Abstract
Trace elements occur in low quantities in the environment but have a crucial importance. Some elements are essential for life whereas many elements exhibit toxicity when exposure to them is too high. In this contribution, trace elements in potato are addressed. Although potato is a crop growing in the soil, the tuber does not intensively accumulate trace elements. Concentrations of trace elements are in the range of other vegetables. Generally, potato exhibits a rather low nutritional value for trace elements, with the exception of Fe, Cr, and Cu, although specific cultivars may provide significant contributions to the intake of several elements. Trace element uptake depends strongly on the plant and also on the growing environment. Soil properties that influence uptake include pH, contents of clay and organic matter, and salinity. In soils with baseline metal concentrations, concentrations of potentially toxic elements are of no concern, but Cd needs to be monitored. Environmental care and good soil management is a must to safeguard the safety of food, including potato.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Trace elements occur in low quantities in the environment, typically below 1 g/kg (Kabata-Pendias 2010). Although not noticeable and sometimes difficult to measure, they are important for various reasons. Specific elements are indispensable for organisms to grow and complete their life cycle. An adequate supply of trace elements through food is an important factor in ensuring public health. Trace elements have numerous applications in various areas and thus are indispensable in our modern society. In contrast to these benefits, trace elements might become toxic when exposure to them exceeds certain limits. Environmental contamination with trace elements is very difficult to revert, because trace elements cannot be degraded and persist in the environment. This warrants the need for a careful use and management of trace elements.
Uptake by food is an important route of exposure of humans to trace elements. Crops such as potato may accumulate trace elements in levels that exceed proposed maximum acceptable levels for human consumption without adverse effects on growth. An increase in uptake of essential trace elements may be desirable in many cases, considering that more than half of the world’s population is estimated to be affected by deficiencies of microelements (Zhao and McGrath 2009).
The potato is the world’s fourth largest food crop and is a staple in many diets around the world (Ozturk et al. 2011). It is thus important to assess the status of trace elements in this crop. In this contribution, various aspects in relation to trace elements in potato are addressed. A first section provides general concepts related to trace elements in the environment. Then, common trace element concentration ranges in soils and in potato are explored, with reference to other vegetables. The subsequent section focuses on the relative importance of potato in contributing to the daily intake of trace elements. Finally, environmental and plant-related factors that affect the uptake of trace elements in potato are explored.
Materials and Methods
Survey
To investigate the relation between trace element contents in potato tuber and soil properties in agricultural soils containing baseline levels of elements, a small survey was conducted. In spring 2014, potato and topsoil (0–30 cm) were sampled on 21 agricultural fields in Flanders, Belgium. All potatoes were of the cultivar Bintje, which is widely grown in Europe. Soil samples were dried to constant weight at 105 °C and ground using a pestle and mortar to pass a 2-mm sieve. The peel of the potato tubers was removed, and the tubers were rinsed using tab water, blotted with a towel, and cut into pieces of about 1 cm3. After determination of wet mass, the potato was dried for several days at 60 °C until constant weight. Subsequently, the dried plant material was powdered in a stainless steel cross beater mill (Culati AG, Zurich, Switzerland) and stored before analysis in polyethylene vials.
Soil conductivity was measured with a WTW LF 537 electrode (Wissenschafltich-Technischen Werkstäten, Weilheim, Germany) after equilibration for 30 min in deionized water at a 5:1 liquid/solid ratio and subsequent filtering (white ribbon; Schleicher & Schuell, Germany). To determine the pH in a water suspension, (pH-H2O), 10 g of air-dry soil was allowed to equilibrate in 50 ml of deionized water for 24 h. For determination of the pH in KCl (pH-KCl), 50 ml of 1 M KC1 was added to 10 g of air-dried soil and allowed to equilibrate for 10 min. The pH of the supernatant was measured using a pH glass electrode (Model 520A, Orion, Boston, MA, USA). Organic matter was assessed using the method of Walkley and Black (Nelson and Sommers 1982). Soil metal content was determined after aqua regia digestion (Van Ranst et al. 1999). Metal contents in potato were determined after wet digestion in HNO3/H2O2 (Du Laing et al. 2003). Trace element analysis was performed using ICP-OES (Varian Vista MPX, Varian, Palo Alto, CA, USA). The quality of the analyses was checked by analyzing procedural blanks, standards, and certified reference material (ryegrass, CRM 281).
