Abstract
Background and Objective
The role of liver resection in advanced hepatocellular carcinoma (multinodular or with macroscopic vascular involvement) is still controversial. The aim of this study is to evaluate the role of surgical resection compared to other therapeutic modalities in patients with advanced hepatocellular carcinoma (HCC).
Methods
Four hundred sixty four patients with HCC observed from 1991 to 2007 were included in the study. All the patients were evaluated for the treatment of HCC in relation to the severity of liver impairment and tumor stage. All the patients included in the study had no evidence of distant metastases.
Results
Median follow up time for surviving patients was 25 months (range 1–155). Two-hundred and eighty-three patients were in Child–Pugh class A, 161 in class B, and 20 in class C. Two-hundred and seventy-one patients had single HCC, 121 patients had two or three HCCs, and 72 more than three HCCs. One-hundred and thirty-six patients (29.3%) were submitted to liver resection (LR), 232 (50.0%) to local ablative therapies (LAT) (ethanol injection, radiofrequency ablation, chemoembolization), eight (1.7%) to liver transplantation (LT), and 88 (19%) to supportive therapy (ST). Median survival time for all patients was 36 months (95% CI 24–36). Median survival time was 57 months for LR, 30 months for LAT, and 8 months for ST, with a 5-year survival of 47%, 20%, and 2.5%, respectively (p = 0.001). Actuarial 5-year survival for patients submitted to LT was 75%. Overall survival was significantly shorter in patients with multiple HCCs compared to single HCC, with median survival times of 39, 16, and 11 months for patients with a single HCC, with two to three HCCs, and with more than three HCCs, respectively (p = 0.01). Survival for patients with single HCC was significantly longer in patients submitted to LR compared to LAT and ST with median survival times of 57, 37, and 14 months, respectively (p = 0.02). Also, in patients with multinodular HCCs (2–3 HCCs) LR showed the best results with a median survival time of 58 months compared to 22 and 8 months for LAT and ST (p = 0.01). In patients with more than three HCCs, LR did not show different results compared to LAT and ST. Seventy-three patients had evidence of macroscopic vascular involvement; median survival in this subgroup of patients was significantly shorter compared to patients without vascular involvement, 10 and 36 months, respectively. Survival for patients with macroscopic vascular involvement submitted to LR or LAT was significant longer compared to ST, with mean survivals of 27, 30, and 12 months, respectively (p = 0.01).
Conclusions
The present study shows that the surgery can achieve good results in patients with single HCC and good liver function. Also, patients with multinodular HCCs (two to three nodules) could benefit from LR where survival is longer than after LAT or ST. In patients with more than three HCCs, LR have similar results of LAT. Macroscopic vascular invasion is a major prognostic factor, and the LR is justified in selected patients, where it can allow good long-term results compared to ST.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hepatocellular carcinoma (HCC) is a common cancer worldwide and is the third most common cause of cancer-related deaths.1 The choice upon different types of treatment depends on tumor stage and the functional status of the liver.2 Liver resection (LR) and liver transplantation (LT) are considered the mainstay of curative therapy, although application of LT is limited by the shortage of organs.
Several local ablative therapies (percutaneous ethanol injection, radiofrequency ablation, microwave ablation, chemoembolization) have been proposed for patients with advanced HCC or severe liver impairment and showed benefits for long-term survival. In patients with multiple HCC or with macroscopic vascular involvement, the choice of treatment is still controversial, and LR is frequently contraindicated.3,4
Several authors have proposed hepatic resection in selected groups of patients for multiple HCC and with macroscopic vascular involvement demonstrating encouraging results.5 The objective of this study is to evaluate the results of LR compared to local ablative therapies (LAT) and to best supportive therapy (ST) in different groups of cirrhotic patients with advanced HCCs (multinodular or with macroscopic vascular involvement).
Patients and Methods
In this study, we review data of a multi-institutional database that included four different departments (one surgical department, two departments of internal medicine, and one department of gastroenterology). This database comprises 464 patients with liver cirrhosis and HCC observed during the period from January 1991 to March 2007.
