Abstract
Background
Zenker’s diverticula (ZD) can be treated by transoral diverticulostomy or open surgery (upper esophageal sphincter myotomy and diverticulectomy or diverticulopexy). The aim of this study was to compare the effectiveness of a minimally invasive (group A) versus a traditional open surgical approach (group B) in the treatment of ZD.
Material and Methods
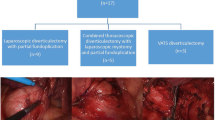
Between 1993 and September 2007, 128 ZD patients underwent transoral diverticulostomy (n = 51) or cricopharyngeal myotomy and diverticulectomy or diverticulopexy (n = 77). All patients were evaluated for symptoms using a detailed questionnaire. Manometry recorded upper esophageal sphincter (UES) pressure, relaxations, and intrabolus pharyngeal pressure. The size of the pouch was measured on the barium swallow. The choice of treatment was based on the size of the diverticulum and the patients’ preference. Long-term follow-up data were available for 121/128 (94.5%) patients with a median follow-up of 40 months (interquartile range, 17–83).
Results
Mortality was nil. Three patients in group A (5.8%) and ten in group B (13%) had postoperative complications (p = n.s.). Hospital stays were markedly shorter for patients after diverticulostomy (p < 0.01). Postoperative manometry showed a reduction in UES pressure, improved UES relaxation, and lower intrabolus pressure in both groups (p < 0.05). Four patients in the open surgery group (5.2%) complained of severe dysphagia after surgery (three of them required endoscopic dilations). In the transoral diverticulostomy group, 11 patients (21.5%) required additional septal reduction (n = 8) or a surgical myotomy (n = 3) for persistent symptoms (p < 0.01); nine of these 11 patients had a ZD ≤ 3 cm in size. After primary and complementary treatments, symptoms disappeared or improved significantly at long-term follow-up in 93.5% of patients in group A and 96% of those in group B.
Conclusion
Diverticulostomy is safe, quick, and effective for most patients with medium-sized ZD, but open surgery offers better long-term results as a primary treatment and should be recommended for younger, healthy patients, especially those with small diverticula. Small ZD may represent a formal contraindication to the transoral approach because an excessively short septum prevents a complete division of the sphincter fibers.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cricopharyngeal diverticula are protrusions of pharyngeal mucosa through an area of relative weakness in the posterior wall of the pharynx, limited laterally by the oblique fibers of the thyropharyngeal muscle and inferiorly by the inferior constrictor muscular sling, the so-called Killian’s triangle1. A higher hypopharyngeal pressure during swallowing and a lower resistance of the posterior wall of the hypopharynx are fundamental factors in the pathogenesis of cricopharyngeal diverticula—also known as Zenker’s diverticula (ZD), from the German pathologist who first described the condition2.
The lower part of the inferior constrictor muscle is a distinct anatomical entity known as the “cricopharyngeal muscle,” which, together with the muscle fibers encircling the upper esophagus, forms the upper esophageal sphincter (UES), which is tonically contracted at rest and relaxes on swallowing. The role of abnormal UES relaxation in causing excessive intrapharyngeal pressures during swallowing was clarified by manometric and cineradiographic studies nearly 60 years ago.3–7 Since then, UES myotomy, to ease the functional obstruction, has become an essential part of surgical treatment for pharyngeal diverticula, together with excision or pexis of the pouch.8
Recent developments in minimally invasive surgery have led to endoscopic-stapling devices being used to divide the septum between the esophagus and the pouch to relieve the outflow obstruction at the pharyngoesophageal junction. This alternative endoscopic approach was introduced by Collard in 1993: The anterior wall of the diverticulum and the posterior wall of the esophagus are divided and sealed using an endostapler inserted through a specially designed endoscope (the Weerda diverticuloscope), thus preventing leakage, mediastinitis, or bleeding.4 This procedure rapidly became widespread and is now often considered the treatment of choice for cricopharyngeal pouches.9–11
In 2003, in a relatively small series of patients, we showed that diverticulostomy was safe, quick, and effective for most patients with medium-sized ZD but that open surgery offered better long-term results and should be recommended for younger, healthy patients with small or very large diverticula.12 The present study thus aims to expand on our experience of treating ZD with both techniques, basing the choice of treatment on the diverticulum’s size.