Compilation of Averages
Data on trace element concentrations in potato were compiled from literature. References were selected that did not involve contamination studies, but that involved surveys or measurements of common vegetables and potato in agriculture and on the market. Elemental contents were recorded as concentrations on wet weight. Data on vegetables and potato expressed on dry weight were converted to wet weights, using dry matter contents provided by the author if available, but mostly using typical dry matter contents for vegetables (Table 1). Published scientific work unfortunately not always indicates explicitly whether reported contents are based on fresh weight or dry weight. These references were not considered.
Reported averages or medians were recorded. In addition, maxima and minima and the number of observations were recorded where available. When more detailed data on individual observations were available, average, minimum, and maximum were calculated. Typical means reported in literature were calculated as the medians from the recorded averages or medians. The range of reported means was calculated as the 10 and 90% percentile of these data. To indicate “typical” reported minima and maxima, the 10% percentile of the reported minima and the 90% percentile of the reported maxima was calculated to eliminate the influence of outliers in evaluating typical ranges.
Plotting of Transfer Factors
The transfer factor was defined as the ratio between the concentration in the vegetable, expressed in milligram/kilogram fresh weight, and that in the soil, expressed in milligram/kilogram dry soil (Kiekens and Camerlynck 1982). To calculate average transfer factors, typical worldwide soil concentrations were calculated as the inverse log of the median of the log-transformed values of minimum and maximum of the range of average concentrations reported worldwide. For concentrations in plant or potato, the medians of averages and medians found in literature were used.
Recommended Daily Intake
The observations of the survey were used to estimate the contribution of potato to the recommended daily intake of selected trace elements. An intake of 150 g of potato was assumed. Values for acceptable daily intake were taken from Anonymous (2001).
Trace Elements in the Environment: General Concepts
The soil is the primary source of trace elements for plants and the entire ecosystem developing on it. Normal concentrations of many trace elements in unpolluted soils are between 1 and 100 mg/kg dry soil (Table 2). Some occur at much lower levels, e.g., Cd, Hg, Mo, Se, and Sn. Exceptions are Fe and Mn, which can be major constituents in particular soils. Wide ranges in the average concentrations indicate a marked variability between regions of the world, which is largely related to the nature of the parent material from which the soils developed (Kabata-Pendias 1993).
Trace elements in the soil are distributed over different compartments and compounds. They exist in a variety of chemical associations and binding forms that strongly differ in reactivity. In the soil solution, elements can exist in their free ionic form, e.g., as the hydrated cations Cd2+, Cu2+, and MoO4 2−, but also as inorganic complexes or complexed to a variety of organic ligands. The latter include small organic compounds such as citrate or acetate and large organic compounds that constitute the dissolved organic carbon in solution. The free elements are believed to be the most available to plants (Parker et al. 2001). Complexes in the soil solution, however, may enhance plant uptake of elements because they contribute to maintain the activities of free ions in the solution (Tack 2010).
Trace elements present in the soil solid phase include adsorbed, complexed, or occluded fractions. Trace elements which are superficially adsorbed or complexed with solid-phase components, such as clay minerals, hydrated oxides of iron and manganese, or organic matter, are more or less exchangeable with the soil solution. When occluded within soil solids such as hydrous oxides of iron and manganese, carbonates, or organic matter, they are at least temporarily unavailable for exchange with the soil solution and hence plant uptake, unless they are again exposed to the soil solution when the phase concerned is partially dissolved or destroyed (Tack 2010). Trace elements that are structurally incorporated in soil minerals are largely immobile. Nevertheless, root exudates of the growing plant still may release the minute amounts they need even from these highly insoluble compounds (Kabata-Pendias 1993).
The distribution of trace elements between these forms is governed by physicochemical reactions of sorption/desorption and dissolution/precipitation. The prevailing reactions and the extent by which they proceed is determined by the physicochemical environment, notably pH and redox potential. As the soil is continuously subject to changes effectuated by environmental conditions and biological activity, and because many reactions in the soil are very slow, a soil system never is in true thermodynamical equilibrium (Sparks 2001).
Many trace elements are not known to have a biological function at low concentrations. A limited number of elements, however, have been shown to be indispensable for the growth and development of organisms. Even if only very low amounts are needed, absence of these will render the organism unable to complete its life cycle (Arnon and Stout 1939). For plants, essential trace elements include boron (B), copper (Cu), iron (Fe), manganese (Mn), molybdenum (Mo), and zinc (Zn). Apart from B, these elements are also essential for animals and humans. These organisms additionally rely on cobalt (Co), chromium (Cr), fluorine (F), nickel (Ni), selenium (Se), and vanadium (V) as essential micronutrients (Alloway 1990; Kabata-Pendias 2010).
