Abstract
Bovine spongiform encephalopathy (BSE), which has been documented in 190,000 cases of BSE-infected cattle, to our knowledge, has not been reported in buffalo. Prion protein (PrP) is critical to susceptibility and development of BSE. Moreover, a new PrP-like protein, Shadoo (Sho), has been shown to have overlapping expression patterns and shared functions with PrP. Therefore, we hypothesize that differences in expression at the transcriptional level and/or the post-transcriptional level of the two genes may be associated with these specific differences between cattle and buffalo. We compared the relative mRNA expression of the prion protein gene (PRNP) and the Shadoo gene (SPRN) in 396 RNA samples using real-time PCR. We also analyzed PrP/Sho protein from 96 samples by Western blot. Our results demonstrated significantly lower PrP expression in the cerebellum, obex, mesenteric lymph node, and bronchial lymph node tissues, but higher relative expression of Sho in the cerebrum and spleen in buffalos compared with cattles. Although these results support our primary assumption, Sho and PrP expressions did not correlate with corresponding mRNA expression, suggesting that the biological modulations of both PrP and Sho proteins are at post-translational levels. Moreover, positive correlations between PRNP and SPRN were found in cattle and buffalo cerebrum as well as in buffalo obex. Conversely, negative correlations between PrP and Sho were detected in buffalo cerebellum and obex tissues. These findings suggest that additional post-transcriptional studies are warranted to elucidate mechanisms behind prion diseases.
摘要
全球有超过 19 万头黄牛被感染疯牛病, 却没有水牛患病的报道。 本研究通过分析与疯牛病关联的PRNP 和SPRN 基因转录水平及其蛋白 (PrP 和 Sho)水平的表达, 揭示了水牛小脑、脑闩、肠系膜淋巴结和支气管淋巴结 PrP 的表达量显著低于黄牛, 而水牛大脑和脾 Sho 表达量显著高于黄牛。然而 PrP 和 Sho 表达与它们相应的 mRNA 表达没有呈现出相同 趋势, 说明这两个蛋白的表达受到了转录后的调控作用。另外, 在水牛和黄牛的大脑和脑闩中, PRNP 和 SPRN mRNA 的表达呈正相关, 而水牛的小脑和脑闩组织中 PrP 和 Sho 蛋白的表达呈负相关, 这些结果提示转录后表达水平的研究在朊病毒疾病研究中的重要性。


Similar content being viewed by others
References
Prusiner SB (1998) Prions. Proc Natl Acad Sci USA 95:13363–13383
Brandner S, Raeber A, Sailer A et al (1996) Normal host prion protein (PrPC) is required for scrapie spread within the central nervous system. Proc Natl Acad Sci USA 93:13148–13151
Westaway D, Mirenda CA, Foster D et al (1991) Paradoxical shortening of scrapie incubation times by expression of prion protein transgenes derived from long incubation period mice. Neuron 7:59–68
Daude N, Westaway D (2011) Biological properties of the PrP-like Shadoo protein. Front Biosci 16:1505–1516
Wang SQ, Zhao H, Zhang YP (2014) Advances in research on Shadoo, shadow of prion protein. Chin Sci Bull 59:821–827
Premzl M, Sangiorgio L, Strumbo B et al (2003) Shadoo, a new protein highly conserved from fish to mammals and with similarity to prion protein. Gene 314:89–102
Watts JC, Drisaldi B, Ng V et al (2007) The CNS glycoprotein Shadoo has PrPC-like protective properties and displays reduced levels in prion infections. EMBO J 26:4038–4050
Lampo E, Van Poucke M, Vandesompele J et al (2009) Positive correlation between relative mRNA expression of PRNP and SPRN in cerebral and cerebellar cortex of sheep. Mol Cell Probes 23:60–64
Collinge J, Sidle KCL, Meads J et al (1996) Molecular analysis of prion strain variation and the aetiology of ‘new variant’ CJD. Nature 383:685–690
Guardo GD (2014) BSE in buffaloes. Vet Rec 174:485
Qing LL, Zhao H, Liu LL (2014) Progress on low susceptibility mechanisms of transmissible spongiform encephalopathies. Dongwuxue Yanjiu 35:436–445 (In Chinese)
Zhao H, Liu LL, Du SH et al (2012) Comparative analysis of the Shadoo gene between cattle and buffalo reveals significant differences. PLoS One 7:e46601
Zhao H, Du YL, Chen SM et al (2015) The prion protein gene polymorphisms associated with bovine spongiform encephalopathy susceptibility differ significantly between cattle and buffalo. Infect Genet Evol 36:531–538
Imran M, Mahmood S, Babar ME et al (2012) PRNP gene variation in Pakistani cattle and buffaloes. Gene 505:180–185
Maignien T, Lasmézas CI, Beringue V et al (1999) Pathogenesis of the oral route of infection of mice with scrapie and bovine spongiform encephalopathy agents. J Gen Virol 80:3035–3042
Prusiner SB, Bolton DC, Groth DF et al (1982) Further purification and characterization of scrapie prions. Biochemistry 21:6942–6950
Farquhar CF, Dornan J, Somerville RA et al (1994) Effect of Sinc genotype, agent isolate and route of infection on the accumulation of protease-resistant PrP in non-central nervous system tissues during the development of murine scrapie. J Gen Virol 75:495–504
Sander P, Hamann H, Drogemuller C et al (2005) Bovine prion protein gene (PRNP) promoter polymorphisms modulate PRNP expression and may be responsible for differences in bovine spongiform encephalopathy susceptibility. J Biol Chem 280:37408–37414