Material and Methods
Patient Population
All patients with ZD referred to our surgical unit between 1993 and September 2007 were included in the study.
Patients were assessed for surgical risk and graded from 1 to 3 according to the risk scale of the American Society of Anesthesiologists (ASA).
Symptom Assessment
Patients’ symptoms were recorded using a standard questionnaire for upper foregut diseases. Dysphagia and regurgitation (the most common symptoms of pharyngeal diverticula) were scored according to severity and frequency. The symptom score was the sum of the severity scores for each symptom (0 = none, 2 = mild, 4 = moderate, 6 = severe) and their frequency (0 = never, 1 = occasionally, 2 = once a month, 3 = every week, 4 = twice a week, 5 = daily); the highest score obtainable was 22. Other symptoms (heartburn, sialorrhea, etc.) were assessed but not counted in the symptom score.
Respiratory symptoms (cough, episodes of pneumonia per year, and asthma) were also recorded.
Diagnostic Studies
Barium Swallow
The diagnosis of pharyngoesophageal diverticula was confirmed by a barium swallow, and the size of the diverticulum was measured in a lateral projection as the distance from the neck of the diverticulum to the bottom of the pouch.
Endoscopy
Upper gastrointestinal endoscopy was performed under mild sedation using a flexible scope to rule out any concomitant anomalies in the esophagus or stomach and, if the diverticulum interfered with the placement of the manometric tube, to pass a guidewire for the manometric probe.
Esophageal Manometry
This was performed using instruments and a technique described in detail elsewhere.13 Briefly, an eight-lumen low-compliance infused system with computerized data acquisition and analysis was employed. A high-frequency data acquisition mode (50-Hz) was used to record the rapid events occurring during swallowing. UES pressure was measured, while the catheter was withdrawn at a constant rate of 5 mm/s. The maximum amplitude recorded by each probe during its passage through the UES was averaged and considered as the UES pressure.
To evaluate pharyngoesophageal function during swallowing, the manometric probe with four radially oriented side holes was positioned at the upper edge of the UES, with two other side holes situated 5 and 10 cm above (in the distal and proximal pharynx, respectively) and one situated 5 cm below the UES (in the cervical esophagus). Ten swallows of 10 ml of water were evaluated, considering the following pharyngeal contraction parameters: amplitude, duration, and intrabolus pressure, i.e., the pressure generated by the passage of the bolus in the distal pharynx and seen at manometry as a slow pressure increase (shoulder) before the major upstroke generated by contractions of the pharyngeal wall, as described by Cook et al.5 We recorded the number of complete UES relaxations (expressed as the percentage of UES relaxations with a residual swallowing pressure <10 mmHg) and the coordination of UES opening with pharyngeal contractions (expressed as the percentage of relaxations with the nadir of the UES pressure coinciding with the pharyngeal wave’s major upstroke).
Treatment of Zenker’s Diverticula
From January 1993 onwards, two options were available for treating patients with ZD: (a) endoscopic diverticulostomy with a stapler or (b) open surgery for UES myotomy with or without diverticulectomy. Both procedures were performed under general anesthesia with endotracheal intubation. Informed consent was routinely obtained from all patients.
Endoscopic Diverticulostomy
With the patient supine and the neck extended, a Weerda diverticuloscope (Karl Storz, Tuttingen, Germany) was positioned with the anterior blade in the esophageal lumen and the posterior blade in the diverticulum. A telescope 5 mm in diameter was passed through the scope. A 30-mm disposable surgical endostapler (Ethicon Endo-surgery) was inserted through the Weerda scope to divide the septum between the diverticulum and the esophageal lumen. One or two stapler applications were used.
