Abstract
Background
DESTINY-Breast01 (NCT03248492) is a phase II single-arm trial evaluating trastuzumab deruxtecan (T-DXd) in adults with human epidermal growth factor receptor 2-positive (HER2+) unresectable or metastatic breast cancer (u/mBC) who have received two or more prior anti-HER2 therapies.
Objectives
Objectives were to explore approaches for estimating long-term overall survival (OS) with T-DXd from immature data (June 2020 data-cut; median follow-up 20.5 months), and compare predicted long-term outcomes with UK-recommended non-targeted therapies eribulin, capecitabine, and vinorelbine.
Methods
Two methods were used to model T-DXd long-term OS: (1) applying a hazard ratio (HR) to the OS curve for another HER2 targeted therapy (third-line trastuzumab emtansine [T-DM1]) with longer trial follow-up; and (2) extrapolating T-DXd OS data directly. Comparator OS was based on direct extrapolation of published data (comparison with vinorelbine OS was not possible). Quality-adjusted life years (QALYs) were calculated using a previously published model of utility.
Results
Both extrapolation methods demonstrated longer mean/median OS with T-DXd versus eribulin, and capecitabine (44.7/32.9 months [applying an HR to the T-DM1 OS curve]; 47.7/29.9 months [using direct extrapolation]; vs 11.3/9.2, and 17.8/13.6 months, respectively), translating to 2.3, 2.3, 0.6, and 0.9 discounted QALYs.
Conclusion
Alternative methods produced consistent results, showing T-DXd is associated with substantial gains in OS and QALYs versus eribulin, and capecitabine. Modelled median OS results were similar to a later data-cut (median of 29.1 months, March 2021 data-cut). The modelling approach in which an HR was applied to the T-DM1 OS curve informed a submission to the National Institute for Health and Care Excellence.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The prognosis of patients with unresectable or metastatic breast cancer (u/mBC) who have received two or more prior anti-human epidermal growth factor receptor 2 (HER2) therapies is poor; UK-recommended non-targeted therapies (eribulin, capecitabine, and vinorelbine) offer limited overall survival (OS) and progression-free survival (PFS). There is an unmet need for targeted therapies. |
The single-arm DESTINY-Breast01 trial investigated trastuzumab deruxtecan (T-DXd, a HER2-targeted antibody drug conjugate) at third or later lines. Interim OS data were relatively immature at the time of this analysis, which presents two approaches to modelling long-term outcomes from immature data. |
This is the first publication reporting methods for extrapolation of long-term survival with T-DXd. Modelled results are consistent, showing T-DXd is associated with substantial gains in life expectancy and discounted quality-adjusted life years versus eribulin, and capecitabine. The modelled median OS (using immature data; June 2020 data-cut) is also consistent with OS data from a later data-cut (March 2021). The modelling approach informed a successful submission to the National Institute for Health and Care Excellence. |
1 Introduction
Breast cancer (BC) is the most common cancer in the UK [1], with a small proportion of patients diagnosed in advanced stages of disease [2]. The most advanced forms, unresectable (inoperable) and metastatic (u/m), are generally incurable [3]. Approximately 13–20% of BCs are human epidermal growth factor receptor 2-positive (HER2+) [4], characterized by HER2 overexpression on the tumor cell surface. This promotes proliferation, activating the PI3K/AKT and RAS/MAPK pathways [5, 6], resulting in more clinically aggressive disease with worse outcomes [7].
BC accounts for 30% of all new cancer cases in UK females (55,545 cases; crude incidence rate 166.0 per 100,000 [2016–2018]) [1] and is the second most common cause of all female cancer deaths (15%, 2018) [8]. The 21-year prevalence of female BC in England was 475,801 cases (diagnosed between 1995 and 2015, alive at the last day of follow-up) [9]. The prevalence of mBC is estimated at 7.39% of BCs, with 34.41% of mBC patients receiving third-line therapy [10].
Until recently, HER2-targeted therapies were available at first and second lines for HER2+ u/mBC, but not at third and later lines. Introduction and expanded use of these first- and second-line targeted therapies for patients with HER2+ u/mBC resulted in survival gains [11], but eventually, nearly all patients develop de novo or acquired resistance and experience disease progression [12].
