Recurrence was diagnosed on the basis of imaging and, if necessary, either cytologic analysis or biopsy was performed. Patient demographics, tumor characteristics, and postoperative course were recorded and analyzed. Perioperative transfusion was defined as allogeneic blood transfusion during surgery or in the first two postoperative days, as in previous press,15 and was performed at the discretion of the treating surgeons and anesthesiologists. The number of total dissected lymph nodes was also classified according to previous press.16 Pathological TNM classification was made according to the UICC (Unio Internationalis Contra Cancrum) staging system.
Patients who received adjuvant chemotherapy for more than 3 months were defined as adjuvant chemotherapy “Yes” group. Adjuvant chemotherapy was consisted of oral administration of 5-fluorouracil (5FU)-based regimens: 5FU, Tegafur/uracil (UFT), or Furtulon (5′-deoxy-5-fluorouridine) alone, or one of them plus PSK (protein-bound polysaccharide K). Although curative operation alone is a standard therapy in stage II CRC at present, oral adjuvant chemotherapy had been recommended to patients with stage II CRC during the term of this patient cohort if they fulfilled the following eligibility criteria: age of 20 to 75 years; the absence of prior chemo-immunotherapy or radiotherapy, and the absence of severe liver dysfunction, heart failure, renal dysfunction, or other severe systemic complications. Therefore, patients who received oral adjuvant chemotherapy reached 180 cases, and the remaining 27 patients declined or did not fulfill the above criteria.
Lk was defined as any clinical or radiological evidence of dehiscence of the anastomosis: the presence of peritonitis caused by anastomosis dehiscence, the presence of feculent discharge from the drainage tube, or the presence of abscess with demonstration of Lk. These were also confirmed by radiography from drainage tube, hydrosoluble enema, or CT-guided abscess drainage except the cases with obvious feculent discharge from the drainage tube (Supplemental Table 1). Anastomotic dehiscence, which was basically diagnosed by, later, routine imagings prior to closure of diverting ileostomy, was not included. We performed routine imagings only for patients with diverting ileostomy prior to ileostomy closure more than 3 months after primary operation. Four patients underwent diverting ileostomy, but no anastomotic dehiscence was detected in such routine diagnosis.
Statistical Analysis
The relationship between Lk and clinicopathological parameters were assessed by Pearson’s chi-square test or Fisher’s exact test, as appropriate, and multivariate logistic regression analysis were performed to obtain an adjusted effect of each factor. The time of follow-up was calculated from the operation date for the primary lesion to the date of recurrence. Cumulative disease-free survival (DFS) of patients was estimated using the Kaplan–Meier method, and statistical significance of the difference of the survival rate between groups was tested using the log-rank test. For the Kaplan–Meier estimate of the survival curves, we truncated the data at a follow-up period of 5 years to avoid the number at risk to be too small. Those with a survival time of more than 5 years were reported to be 5 years, and events occurring after the end of the 5-year follow-up period were computed as censored data. Five-year cumulative DFS probability was estimated using the life table method with the interval length set at 1 month. Multivariable analysis was performed by employing the Cox proportional hazards model to examine the interaction between Lk and other clinicopathological variables and estimate the independent prognostic effect of Lk on survival by adjusting for confounding factors. For ordinal variable, when zero event was detected in the lowest exposure group, analyses was designed to be performed by grouping categories together, treating it as ordinal data to get an average effect, or by confounding sensitivity analyses excluding it from analysis. Within the present study population, there were 27 recurrences of stage II CRC which allows up to three variables to be included in a multivariable regression model. To avoid over-fitting, all potential confounding factors of Lk were reduced to one single composite characteristic by applying a propensity score.17 The conventional P value of 0.05 or less was used to determine the level of statistical significance. All reported P values are two-sided. Analyses were performed independently at our clinical research center using SPSS version 17.0 software (SPSS Inc., Chicago, IL).