UES myotomy
This was performed through a left laterocervical approach, anteromedial to the sternocleidomastoid muscle. The diverticulum was isolated, and the cricopharyngeal muscle fibers were divided at the midline posteriorly from the neck of the sac down to the esophagus over a length of 4 cm. After completing the myotomy, diverticula >3 cm were transected using a stapler; diverticula of 1.5 cm or less were left in place; diverticula nearing 2 cm in size were inverted below the pharyngeal muscles and sutured to the muscle layer with two non-absorbable stitches.
Patient Stratification
The endoscopic procedure was suggested for patients with diverticula >3 and <5 cm in length; open surgery was recommended for patients with diverticula <3 or >5 cm. Postoperative course and any adverse events occurring after surgery were recorded.
Additional Procedures
A Nissen fundoplication was also performed in three patients with gastroesophageal reflux disease and hiatus hernia; an intrathoracic esophagectomy was performed in one patient for esophageal cancer.
Follow-up
Patients had a barium swallow a month after the operation and esophageal manometry after 6 months, when symptoms were reassessed using the same questionnaire, and patients were also asked if they were entirely or partially satisfied or dissatisfied with their treatment. Follow-up was yearly thereafter. Patients who failed to show up at the outpatient clinic were interviewed by phone. A procedure was considered a failure whenever patients complained of persistent dysphagia or (in cases treated with open surgery) recurrent diverticula.
Statistical Analysis
Data were collected in a database and analyzed using commercially available statistical software (Statview; SAS Institute, Inc., SAS Campus Drive, Cary, NC, USA). Data are expressed as medians and interquartile ranges (IQR). The Mann–Whitney and Wilcoxon tests were used as appropriate. Fisher’s exact test and the chi-square test were used to compare categorical data, as appropriate. A difference <0.05 was considered statistically significant.
The present study was approved by the local Bioethic Service for human study of the University of Padua.
Results
Clinical Data and Morbidity
During the study period, 128 patients with ZD were referred to our surgical unit: They included 90 men and 38 women with a median age of 66 years (IQR, 59.5–74). Fifty-one patients (38 men and 13 women), a median of 68 years old (IQR 60–75), were treated with endoscopic diverticulostomy (group A), and 77 patients (52 men and 25 women), a median 66 years old (IQR, 59–72), were treated with open UES myotomy (group B), p = n.s. In the second group, myotomy was performed alone in eight patients (10.4%) and combined with a diverticulectomy in 41 patients (53.2%) and with a diverticulopexy in 28 (36.4%). Eight patients explicitly asked for an endoscopic treatment: Four had a diverticulum shorter than 3 cm, one had a diverticulum 3 cm in length, and three had a diverticulum larger than 5 cm.
The main demographic and clinical data on the patient population are given in Table 1. The median symptom score was 13 in group A (IQR, 10–16) and 14 in group B (10–15.5), and symptom duration was 18 months in group A (IQR, 12–36) and 18 months in group B (IQR, 12–24), p = n.s. The presenting symptoms are listed in Table 2. As expected, dysphagia was the most common symptom, followed by regurgitation. Other symptoms included throat lump and throat pain. Recurrent lung infections were reported by almost one in five patients.
In group A, the pouch was a median 3.5 cm in size (IQR, 3–5): Four patients had diverticula shorter than 3 cm (and requested a transoral approach); 21 had diverticula approximately 3 cm long; nine had diverticula between 4 and 5 cm long; and three had diverticula larger than 5 cm (two were 6 cm long, one was 7 cm).