Previously, at third line, the National Institute for Health and Care Excellence (NICE) pathways recommended non-targeted chemotherapies, eribulin, capecitabine, or vinorelbine for both HER2+ and HER2-negative (HER2−) locally advanced or metastatic disease [13, 14] (however, only eribulin has been appraised by NICE in this patient population [13]). The prognosis of patients who have received two or more prior anti-HER2 therapies is poor; eribulin, capecitabine, and vinorelbine offer limited overall survival (OS; median 10.2–15.2 months) [15,16,17,18,19,20,21,22,23] and progression-free survival (PFS; median 2.6–4.9 months) [15,16,17,18,19,20,21,22,23,24].
Trastuzumab deruxtecan (T-DXd) is indicated for adults with HER2+ u/mBC who have received two or more prior anti-HER2-based regimens and has been granted marketing authorization by the European Medicines Agency (conditional approval) and Medicines and Healthcare products Regulatory Agency [25, 26]. T-DXd is a HER2-targeted antibody drug conjugate (ADC) designed to deliver optimal anti-tumor efficacy and minimize off-target toxicity versus earlier ADCs [27,28,29,30]. T-DXd selectively binds to HER2, and following internalization, releases a cytotoxic topoisomerase I inhibitor payload, causing DNA damage and tumor cell death [27, 29].
The efficacy and safety of T-DXd in patients with HER2+ u/mBC who have received two or more prior anti-HER2 therapies was assessed in the phase II, multicenter, single-arm trial, DESTINY-Breast01 [31, 32]. In total, 184 female patients (median of six previous lines of therapy) were treated with T-DXd. At the June 2020 data-cut (the longest follow-up available at time of analysis; median 20.5 months), T-DXd was associated with a confirmed objective response rate (ORR) of 61.4% (95% confidence interval [CI] 54.0, 68.5) [32]. Median duration of response (DOR) was 20.8 months (95% CI 15.0, not evaluable [NE]), with a median PFS of 19.4 months (95% CI 14.1, NE). Median OS data were immature but encouraging (24.6 months; 95% CI 23.1, NE).
Clinical experts and trial data for comparators suggested life expectancy was < 24 months for patients progressing after trastuzumab emtansine (T-DM1) and preliminary OS results from DESTINY-Breast01 indicate a likely extension to life of > 3 months [33]. Therefore, NICE considers T-DXd to meet the end-of-life criteria. T-DXd helps address the substantial unmet need for a therapy with demonstrated efficacy and tolerability in patients with HER2+ u/mBC, and helps overcome treatment resistance. In April 2021, T-DXd was recommended by NICE for use within the Cancer Drugs Fund (CDF) for patients with HER2+ u/mBC who have received two or more prior anti-HER2 therapies [34]. Prior to this, no third-line targeted treatments were available for this population on the National Health Service (NHS) in England.
The objectives of this study were to explore two different methods for estimating long-term survival with T-DXd from immature OS data, and estimate the discounted quality-adjusted life year (QALY) and life year (LY) gains associated with T-DXd versus the comparators.
2 Methods
2.1 Overview
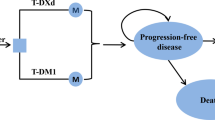
A de novo partitioned survival model consisting of four health states (progression-free [on- and off-treatment], progressed, dead) was developed to model discounted LYs and QALYs with T-DXd versus comparators. To model discounted LYs, estimates of OS over a lifetime horizon are required. Quality of life (QoL) estimates are informed by assigning utilities based on treatment status (on/off treatment), progression status, response status, and adverse events (AEs); thus, estimates of PFS and time to discontinuation (TTD) over a lifetime horizon are also required. Discounting was applied at a rate of 3.5% per annum; a 1-week cycle length was assumed. Modeling approaches are summarized in Table 1.
Relevant studies reporting efficacy and safety data for the comparators (eribulin, capecitabine, vinorelbine) were identified by systematic literature review (SLR). Multiple studies were identified for eribulin [15,16,17,18] and capecitabine [19,20,21,22,23]. For eribulin, Cortes et al. 2011 [17] was selected for the model base case, as it reports outcomes from the pivotal EMBRACE trial, the primary source of evidence in NICE technology appraisal 423 (TA423) investigating eribulin in patients with mBC [10, 13]; a scenario analysis considers eribulin data from Barni et al. [15], which presented data in a HER2+ BC population. For capecitabine, Cameron et al.2010 [23] was chosen as the base case as it was the only study that reported data in a HER2+ BC population; a scenario analysis considers capecitabine data from Fumoleau et al. [20], the most recent of the other identified studies. Only Sim et al. [24] was available to inform comparisons against vinorelbine. Clinical experts did not consider the OS data from this study to be clinically plausible as compared with the reported PFS or expected OS in UK patients; subsequent therapies may be driving the reported OS. Due to the concerns regarding plausibility of the OS data, it was not possible to generate long-term outcomes for vinorelbine.