In the 1950–1960s, some serious incidents involving severe trace element toxicity problems have occurred. These have triggered public and scientific attention to issues of trace element contamination. In Toyama, Japan, a region downstream of a mining area was contaminated with metals causing the downstream population to be chronically exposed to elevated concentrations of Cd in rice. This was shown to be at the origin of an endemic disease. Serious deformities of the bones was one of the most striking symptoms of the so-called itae-itae disease (Nogawa 1981). Also in Japan, widespread mercury poisoning in the region around Minamata Bay caused serious health problems, referred to as “Minamata disease.” Mainly fishermen and their families were affected. They consumed local fish and shellfish that proved to be strongly contaminated with mercury discharged from a local chemical plant (Sakamoto et al. 2001). Unfortunately, the “Arsenic Crisis” in Bangladesh and West Bengal is an example of a contemporary issue with trace elements. It is caused by the chronic exposure of about ten million people to excessive As concentrations in water from tube wells, which was consumed as drinking water and used for irrigation in agriculture. Symptoms include arsenic-induced skin lesions and hyperkeratoses, i.e., hardened patches of skin that may develop into cancers (Bagla and Kaiser 1996; Tondel et al. 1999).
Trace Element Contents in Vegetables and Potato
Table 3 presents a compilation of concentration data of trace elements in vegetables in general, bulbs and roots, and potato in particular. Expressed on a fresh weight basis, trace elements in vegetables usually are at levels below 1 mg/kg. Some elements that are essential for plants are found in levels in the order of 10 mg/kg fresh weight (Cu, Fe, Mn, Zn).
The extent by which vegetables in general and potato in particular take up elements from soils may be inferred by plotting the overall ratio of plant metal contents versus soil contents (Fig. 1). The transfer factor, i.e., the ratio between metal concentrations in the plant and that in the soil (Kiekens and Camerlynck 1982), is highest for the four essential elements Mo, Fe, Zn, and Cu. The first is an essential element that naturally occurs in very low concentrations in the soil. Molybdenum plays a role in the nitrogen assimilation of plants. Cadmium as a non-essential trace element also exhibits a relatively high transfer factor. Geochemically, it behaves very much similar to Zn (Smies 1983). Its relatively high mobility and availability to plants is a reason why this element is of concern.
The transfer factors toward the potato tuber are superimposed on the graph and reveal similarities and differences with the trend observed for vegetables in general. Potato appears to have a higher tendency to accumulate Fe, Zn, Mn, and Cr than average vegetables. Accumulation of the essential element Cu tends to be lower than that for vegetables in general.
Figure 2 provides a more detailed view of the average content of selected trace elements compared to other vegetables. Vegetables are plotted in order of decreasing average concentrations. Except for Cr and Cu, typical concentrations in potato tuber are in a mid-range, common to many other vegetables. Intervals indicate typical ranges that have been reported in literature. They may be limited by the available data and thus do not necessarily indicate a true tendency of a vegetable to exhibit more or less variability in metal contents. Overall, potato tubers exhibit moderate trace element concentrations, despite being a crop that grows in the soil. It should be noted that potato is a vegetable with a relatively high dry matter content (about 20%) compared to many other vegetables (e.g., leek and carrot, about 10%; leafy vegetables and tomato, 5% and below). Hence, when expressed on the basis of dry matter content, potato would shift to lower positions on these graphs.
Daily Intake of Trace Elements Through Potato
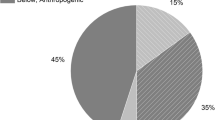
Potato is a staple food in many countries and thus has the potential to contribute significantly to the intake of trace elements through the diet in these countries. Although potato usually is considered to contribute primarily carbohydrates and energy to the diet, it is also an important contributor for a few essential elements, notably Fe, Cu, and Cr. For Cu, potato ranked after bread as the second most important contributor of Cu to the diet in the Netherlands (13% of the total daily intake) and in Germany (7%) (Van Dokkum et al. 1989; Van Dokkum 1995). For Fe, bread was estimated to contribute most, followed by meat, and then by potato (9% for The Netherlands, 5% for Germany) (Van Dokkum et al. 1989). Although Cr was not included in these studies, it may be concluded from evaluating the trace element contents that potato may also significantly contribute. Based on the concentrations of Cr of potato in Belgium, Cr was estimated to contribute by 35% to the daily intake of the element. Contributions for other elements were estimated as 12% for Cu, 8% for Fe, 6% for Mn and Zn, and 1% for Ni.