Results
Patients’ Characteristics and Their Association with Lk
The clinicopathological characteristics were shown in Table 1. One hundred twenty-seven males and 80 females were analyzed with age being 61.0 ± 11.1 years. Lk occurred in 12 (5.8%) cases, and, among them, only one patient had a particularly preoperative complication (diabetes mellitus). The diabetes of this patient was well-controlled by insulin from preoperation through postoperation. And, there was no patient with other factors for poor nourishment such as medication of steroids. Lk occurred in 22.2% of patients with perioperative blood transfusion and in 1.2% of those without perioperative blood transfusion. Lk was significantly related to perioperative blood transfusion (P < 0.001, Fisher’s exact test), followed by T4 factor (direct invasion into other organ; P = 0.071), the elevation of preoperative CEA (P = 0.110), and tumor position (P = 0.129). Preoperative obstruction was observed in only one patient with Lk (Table 1). There was also no significance in relationship between Lk and obstruction in the present study population. Lk occurred in five cases (3.8%) in colon cancer and seven in rectal cancer (9.2%). Among them, two patients required ileostomy (reoperation) for Lk in colon cancer and five in rectal cancer, and one patient (colon cancer) underwent ileostomy before curative resection (two-stage operation) for obstruction, one patient (rectal cancer) underwent diverting ileostomy, and the remaining three patients were conservatively observed with percutaneous drainage and finally cured. The multivariable logistic regression analysis of these factors indicated that Lk was independently associated with perioperative blood transfusion (P < 0.001).
Kaplan–Meier Estimate of 5-Year DFS
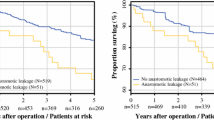
All the patients were included in the survival analysis. The overall follow-up period ranged from 2 to 207 months (median, 116 months), and the mean DFS was 55.4 months corresponding to a 5-year follow-up. Because a cumulative DFS probability of 50% was not yet reached by the end of 5-year follow-up, the overall median DFS time was not determined. The overall DFS rate was 87.0% (27 cases with recurrence and 180 cases without recurrence). Five-year cumulative DFS of patients with Lk was remarkably worse (58.3%), which corresponded to stage III CRC (63.2%), compared with those without Lk (88.7%; P < 0.001, Fig. 1a). Lymphatic involvement (ly; P = 0.119) and vascular involvement (v; P = 0.086) tended to indicate poor prognosis (Supplemental Fig. 1a, b), and patients with both ly and v involvement (n = 28) showed significantly poor prognosis (DFS, 84.9%) compared with the counterpart (n = 179; 100.0%; P = 0.033; Supplemental Fig. 1c).
When separately analyzed on tumor position, Lk still significantly affected adversely on long-term prognosis in both colon and rectum (Fig. 1b, c), and there was no significant difference between DFS of patients with Lk in colon cancer (60.0%) and that in rectal cancer (57.1%). In addition, Lk was the only significant prognostic factor, and there was no factor which had prognostic potential (P < 0.1) both in colon and rectum when separately analyzed (data not shown).
Contribution of Lk to the Risk of Recurrence with Multivariable Analysis
Cox proportional hazards model was applied to estimate the effect of Lk on DFS. Lk was the only significant prognostic factor, and there was no other factor which had prognostic potential (P < 0.1). The crude hazard ratio (HR) of Lk-positive compared to Lk-negative was 4.38 (95% confidence interval (CI), 1.66–11.58; P = 0.003), which indicated Lk increased the risk of recurrence of CRC and cancer-related death by more than four times that of without Lk. The effect of Lk on recurrence in colon and rectal cancer group gave similar results: crude HR (95%CI) was 4.1 (0.9–17.9) for the colon group and 4.9 (1.3–19.0) for the rectal group.