All patients in DESTINY-Breast01 were HER2+. Of the studies selected for the base-case and scenario analyses, Cortes et al. 2011 had a population with mixed HER2 status, but provided the distribution of HER2 expression (18.2% of patients with known HER2 status were HER2+) [17]. Adjustments for HER2 status were made to the trial outcomes to compare with a 100% HER2+ population. Lv et al. retrospectively compared clinical outcomes of HER2+ patients with or without trastuzumab versus HER2− patients [35]. Patients with HER2+ disease experienced poorer OS outcomes compared with HER2– patients (hazard ratio [HR] 1.84, 95% CI 1.33, 2.56); this is consistent with clinical expert opinion. The HR from Lv et al. was applied to the OS and PFS curves for Cortes et al. 2011 in the proportion of patients with HER2− disease (81.8%); an HR of 1 (i.e. no adjustment) was applied to those with HER2+ disease (18.2%), resulting in an overall HR of 1.69. Fumoleau et al. did not present the proportion of HER2+ patients; in the scenario where this study is used to inform outcomes for capecitabine, 20% of patients were assumed to be HER2+, as observed in UK clinical practice [4]. No adjustment was required for data from Cameron et al. 2010, as capecitabine was evaluated in a HER2+ population, or from Barni et al., as data were available in the HER2+ sub-population.
For extrapolation of outcomes, six parametric distributions (exponential, Weibull, Gompertz, log-normal, log-logistic, and generalized gamma) were fitted to the data for each outcome. These were assessed for the lowest Akaike information criterion/Bayesian information criterion (AIC/BIC) scores and most plausible clinical outcomes, as validated by clinical experts.
2.2 Overall Survival
At the time of analysis, OS data for T-DXd from DESTINY-Breast01 were relatively immature; 64.7% of patients were alive at the June 2020 data-cut (median follow-up 20.5 months) [32]. Two alternative methods were used to model long-term OS with T-DXd: (1) applying an HR to the OS curve for another HER2-targeted therapy with longer trial follow-up, and (2) extrapolating T-DXd OS data directly.
For the first approach, clinical experts advised that the shape of the OS curve for T-DXd is likely to be more similar to another anti-HER2 therapy, such as T-DM1, than the non-HER2 targeted model comparators (eribulin, capecitabine, vinorelbine). T-DM1 data from the TH3RESA trial were considered the most relevant to inform T-DXd OS, as it investigated this anti-HER2 therapy in patients with HER2+ u/mBC at third line [36], and had longer follow-up than available at the DESTINY-Breast01 June 2020 data-cut. An OS HR for T-DXd versus T-DM1 was generated using a Cox proportional hazards (PH) model applied to T-DXd data from DESTINY-Breast01 and digitized T-DM1 data from TH3RESA. Predictions of long-term OS with T-DXd were generated by applying this HR to extrapolated digitized third-line T-DM1 data from TH3RESA. Of the alternative extrapolations of TH3RESA data, clinical experts considered the generalized gamma and exponential distributions to result in the most plausible OS curves for T-DXd; the generalized gamma distribution results in a more conservative estimate of long-term OS, thus was selected for the base case. The exponential distribution was considered in a scenario analysis.
For the second approach (direct extrapolation of T-DXd OS data), the log-logistic, log-normal, and exponential distributions resulted in OS curves considered plausible by clinical experts; the log-logistic distribution results in a more conservative estimate of long-term OS and was selected for the base case. All other survival distributions were considered in scenario analyses, except the Gompertz and generalized gamma distributions, which were not considered clinically plausible.
OS for the comparator treatments was estimated by fitting parametric survival curves to digitized Kaplan–Meier (KM) data from the relevant studies. Unadjusted KM data was used from the comparator trials, rather than utilizing the results of an indirect treatment comparison such as matching-adjusted indirect comparison (MAIC), as clinical experts indicated the shape of the OS curve would be different for T-DXd versus the non-targeted model comparators, and thus calculating an HR on the basis of an assumption of PH would not be appropriate.
For the base case, the generalized gamma and Weibull distributions were selected for eribulin, and capecitabine, respectively. For eribulin, this distribution represents a reasonable midpoint of the available extrapolations and has the second lowest AIC/BIC score; for capecitabine, the KM was relatively complete, and so this distribution was selected based on having the lowest AIC/BIC score. Model diagnostics are presented in electronic supplementary material (ESM) Table S3. Survival curves for the base-case extrapolations of OS are presented in Fig. 1.