All patients had liver cirrhosis. The diagnosis of liver cirrhosis was made with biopsy or with clinical and laboratory criteria of chronic hepatic disease associated with portal hypertension. The diagnosis of HCC was made with cytological or histological criteria or with radiological criteria. From 2001 the diagnosis of HCC was based on concordance between two imaging techniques [ultrasonography, computed tomography (CT), or magnetic resonance imaging (MRI)] showing arterial hypervascularity in a focal lesion ≥2 cm or with the combined criteria of an imaging technique and a serum alpha-fetoprotein (AFP) level greater than 400 ng/ml, according to the criteria of the consensus conference of the European Association for Study of the Liver.6 A fine-needle cytology was performed only in patients with an otherwise uncertain diagnosis.
Before treatment, all patients had serum liver function tests (bilirubin, alkaline phosphatase, transaminase, albumin, prothrombin time), blood count, and serum creatinine level. All patients were staged according to the Child–Pugh classification. The assessment of tumor stage was made with different imaging techniques: ultrasonography, contrast-enhanced CT, and contrast-enhanced MRI.
We reviewed patients’ records for demographic variables (age, gender, etiology of liver cirrhosis), severity of liver cirrhosis (Child–Pugh class), and tumor stage (size, number, macroscopic vascular invasion and AFP). The patients were evaluated for different therapies (LT, LR, LAT, or ST) according to the degree of liver dysfunction and the stage of tumor. All the patients included into the study did not have extrahepatic metastasis.
LT was considered for patients within Milan criteria and for patients with absence of macroscopic vascular invasion or extrahepatic metastasis by imaging techniques.
LR was the treatment of choice for patients with single HCC and well preserved liver function (Child–Pugh A) without portal vein hypertension. Surgical resection was also applied in a selected group of patients with multiple HCCs or with Child–Pugh class B liver dysfunction.
During surgery, intraoperative ultrasonography was routinely used in order to confirm preoperative diagnosis, to evaluate relationship between tumor and blood vessel and to evaluate the presence of additional tumors.
LAT (radiofrequency ablation, ethanol injection, chemoembolization) were indicated for patients excluded from surgery with single or multiple HCCs with Child–Pugh A and B liver cirrhosis. From 1991, percutaneous ethanol injection (PEI) was applied to lesions up to 3 cm, and from 1998, radiofrequency ablation (RFA) was introduced to treat lesions up to 6 cm. PEI and RFA were applied in patients with fewer than four nodules of HCCs. Chemoembolization (TACE) was indicated for patients excluded from surgical therapy and for patients with single or multiple HCCs and preserved liver function (Child–Pugh class A and B) without main portal vein thrombosis. TACE was also applied in selected patients in conjunction with other LAT (PEI, RFA). Patients excluded from other treatment due to severe liver dysfunction (Child–Pugh C class) or advanced tumor stage (multinodular, main portal vein thrombosis) were submitted to ST.
After treatment, all patients underwent regular follow up with serum AFP and ultrasonography every 6 months. Suspect recurrences were confirmed with CT or MRI. Chest CT or bone scan were performed in case of recurrence or of clinical suspect of distant metastases. All recurrences were evaluated for new treatment; the choice of the type of treatment was related to the number and size of tumors, the presence of extrahepatic disease, the liver function, and the general status of the patient.
Statistical Analysis
Data were collected and analyzed with SPSS statistical software (SPSS version 16.0, Chicago, IL, USA). The differences between categorical variables were analyzed with a chi-square test. The differences between continuous variables were analyzed with t test.
Survival analysis was carried out with Kaplan–Meier method; we considered the treatment day as time zero, and patients that were alive at the end of follow-up were considered censored.
Univariate analysis for survival was performed with the Kaplan–Meier method with the log rank test to verify significance of differences. Cox’s regression model was utilized for multivariate analysis.
Results
The clinical features of the 464 patients included in the study are reported in Table 1. There were 381 males and 83 females, for a male-to-female ratio of 4.6:1. The median patients’ age was 68 years (range 28–90). The preoperative liver function according to Child–Pugh classification classified 283 patients in class A, 161 patients in class B, and 20 patients in class C. The liver cirrhosis was related to alcohol intake in 157 patients, to HCV infection in 222, to HBV infection in 45, and to other causes in 40. Tumors were single in 271 patients and multiple in 193. The mean number of tumors was 1.9 (range 1–5), with 121 patients with two to three tumors and 72 with more than three tumors. The mean tumor diameter was 4.3 cm (range 1.5–20 cm). The mean AFP level was 385 ng/ml (range 1–21,000), with 357 patients (77%) with AFP level lower than 100 ng/ml.