In group B, eight patients underwent UES myotomy alone [they all had diverticula ≤1.5 cm (median 1 cm, IQR, 1–1.5)]. Twenty-eight patients had diverticula approximately 2 cm in size [median 2 cm (IQR, 1.5–3)] and, after the myotomy, their diverticulum was inverted below the pharyngeal muscle layer. Forty-one patients had myotomy plus diverticulectomy for diverticula that were a median 3 cm in size (IQR, 2.5–5).
In the diverticulostomy group, nine patients had an ASA risk grade of 3, and 26 were grade 2; in the open surgery group, six patients were grade 3 and 40 were grade 2. The distribution of the risk did not differ statistically between the two groups (p = n.s.).
The duration of the operation was shorter in the endoscopic group (31 min; IQR, 18–36) than in the open surgery group (80 min; IQR, 61–122; p < 0.05). There were no deaths in either group. The overall morbidity rate was 10% (13/128 patients), with three patients in group A (5.8%) and ten in group B (13%), p = n.s. The postoperative complications are summarized in Table 3. All the complications in group A patients were due to difficulties encountered in inserting the diverticuloscope or stapler; they included two conversions to open surgery (3.9%) because it proved difficult to expose the septum in one case and a mucosal tear occurred while inserting the endostapler in the other. In group B, there were three leaks, four cases of bleeding with cervical hematoma (requiring surgical drainage in one case), one pericarditis (probably of viral etiology), one transient left recurrent palsy, and one injury to the recurrent laryngeal nerve. Two of the three leaks were detected by the Gastrografin® swallow obtained postoperatively: They were treated conservatively and healed within 2 weeks; one was detected during the operation and was sutured. The hospital stay was shorter in group A (5 days; IQR, 4–5 vs 9 days; IQR, 7–10; p < 0.05).
Follow-up and Early and Late Results
An adequate follow-up was obtained in 121/128 patients (94.5%): Of the seven patients lost to follow-up, three (6%) were in group A and four (5.2%) were in group B. Five patients died of unrelated causes. The median follow-up was 40 months (IQR, 17–83) and was similar in the two groups [36.5 months (IQR, 15.5–80.5) vs 41 months (IQR, 18.5–88); p = n.s.].
The median symptom score decreased from 13 (IQR, 10–16) to 0 (IQR, 0–2) in group A and from 14 (IQR, 10–15.5) to 0 (IQR, 0–0) in group B, p < 0.05.
A barium swallow was obtained in 40 of 51 patients (78.5%) after diverticulostomy and in 57 of 77 patients (74%) after open surgery. In all cases, a posterior pouch was still evident after diverticulostomy, though most of these patients were symptom-free. A small indentation was apparent in one patient treated with myotomy alone, in another treated with myotomy plus diverticulectomy (both these patients were symptom-free), and in three who had also undergone diverticulopexy (and one of these had recurrent dysphagia).
Preoperative esophageal manometry was performed in 36 of 51 (70.6%) group A patients and 64 of 77 (83.1%) group B patients; it was repeated, a median of 8 months, after the operation in 19 of 36 (53%) of the former and 32 of 64 (50%) of the latter, showing significantly lower UES resting and intrabolus pressures in both groups (Figs. 1 and 2). The percentage of complete UES relaxations increased from 30% (IQR, 0–80) to 100% (IQR, 50–100; p < 0.005) and from 20% (IQR, 0–80) to 80% (IQR, 57–100; p < 0.005) in groups A and B, respectively. No differences were observed in pharyngeal/UES coordination before and after the treatment (pharyngeal/UES coordination was normal in most patients).
UES resting pressure decreased significantly in both groups. Group A: preoperative 67 mmHg (IQR 45–85) vs postoperative 29 mmHg (IQR 24–40); p < 0.05. Group B: preoperative 69 mmHg (IQR 47.5–93) vs postoperative 50 mmHg (IQR 38.5–73); p < 0.05. Boxes represent the interquartile range with the horizontal line representing the median value. Error bars represent maximum and minimum values.
