Abstract
Introduction
Intraabdominal schwannomas are rare, benign tumors. This study presents a single institution experience with 12 such tumors.
Methods
Between 1991 to 2006, 12 patients with a pathologically proven intraabdominal schwannoma were identified from a series of 216 mesenchymal tumors and were reviewed retrospectively.
Results
There were nine females and three male patients with a median age of 58 years (range 35–88 years). Eleven patients were symptomatic, and the tumors were located in the stomach (n = 8), jejunum, colon, rectum, and lesser sac. Multiple preoperative investigations including endoscopies with biopsies and computed tomography (CT) scans were performed, but none yielded a correct definitive preoperative diagnosis. The median tumor size was 52 mm (range 18–95 mm). Pathological examination demonstrated the 11 gastrointestinal tract (GIT) schwannomas to be solid homogenous tumors, which were highly cellular and were composed of spindle cells with positive staining for S100 protein. The pathological appearance of the lesser sac schwannoma was distinct as it demonstrated cystic degeneration with hemorrhage and Antoni A and B areas on microscopy typical of soft tissue schwannomas. All 12 patients were disease-free at a median follow-up of 22 months (range 1–120 months).
Conclusion
Intraabdominal schwannomas are rare tumors, which are most frequently located within the GIT. GIT schwannomas are difficult if not impossible to diagnose preoperatively as endoscopic and radiologic findings are nonspecific. The treatment of choice is complete surgical excision because of diagnostic uncertainty, and the long-term outcome is excellent as these lesions are uniformly benign.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Nerve sheath tumors are a subclass of soft-tissue neoplasms that include benign and malignant schwannomas and neurofibromas.1 Schwannomas are common tumors, which are most frequently detected in cranial and peripheral nerves. The occurrence of intraabdominal2 and retroperitoneal schwannomas3 are, however, extremely rare. Intraabdominal schwannomas occur most frequently in the alimentary tract, and the most common site is the stomach.4–6 Other intraabdominal sites are even rarer and these have been reported in the greater omentum,7 lesser sac,8 and the biliary tree.9 Gastrointestinal tract (GIT) schwannomas have been shown to demonstrate distinct histological features from conventional soft tissue schwannomas.10,11 These tumors belong to the family of GI mesenchymal tumors of which the most common are gastrointestinal stromal tumors (GIST) followed by smooth muscle tumors. Schwannomas have been reported to represent only 3% of all GI mesenchymal tumors.5
GIT schwannomas were first reported by Daimaru et al. in 1988,12 and since then, only a few series’ have been reported in the pathological literature, which were often multiinstitution reviews.5,6,11–13 Reports of this condition outside the pathology literature have been limited to case reports2,4 and a single multiinstitution experience (from the Armed Forces Institute of Pathology files) of eight patients.10 This report details the experience with intraabdominal schwannomas at a large tertiary referral center. To the best of our knowledge, this is the largest single institution review of these unusual tumors.
Methods
The records of 216 patients who underwent surgical resection of an intraabdominal mesenchymal tumor (not including retroperitoneal tumors) between 1991 to 2006 at the Department of General Surgery and Colorectal Surgery, Singapore General Hospital were retrospectively reviewed. All pathology slides and paraffin blocks of the patients were retrieved and reexamined by one of the above two pathologists (SMK, WMY). In addition, immunohistochemical staining was performed for cases that were not immunostained previously. Of these 216 patients, 12 (5.6%) had a pathologically proven schwannoma, and their case notes and radiological reports were reviewed retrospectively. One of these patients (patient 9) has been reported previously.14
Results
The patients’ clinicopathological, surgical data and outcome are summarized in Tables 1 and 2. There were nine females and three males with a median age of 58 years (range 35–88 years). Eleven patients were symptomatic and the most common symptoms were epigastric discomfort (n = 4) and upper GI bleed (n = 2). None of the patients had von Recklinghausen’s disease. The tumors were located in the stomach (n = 8), jejunum, colon, rectum, and lesser sac.