Among 464 patients, eight underwent LT (1.7%), 136 underwent LR (29.3%), 232 underwent LAT (50%), and 88 (19%) underwent ST. Among patients submitted to LAT, 29 patients underwent PEI, 128 underwent RFA, 39 underwent TACE, and 36 underwent TACE associated with RFA.
The survival analysis of the entire group of study identified a median survival time of 30 months, with actuarial 3-, 5-, and 10-year survivals of 46%, 25%, and 10%, respectively. The univariate analysis for factors related with survival identified that the number and the size of HCC, Child–Pugh class, serum AFP level, and presence of macroscopic vascular involvement were significantly related with shorter survival time (Table 2). Cox’s multivariate regression model identified that survival-related factors were AFP level, type of therapy, and size and number of HCC, with hazard ratios of 1.73, 0.71, 1.40, and 1.29, respectively (Table 3).
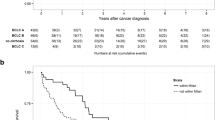
The survival according to the type of treatment showed median survival times for patients submitted to LR, LAT, and ST of 57, 30, and 8 months and with 5-year actuarial survival rates of 47%, 20%, and 2.5%, respectively. Actuarial 5-year survival for patients submitted to LT was 75%. Patients submitted to LT were not included in further statistical analysis due to small sample size in this group.
The univariate analysis for prognostic factor for survival in patients submitted to LR identified that the number and the size of HCC, Child–Pugh class, and presence of macroscopic vascular involvement were significantly related with shorter survival time (Table 4). Cox’s multivariate regression model identified that the presence of macroscopic vascular involvement was the most significant factor related with survival with a hazard ratio of 7.1.
During the follow up, 223 patients that submitted to LR or LAT showed recurrence of the HCC; the median disease-free survival was 16 months, with actuarial 3- and 5-year disease-free survival rates of 30% and 15%. The recurrence rate was significantly different among patients submitted to LR and LAT, with 5-year disease-free survivals of 22% and 12%, respectively (p < 0.001).
Subgroup Analysis
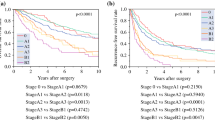
Further survival analyses among different categories are reported in Table 5. In patients with well preserved liver function (Child–Pugh A) and single HCC, LR showed the best results compared to LAT or ST, with median survival times of 63, 41, and 4 months, respectively (p = 0.01). In patients with single HCC and Child Pugh B cirrhosis, LR and LAT did not show significant differences, with median survival times of 24 and 30 months, respectively.
Patients with multiple HCCs (two to three HCCs) submitted to LR showed a longer survival compared to LAT and ST, with median survival times of 58, 22, and 8 months, respectively (p = 0.01). In patients with more than three HCCs, the results of LR and LAT did not show significant differences.
In the study group, 73 patients showed macroscopic vascular involvement at preoperative imaging. Among these patients, 17 were submitted to LR, 17 to LAT, and 39 to ST. Macroscopic vascular involvement was a negative prognostic factor with a 5-year survival of 5% compare to 28% for patients without macroscopic vascular involvement. Survival analysis showed no differences in survival between LR and LAT, whereas we identified a significantly longer survival for patients submitted to LR compared to ST, with median survival times of 10 and 7, respectively (p = 0.05).