A significant drop in intrabolus pressure was recorded in both groups. Group A: preoperative 15 mmHg (IQR 10–18) vs postoperative 0 mmHg (IQR 0–10); p < 0.05. Group B: preoperative 17 mmHg (IQR 6.25–23.5) vs postoperative 7 mmHg (IQR 0–12.25); p < 0.05. Boxes represent the interquartile range with the horizontal line representing the median value. Error bars represent maximum and minimum values.
Analysis of Failures
On a single-patient basis, 11 patients (21.5%) in group A complained of persistent postoperative dysphagia, which required additional endoscopic procedures (to reduce the septum between the pouch and the esophagus) in eight cases. Three patients eventually required a surgical UES myotomy. On the other hand, only four patients (5.2%) in group B had recurrent dysphagia (p < 0.05); they were treated with UES pneumatic dilations in three of four cases, while one refused any further treatment.
The demographics and clinical characteristics of patients in groups A and B with and without recurrent symptoms are presented in Table 4. Among those who had transoral diverticulostomy, three parameters were statistically influential in the patients who experienced a recurrence, i.e., age, size of diverticulum, and duration of symptoms. Nine of 11 symptomatic patients of group A had a diverticulum no more than 3 cm in length, giving a 36% chance of recurrence in the subgroup of patients with small diverticula treated transorally. (Table 5). These patients were also significantly younger and had lower overall symptom scores. In group B (open surgery), no parameters appeared to discriminate between patients whose treatment failed and those with a good outcome.
When preoperative manometric findings in patients with and without recurrence were compared, neither resting nor intrabolus UES pressures could discriminate between the two groups. Postoperative physiological findings in patients with and without recurrence are compared in Table 6: In both treatment groups, patients with recurrences had higher postoperative UES intrabolus pressures, that is to say, a smaller reduction in their UES intrabolus pressure, than patients whose surgery had been successful.
Final Results after Additional Treatments
Overall, after the primary treatment and any additional treatments for recurrent dysphagia, symptoms disappeared or improved significantly in 45 of 48 patients in group A (93.5%) and 70 of 73 in group B (96%).
Discussion
Though it was first described by Ludlow14, the cricopharyngeal pouch is better known by the name of the German pathologist, Frederick Albert von Zenker, who published a review of 27 patients with this disease together with von Ziemssen. It is now accepted that Zenker’s diverticulum is due to an outflow obstruction caused by a noncompliant fibrotic cricopharyngeal sphincter. Histological and functional studies on the muscle have revealed fibrosis, atrophy, hypertrophy, and inflammation.15–17 Inadequate UES opening considerably increases hypopharyngeal intrabolus pressure and leads to the formation of a pulsion (Zenker’s) diverticulum.5,17,18
For several decades, surgical therapy for cricopharyngeal diverticula focused on treating the sac by excision or pexis to the prevertebral fascia. But simple resection (or the pouch suspension) without a concomitant myotomy often caused severe complications, such as leakage from the suture line and failing to relieve dysphagia and being associated with a high rate of diverticulum recurrence. Cricopharyngeal myotomy directly addressed the pathogenesis of pulsion diverticula and soon came to be included as a fundamental part of the procedure. Thirty years later, an old endoscopic technique used to transect the septum between the diverticulum and the esophagus (initially with a cautery, but the rate of dehiscence was unacceptable)6 remerged but using a laparoscopic stapler to simultaneously divide and suture the septum. Using this technique, the UES was divided too, thus obtaining physiological results similar to those of the open surgery, the only difference being that the pouch was not removed, a common cavity was created. This technique had its appeal because no incision was necessary, the procedure was quick to complete, risks related to transcervical approach were reduced, and patients recovered quickly, but it was not clear whether the results were fully comparable with those of open surgery and whether the method could be applied to all patients with ZD.