The above are only averaged trends. The actual value of a particular potato harvest in mineral nutrition depends primarily on the potato cultivar and also on the location where the plant is grown. From an extensive survey involving 16 cultivars grown at five locations, it was concluded that particular cultivars may provide significant contributions to the recommended daily intake of Cu, Fe, Se, and Zn. Whereas Cu and Fe more consistently exhibited a significant contribution to the ADI, this was not the case for Se and Zn (Nassar et al. 2012).
Where potato is consumed as a staple food, it invariably constitutes one of the most important contributors of the element Cd in the daily intake (López-Artíguez et al. 1993; Van Dokkum 1995; Ysart et al. 2000; Llobet et al. 2003; Santos et al. 2004). Current European guidelines (European Commission 2006) restrict maximum levels of Cd in the potato tuber to 0.1 mg/kg fresh weight. In a survey conducted in Flanders, none of the samples exceeded this value (Cd concentrations in mg/kg fresh weight: median, 0.017, min, 0.005, max, 0.078 mg/kg). The median average concentration for Cd reported in literature is 0.021 (Table 3). One out of 29 references reported an average Cd concentration exceeding 0.1 mg/kg. From the 20 references reporting maxima, 15 or 75% reported maximum values below 0.1 mg/kg fresh weight. Although this suggests that there is essentially no general problem with Cd levels in potato, monitoring is needed because there remains a possibility that reference values are exceeded even in soils with baseline metal contents. There is a need for a good control on fertilizers and soil amendments that, through their impurities, may introduce available Cd in the soil.
Factors Influencing Trace Element Concentrations in Potato
In cultivating crops, focus on management of trace elements is increasing. It is estimated that more than half of the world’s population is affected by deficiencies of the microelements Fe, Zn, Se, and I. Other elements that can be deficient include Ca, Mg, and Cu (Zhao and McGrath 2009). Traditional approaches to address malnutrition include supplementation, food fortification, and diversification of the diet. A complementary approach has recently been proposed, where attempts are made to increase the bioavailable concentrations of essential elements in edible portions of crop plants by means of either breeding, plant biotechnology, the use of micronutrient fertilizers, or enhancing of the bioavailability of micronutrients in the crop by manipulating the compounds that promote or decrease their uptake (Frossard et al. 2000; White and Broadley 2005; Zhao and McGrath 2009). Conversely, there may be a need to reduce the uptake of potentially toxic non-essential metals such as Cd and Pb in order to safeguard food safety and protect the population against excessive exposure to these elements. Current European regulations stipulate that the potato tuber should not contain Cd or Pb in excess of 100 μg/kg fresh weight (European Commission 2006). As shown above, baseline total contents of these elements are not strongly below that level, and considering the natural variability in metal concentrations, these levels may be exceeded at times. In this context, it is increasingly important to manage trace elements in soils and crops. This may not be easy because the uptake of trace elements in vegetables is in a complex way governed by a multitude of factors, related to the soil properties (soil components, nutritional state, soil chemistry), the plant (plant species and cultivar, plant physiology), and the growing conditions (temperature, weather, moisture regimes).
Soil Properties
The uptake of several trace elements in plants tends to be reduced at higher pH. Specifically for potato, data from field surveys in Sweden revealed significant negative correlations between Cd, Ni, Zn, Mn, Cu, and Al concentration in potatoes and surface soil pH (0–25 cm) (Öborn et al. 1995).
In an extensive field study in Southern Australia, soil salinity was shown to markedly increase the uptake of Cd in potato tubers (McLaughlin et al. 1994). This was attributed to the use of saline irrigation waters. The observations included a wide range of salinities (electrical conductivity between 0.07 and 1.43 dS/m in a 1:5 soil/water extract). The chloride anion forms relatively strong complexes with the Cd2+ ion and therefore mobilizes Cd and increases its availability.
Limited data from a survey in Flanders provide a first look into the relations between soil properties, soil element contents, and concentrations in potato grown in uncontaminated fields with baseline element concentrations. Contents for Cd, Cr, Cu, and Zn in potato tubers are plotted in relation to soil properties in Fig. 3. Trace element contents in potato tuber were within the general ranges found in literature (Table 3). Two observations for Cd exceeded 0.06 mg/kg fresh weight. Although still below the maximum limit for potato (0.1 mg/kg fresh weight), these concentrations are markedly higher than typical averages reported in literature, indicating a need to closely monitor concentrations of this element. All Pb concentrations observed, in contrast, were low compared to typical averages reported in literature and significantly below the maximum limits.