Discussion
HCC is the most common primary liver cancer and is the most severe complication of chronic liver diseases.7 The prognosis is poor even after potentially curative treatments, with a 5-year survival rate of 47% and a 5-year recurrence rate of 80%, respectively.8
A peculiar feature of HCC is that several therapeutic approaches (liver transplantation, surgical resection, LAT, chemoembolization) can be chosen in relation to the stage of disease and severity of liver impairment. Liver transplantation is indicated for early HCC in order to treat both the neoplastic disease and the liver impairment.9 The Milan criteria are widely accepted for liver transplantation in patients with HCC, and when these criteria are fulfilled, 5-year survival reaches 60–80%, with a recurrence rate lower than 20%.10
The local ablative treatments (PEI and RFA) are widespread and allow good results, with 3-year survivals of 83% in Child–Pugh A cirrhotic patients and 31% in Child–Pugh B patients.11
The TACE is indicated for patients with multifocal, asymptomatic liver tumor, with a Child A–B liver function, without extrahepatic spread.7 After TACE, a significant tumor response is achieved in 17–61.9%, but a complete tumor response is rare (0%–4.8%); however, significant improvement in long-term survival had been demonstrated in meta-analysis studies.12,13
Resective surgery with curative intent is applied for early HCC in patients with well preserved liver function. However, only less than 30% of patients can be submitted to surgery for the advanced stage of tumor or severe liver function impairment. According to literature, surgery can achieve the best results in patients with a single nodule, smaller than 5 cm, without vascular invasion in patients with compensated liver cirrhosis (class A according to the Child–Pugh classification) and without portal hypertension. In these patients, the 5-year survival can reach 70%, but the major issue of surgical treatment of HCC is sill the high recurrence rate (80–100% after 5 years).14
The role of resective surgery in patients with advanced HCC (large, multifocal, or with macroscopic vascular invasion) is still under debate, and few studies in literature analyze the long-term results of surgery in these patients.4,15–17
In the 17th Nationwide Follow-up Survey of Primary Liver Cancer in Japan, 27,062 patients submitted to hepatic resection for HCC in the period from 1992 to 2003 were followed up. The 5-year survival rate was 59.2% for patients with a single HCC, 46.4% for patients with two nodules, and 30.0% for patients with three nodules.8
In a multi-institutional study, 308 patients with large (more than 3 cm) or multiple HCC (more than three nodules) were compared to 404 patients with small HCC. This study reported a 5-year survival of 26% for advanced HCC compared to 39% for early HCC.5 A recent study of by Ishizawa et al. in 434 patients who underwent to LR for single or multiple HCC (more than two nodules) reported a 5-year survival of 58% for Child A patients with multiple nodules, compared to 68% for Child A patients with a single nodule (p = 0.035).18 In Child B patients, 5-year survival decreased to 19% for patients with multiple nodules compared to 45% for patients with a single nodule (p = 0.13).18 Patients with HCC with multiple tumors in both hepatic lobes (TNM stage IVa) are generally considered unsuitable for surgery. However, recent data of the literature showed a significant improvement in survival after resective surgery compared to nonsurgical treatments, with median survivals of 19.5 and 7.1 months, respectively (p = 0.08).19
In our study, we analyzed a large group (464 patients) of cirrhotic patients with HCC, who underwent different therapeutic approaches in relation to the tumor stage and degree of liver impairment. The major limitation of our study is its retrospective design that implies biases of selection of patients, although the large number of patients included in the study and the subgroup analyses of patients with different tumor and liver function stages should lower the impact of these limitations. Our data confirm the good results of LR in patients with single HCC and class Child–Pugh A cirrhosis with 5-year survival of 54%. Even in patients with two or three nodules, the LR shows good performance compared to LAT or ST with actuarial 5-year survival of 46%, 10%, and 0%, respectively. In patients with more than three nodules, the LR decreases their efficacy and it is probably not justified. According to our study, surgical resection in patients with well preserved liver function and single or oligonodular HCC (two to three nodules) seem to have superior results compared to other therapies, and the presence of multiple HCC (up to three nodules) should not be considered an absolute contraindication to surgical resection.
The presence of macroscopic vascular invasion is an important prognostic factor in patients affected by HCC. The median of survival in patients with macroscopic vascular involvement without treatment is very poor: about 10 weeks.20,21 The choice of the best treatment for these patients is still under debate. Nonsurgical treatments such as TACE or RFA give a 1-year overall survival of 14%.22 Transplantation is contraindicated because of a high frequency of recurrences and short survival. Also, surgical resection in patients with macroscopic vascular invasion is contraindicated in the majority of patients, and few data in literature report long-term results in these patients. Poon et al., in a retrospective analysis, showed a 5-year prognosis of 13% in patients with macroscopic vascular invasion.23 Chen et al. reported a median survival of patients with portal vein tumor thrombosis located in the hepatic resection area or in the first division branch of the portal vein of 18.8 months.4 The author showed a reduction in survival, 10.1 months, in patients who underwent to thrombectomy of main portal vein tumor thrombus (p = 0.0275). Minigawa et al. proposed a combined therapeutic approach with preoperative TACE, obtaining a 5-year survival of 42% in 18 patients with no more than two nodules HCC and macroscopic portal invasion with a good liver function.24 Also, Fan et al. reported that surgical resection associated with thrombectomy followed by adjuvant TACE has better results than TACE alone, with median survivals of 12 and 5 months, respectively, with an actuarial 5-year survival of 16.6% in patients undergoing surgical resection followed by TACE vs 0% in patients undergoing only TACE.25
In our study, we confirmed the prognostic significance of macroscopic vascular involvement. Less than 25% of patients with macroscopic vascular involvement were submitted to LR. In this group, survival was not significantly different from patients submitted to LAT with actuarial 3-year survival of 40% and 47%, respectively (p = 0.46). However, survival after LR and LAT was significantly longer than in patients submitted to ST who showed actuarial 3-year survival of 10%. Our results suggest that, even if the prognosis of patients with macroscopic vascular involvement is very poor, the presence of macroscopic vascular invasion should not considered an absolute contraindication to surgery because LR in selected cases can improve survival compared to ST.