Before multivariable analyses were adopted to estimate adjusted effect of Lk on DFS, we further confirmed that there was no interaction effect between cancer position (colon or rectum) and Lk (P = 0.874); taking into account that evaluation in each group would result in a small sample size and thus decrease the power of the study, we finally combined them together. Potential confounders of variables were included in the multivariable analysis (Table 2). The adjusted HR of Lk became 5.27 (95%CI, 1.54–18.10; P = 0.008) in comparison to Lk-negative. We also performed an analysis by using propensity score to adjust the effect of Lk by transforming all other confounding variables into a single estimator and revealed that, after the adjustment, the HR of Lk became 4.21 (95%CI, 1.24–14.33; P = 0.021). These findings suggested that Lk seems to be an independent and significant risk factor of poorer DFS (Table 2).
Lk was Associated with Hematogenic Recurrence Rather than Local or Peritoneal Recurrence in Stage II CRC
Next, first recurrence site in patients with stage II CRC was analyzed according to Lk. Interestingly, Lk was correlated with hematogenic recurrence (P = 0.003 by Fisher’s exact test) rather than local recurrence or peritoneal dissemination (P = 0.605; Table 3). Therefore, Lk may cause systemic micrometastasis, leading to systemic recurrence.
Effect of Lk on DFS When Taking Systemic Inflammatory Response into Account
Recently, a systemic inflammatory response, as evidenced by raised circulating levels of C-reactive protein (CRP), has been reported to be associated with poor survival in patients who underwent potentially curative resection for CRC.18 These reports may explain the above implication of Lk in systemic recurrences, hence circulating level of CRP was analyzed, which was measured as a part of routine blood examination either before or after potentially curative resection for stage II CRC. CRP level was classified as raised (≥1.0 mg/dl) or normal (<1.0 mg/dl) from a clinical practice view. Lk was significantly correlated with CRP level at 1 or 2 weeks after curative operation (P = 0.018, 0.003, respectively, by Fisher’s exact test; Supplemental Table 2). Moreover, the sustained elevation of CRP level at 2 weeks after operation predicted significantly worse prognosis (DFS, 75.0%) than its counterpart (89.3%; P = 0.022, compared by log-rank test, Supplemental Fig. 2), while preoperative CRP and CRP at 1 week after operation did not show prognostic significance (data not shown). The multivariable prognostic analysis including CRP at 2 weeks after operation (n = 175) showed that Lk still indicated prognostic potential (HR, 3.70, P = 0.075; Table 4). This result suggests that Lk is more strongly associated with recurrence independent of sustained systemic inflammation.
Discussion
The present study showed that an anastomotic leakage (Lk) was closely associated with an adverse impact on long-term DFS (5-year DFS, 58.3%) in patients who underwent potentially curative resection for stage II CRC, and it was the most robust independent prognostic factor. This DFS was comparable to that of patients with stage III CRC. Although intramural vessel involvement may be available for the selection of low-risk patients (DFS, 100.0%), it was insufficient for the patient selection who have high risk of recurrence and would be rather low-risk selection (Supplemental Fig. 1). Therefore, with regard to patient selection, Lk alone may be potential classifier of stage II CRC. Lk has been reported to be a risk factor of local recurrences in curatively operated CRC patients10–12,19 which included several stage CRCs. However, to our knowledge, our study is the first report concerning Lk with high risk of recurrence limited in stage II disease. Interestingly, in our study, Lk was significantly implicated in systemic recurrence (P = 0.003) rather than local recurrence in stage II.
In our study, there was no prognostic difference between colon cancer and rectal cancer. Although tumor position did not affect Lk and long-term prognosis in this study, anastomosis and prognosis in rectal cancer is thought to be affected by various factors compared with that in colon cancer.10,20–23 However, even when separately analyzed on tumor positions, Lk was still significant prognostic factor (Fig. 1b, c).