Data from published life tables for England and Wales (2019) [37] were used in the model to prevent the weekly probability of mortality falling below that of the general population.
2.3 Progression-Free Survival
Parametric survival curves for PFS were generated for T-DXd and HRs from unanchored MAICs (ESM Table S2) were applied to generate long-term outcomes for the comparators. For the base case, the log-normal distribution was considered the most clinically plausible by experts, and had the lowest AIC/BIC (ESM Table S3). Extrapolated survival curves are presented in ESM Fig. S1a.
2.4 Time to Discontinuation
Parametric survival curves for TTD were generated for T-DXd (see ESM Table S3 for model diagnostics). Of the six extrapolations, two groups were present; one implies a proportion of patients would remain on treatment beyond 5 years, and the other that all patients would discontinue after 5 years.Clinical experts confirmed some patients would continue beyond 5 years, but it was not clear which group was more representative; thus, the exponential was selected for the base case as it represented an approximate mid-point between the two groups. No KM data for TTD was published for comparator studies, so treatment to progression was assumed in the base case. Extrapolated survival curves are presented in ESM Fig. S1b.
2.5 Derivation of Utilities
2.5.1 Health State Utilities
Health state utilities were derived using a similar approach to that described in NICE TA423 (investigating eribulin in u/mBC) [10]. In TA423, progression-free, on-treatment utility values were calculated as a function of ORR (patients experience a best overall response of complete response or partial response) and AE rates from the eribulin and treatment of physician’s choice (TPC) arms of the EMBRACE trial [17]. In the current analysis, AE disutilities are modelled in the first cycle only; health state utility values therefore incorporate response only and AE disutilities are modelled separately. The ORR from DESTINY-Breast01 (61.4%) was used for T-DXd [32], and ORR values generated from unanchored MAICs (ESM Table S2) were used for each comparator, resulting in a higher on-treatment utility value for T-DXd versus the comparators (ESM Table S4). A scenario analysis was considered in which the progression-free, on-treatment utility value for T-DXd was assumed equal to that for eribulin.
The progression-free, off-treatment utility values were assumed to be equal to the baseline utility values presented in TA423 (0.704) [10]. The progressed disease utility value for T-DXd was aligned with the committee comments from TA423 [10] (taking the average of the company and evidence review group [ERG] values for progressed disease; 0.588); scenario analyses considered the company (0.679), and ERG (0.496) values. An additional scenario considered progression-free and progressed utility values from Le et al. [38].
2.5.2 Adverse Event Disutilities
AEs of Grade 3 or 4 occurring in ≥ 5% of patients were included for T-DXd and the comparators from respective studies. AEs of special interest in DESTINY-Breast01 or deemed of clinical importance by clinicians were also included in the model. The AE inputs are presented in ESM Table S5–Table S7.
For simplicity, the impact of AEs on health-related quality of life (HRQoL) is captured as a one-off QALY loss in the first cycle of the model. For each treatment, this was calculated using the AE frequencies from relevant studies, the duration of each AE reported in DESTINY-Breast01, and disutilities sourced from the literature.
2.5.3 General Population Utility Multiplier
Age-specific utility multipliers were derived based on the relationship between age and utility values observed in the general population [39]. Health state utilities described previously were assumed to apply at the start of the model; for every year after this, a multiplier was applied based on the ratio between the general population utilities for current age and starting age. The baseline starting age in the model was 56 years, to align with DESTINY-Breast01.
3 Results
3.1 Base-Case Analysis
In the base case, OS was predicted to be substantially longer for patients receiving T-DXd, with a mean/median of 44.7/32.9 months (applying an HR to the T-DM1 OS curve), and 47.7/29.9 months (using direct extrapolation) versus 11.3/9.2 months for eribulin and 17.8/13.6 months for capecitabine (Table 2). T-DXd is associated with 3.3 (applying an HR to the T-DM1 OS curve) and 3.4 (using direct extrapolation) discounted LYs versus 0.9 for eribulin and 1.4 for capecitabine, translating to 2.3, 2.3, 0.6, and 0.9 discounted QALYs, respectively.
3.2 Scenario Analyses
Scenario analyses were performed with reference to each of the alternative modelling approaches: (1) applying an HR to the T-DM1 OS curve, and (2) direct extrapolation of T-DXd OS (results presented in Table 3).