Relations between metal contents in potato tuber and soil properties are practically absent in these observations relating to soils with baseline metal contents and a relatively limited variability in soil properties. Influences of soil properties, if any, are thus masked by the variability in the data. The lack of a relation between total contents in soils and plants is evident. For example, for Cd, the two observations with relatively high Cd contents were observed in soils with low total Cd contents. The pH of these soils was also among the higher ones, which is contradictory to the general notion that trace elements tend to be less mobile with higher pH. In the context of food safety, where strict limits on total element contents in the crops may be imposed, it is useful to be able to predict the chances of exceeding threshold levels in specific soils. More research will be needed to develop practical models that allow to estimate the likelihood of exceeding certain levels in the plant.
The relation between Cu and organic matter is the only exception where a generally expected trend is also reflected in the data. Copper is known to be strongly associated with organic matter (Alloway 1990; Kabata-Pendias 2010). High organic matter contents in a soil are therefore expected to result in a stronger binding of Cu in the soil and a reduced uptake in the plant. Although current data are limited, a reduced range in Cu concentrations in the plant is observed with a higher organic matter content in the soil.
Cultivar
Plant-specific mechanisms of plant metal tolerance and homeostasis play a major role in determining the effective uptake of elements in a given growing environment (Clemens 2001). There are large differences between plant species and even within the same species (Florijn and van Beusichem 1993). Specifically for potato, the strong differences in elemental content of potato between cultivars and within cultivars when grown in different locations were convincingly shown in the field study cited before (Nassar et al. 2012). In Tenerife, traditional cultivars were shown to accumulate higher levels of Fe, Zn, and Cu than recently introduced cultivars (Rivero et al. 2003). Sixteen potato cultivars grown at Erzurum, Turkey, showed a considerable variation in trace element concentrations (Ozturk et al. 2011).
Influence of Agricultural Practices
There has been concern that modern cultivars and/or agronomic practices have resulted in reduced concentrations of trace elements essential to human nutrition in edible crops. This was found to be only partially true (White et al. 2009). The application of fertilizers influences the trace element content in potato in a complex manner that depends on effects of soil chemistry and interactions between trace elements in the plant. Higher-yielding varieties occasionally have lower concentrations of some mineral elements in their edible tissues than lower-yielding varieties (White et al. 2009). The concentrations of As, Cd, Cr, Cu, Fe, Mn, Pb, and Zn in tubers were not significantly affected by fertilizer treatment although pig slurry supplied substantially higher amounts of Cu and Zn to the soil than were taken up by the tubers (Šrek et al. 2010). The effect of organic farming versus conventional farming was investigated in a couple of studies (Jorhem and Slanina 2000; Hajšlová et al. 2005). Both studies concluded that there were no systematic differences in trace element contents between potato grown conventionally or organically and that organic farming, at least in the short term, does not necessarily result in reduced levels of Cd and other potentially harmful elements in the crops. Variabilities among years, variety, and geographical factors were considered equally or more important in determining the quality of potatoes than the farming system (Hajšlová et al. 2005).
Conclusions
Trace elements, despite their low concentrations, have a crucial importance. Some elements are indispensable for life. Many elements have various uses and are indispensable in modern society. However, trace elements are potentially toxic, and wrong handling may cause serious environmental issues that are very difficult to revert. Trace elements thus must be carefully managed.
Although potato is a product growing in the soil, the tuber does not intensively accumulate trace elements. Concentrations of trace elements are in the range of other vegetables. Generally, potato exhibits a rather low nutritional value for trace elements, with the exception of Fe, Cr, and Cu. In soils with baseline metal concentrations, there are no indications for concern with potentially toxic elements although Cd needs to be monitored. Environmental care and good soil management is a must to safeguard the safety of food, including potato.