Conclusions
The present study shows that the surgery can achieve good results in patients with single HCC and good liver function. Also, patients with multinodular HCCs (two to three nodules) could benefit from LR where survival is longer than after LAT or ST, whereas, in patients with more than three HCCs, LR have results similar to those of LAT. Macroscopic vascular invasion is a major prognostic factor, and LR can be applied in highly selected patients, where it can allow good long-term results compared to ST.
References
Parkin DM, Bray F, Ferlay J, Pisani P. Estimating the world cancer burden: Globocan 2000. Int J Cancer. 2001;94(2):153–156. doi:10.1002/ijc.1440.
Abdalla EK, Denys A, Hasegawa K, Leung TW, Makuuchi M, Murthy R, et al. Treatment of large and advanced hepatocellular carcinoma. Ann Surg Oncol. 2008;15(4):979–985. doi:10.1245/s10434-007-9727-7.
Wakabayashi H, Ushiyama T, Ishimura K, Izuishi K, Karasawa Y, Masaki T, et al. Significance of reduction surgery in multidisciplinary treatment of advanced hepatocellular carcinoma with multiple intrahepatic lesions. J Surg Oncol. 2003;82(2):98–103. doi:10.1002/jso.10203.
Chen XP, Qiu FZ, Wu ZD, Zhang ZW, Huang ZY, Chen YF, et al. Effects of location and extension of portal vein tumor thrombus on long-term outcomes of surgical treatment for hepatocellular carcinoma. Ann Surg Oncol. 2006;13(7):940–946. doi:10.1245/ASO.2006.08.007.
Ng KK, Vauthey JN, Pawlik TM, Lauwers GY, Regimbeau JM, Belghiti J, et al. Is hepatic resection for large or multinodular hepatocellular carcinoma justified? Results from a multi-institutional database. Ann Surg Oncol. 2005;12(5):364–373. doi:10.1245/ASO.2005.06.004.
Bruix J, Sherman M, Llovet JM, Beaugrand M, Lencioni R, Burroughs AK, et al. Clinical management of hepatocellular carcinoma. Conclusions of the Barcelona-2000 EASL conference. European Association for the Study of the Liver. J Hepatol. 2001;35(3):421–430. doi:10.1016/S0168-8278(01)00130-1.
Llovet JM, Burroughs A, Bruix J. Hepatocellular carcinoma. Lancet 2003;362(9399):1907–1917. doi:10.1016/S0140-6736(03)14964-1.
Ikai I, Arii S, Okazaki M, Okita K, Omata M, Kojiro M, et al. Report of the 17th Nationwide Follow-up Survey of Primary Liver Cancer in Japan. Hepatol Res. 2007;37(9):676–691. doi:10.1111/j.1872-034X.2007.00119.x.
Llovet JM, Schwartz M, Mazzaferro V. Resection and liver transplantation for hepatocellular carcinoma. Semin Liver Dis. 2005;25(2):181–200. doi:10.1055/s-2005-871198.
Benckert C, Jonas S, Thelen A, Spinelli A, Schumacher G, Heise M, et al. Liver transplantation for hepatocellular carcinoma in cirrhosis: prognostic parameters. Transplant Proc. 2005;37(4):1693–1694. doi:10.1016/j.transproceed.2005.03.143.