Adjuvant chemotherapy for stage II CRC has been controversial at present because stage II patients show good prognosis and only a part of high-risk stage II patients may benefit in prognosis from previous studies.6,24,25 Nevertheless, at present, standard chemotherapy is not recommended for stage II CRC patients because of excellent prognosis. Our current study included many such patients even with Lk who actually underwent adjuvant chemotherapy, but which did not include the most active agents such as oxaliplatin, CPT-11, bevacizumab, or cetuximab, suggesting that Lk anyway showed high risk for stage II CRC irrespective of adjuvant therapy. Therefore, our current result is worthy of further study on high-risk patient selection in stage II CRC and also on more powerful adjuvant chemotherapy such as FOLFOX in stage II patients with Lk in order to elucidate the benefit of adjuvant chemotherapy for these patients. In addition, neoadjuvant chemo-radiotherapy for locally advanced rectal cancer is now becoming standard. However, during the terms of this current study, we did not think that neoadjuvant treatment is really effective for such patients from a prognostic point of view. Thus, Lk in patients with neoadjuvant treatment should be also studied in the future.
Several parameters have been reported as independent prognostic factor or chemosensitive marker for patient selection allowing for the application of adjuvant chemotherapy in stage II CRC.6,24,26 The number of evaluated lymph nodes,27 T4 factor (direct invasion into adjacent structure),16,28 tumor budding/infiltrating,29 vascular involvement,16,28 or perforation through the tumor28 were such high-risk potential markers. In the present study, vascular involvement tended to be a prognostic factor, however, it was not insufficient to select high-risk patients. On the other hand, the number of evaluated lymph nodes and T4 factor did not indicate any prognostic significance in our current cohort of stage II CRC. Several molecular and genetic markers have also been reported to indicate poor prognosis of stage II CRC such as the DNA aneuploid,30 17p or 18q allelic imbalance,7 gene expression profiling by cDNA microarray,8 and micrometastasis detected by reverse transcriptase-polymerase chain reaction of CEA31 or CK20.32 In addition, microsatellite instability (MSI) has been reported as chemoresistant marker.9 Actually, the largest stage II colon cancer trial (ECOG 5202, the US Gastrointestinal Intergroup including the National Cancer Institute of Canada) is ongoing, in which patients are now selected prospectively for adjuvant chemotherapy based on 18q loss of heterozygosity and MSI status.33 Nevertheless, all such genetic and molecular tools are unsuitable for routine application at present because they are costly and time-consuming methods and have not been validated yet. In this meaning, Lk is easily available for patient selection at any minute.
Viable cancer cells in the lumen may be present at the site of the anastomosis at the time of surgery, which can be detected on suture or staple lines of anastomosis,34 and on the occasion of Lk, those may be capable of implantation and subsequent local recurrence.35 However, this theory alone did not explain the association of Lk with systemic recurrence in the present study. Systemic inflammatory response, as evidenced by raised circulating concentrations of CRP, has been reported to predict recurrence and disease-specific survival in curatively operated CRC patients.18 Consistently, the sustained CRP elevation at either 1 or 2 weeks after operation was significantly associated with Lk, and especially, CRP at 2 weeks after operation per se predicted poor prognosis (P = 0.022) in the present study. CRP may reflect the inflammatory response promoted by various cytokines which are presumably released from leukocytes in the malignant process.36 On the other hand, a raised CRP level was thought to be related to the reduction of circulating lymphocytes.37 In addition, the reduction of lymphocytes in the peripheral blood was shown to reflect the immune suppression in patients with malignant tumor,38 and tumor-induced immune suppression adversely affects their prognosis.39
Perioperative allogeneic blood transfusion was reported to be an independent risk factor for Lk in a dose-dependent manner.23 Also in the present study, perioperative blood transfusion affected Lk most robustly even when CRP was included in the multivariable logistic analysis (data not shown). Allogenic blood transfusion impairs the cell-mediated immune response40 and predisposes to postoperative infectious complication,41 and cell-mediated immune responses, which include mainly macrophage and T-lymphocyte, has been thought to affect the healing process.42 Tadros T. et al. reported that perioperative blood transfusion impaired the healing of experimental intestinal anastomosis in an animal model using bursting pressure of anastomosis, in addition, cell-mediated immune response, as evidenced by exogenous IL-2, reversed the negative effects of blood transfusion on anastomotic repair.43 Taken together, Lk may lead to systemic recurrences partly through cancer immune suppression together with sustained CRP elevation and perioperative blood transfusion. Conversely, we could also say that Lk is favored by a local depression of the immune system for the presence of undetected micrometastasis.