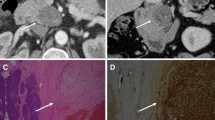
Most of the patients underwent multiple preoperative investigations, but none had a correct definitive preoperative diagnosis. The tumors were most frequently thought to be GISTs (n = 9). All eight of the patients with gastric tumors underwent upper GI endoscopy, which demonstrated a submucosal lesion, three of which had central ulceration. Six patients had an endoscopic biopsy of the lesion, which was nondiagnostic in five (too superficial) and suggestive of a stromal tumor in one (small number of spindle cells). Patient 5 had an endoscopic ultrasound with fine-needle aspirate, which was also nondiagnostic (inadequate cells). Computed tomography (CT) scan was performed in seven patients and ultrasonography (US) in two patients. The six gastric tumors appeared on CT as a solid homogenous exophytic or intraluminal lesion arising from the stomach. The lesser sac schwannoma was thought to be a septated pancreatic cyst on both CT and US.14
Eleven patients underwent laparotomy and resection of tumor. One patient underwent transanal resection of rectal schwannoma. The median tumor size was 52 mm (range 18–95 mm). Pathological examination demonstrated the 11 GI schwannomas to be solid homogeneous tumors, which were highly cellular and were composed of spindle cells. These stained uniformly for the S100 protein. The pathological appearance of the lesser sac schwannoma was, however, distinct as it demonstrated cystic degeneration with hemorrhage with Antoni A and B areas on microscopy. None of the schwannomas demonstrated dysplastic or malignant cells. All 12 specimens demonstrated positive immunostaining for S100. The remaining immunohistochemical staining results were as follows: CD117 was negative in 11 of 11 cases, CD34 was negative in 11 of 11 cases, smooth muscle actin was positive in 1 of 11 cases, and desmin was negative in 10 of 10 patients. All 12 patients were disease-free at a median follow-up of 22 months (range 1–120 months).
Discussion
Intraabdominal schwannomas are rare tumors. In our experience, these comprised of 5.6% of mesenchymal tumors, which mirrored the incidence of 2.9% to 6% reported by others.5,11,13 These tumors are most frequently located in the GIT of which the vast majority (73%) are found in the stomach.5,11,13 Extremely rare cases of extragastrointestinal intraabdominal schwannomas have been reported in the lesser sac,8 biliary tree,9 liver,15 and greater omentum.7 Because of the rarity of extragastrointestinal intraabdominal schwannomas, the following discussion will focus mainly on the clinicopathological features of GIT schwannomas.
GIT schwannomas have been reported to occur in patients over a wide range of age groups with a median age of 50 to 60 years.5, 11 Most series’ report a female preponderance.6,11,12,16 These tumors range in size from 0.5 to 11 cm, which is markedly smaller than GISTs and which, not infrequently, grow to more than 20 cm in size.11,17
Pathologically, GIT schwannomas are regarded as distinct tumors from conventional schwannomas, which arise from the central nervous system and soft tissues.10,11 These tumors are assumed to arise from the nerve plexus of the gut wall.10–12 Macroscopically, these are round or fusiform and are often described as homogenous, firm, or rubbery.5,11 Degenerative changes such as necrosis, hemorrhage, and cystic change, which are frequently found in soft tissue schwannomas such as those in the retroperitoneum,3 are seldom present.5,11 Microscopically, unlike conventional schwannomas, GIT schwannomas are not encapsulated, although most are well circumscribed. These are frequently surrounded by a cuff of lymphoid aggregates,5,6,12,13,18 are highly cellular, and are composed mainly of bipolar spindle cells. Verocay bodies, vascular hyalinization, Antoni A and B areas, and a typical palisading structure are typically absent unlike conventional schwannomas. The pathologic findings of the GIT schwannomas in the present analysis were consistent with these previously described findings.
On the other hand, the lesser sac schwannoma in this study14 demonstrated the typical pathologic features of peripheral and soft tissue schwannomas3 including cystic degeneration with hemorrhage and typical Antoni and B areas. This observation suggests that the lesser sac schwannoma did not arise from extensive extramural growth of a gastric schwannoma resulting in loss of contact with the external muscle coat of the gut as has been suggested for some extragastrointestinal GISTs.19 Instead, it probably originated from one of the branches of the vagus nerve at the lesser curvature of the stomach.14 Based on cases reported in the literature, the pathologic appearance of extragastrointestinal intraabdominal schwannomas are variable with some cases in the omentum7 or lesser sac8 having the typical appearance of conventional schwannomas, whereas those in the liver15 and biliary tree9 were reported to have features similar to GIT schwannomas.