The results of this study confirm that both transoral and surgical procedures are safe and effective in treating Zenker’s diverticulum. When stapling diverticulostomy is performed and the cricopharyngeal muscle fibers are divided, pharyngoesophageal manometry demonstrates a substantial reduction in subsequent UES resting pressures with both techniques, as reported by Ishioka et al.19, too. The functional efficacy of the transoral approach in improving pharyngoesophageal function was also confirmed by the elimination of intrabolus pressure and the disappearance of any obstacle to bolus outflow (the advantage of endostapling over traditional surgery can be explained by the presence of a large common cavity, where small pressures are not easy to identify by perfusion manometry).
Our study confirmed the advantages of endostapling over conventional surgery, given the shorter operating time, little or no postoperative pain, quicker return to oral feeding, less severe complications, and shorter hospital stay.
The drawbacks of endoscopic diverticulostomy relate to certain patient features, e.g., the inability to open the mouth wide or to (over)extend the neck in cases of severe kyphosis and, more importantly, the size of the diverticulum. This study strongly suggests that if the diverticulum is ≤3 cm in size, recurrence may occur in 36% of patients. When a diverticulum is small, the anvil of the stapler is too long to be accommodated properly inside the pouch, so probably not all the UES fibers can be transected. Manometric studies in nine of 11 patients with severe dysphagia after their operation revealed incomplete UES relaxation and persistently high pharyngeal intrabolus pressure in four cases (44.5%): Three of these patients were reoperated and revealed uncut muscle fibers just below the end of the stapler line (and after myotomy they became asymptomatic). The strongest indication for endoscopic diverticulostomy is therefore a medium-sized diverticulum in which the stapler cartridge can be accommodated and the stapler can achieve an adequate cricopharyngeal myotomy, whereas diverticula shorter than 3 cm should be seen as a formal contraindication to the transoral approach.
The major drawbacks of open surgery are the related morbidity, mainly bleeding (a small drain is routinely left in place) and leakage from the suture line. Although our three leaks required no further surgery and healed spontaneously (after we had left the nasogastric tube in place, avoided oral feeding, and administered antibiotic therapy), it is nonetheless a severe potential complication in patients with concurrent respiratory or heart disease. Open surgery assures a complete and effective myotomy of the UES, especially in the subset of patients with small diverticula, for whom it should be considered the treatment of choice. After UES myotomy, small sacs (≤1.5cm) could be left in place, and they tend to disappear once the obstacle to outflow has been removed. Slightly larger diverticula (≤2 cm) can be introflected below the pharyngeal muscle layer to further reduce the risk of suture leakage. In our opinion, surgical myotomy is the therapy of choice for diverticula smaller than 3 cm.
In conclusion, this study on a larger number of patients confirms our previous observations: ZD can be treated effectively by endoscopic diverticulostomy or open surgery. Moreover, our analysis of treatment failures demonstrates that these are caused by a persistently noncompliant UES opposing the bolus outflow, as revealed indirectly by an unchanged pharyngeal intrabolus pressure. Based on these results, endoscopic diverticulostomy is better suited to medium-sized diverticula (3–5 cm). When applied to small diverticula, it carries a greater risk of failure in terms of persistent severe dysphagia because of an incomplete dissection of the UES. Open surgical myotomy, with or without diverticulectomy, is effective for diverticula of all sizes and should be considered the treatment of choice for small diverticula. It is important to bear in mind, however, that some of its complications, e.g. leakage or laryngeal nerve palsy, may have disastrous effects in elderly patients with comorbidities.
References
Killian G. The mouth of the esophagus. Laryngoscope 1907;17:421–428.
Zenker FA, von Ziemssen H. Krankheiten des Oesophagus. In: von Ziemsen H (ed) Handbuch der Speciellen Pathologie und Therapie 1874;7(Suppl). Vogel, Leipzig, pp 1–87
Belsey R. Functional disease of the esophagus. J Thorac Cardiovasc Surg. 1966;52:164–188.