Scenario analysis using the exponential distribution for extrapolation of T-DXd OS using the first modelling method predicted longer mean OS versus the base-case distribution for that approach (generalized gamma) (Table 3). The different distributions selected for extrapolation of T-DXd OS using the second modelling method predicted a similar mean OS versus the base-case distribution, except for the Weibull, which resulted in slightly shorter mean OS.
Scenarios considering data from Barni et al. [15] or Fumoleau et al. [20] resulted in longer mean OS for eribulin and shorter mean OS for capecitabine, respectively, versus the base case. In scenarios where the progression-free, on-treatment utility value for T-DXd was assumed equal to that for eribulin, the progressed utility value was aligned with either the ERG or company approach from TA423, or all utility values were aligned with Le et al. [38], there was minimal impact on the QALYs versus the base case. In all considered scenarios, T-DXd was associated with substantial gains in life expectancy and QALYs versus eribulin, and capecitabine.
4 Discussion
In the current study, modelling long-term survival with T-DXd, either by applying an HR to the T-DM1 OS curve or using direct extrapolation, predicts mean OS for patients receiving T-DXd to be significantly longer versus the model comparators (approximately 4.0/4.2 times longer than eribulin, respectively, and approximately 2.5/2.7 times longer than capecitabine, respectively; comparison with vinorelbine was not possible). T-DXd is associated with greater discounted LYs versus eribulin, and capecitabine, and a substantial discounted QALY gain. In addition, scenario analyses showed that the results are relatively robust to alternative assumptions.
Eribulin, capecitabine, and vinorelbine were selected as the model comparators, as these were the only UK-approved therapies for patients who had received two or more prior anti-HER2 therapies at the time of the analysis and submission to NICE. Since the reimbursement of T-DXd through the CDF, tucatinib with trastuzumab and capecitabine has now also been approved for the treatment of patients with HER2+ advanced BC at third line [40]. With new therapies emerging to help address the unmet need for therapies for patients with HER2+ u/mBC who have received two prior anti-HER2-based regimens, the modelling methods presented in this study may help to inform future health technology appraisals for other therapies with immature OS data.
The strengths of this analysis were that key model components were informed and validated by clinical experts with specialized knowledge of u/mBC. The MAICs, which informed PFS and ORR for the comparators, were conducted following NICE guidance for such analyses, and with all available confounding factors adjusted for in the comparisons. Key limitations included the fact that all patients in DESTINY-Breast01 had HER2+ disease, while the eribulin study used in the base case had a population with mixed HER2 status. Adjustments were made in the model to try to account for this. In addition, long-term extrapolation of vinorelbine OS was not possible; however, given that vinorelbine is associated with a similar PFS to capecitabine, we would expect OS results to be similar to those for capecitabine. Due to the current uncertainties, T-DXd has been approved for use within the CDF [33], rather than routine NHS commissioning, until further clinical data becomes available. Future research will include the analysis of subsequently published datasets, including the March 2021 DESTINY-Breast01 data-cut (median follow-up 26.5 months) [41]. The modelled medians presented here are consistent with the median OS from the latest available data-cut (29.1 months [March 2021 data-cut], versus 32.9 [applying an HR to the T-DM1 OS curve] and 29.9 [modelling based on direct extrapolation]).
In future, comparative data from the DESTINY-Breast02 study can be used to validate the model results. This is an ongoing phase III, multicenter, randomized, open-label, active controlled study in patients with u/mBC who have previously received T-DM1, investigating T-DXd as third-line therapy versus TPC (either of the anti-HER2 therapies trastuzumab or lapatinib, both in combination with capecitabine) [42].
5 Conclusions
This study presents methodology to overcome a lack of direct comparative efficacy data and mature OS data when evaluating LY and QALY changes for new therapies. This is the first analysis modelling long-term outcomes for T-DXd. Modelled results show that T-DXd is associated with significant gains in life expectancy, with a substantial discounted QALY gain, versus eribulin, and capecitabine; vinorelbine was not analyzed due to lack of appropriate data. The alternative methods (applying an HR to T-DM1 data versus direct extrapolation) used for modelling survival produce consistent results, with a similar median OS compared with the latest available March 2021 DESTINY-Breast01 data-cut. T-DXd helps to address the substantial unmet need for targeted third-line therapies with proven efficacy and tolerability in patients with HER2+ u/mBC and is expected to be a life-extending treatment for these patients. The modelling approach described informed a successful cost-effectiveness analysis versus previously UK-recommended non-targeted chemotherapies.
References
Cancer Research UK. Breast cancer incidence by sex and country (UK 2016–2018). 2021. https://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/breast-cancer/incidence-invasive#heading-Zero. Accessed 14 June 2022.