Recently, it has been suggested that tumor progression such as invasion and metastasis is coordinated by both cancer cells and host stromal cells, which consist tumor microenvironment.44–46 A variety of host bone marrow-derived cells, which include inflammatory cells, cancer-associated fibroblasts, and endothelial progenitor cells compose of a tumor microenvironment.47–49 Host inflammatory cells produce much more TGF-β than tumor cells, leading to inhibition of host tumor immune surveillance,50,51 which may lead to cancer cell escape and intravasate into circulation. Local inflammation caused by Lk may additionally affect the above mechanism and may result in metastasis-prone phenotype. However, in order to answer the reason why Lk was associated with systemic recurrence, further experimental studies, such as comparison of circulating cancer cells or cytokines in both patients and experimental model, may be needed.
In conclusion, we showed that Lk was the most robust independent prognostic factor among the clinicopathological factors in stage II CRC. These results suggest that Lk may be appropriate for the selection of high-risk patients. And, Lk was associated with systemic recurrence in both colon and rectal cancer. Because Lk necessarily occurs at a given rate in spite of perioperative treatment with maximal attention and it is immediately available for clinical use from cost and technical point of view, Lk could be a factor for selecting high-risk patients. As only 12 patients (out of 207) had an Lk in this study, the prognostic impact of Lk should be validated in a larger study. On the other hand, because the DFS of patients without Lk was still 88.7%, further molecular tools would be necessary.
References
Parkin DM, Bray F, Ferlay J, Pisani P: Global cancer statistics, 2002. CA Cancer J Clin 2005;55:74–108.
Cunningham D, Starling N: Adjuvant chemotherapy of colorectal cancer. Lancet 2007;370:1980–1981.
Goldberg RM, Sargent DJ, Morton RF, Fuchs CS, Ramanathan RK, Williamson SK, Findlay BP, Pitot HC, Alberts SR: A randomized controlled trial of fluorouracil plus leucovorin, irinotecan, and oxaliplatin combinations in patients with previously untreated metastatic colorectal cancer. J Clin Oncol 2004;22:23–30. Epub 2003 Dec 2009.
Kuebler JP, Wieand HS, O’Connell MJ, Smith RE, Colangelo LH, Yothers G, Petrelli NJ, Findlay MP, Seay TE, Atkins JN, Zapas JL, Goodwin JW, Fehrenbacher L, Ramanathan RK, Conley BA, Flynn PJ, Soori G, Colman LK, Levine EA, Lanier KS, Wolmark N: Oxaliplatin combined with weekly bolus fluorouracil and leucovorin as surgical adjuvant chemotherapy for stage II and III colon cancer: results from NSABP C-07. J Clin Oncol 2007;25:2198–2204.
Andre T, Sargent D, Tabernero J, O’Connell M, Buyse M, Sobrero A, Misset JL, Boni C, de Gramont A: Current issues in adjuvant treatment of stage II colon cancer. Ann Surg Oncol 2006;13:887–898. Epub 2006 Apr 2014.
Benson AB, 3 rd, Schrag D, Somerfield MR, Cohen AM, Figueredo AT, Flynn PJ, Krzyzanowska MK, Maroun J, McAllister P, Van Cutsem E, Brouwers M, Charette M, Haller DG: American Society of Clinical Oncology recommendations on adjuvant chemotherapy for stage II colon cancer. J Clin Oncol 2004;22:3408–3419. Epub 2004 Jun 3415.
Diep CB, Thorstensen L, Meling GI, Skovlund E, Rognum TO, Lothe RA: Genetic tumor markers with prognostic impact in Dukes’ stages B and C colorectal cancer patients. J Clin Oncol 2003;21:820–829.