References
Alloway BJ (1990) Heavy metals in soils. Blackie and Son, Glasgow
Anderson KA, Magnuson BA, Tschirgi ML, Smith B (1999) Determining the geographic origin of potatoes with trace metal analysis using statistical and neural network classifiers. J Agric Food Chem 47:1568–1575. doi:10.1021/jf980677u
Anonymous (2001) Dietary reference intakes for vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium, and zinc. National Academy Press, Washington, DC
Arnich N, Sirot V, Rivière G, et al (2012) Dietary exposure to trace elements and health risk assessment in the 2nd French Total Diet Study. Food Chem Toxicol 50:2432–2449. doi:10.1016/j.fct.2012.04.016
Arnon DI, Stout PR (1939) The essentiality of certain elements in minute quantity for plants with special reference to copper. Plant Physiol 14:371–375
Bagla P, Kaiser J (1996) India’s spreading health crisis draws global arsenic experts. Science 274:174
Becker W, Jorhem L, Sundström B, Grawé KP (2011) Contents of mineral elements in Swedish market basket diets. J Food Compos Anal 24:279–287. doi:10.1016/j.jfca.2010.10.001
Biego GH, Joyeux M, Hartemann P, Debry G (1999) Determination of dietary tin intake in an adult French citizen. Arch Environ Contam Toxicol 36:227–232
Choi M-K, Jun Y-S (2008) Analysis of boron content in frequently consumed foods in Korea. Biol Trace Elem Res 126:13–26. doi:10.1007/s12011-008-8179-7
Clemens S (2001) Molecular mechanisms of plant metal tolerance and homeostasis. Planta 212:475–486
De Temmerman L, Waegeneers N, Thiry C, et al (2014) Selenium content of Belgian cultivated soils and its uptake by field crops and vegetables. Sci Total Environ 468–469:77–82. doi:10.1016/j.scitotenv.2013.08.016
Di Giacomo F, Del Signore A, Giaccio M (2007) Determining the geographic origin of potatoes using mineral and trace element content. J Agric Food Chem 55:860–866. doi:10.1021/jf062690h
Du Laing G, Tack FMG, Verloo MG (2003) Performance of selected destruction methods for the determination of heavy metals in reed plants (Phragmites australis). Anal Chim Acta 497:191–198
Ekholm P, Reinivuo H, Mattila P, et al (2007) Changes in the mineral and trace element contents of cereals, fruits and vegetables in Finland. J Food Compos Anal 20:487–495. doi:10.1016/j.jfca.2007.02.007
European Commission (2006) Commission Regulation (EC) No 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs.
Florijn PJ, van Beusichem ML (1993) Uptake and distribution of cadmium in maize inbred lines. Plant Soil 150:25–32. doi:10.1007/BF00779172
Frossard E, Bucher M, Mächler F et al (2000) Potential for increasing the content and bioavailability of Fe, Zn and Ca in plants for human nutrition. J Sci Food Agric 80:861–879. doi:10.1002/(SICI)1097-0010(20000515)80:7<861::AID-JSFA601>3.0.CO;2-P
Hajšlová J, Schulzová V, Slanina P et al (2005) Quality of organically and conventionally grown potatoes: four-year study of micronutrients, metals, secondary metabolites, enzymic browning and organoleptic properties. Food Addit Contam 22:514–534. doi:10.1080/02652030500137827
Ismail F, Anjum MR, Mamon AN, Kazi TG (2011) Trace metal content of vegetables and fruits of hyderabad retail market. Pak J Nutr 10:365–372
Jinadasa KBPN, Milham PJ, Hawkins CA, et al (1997) survey of cadmium levels in vegetables and soils of Greater Sydney, Australia. J Environ Qual 26:924–933. doi:10.2134/jeq1997.00472425002600040002x
Jorhem L, Slanina P (2000) Does organic farming reduce the content of Cd and certain other trace metals in plant foods? A pilot study. J Sci Food Agric 80:43–48. doi:10.1002/(SICI)1097-0010(20000101)80:1<43::AID-JSFA482>3.0.CO;2-Y
Kabata-Pendias A (1993) Behavioural properties of trace metals in soils. Appl Geochem 8(Supplement 2):3–9. doi:10.1016/S0883-2927(09)80002-4
Kabata-Pendias A (2010) Trace elements in soils and plants. CRC, Boca Raton, FL
Kiekens L, Camerlynck R (1982) Transfer characteristics for uptake of heavy metals by plants. Landwirtsch Forsch 39:255–261
Larsen EH, Moseholm L, Nielsen MM (1992) Atmospheric deposition of trace elements around point sources and human health risk assessment. II: Uptake of arsenic and chromium by vegetables grown near a wood preservation factory. Sci Total Environ 126:263–275. doi:10.1016/0048-9697(92)90201-3