Cancer Research UK. Proportion of cancer cases diagnosed by stage (stacked chart). 2022. https://crukcancerintelligence.shinyapps.io/EarlyDiagnosis/. Accessed 14 June 2022.
Cardoso F, Senkus E, Costa A, Papadopoulos E, Aapro M, André F, et al. 4th ESO–ESMO international consensus guidelines for advanced breast cancer (ABC 4). Ann Oncol. 2018;29(8):1634–57.
Rakha EA, Pinder SE, Bartlett JM, Ibrahim M, Starczynski J, Carder PJ, et al. Updated UK Recommendations for HER2 assessment in breast cancer. J Clin Pathol. 2015;68(2):93–9.
Esteva FJ, Guo H, Zhang S, Santa-Maria C, Stone S, Lanchbury JS, et al. PTEN, PIK3CA, p-AKT, and p-p70S6K status: association with trastuzumab response and survival in patients with HER2-positive metastatic breast cancer. Am J Pathol. 2010;177(4):1647–56. https://doi.org/10.2353/ajpath.2010.090885.
Archer S, Eliopoulos A, Spandidos D, Barnes D, Ellis I, Blamey R, et al. Expression of ras p21, p53 and c-erbB-2 in advanced breast cancer and response to first line hormonal therapy. Br J Cancer. 1995;72(5):1259.
Tovey SM, Brown S, Doughty JC, Mallon EA, Cooke TG, Edwards J. Poor survival outcomes in HER2-positive breast cancer patients with low-grade, node-negative tumours. Br J Cancer. 2009;100(5):680–3. https://doi.org/10.1038/sj.bjc.6604940.
Cancer Research UK. Breast cancer mortality by sex and UK country. 2018. https://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/breast-cancer/mortality#heading-Zero. Accessed 14 June 2022.
The Transforming Cancer Services Team in partnership with Macmillan Cancer Support and NCRAS. Cancer Prevalence in England—21 year prevalence by demographic and geographic measures. Excel workbook. 2018. http://www.ncin.org.uk/about_ncin/releases. Accessed 14 June 2022.
National Institute for Health and Care Excellence (NICE). Eribulin for treating locally advanced or metastatic breast cancer after two or more prior chemotherapy regimens [ID964]. Committee papers. 2016. https://www.nice.org.uk/guidance/ta423/documents/committee-papers. Accessed 14 June 2022.
Roth JA, Mahtani R. Survival gains from advances in first-line systemic therapy for HER2-positive metastatic breast cancer in the US, 1995–2015. Ann Oncol. 2017;28(5):V85. https://doi.org/10.1093/annonc/mdx365.
Pernas S, Tolaney SM. HER2-positive breast cancer: new therapeutic frontiers and overcoming resistance. Ther Adv Med Oncol. 2019;11:1758835919833519. https://doi.org/10.1177/1758835919833519.
National Institute for Health and Care Excellence (NICE). TA423. Eribulin for treating locally advanced or metastatic breast cancer after 2 or more chemotherapy regimens. 2016. https://www.nice.org.uk/guidance/TA423. Accessed 14 June 2022.
National Institute for Health and Care Excellence (NICE). CG81. Advanced breast cancer: diagnosis and treatment. 2017. https://www.nice.org.uk/guidance/cg81/resources/advanced-breast-cancer-diagnosis-and-treatment-pdf-975683850181. Accessed 14 June 2022.
Barni S, Livraghi L, Morritti M, Vici P, Michelotti A, Cinieri S, et al. Eribulin in the treatment of advanced breast cancer: real-world scenario from 39 Italian centers–ESEMPiO study. Future Oncol. 2019;15(1):33–44.
Cortes J, Vahdat L, Blum JL, Twelves C, Campone M, Roché H, et al. Phase II study of the halichondrin B analog eribulin mesylate in patients with locally advanced or metastatic breast cancer previously treated with an anthracycline, a taxane, and capecitabine. J Clin Oncol. 2010;28(25):3922–8. https://doi.org/10.1200/jco.2009.25.8467.
Cortes J, O’Shaughnessy J, Loesch D, Blum JL, Vahdat LT, Petrakova K, et al. Eribulin monotherapy versus treatment of physician’s choice in patients with metastatic breast cancer (EMBRACE): a phase 3 open-label randomised study. Lancet. 2011;377(9769):914–23. https://doi.org/10.1016/s0140-6736(11)60070-6.