Collard JM, Otte IB, Kestens PJ. Endoscopic stapling technique of esophagodiverticulostomy for Zenker’s diverticulum. Ann Thorac Surg 1993;56:573–576.
Cook IJ, Gabb M, Panagopoulos V. Zenker’s diverticulum is a disorder of upper esophageal sphincter opening. Gastroenterology 1992;103:1229–1235.
Dohlman G, Mattson O. The endoscopic operation for hypopharyngeal diverticula. Arch Otolaryngol 1960;71:744–752.
Ellis FH Jr, Schlegal JF, Lynch VP, Payne WS. Cricopharyngeal myotomy for pharyngo-oesophageal diverticula. Ann Surg. 1969;170:340–349. doi:10.1097/00000658-196909010-00004.
Payne WS. The treatment of pharyngoesophageal diverticulum: the simple and the complex. Hepatogastroenterology 1992;39:109–114.
Omote K, Feussner H, Stein HJ, Ungeheuer A, Siewert JR. Endoscopic stapling diverticulostomy for Zenker’s diverticulum. Surg Endosc 1999;13:535–538. doi:10.1007/s004649901031.
Peracchia A, Bonavina L, Narne S, Segalin A, Antoniazzi L, Marotta G. Minimally invasive surgery for Zenker diverticulum: analysis of results in 95 consecutive patients. Arch Surg 1998;133:695–700. doi:10.1001/archsurg.133.7.695.
Bonavina L, Bona D, Abraham M, Saino G, Abate E. Long-term results of endosurgical and open surgical approach for Zenker diverticulum. World J Gastroenterol 2007;13:2586–2589.
Zaninotto G, Narne S, Costantini M, Molena D, Cutrone C, Portale G, et al. Tailored approach to Zenker’s diverticula. Surg Endosc 2003;17:129–133. doi:10.1007/s00464-002-8806-x.
Passaretti S, Zaninotto G, DiMartino N, Leo P, Costantini M, Baldi F. Standards for oesophageal manometry: a position statement from the Gruppo Italiano di Studio per la Motilita’ Apparato Digerente. Dig Liver Dis 2000;32:46–55. doi:10.1016/S1590-8658(00)80044-4.
Ludlow A. A case of obstructed deglutition from a preternatural dilatation of and bag formed in the pharynx. Med Observ Inq Soc Phys 1769;3:85–101.
Zaninotto G, Costantini M, Boccù C, Anselmino M, Parenti A, Guidolin D, et al. Functional and morphological study of the cricopharyngeal muscle in patients with Zenker’s diverticulum. Br J Surg 1996;83:1263–1267. doi:10.1002/bjs.1800830928.
Lerut T, van Raemdonck D, Guelinckx P, Dom R, Geboes K. Zenker’s diverticulum: is a myotomy of the cricopharyngeus useful? How long should it be? Hepatogastroenterology 1992;39:127–131.
Venturi M, Bonavina L, Colombo L, Antoniazzi L, Bruno A, Mussini E, et al. Biochemical markers of upper esophageal sphincter compliance in patients with Zenker’s diverticulum. J Surg Res 1997;70:46–48. doi:10.1006/jsre.1997.5049.
Shaw DW, Cook IJ, Jamieson GG, Gabb M, Simula ME, Dent J. Influence of surgery on deglutitive upper oesophageal sphincter mechanics in Zenker’s diverticulum. Gut 1996;38:806–811. doi:10.1136/gut.38.6.806.
Ishioka S, Felix VN, Sakai P, Melo J, Pinotti HW. Manometric study of the upper esophageal sphincter before and after endoscopic management of Zenker’s diverticulum. Hepatogastroenterology 1995;42:628–632.