Wang Y, Jatkoe T, Zhang Y, Mutch MG, Talantov D, Jiang J, McLeod HL, Atkins D: Gene expression profiles and molecular markers to predict recurrence of Dukes’ B colon cancer. J Clin Oncol 2004;22:1564–1571.
Ribic CM, Sargent DJ, Moore MJ, Thibodeau SN, French AJ, Goldberg RM, Hamilton SR, Laurent-Puig P, Gryfe R, Shepherd LE, Tu D, Redston M, Gallinger S: Tumor microsatellite-instability status as a predictor of benefit from fluorouracil-based adjuvant chemotherapy for colon cancer. N Engl J Med 2003;349:247–257.
Branagan G, Finnis D: Prognosis after anastomotic leakage in colorectal surgery. Dis Colon Rectum 2005;48:1021–1026.
Fujita S, Teramoto T, Watanabe M, Kodaira S, Kitajima M: Anastomotic leakage after colorectal cancer surgery: a risk factor for recurrence and poor prognosis. Jpn J Clin Oncol 1993;23:299–302.
Ptok H, Marusch F, Meyer F, Schubert D, Gastinger I, Lippert H: Impact of anastomotic leakage on oncological outcome after rectal cancer resection. Br J Surg 2007;94:1548–1554.
McArdle CS, McMillan DC, Hole DJ: Impact of anastomotic leakage on long-term survival of patients undergoing curative resection for colorectal cancer. Br J Surg 2005;92:1150–1154.
Walker KG, Bell SW, Rickard MJ, Mehanna D, Dent OF, Chapuis PH, Bokey E L: Anastomotic leakage is predictive of diminished survival after potentially curative resection for colorectal cancer. Ann Surg 2004;240:255–259.
Yamashita K, Sakuramoto S, Kikuchi S, Katada N, Kobayashi N, Watanabe M: Transfusion alert for patients with curable cancer. World J Surg 2007;31:2315–2322.
Morris M, Platell C, de Boer B, McCaul K, Iacopetta B: Population-based study of prognostic factors in stage II colonic cancer. Br J Surg 2006;93:866–871.
Rubin DB: Estimating causal effects from large data sets using propensity scores. Ann Intern Med 1997;127:757–763.
McMillan DC, Canna K, McArdle CS: Systemic inflammatory response predicts survival following curative resection of colorectal cancer. Br J Surg 2003;90:215–219.
Law WL, Choi HK, Lee YM, Ho JW, Seto CL: Anastomotic leakage is associated with poor long-term outcome in patients after curative colorectal resection for malignancy. J Gastrointest Surg 2007;11:8–15.
Alves A, Panis Y, Trancart D, Regimbeau JM, Pocard M, Valleur P: Factors associated with clinically significant anastomotic leakage after large bowel resection: multivariate analysis of 707 patients. World J Surg 2002;26:499–502.
Lipska MA, Bissett IP, Parry BR, Merrie AE: Anastomotic leakage after lower gastrointestinal anastomosis: men are at a higher risk. ANZ J Surg 2006;76:579–585.
Peeters KC, Tollenaar RA, Marijnen CA, Klein Kranenbarg E, Steup WH, Wiggers T, Rutten HJ, van de Velde CJ: Risk factors for anastomotic failure after total mesorectal excision of rectal cancer. Br J Surg 2005;92:211–216.
Yeh CY, Changchien CR, Wang JY, Chen JS, Chen HH, Chiang JM, Tang R: Pelvic drainage and other risk factors for leakage after elective anterior resection in rectal cancer patients: a prospective study of 978 patients. Ann Surg 2005;241:9–13.
Figueredo A, Charette ML, Maroun J, Brouwers MC, Zuraw L: Adjuvant therapy for stage II colon cancer: a systematic review from the Cancer Care Ontario Program in evidence-based care’s gastrointestinal cancer disease site group. J Clin Oncol 2004;22:3395–3407.
Quasar Collaborative G, Gray R, Barnwell J, McConkey C, Hills RK, Williams NS, Kerr DJ (2007) Adjuvant chemotherapy versus observation in patients with colorectal cancer: a randomised study. Lancet;370:2020–2029.