Gamucci T, Michelotti A, Pizzuti L, Mentuccia L, Landucci E, Sperduti I, et al. Eribulin mesylate in pretreated breast cancer patients: a multicenter retrospective observational study. J Cancer. 2014;5(5):320–7. https://doi.org/10.7150/jca.8748.
Blum JL, Dieras V, Lo Russo PM, Horton J, Rutman O, Buzdar A, et al. Multicenter, phase II study of capecitabine in taxane-pretreated metastatic breast carcinoma patients. Cancer. 2001;92(7):1759–68. https://doi.org/10.1002/1097-0142(20011001)92:7%3c1759::aid-cncr1691%3e3.0.co;2-a.
Fumoleau P, Largillier R, Clippe C, Dièras V, Orfeuvre H, Lesimple T, et al. Multicentre, phase II study evaluating capecitabine monotherapy in patients with anthracycline- and taxane-pretreated metastatic breast cancer. Eur J Cancer. 2004;40(4):536–42. https://doi.org/10.1016/j.ejca.2003.11.007.
Geyer CE, Forster J, Lindquist D, Chan S, Romieu CG, Pienkowski T, et al. Lapatinib plus capecitabine for HER2-positive advanced breast cancer. N Engl J Med. 2006;355(26):2733–43. https://doi.org/10.1056/NEJMoa064320.
Cameron D, Casey M, Press M, Lindquist D, Pienkowski T, Romieu CG, et al. A phase III randomized comparison of lapatinib plus capecitabine versus capecitabine alone in women with advanced breast cancer that has progressed on trastuzumab: updated efficacy and biomarker analyses. Breast Cancer Res Treat. 2008;112(3):533–43. https://doi.org/10.1007/s10549-007-9885-0.
Cameron D, Casey M, Oliva C, Newstat B, Imwalle B, Geyer CE. Lapatinib plus capecitabine in women with HER-2-positive advanced breast cancer: final survival analysis of a phase III randomized trial. Oncologist. 2010;15(9):924.
Sim SH, Park IH, Jung KH, Kim S-B, Ahn J-H, Lee K-H, et al. Randomised phase 2 study of lapatinib and vinorelbine vs vinorelbine in patients with HER2+ metastatic breast cancer after lapatinib and trastuzumab treatment (KCSG BR11-16). Br J Cancer. 2019;121(12):985–90.
European Medicines Agency (EMA). Enhertu, trastuzumab deruxtecan. 2020. https://www.ema.europa.eu/en/medicines/human/summaries-opinion/enhertu. Accessed 14 June 2022.
Medicines and Healthcare Products Regulatory Agency (MHRA). Summary of product characteristics. Enhertu 100 mg powder. 2022. https://mhraproducts4853.blob.core.windows.net/docs/e35c7ffebbb6b8fe24de65fa715f53d779836754. Accessed 14 June 2022.
Nakada T, Sugihara K, Jikoh T, Abe Y, Agatsuma T. The latest research and development into the antibody–drug conjugate, [fam-] trastuzumab deruxtecan (DS-8201a), for HER2 cancer therapy. Chem Pharm Bull. 2019;67(3):173–85.
Yver A, Agatsuma T, Soria JC. The art of innovation: clinical development of trastuzumab deruxtecan and redefining how antibody-drug conjugates target HER2-positive cancers. Ann Oncol. 2020;31(3):430–4. https://doi.org/10.1016/j.annonc.2019.11.019.
Ogitani Y, Aida T, Hagihara K, Yamaguchi J, Ishii C, Harada N, et al. DS-8201a, a novel HER2-targeting ADC with a novel DNA topoisomerase I inhibitor, demonstrates a promising antitumor efficacy with differentiation from T-DM1. Clin Cancer Res. 2016;22(20):5097–108. https://doi.org/10.1158/1078-0432.CCR-15-2822.
Trail PA, Dubowchik GM, Lowinger TB. Antibody drug conjugates for treatment of breast cancer: novel targets and diverse approaches in ADC design. Pharmacol Ther. 2018;181:126–42. https://doi.org/10.1016/j.pharmthera.2017.07.013.
Modi S, Saura C, Yamashita T, Park YH, Kim SB, Tamura K, et al. Trastuzumab deruxtecan in previously treated HER2-positive breast cancer. N Engl J Med. 2020;382(7):610–21. https://doi.org/10.1056/NEJMoa1914510.
Modi S, Saura C, Yamashita T, Park H, Kim S-B, Tamura K, et al. PD3-06: updated results from DESTINY-breast01, a phase 2 trial of trastuzumab deruxtecan (T-DXd) in HER2 positive metastatic breast cancer. Cancer Res. 2021;81(4_Supplement):PD3-06.