Uboldi C, Paulis M, Guidi E et al (2006) Cloning of the bovine prion-like Shadoo (SPRN) gene by comparative analysis of the predicted genomic locus. Mamm Genome 17:1130–1139
Haase B, Doherr MG, Seuberlich T et al (2007) PRNP promoter polymorphisms are associated with BSE susceptibility in Swiss and German cattle. BMC Genet 8:15
Juling K, Schwarzenbacher H, Williams JL et al (2006) A major genetic component of BSE susceptibility. BMC Biol 4:33
Kashkevich K, Humeny A, Ziegler U et al (2007) Functional relevance of DNA polymorphisms within the promoter region of the prion protein gene and their association to BSE infection. FASEB J 21:1547–1555
Msalya G, Shimogiri T, Ohno S et al (2011) Evaluation of PRNP expression based on genotypes and alleles of two indel loci in the medulla oblongata of Japanese Black and Japanese Brown cattle. PLoS One 6:e18787
Zhao H, Wang XY, Zou W et al (2010) Prion protein gene (PRNP) polymorphisms in native Chinese cattle. Genome 53:138–145
Yedidia Y, Horonchik L, Tzaban S et al (2001) Proteasomes and ubiquitin are involved in the turnover of the wild-type prion protein. EMBO J 20:5383–5391
Yoshioka Y, Ishiguro N, Inoshima Y (2010) Proteasome activity and biological properties of normal prion protein: a comparison between young and aged cattle. J Vet Med Sci 72:1583–1587
Ma J, Lindquist S (2002) Conversion of PrP to a self-perpetuating PrPSc-like conformation in the cytosol. Science 298:1785–1788
Gossner AG, Bennet N, Hunter N et al (2009) Differential expression of Prnp and Sprn in scrapie infected sheep also reveals Prnp genotype specific differences. Biochem Biophys Res Commun 378:862–866
Watts JC, Stöhr J, Bhardwaj S et al (2011) Protease-resistant prions selectively decrease Shadoo protein. PLoS Pathog 7:e1002382
Westaway D, Genovesi S, Daude N et al (2011) Down-regulation of Shadoo in prion infections traces a pre-clinical event inversely related to PrPSc accumulation. PLoS Pathog 7:e1002391
Bartel DP (2004) MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116:281–297
He L, Hannon GJ (2004) MicroRNAs: small RNAs with a big role in gene regulation. Nat Rev Genet 5:522–531
Peletto S, Bertolini S, Maniaci MG et al (2012) Association of an indel polymorphism in the 3′ UTR of the caprine SPRN gene with scrapie positivity in the central nervous system. J Gen Virol 93:1620–1623
Wan JY, Hao Z, Xu M et al (2010) Mapping the interaction site of prion protein and Sho. Mol Biol Rep 37:2295–3300
Cric D, Richard CA, Moudjou M et al (2015) Interaction between Shadoo and PrP affects the PrP-folding pathway. J Virol 89:6287–6293
Acknowledgments
This work was supported by the National Natural Science Foundation of China (31060302, 31260032 and 31460038), and the Program for Innovative Research Team (in Science and Technology) in the University of Yunnan Province.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Hui Zhao and Si-Qi Wang contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11434_2016_1130_MOESM1_ESM.pdf
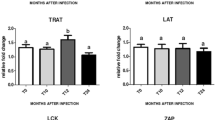
Figure S1. Relative expression of PRNP in cattle and buffalo. RNA was isolated from cerebrum (a), cerebellum (b), obex (c), mesenteric lymph node (d), bronchial lymph node (e), and spleen (f) of 34 cattle and 32 buffalo. Transcript quantities were analyzed by qPCR. The vertical ordinates show the relative expression of PRNP normalized to ACTB. The mean values of each species are indicated by horizontal lines. * Indicates significant differences for PRNP expression between cattle and buffalo. (PDF 27 kb)
11434_2016_1130_MOESM2_ESM.pdf
Figure S2. Relative expression of SPRN in cattle and buffalo. RNA was isolated from cerebrum (a), cerebellum (b), obex (c), mesenteric lymph node (d), bronchial lymph node (e), and spleen (f) of 34 cattles and 32 buffaloes. The vertical ordinates depict relative expression of SPRN normalized to ACTB. The mean values of each species are indicated by horizontal lines. * Indicates significant differences for SPRN expression between cattles and buffalos. (PDF 23 kb)
11434_2016_1130_MOESM3_ESM.png
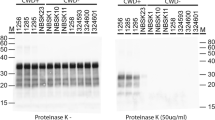
Figure S3. The expression levels of PrP and Shadoo proteins. Six tissues, including cerebrum (a), cerebellum (b), obex (c), MLN (d), BLN (e) and spleen (f), from 8 cattles and 8 buffaloes were prepared and then immunoblotted for PrP and Sho proteins. (PNG 276 kb)
About this article
Cite this article
Zhao, H., Wang, SQ., Qing, LL. et al. Expression of BSE-associated proteins in the CNS and lymphoreticular tissues of cattle and buffalo. Sci. Bull. 61, 1377–1383 (2016). https://doi.org/10.1007/s11434-016-1130-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-016-1130-y