Lendinez E, Lorenzo ML, Cabrera C, López MC (2001) Chromium in basic foods of the Spanish diet: seafood, cereals, vegetables, olive oils and dairy products. Sci Total Environ 278:183–189. doi:10.1016/S0048-9697(01)00647-7
Llobet JM, Falco G, Casas C et al (2003) Concentrations of arsenic, cadmium, mercury, and lead in common foods and estimated daily intake by children, adolescents, adults, and seniors of Catalonia, Spain. J Agric Food Chem 51:838–842
López-Artíguez M, Soria ML, Cameán A, Repetto M (1993) Cadmium in the diet of the local population of Seville (Spain). Bull Environ Contam Toxicol 50:417–424
Mansour SA, Belal MH, Abou-Arab AAK, et al (2009) Evaluation of some pollutant levels in conventionally and organically farmed potato tubers and their risks to human health. Food Chem Toxicol 47:615–624. doi:10.1016/j.fct.2008.12.019
McLaughlin MJ, Palmer LT, Tiller KG et al (1994) Increased soil salinity causes elevated cadmium concentrations in field-grown potato tubers. J Environ Qual 23:1013–1018
Milacic R, Kralj B (2003) Determination of Zn, Cu, Cd, Pb, Ni and Cr in some Slovenian foodstuffs. Eur Food Res Technol Z Für Lebensm-Unters -Forsch A 217:211–214. doi:10.1007/s00217-003-0755-7
Muñoz O, Bastias JM, Araya M, et al (2005) Estimation of the dietary intake of cadmium, lead, mercury, and arsenic by the population of Santiago (Chile) using a Total Diet Study. Food Chem Toxicol 43:1647–1655. doi:10.1016/j.fct.2005.05.006
Nassar AMK, Sabally K, Kubow S et al. (2012) Some Canadian-grown potato cultivars contribute to a substantial content of essential dietary minerals. J Agric Food Chem 60:4688–4696. doi:10.1021/jf204940t
Nelson DW, Sommers LE (1982) Total carbon, organic carbon, and organic matter. In: Nelson DW, Miller RH, Keeney DR (eds) Methods of soil analysis, Part 2. Chemical and microbiological properties. ASA, Madison, Wisconsin, pp 539–579
Nielson KK, Mahoney AW, Williams LS, Rogers VC (1991) Screening for elevated lead and tin in fruits and vegetables by Nondestructive X-ray fluorescence. J Food Compos Anal 4:206–215. doi:10.1016/0889-1575(91)90032-2
Nogawa K (1981) Itai-Itai disease and follow-up studies. In: Nriagu, JO (ed) Cadmium in the Environment, Part 11, Health Effects. Wiley, New York, p 1–37
Öborn I, Jansson G, Johnsson L (1995) A field study on the influence of soil ph on trace element levels in spring wheat (Triticum aestivum), potatoes (Solanum tuberosum) and carrots (Daucus carota). Water Air Soil Pollut 85:835–840. doi:10.1007/BF00476933
Oporto C, Vandecasteele C, Smolders E (2007) Elevated cadmium concentrations in potato tubers due to irrigation with river water contaminated by Mining in Potosí, Bolivia. J Environ Qual 36:1181. doi:10.2134/jeq2006.0401
Ozturk E, Atsan E, Polat T, Kara K (2011) Variation in heavy metal concentrations of potato (Solanum tuberosum L.) cultivars. J Anim Plant Sci 21:235–239
Parker DR, Pedler JF, Ahnstrom ZAS, Resketo M (2001) Reevaluating the free-ion activity model of trace metal toxicity toward higher plants: experimental evidence with copper and zinc. Environ Toxicol Chem 20:899–906
Parveen Z, Khuhro MI, Rafiq N (2003) Market Basket Survey for Lead, Cadmium, Copper, Chromium, Nickel, and Zinc in Fruits and Vegetables. Bull Environ Contam Toxicol 71:1260–1264. doi:10.1007/s00128-003-8640-4
Queirolo F, Stegen S, Restovic M, et al (2000) Total arsenic, lead, and cadmium levels in vegetables cultivated at the Andean villages of northern Chile. Sci Total Environ 255:75–84. doi:10.1016/S0048-9697(00)00450-2
Radwan MA, Salama AK (2006) Market basket survey for some heavy metals in Egyptian fruits and vegetables. Food Chem Toxicol 44:1273–1278. doi:10.1016/j.fct.2006.02.004
Rainey CJ, Nyquist LA, Coughlin JR, Downing RG (2002) Dietary boron intake in the United States: CSFII 1994–1996. J Food Compos Anal 15:237–250
Rivero RC, Hernández PS, Rodrı́guez EMR et al (2003) Mineral concentrations in cultivars of potatoes. Food Chem 83:247–253. doi:10.1016/S0308-8146(03)00087-6