On immunohistochemistry, the cells of GIT schwannomas diffusely and strongly express vimentin and S100 proteins.11 The S100 immunostaining pattern is both in a nuclear and cytoplasmic distribution.5 GIT schwannomas may rarely express CD34 cells, but CD117, SMA, and desmin are uniformly negative.20 Hence, immunohistochemistry is extremely useful in distinguishing GIT schwannomas from the other GI mesenchymal tumors such as GISTs, which express CD117 (almost always) and CD34 (frequently) and true smooth muscle tumors, which express smooth muscle actin (SMA) and desmin.17
GIT schwannomas are usually detected preoperatively via cross-sectional imaging or endoscopy. However, preoperative diagnosis is difficult as none of these modalities have shown any pathognomonic features unique to this tumor. Presently, because of its rarity, there are limited data reporting the CT features of GIT schwannomas in the literature with only a single-case series of eight patients to date.10 On CT scan, these tumors have a homogeneous pattern of attenuation on both unenhanced and contrast-enhanced scans with tumor enhancement occurring in the equilibrium phase. The main differential diagnoses of GIT schwannomas are GISTs, which are the most common mesenchymal tumors of the GI tract.10 Although these tumors most frequently have a heterogeneous appearance on CT because of hemorrhage, necrosis or cystic change, 8–13% of GISTs may appear as homogeneous tumors, making them indistinguishable from GI schwannomas.21,22 Other neoplasms such as lymphomas and GI adenocarcinomas may also have overlapping features with GIT schwannomas.10 In this study, all six gastric schwannomas appeared as solid homogenous tumors on CT. Presently, experience with the US features of GIT schwannomas is extremely limited.23 Gastric schwannomas have been reported to appear as a solid homogeneous hypoechoic mass,23 which was similar to the US appearance of patient 6.
Similar to cross-sectional imaging, the endoscopic features of GI schwannomas are nonpathognomonic.23,24 The endoscopic findings are almost always nonspecific as these tumors appear grossly as submucosal lesions, which are indistinguishable from other mesenchymal tumors. Futhermore, endoscopic biopsies are usually not representative of the deeper submucosal tissue. Even when the endoscopist succeeds in obtaining samples from the deeper tissues, these usually demonstrate nonspecific spindle cells, and there is usually insufficient tissue for the pathologist to obtain a definite diagnosis. These problems were well-illustrated in the present analysis whereby none of eight patients who underwent gastroscopy had a definitive diagnosis.
Thus far, all series5,10–13,18 in the literature addressing GIT schwannomas regard these tumors as uniformly benign. However, isolated case reports of “malignant schwannomas” also termed malignant peripheral nerve sheath tumors have been reported.25 Whichever, these malignant tumors arise from benign schwannomas remains controversial.24 Presently, most pathologists regard these malignant tumors with neural differentiation as distinct tumors from GIT schwannomas, giving them the term gastrointestinal autonomic nerve tumors (GANTs).6 Nonetheless, although benign, the treatment of choice of GIT schwannomas is complete surgical excision in fit, healthy patients as it is frequently impossible to distinguish these tumors from other GIT mesenchymal tumors such as GIST and smooth muscle tumors, which are malignant or have malignant potential. The outcome after surgical resection is excellent and to date, there is no evidence in the literature to suggest that GIT schwannomas have malignant potential.5,6, 10–13,18
In conclusion, intraabdominal schwannomas are rare tumors, which are most frequently located within the GIT. Very rarely, these may arise from outside the GIT. GIT schwannomas are difficult, if not impossible, to diagnose preoperatively as endoscopic and radiologic findings are nonspecific. The treatment of choice is complete surgical excision because of diagnostic uncertainty, and the long-term outcome is excellent as these lesions are uniformly benign.
References
Gubbay AD, Moschilla G, Gray BN, Thompson I. Retroperitoneal schwannoma: A case series and review. Aust N Z J Surg 1995;65:197–200.
Khan AA, Schizas AM, Cresswell AB, Khan MK, Khawaja HT. Digestive tract schwannoma. Dig Surg 2006;23:265–269.
Goh BK, Tan YM, Chung YF, Chow PK, Ooi LL, Wong WK. Retroperitoneal schwannoma. Am J Surg 2006;192:14–18.
Melvin WS, Wilkinson MG. Gastric schwannoma. Clinical and pathologic considerations. Am Surg 1993;59:293–296.
Hou YY, Tan YS, Wang XN, Lu SH, Ji Y, Wang J, Zhu XZ. Schwannoma of the gastrointestinal tract: A clinicopathological, immunohistochemical and ultrastructural study of 33 cases. Histopathology 2006;48:536–545.