Author information
Authors and Affiliations
Corresponding author
Additional information
Tom R. DeMeester, M.D. (Los Angeles, CA, USA): Dr. Rizzetto, I congratulate you on a well constructed manuscript; I enjoyed reading it. Similarly, I enjoyed listening to your excellent presentation on this rather common problem of Zenker’s diverticulum. The pathophysiology of the problem is largely understood at the present time. I appreciate the focus of your study on the therapeutic approach to the problem. Your study population was large, the follow-up long, and the extent of function studies obtained unique. I commend you on getting preoperative motility studies on these patients. It can be very difficult to get patient consent and to perform. You have nicely shown us and, I think for the first time, documented that a 3-cm diverticulum is not well managed with the endoscopic staple technique and leads to a high rate of recurrence.
I have four questions. First, you had two patients in whom you could not get the staple into the hypopharynx and into the diverticulum and had to convert to an open procedure. In the manuscript, you implied that some patients were difficult to do. Can you describe how hard you tried before you convert, and does the cervical spine or the ability of the patient to extend his head effect your decision to do the transoral procedure?
Second, is a motility study necessary in the common care of these patients at the present time?
Third, Dr. Jean-Marie Collard, who introduced the stapling technique, showed that there were some minor symptoms that persisted in the stapled group other than dysphagia and were due to the large common cavity created when you cut the septum between the diverticulum and the esophagus. Was your symptom evaluation careful enough to pick up those subtleties, or how do you explain the difference between his observation and yours?
Lastly, although this transoral approach suggests that it would protect the recurrent laryngeal nerves, we have seen some nerve palsy following the procedure. They were likely due to stretching of the nerve in trying to get the stapler in the hypopharynx. Would you comment on this and have you seen short-term palsies with this approach?
Christian, it was a superb presentation and, as a previous research fellow in our unit, you have given us reason to be very proud of you.
Christian Rizzetto, M.D. (Padova, Italy): Thank you very much for your kind words and comments. There are several issues to consider in deciding whether or not to perform the transoral procedure. The main appeal of this technique is that no incision is needed and the procedure is quick to complete. Zenker’s diverticula frequently affect elderly patients, however, and the transoral procedure has its drawbacks in certain patients, e.g., if they are unable to open their mouth wide or to overextend their neck (in cases of severe kyphosis). In the two cases you mentioned, it was not easy to position the diverticular scope, and we had a mucosal perforation in one case. Basically, if we consider a patient a suitable candidate for the transoral approach, we normally try this procedure, but if we have trouble inserting the diverticular scope, then we usually opt to convert the procedure.
The second question addresses the manometry issue. I would say that this has been extremely important to our understanding of the pathophysiology of Zenker’s diverticulum and is still important in the preoperative diagnostic work-up, especially in the case of small diverticula. Manometry can also play a part in patients experiencing recurrent dysphagia, to help us understand how it can be managed and the intra-bolus pressure adequately reduced.
Your third question refers to Collard’s study. Our symptom questionnaire focused mainly on dysphagia and regurgitation, but other symptoms were assessed even though they did not count in the symptom score. We observed no differences, however, and the two patient groups were equally satisfied, in our experience at least. I think you have raised an intriguing point that warrants a prospective assessment.
Your last question was about laryngeal nerve palsy. We experienced transient palsy in one case and permanent recurrent laryngeal nerve injury in another: Both patients were in the open surgery group. It is hard to say how this might happen using the transoral approach, but—as you said—they were probably due to stretching of the nerve to accommodate the anvil of the stapler in the hypopharynx. I think exposure is the key issue in this type of surgery: The only way to avoid complications is to ensure adequate vision of the hypopharynx, the diverticulum, and the septum between the sac and the esophagus. In our opinion, the stapler should not be inserted and fired if the exposure conditions are less than optimal.
Rights and permissions
About this article
Cite this article
Rizzetto, C., Zaninotto, G., Costantini, M. et al. Zenker’s Diverticula: Feasibility of a Tailored Approach Based on Diverticulum Size. J Gastrointest Surg 12, 2057–2065 (2008). https://doi.org/10.1007/s11605-008-0684-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-008-0684-7