Guglielmi A, Ruzzenente A, Battocchia A, Tonon A, Fracastoro G, Cordiano C. Radiofrequency ablation of hepatocellular carcinoma in cirrhotic patients. Hepatogastroenterology 2003;50(50):480–484.
Lau WY, Lai EC. Hepatocellular carcinoma: current management and recent advances. Hepatobiliary Pancreat Dis Int. 2008;7(3):237–257.
Llovet JM, Bruix J. Systematic review of randomized trials for unresectable hepatocellular carcinoma: Chemoembolization improves survival. Hepatology 2003;37(2):429–442. doi:10.1053/jhep.2003.50047.
Lau W. Future perspectives for hepatocellular carcinoma. HPB Oxf. 2003;5(4):206–213.
Fan J, Zhou J, Wu ZQ, Qiu SJ, Wang XY, Shi YH, et al. Efficacy of different treatment strategies for hepatocellular carcinoma with portal vein tumor thrombosis. World J Gastroenterol. 2005;11(8):1215–1219.
Fan J, Wu ZQ, Zhou J, Qiu SJ, Shi YH, Chen RX, et al. Hepatocellular carcinoma associated with tumor thrombosis in the portal vein: the effects of different treatments. Hepatobiliary Pancreat Dis Int. 2003;2(4):513–519.
Pawlik TM, Delman KA, Vauthey JN, Nagorney DM, Ng IO, Ikai I, et al. Tumor size predicts vascular invasion and histologic grade: Implications for selection of surgical treatment for hepatocellular carcinoma. Liver Transpl. 2005;11(9):1086–1092. doi:10.1002/lt.20472.
Ishizawa T, Hasegawa K, Aoki T, Takahashi M, Inoue Y, Sano K, et al. Neither multiple tumors nor portal hypertension are surgical contraindications for hepatocellular carcinoma. Gastroenterology 2008;134(7):1908–1916. doi:10.1053/j.gastro.2008.02.091.
Liu CL, Fan ST, Lo CM, Ng IO, Poon RT, Wong J. Hepatic resection for bilobar hepatocellular carcinoma: is it justified? Arch Surg. 2003;138(1):100–104.
Pawarode A, Voravud N, Sriuranpong V, Kullavanijaya P, Patt YZ. Natural history of untreated primary hepatocellular carcinoma: a retrospective study of 157 patients. Am J Clin Oncol. 1998;21(4):386–391. doi:10.1097/00000421-199808000-00014.
Llovet JM, Bustamante J, Castells A, Vilana R, Ayuso Mdel C, Sala M, et al. Natural history of untreated nonsurgical hepatocellular carcinoma: rationale for the design and evaluation of therapeutic trials. Hepatology 1999;29(1):62–67. doi:10.1002/hep.510290145.
Liver Cancer Study Group of Japan. Primary liver cancer in Japan. Clinicopathologic features and results of surgical treatment. Ann Surg. 1990;211(3):277–287.
Poon RT, Fan ST. Evaluation of the new AJCC/UICC staging system for hepatocellular carcinoma after hepatic resection in Chinese patients. Surg Oncol Clin N Am. 2003;12(1):35–50, viii. doi:10.1016/S1055-3207(02)00086-8.
Minagawa M, Makuuchi M, Takayama T, Ohtomo K. Selection criteria for hepatectomy in patients with hepatocellular carcinoma and portal vein tumor thrombus. Ann Surg. 2001;233(3):379–384. doi:10.1097/00000658-200103000-00012.
Fan J, Wu ZQ, Tang ZY, Zhou J, Qiu SJ, Ma ZC, et al. Multimodality treatment in hepatocellular carcinoma patients with tumor thrombi in portal vein. World J Gastroenterol. 2001;7(1):28–32.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was presented as Quick Shot oral presentation of the Surgical Society Alimentary Tract at Digestive Disease Week, May 20, 2008, San Diego Convention Center, San Diego, CA, USA and as oral presentation at 23rd Annual SSAT Residents & Fellows Research Conference, May 17, 2008, Omni San Diego, San Diego, CA.
Rights and permissions
About this article
Cite this article
Ruzzenente, A., Capra, F., Pachera, S. et al. Is Liver Resection Justified in Advanced Hepatocellular Carcinoma? Results of an Observational Study in 464 Patients. J Gastrointest Surg 13, 1313–1320 (2009). https://doi.org/10.1007/s11605-009-0903-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-009-0903-x