Andre T, Boni C, Mounedji-Boudiaf L, Navarro M, Tabernero J, Hickish T, Topham C, Zaninelli M, Clingan P, Bridgewater J, Tabah-Fisch I, de Gramont A: Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. N Engl J Med 2004;350:2343–2351.
Caplin S, Cerottini JP, Bosman FT, Constanda MT, Givel JC: For patients with Dukes’ B (TNM Stage II) colorectal carcinoma, examination of six or fewer lymph nodes is related to poor prognosis. Cancer 1998;83:666–672.
Petersen VC, Baxter KJ, Love SB, Shepherd NA: Identification of objective pathological prognostic determinants and models of prognosis in Dukes’ B colon cancer. Gut 2002;51:65–69.
Nakamura T, Mitomi H, Kanazawa H, Ohkura Y, Watanabe M: Tumor budding as an index to identify high-risk patients with stage II colon cancer. Dis Colon Rectum 2008;51:568–572.
Garrity MM, Burgart LJ, Mahoney MR, Windschitl HE, Salim M, Wiesenfeld M, Krook JE, Michalak JC, Goldberg RM, O’Connell MJ, Furth AF, Sargent DJ, Murphy LM, Hill E, Riehle DL, Meyers CH, Witzig TE: Prognostic value of proliferation, apoptosis, defective DNA mismatch repair, and p53 overexpression in patients with resected Dukes’ B2 or C colon cancer: a North Central Cancer Treatment Group Study. J Clin Oncol 2004;22:1572–1582.
Noura S, Yamamoto H, Ohnishi T, Masuda N, Matsumoto T, Takayama O, Fukunaga H, Miyake Y Ikenaga M, Ikeda M, Sekimoto M, Matsuura N, Monden M: Comparative detection of lymph node micrometastases of stage II colorectal cancer by reverse transcriptase polymerase chain reaction and immunohistochemistry. J Clin Oncol 2002;20:4232–4241.
Koch M, Kienle P, Kastrati D, Antolovic D, Schmidt J, Herfarth C, von Knebel Doeberitz M, Weitz J: Prognostic impact of hematogenous tumor cell dissemination in patients with stage II colorectal cancer. Int J Cancer 2006;118:3072–3077.
Benson AB, 3rd: New approaches to assessing and treating early-stage colon and rectal cancers: cooperative group strategies for assessing optimal approaches in early-stage disease. Clin Cancer Res 2007;13:6913 s–6920 s.
Gertsch P, Baer HU, Kraft R, Maddern GJ, Altermatt HJ: Malignant cells are collected on circular staplers. Dis Colon Rectum 1992;35:238–241.
Umpleby HC, Fermor B, Symes MO, Williamson RC: Viability of exfoliated colorectal carcinoma cells. Br J Surg 1984;71:659–663.
Gabay C, Kushner I: Acute-phase proteins and other systemic responses to inflammation. N Engl J Med 1999;340:448–454.
Nozoe T, Matsumata T, Sugimachi K: Preoperative elevation of serum C-reactive protein is related to impaired immunity in patients with colorectal cancer. Am J Clin Oncol 2000;23:263–266.
Oka M, Hirazawa K, Yamamoto K, Iizuka N, Hazama S, Suzuki T, Kobayashi N: Induction of Fas-mediated apoptosis on circulating lymphocytes by surgical stress. Ann Surg 1996;223:434–440.
Eilber FR, Morton DL: Impaired immunologic reactivity and recurrence following cancer surgery. Cancer 1970;25:362–367.
Waymack JP, Rapien J, Garnett D, Tweddell JS, Alexander JW: Effect of transfusion on immune function in a traumatized animal model. Arch Surg 1986;121:50–55.