Sakamoto M, Nakano A, Akagi H (2001) Declining Minamata male birth ratio associated with increased male fetal death due to heavy methylmercury pollution. Environ Res 87:92–98
Sanchez-Castillo CP, Dewey PJS, Aguirre A, et al (1998) The mineral content of Mexican fruits and vegetables. J Food Compos Anal 11:340–356. doi:10.1006/jfca.1998.0598
Santos EE, Lauria DC, Porto da Silveira CL (2004) Assessment of daily intake of trace elements due to consumption of foodstuffs by adult inhabitants of Rio de Janeiro city. Sci Total Environ 327:69–79. doi:10.1016/j.scitotenv.2004.01.016
Simsek A, Velioglu YS, Coskun AL, Sayli BS (2003) Boron concentrations in selected foods from borate-producing regions in Turkey. J Sci Food Agric 83:586–592. doi:10.1002/jsfa.1408
Singh V, Garg AN (2006) Availability of essential trace elements in Indian cereals, vegetables and spices using INAA and the contribution of spices to daily dietary intake. Food Chem 94:81–89. doi:10.1016/j.foodchem.2004.10.053
Smies M (1983) Biological aspects of trace element speciation in the aquatic environment. In: Leppard G G (ed). Trace element speciation in surface waters and its ecological implications. Plenum, New York, p 177–191
Sobukola OP, Adeniran OM, Odedairo AA, Kajihausa OE (2010) Heavy metal levels of some fruits and leafy vegetables from selected markets in Lagos, Nigeria. Afr J Food Sci 4:389–393
Sparks DL (2001) Elucidating the fundamental chemistry of soils: past and recent achievements and future frontiers. Geoderma 100:303–319
Šrek P, Hejcman M, Kunzová E (2010) Multivariate analysis of relationship between potato (Solanum tuberosum L.) yield, amount of applied elements, their concentrations in tubers and uptake in a long-term fertilizer experiment. Field Crops Res 118:183–193. doi:10.1016/j.fcr.2010.05.009
Sungur Ş, Okur R (2009) Using azomethine-H method determination of boron contents of various foods consumed in Hatay Region in Turkey. Food Chem 115:711–714. doi:10.1016/j.foodchem.2008.11.103
Tack FMG (2010) Trace elements: general soil chemistry, principles and processes. In: Hooda P (ed) Trace elements in soils. Wiley-Blackwell, Chichester, pp 9–37
Tahvonen R (1993) Contents of selected elements in some fruits, berries, and vegetables on the Finnish market in 1987–1989. J Food Compos Anal 6:75–86. doi:10.1006/jfca.1993.1009
Tondel M, Rahman M, Magnuson A et al (1999) The relationship of arsenic levels in drinking water and the prevalence rate of skin lesions in Bangladesh. Environ Health Perspect 107:727
Tsongas TA, Meglen RR, Walravens PA, Chappell WR (1980) Molybdenum in the diet: an estimate of average daily intake in the United States. Am J Clin Nutr 33:1103–1107
Van Dokkum W (1995) The intake of selected minerals and trace elements in European countries. Nutr Res Rev 8:271–302. doi:10.1079/NRR19950016
Van Dokkum WV, De Vos RH, Muys T, Wesstra JA (1989) Minerals and trace elements in total diets in The Netherlands. Br J Nutr 61:7–15. doi:10.1079/BJN19890087
Van Ranst E, Verloo M, Demeyer A, Pauwels JM (1999) Manual for the soil chemistry and fertility laboratory. International Training Centre for Post-Graduate Soil Scientists, Gent, Belgium
White PJ, Broadley MR (2005) Biofortifying crops with essential mineral elements. Trends Plant Sci 10:586–593. doi:10.1016/j.tplants.2005.10.001
White PJ, Bradshaw JE, Finlay M et al (2009) Relationships Between Yield and Mineral Concentrations in Potato Tubers. HortSci 44:6–11
Ysart G, Miller P, Croasdale M et al (2000) 1997 UK Total Diet Study dietary exposures to aluminium, arsenic, cadmium, chromium, copper, lead, mercury, nickel, selenium, tin and zinc. Food Addit Contam 17:775–786. doi:10.1080/026520300415327
Zhao F-J, McGrath SP (2009) Biofortification and phytoremediation. Curr Opin Plant Biol 12:373–380
Acknowledgements
The author acknowledges Arne Demey for his involvement in the survey.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tack, F.M.G. Trace Elements in Potato. Potato Res. 57, 311–325 (2014). https://doi.org/10.1007/s11540-014-9268-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11540-014-9268-y