Prevot S, Bienvenu L, Vaillant JC, de Saint-Maur PP. Benign schwannoma of the digestive tract. A clinicopathologic and immunohistochemical study of five cases, including a case of esophageal tumor. Am J Surg Pathol 1999;23:431–436.
Bankier AA, Stanek C, Hubsch P. Case report: benign solitary schwannoma of the greater omentum: A rare cause of acute intraperitoneal bleeding—diagnosis by CT. Clin Radiol 1996;51:517–518.
Noonan JD, Minagi H, Margolin R. Benign solitary schwannoma of the lesser peritoneal sac. AJR Am J Roentgenol 1976;125:391.
Fenoglio L, Severini S, Cena P, Migliore E, Bracco C, Pomero F, Panzone S, Cavallero GB, Silvestri A, Brizio R, Borghi F. Common bile duct schwannoma: A case report an review of literature. World J Gastroenterol 2007;13:1275–1278.
Levy AD, Quiles AM, Miettinen M, Sobin LH. Gastrointestinal schwannomas: CT features with clinicopathologic correlation. AJR Am J Roentgenol 2005;184:797–802.
Kwon MS, Lee SS, Ahn GH. Schwannomas of the gastrointestinal tract: Clinicopathological features of 12 cases including a case of esophageal tumor compared with those of gastrointestinal stromal tumors and leiomyomas of the gastrointestinal tract. Pathol Res Pract 2002;198:605–613.
Daimaru Y, Kido H, Hashimoto H, Enjoji M. Benign schwannoma of the gastrointestinal tract: A clinicopathologic and immunohistochemical study. Hum Pathol 1988;19:257–264.
Sarlomo-Rikala M, Miettinen M. Gastric schwannoma—a clinicopathological analysis of six cases. Histopathology 1995;27:355–360.
Toh LM, Wong SK. A case of cystic lesser sac schwannoma. Ann Acad Med Singapore 2006;35:45–48.
Flemming P, Frerker M, Klempnauer J, Pichlmayr R. Benign schwannoma of the liver with cystic changes misinterpreted as hydatid disease. Hepatogastroenterology 1998;45:1764–1766.
Yagishashi N, Kaimori M, Katayama Y, Yagihashi S. Crystalloid formation in gastrointestinal schwannoma. Hum Pathol 1997;28:304–308.
Fletcher CD, Berman JJ, Corless C, et al. Diagnosis of gastrointestinal stromal tumors: A consensus approach. Hum Pathol 2002;33:459–465.
Miettinen M, Shekitka KM, Sobin LH. Schwannomas in the colon and rectum: A clinicopathologic and immunohistochemical study of 20 cases. Am J Surg Pathol 2001;25:846–855.
Agaimy A, Wunsch PH. Gastrointestinal stromal tumors: A regular origin in the muscularis propria, but an extremely diverse gross presentation. A review of 200 cases to critically re-evaluate the concept of so-called extra-gastrointestinal stromal tumours. Langenbecks Arch Surg 2006;391:322–329.
Meittinen M, Virolainen M, Rikala MS. Gastrointestinal stromal tumors. Value of CD34 antigen in their identification and separation from true leiomyomas and schwannomas. Am J Surg Pathol 1995;19:207–216.
Burkill GJ, Badran M, Al-Muderis O, et al. Malignant gastrointestinal stromal tumor: Distribution, imaging features, and pattern of metastatic spread. Radiology 2003;226:527–532.
Levy AD, Remotti HE, Thompson WM, Sobin LH, Miettinen M. Gastrointestinal stromal tumors: Radiologic features with pathologic correlation. Radiographics 2003;23:283–304, 456,quiz 532.
Fujii Y, Taniguchi N, Hosoya Y, Yoshizawa K, Yasuda Y, Nagai H, Itoh K. Gastric schwannoma: Sonographic findings. J Ultrasound Med 2004;23:1527–1530.
Rodriguez SA, Faigel DO. Endoscopic diagnosis of gastrointestinal stromal cell tumors. Curr Opin Gastroenterol 2007;23:539–543.
Bees NR, Ng CS, Dicks-Mireaux C, Kiely EM. Gastric malignant schwannoma in a child. Br J Radiol 1997;70:952–955.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Goh, B.K.P., Chow, P.K.H., Kesavan, S. et al. Intraabdominal Schwannomas: A Single Institution Experience. J Gastrointest Surg 12, 756–760 (2008). https://doi.org/10.1007/s11605-007-0441-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-007-0441-3