Jensen LS, Andersen AJ, Christiansen PM, Hokland P, Juhl CO, Madsen G, Mortensen J, Moller-Nielsen C, Hanberg-Sorensen F, Hokland M: Postoperative infection and natural killer cell function following blood transfusion in patients undergoing elective colorectal surgery. Br J Surg 1992;79:513–516.
Barbul A, Breslin RJ, Woodyard JP, Wasserkrug HL, Efron G: The effect of in vivo T helper and T suppressor lymphocyte depletion on wound healing. Ann Surg 1989;209:479–483.
Tadros T, Wobbes T, Hendriks T: Opposite effects of interleukin-2 on normal and transfusion-suppressed healing of experimental intestinal anastomoses. Ann Surg 1993;218:800–808.
Kaplan RN, Rafii S, Lyden D: Preparing the “soil”: the premetastatic niche. Cancer Res 2006;66:11089–11093.
Lyden D, Hattori K, Dias S, Costa C, Blaikie P, Butros L, Chadburn A, Heissig B, Marks W, Witte L, Wu Y, Hicklin D, Zhu Z, Hackett NR, Crystal RG, Moore MA, Hajjar KA, Manova K, Benezra R, Rafii S: Impaired recruitment of bone-marrow-derived endothelial and hematopoietic precursor cells blocks tumor angiogenesis and growth. Nat Med 2001;7:1194–1201.
Rafii S, Lyden D: S100 chemokines mediate bookmarking of premetastatic niches. Nat Cell Biol 2006;8:1321–1323.
Du R, Lu KV, Petritsch C, Liu P, Ganss R, Passegue E, Song H, Vandenberg S, Johnson RS, Werb Z, Bergers G: HIF1alpha induces the recruitment of bone marrow-derived vascular modulatory cells to regulate tumor angiogenesis and invasion. Cancer Cell 2008;13:206–220.
Katoh H, Hosono K, Ito Y, Suzuki T, Ogawa Y, Kubo H, Kamata H, Mishima T, Tamaki H, Sakagami H, Sugimoto Y, Narumiya S, Watanabe M, Majima M: COX-2 and prostaglandin EP3/EP4 signaling regulate the tumor stromal proangiogenic microenvironment via CXCL12-CXCR4 chemokine systems. Am J Pathol 2010;176:1469–1483.
Yang L, DeBusk LM, Fukuda K, Fingleton B, Green-Jarvis B, Shyr Y, Matrisian LM, Carbone DP, Lin PC: Expansion of myeloid immune suppressor Gr + CD11b + cells in tumor-bearing host directly promotes tumor angiogenesis. Cancer Cell 2004;6:409–421.
Li MO, Flavell RA: TGF-beta: a master of all T cell trades. Cell 2008;134:392–404.
Yang L, Huang J, Ren X, Gorska AE, Chytil A, Aakre M, Carbone DP, Matrisian LM, Richmond A, Lin PC, Moses HL: Abrogation of TGF beta signaling in mammary carcinomas recruits Gr-1 + CD11b + myeloid cells that promote metastasis. Cancer Cell 2008;13:23–35.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Materials
Below is the link to the electronic supplementary material.
Supplemental Fig. 1
Kaplan–Meier analysis of 5-year DFS according to (A) lymphatic involvement (ly), (B) vascular involvement (v), and (C) both ly and v involvement (VI) (GIF 22 kb)
Supplemental Fig. 2
Kaplan–Meier analysis of 5-year DFS according to CRP at 2 week after operation [post-CRP (2w)] (n = 175) (GIF 18 kb)
Supplemental Table 1
Diagnostic details of the patients with anastomotic leakage (XLS 14.5 kb)
Supplemental Table 2
Correlation between CRP and clinicopathological factors in stage II patients (XLS 46.0 kb)
Rights and permissions
About this article
Cite this article
Katoh, H., Yamashita, K., Wang, G. et al. Anastomotic Leakage Contributes to the Risk for Systemic Recurrence in Stage II Colorectal Cancer. J Gastrointest Surg 15, 120–129 (2011). https://doi.org/10.1007/s11605-010-1379-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-010-1379-4