National Institute for Health and Care Excellence (NICE). Trastuzumab deruxtecan for treating HER2-positive unresectable or metastatic breast cancer after 2 or more anti-HER2 therapies (TA704). 2021. https://www.nice.org.uk/guidance/TA704. Accessed 14 June 2022.
National Institute for Health and Care Excellence (NICE). NICE draft guidance provides Cancer Drugs Fund first in Europe access to new treatment option for advanced breast cancer 2021. https://www.nice.org.uk/news/article/nice-draft-guidance-provides-cancer-drugs-fund-first-in-europe-access-to-new-treatment-option-for-advanced-breast-cancer. Accessed 14 June 2022.
Lv S, Wang Y, Sun T, Wan D, Sheng L, Li W, et al. Overall survival benefit from trastuzumab-based treatment in HER2-positive metastatic breast cancer: a retrospective analysis. Oncol Res Treat. 2018;41(7–8):450–5.
Krop IE, Kim SB, Martin AG, LoRusso PM, Ferrero JM, Badovinac-Crnjevic T, et al. Trastuzumab emtansine versus treatment of physician’s choice in patients with previously treated HER2-positive metastatic breast cancer (TH3RESA): final overall survival results from a randomised open-label phase 3 trial. Lancet Oncol. 2017;18(6):743–54. https://doi.org/10.1016/S1470-2045(17)30313-3.
Office for National Statistics. National life tables: England and Wales. 2019. https://www.ons.gov.uk/peoplepopulationandcommunity/birthsdeathsandmarriages/lifeexpectancies/datasets/nationallifetablesenglandandwalesreferencetables. Accessed 14 June 2022.
Le QA, Bae YH, Kang JH. Cost-effectiveness analysis of trastuzumab emtansine (T-DM1) in human epidermal growth factor receptor 2 (HER2): positive advanced breast cancer. Breast Cancer Res Treat. 2016;159(3):565–73. https://doi.org/10.1007/s10549-016-3958-x.
Ara R, Brazier JE. Populating an economic model with health state utility values: moving toward better practice. Value Health. 2010;13(5):509–18. https://doi.org/10.1111/j.1524-4733.2010.00700.x.
National Institute for Health and Care Excellence (NICE). TA786. Tucatinib with trastuzumab and capecitabine for treating HER2-positive advanced breast cancer after 2 or more anti-HER2 therapies. 2022. https://www.nice.org.uk/guidance/ta786. Accessed 14 Sept 2022.
Manich CS, Modi S, Krop I, Park YH, Kim S, Tamura K, et al. Trastuzumab deruxtecan (T-DXd) in patients with HER2-positive metastatic breast cancer (MBC): updated survival results from a phase II trial (DESTINY-Breast01). Ann Oncol. 2021;32(SUPPLEMENT 5):S485–6.
Clinicaltrials.gov. DS-8201a in pre-treated HER2 breast cancer that cannot be surgically removed or has spread [DESTINY-Breast02] (NCT03523585). 2020. https://clinicaltrials.gov/ct2/show/NCT03523585. Accessed 14 June 2022.
Acknowledgements
The authors would like to acknowledge Keyur Patel and Safa McLachlan of Daiichi Sankyo UK Ltd for input into the methods applied, Dr. Michelle Orme of ICERA Consulting Ltd on behalf of Source Health Economics for support with statistical analyses, and Dr. Emma Lones of Source Health Economics for medical writing and editorial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by Daiichi Sankyo Europe GmbH.
Conflict of interest
AP received consultancy fees for the statistical analyses presented in this manuscript. EH, MP, and OB are employees of Source Health Economics, which received consultancy fees for the extrapolation of long-term outcomes presented in this manuscript.
Ethics approval
Not required.
Consent to participate
Not required.
Consent for publication
Not applicable.
Availability of data and material
No additional data or materials are available.
Code availability
No code is available.
Author contributions
All authors contributed to the study conception and design. Statistical analyses were performed by AP. Extrapolation of long-term outcomes were performed by EH, MP, and OB. All authors commented on the manuscript, and read and approved the final version of the manuscript.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Dunton, K., Vondeling, G., Hancock, E. et al. Methods for Estimating Long-Term Outcomes for Trastuzumab Deruxtecan in HER2-Positive Unresectable or Metastatic Breast Cancer After Two or More Anti-HER2 Therapies. Targ Oncol 17, 655–663 (2022). https://doi.org/10.1007/s11523-022-00923-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11523-022-00923-9