Abstract
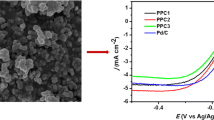
In this paper, we synthesized cathode catalysts (PANI-PPYR, Fe/PANI-PPYR, Co/PANI-PPYR and Fe-Co/PANI-PPYR) with high performance oxygen reduction by using a simple heat treatment process. These catalysts were fabricated by directly calcining the Fe and/or Co doped polyaniline (PANI)-polypyrrole (PPYR) composites. Their electrocatalytic activity for ORR both in acidic and in alkaline media was investigated by voltammetric techniques. Among the prepared catalysts, Co/PANI-PPYR presents the most positive ORR onset potential of 0.62 V (vs. SCE) in 0.5 mol/L H2SO4 solution or −0.09 V (vs. SCE) in 1 mol/L NaOH solution. In addition, the Co/PANI-PPYR catalyst shows the largest limiting-diffusion current density for ORR, which is 4.3 mA/cm2@0.2 V (vs. SCE) in acidic and 2.3 mA/cm2@−0.3 V (vs. SCE) in alkaline media. In acidic media, a four-electron reaction of ORR on the Co/PANI-PPYR and Fe/PANI-PPYR catalysts is more dominant than a two-electron reaction. In alkaline media, however, a four-electron and a two-electron mechanisms are co-present for the ORR on all the prepared catalysts. Co/PANI-PPYR catalyst also presents good electrocatalytic activity stability for ORR both in acidic and in alkaline media.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Jasinski R. A new fuel cell cathode catalyst. Nature, 1964, 201: 1212–1213
Bashyam R, Zelenay P. A class of non-precious metal composite catalysts. Nature, 2006, 443: 63–66
Egli M, Pallan PS. Insights from crystallographic studies into the structural and pairing properties of nucleic acid analogs and chemically modified DNA and RNA oligonucleotides. Annu Rev Biophys Biomol Struct, 2007, 36: 281–305
Seeliger W, Hamnett A. Novel electrocatalysts for oxygen reduction. Electrochim Acta, 1992, 37: 763–765
Jahnke H, Schonbron M, Zimmerman G. Organic Dyestuffs as Catalysts for Fuel Cells, Physical and Chemical Applications of Dyestuffs. Topics in Current Chemistry. Berlin: Springer Berlin/Heidelberg 1976. 165–168
Zhong H, Zhang H, Liu G, Liang Y, Hu J, Yi B. A novel non-noble electrocatalyst for PEM fuel cell based on molybdenum nitride. Electrochem Commun, 2006, 8: 707–712
Ishihara A, Lee K, Doi S, Mitsushima S, Kamiya N, Hara M, Domen K, Fukuda K, Ota K-ichiro. Tantalum oxynitride for a novel cathode of PEFC. Electrochem Solid-State Lett, 2005, 8: A201–A203
Liu G, Zhang HM, Wang MR, Zhong HX, Chen J. Preparation characterization of ZrOxNy-C and its application in PEMFC as an electrocatalyst for oxygen reduction. J Power Sources, 2007, 172: 503–510
Biddinger EJ, Ozkan US. Methanol tolerance of CNx oxygen reduction catalysts. Top Catal, 2007, 46: 339–348
Liu G, Li X, Ganesan P, Popov BN. Development of non-preciousmetal oxygen-reduction catalysts for PEM fuel cells based on N-doped ordered porous carbon. Appl Catal B: Environ, 2009, 93: 156–165
Lei M, Li PG, Li LH, Tang WH. A highly ordered Fe-N-C nanoarray as a non-precious oxygen-reduction catalyst for proton exchange membrane fuel cells. J Power Sources, 2011, 196: 3548–3552
Deng L, Zhou M, Liu C, Liu L, Liu C, Dong S. Development of high performance of Co/Fe/N/CNT nanocatalyst for oxygen reduction in microbial fuel cells. Talanta, 2010, 81: 444–448
Byon HR, Suntivich J, Crumlin EJ, Shao-Horn Y. Fe-N-modified multi-walled carbon nanotubes for oxygen reduction reaction in acid. Phys Chem Chem Phys, 2011, 13: 21437–21445
Morozan A, Jegou P, Jousselme B, Palacin S. Electrochemical performance of annealed cobalt-benzotriazole/CNTs catalysts towards the oxygen reduction reaction. Phys Chem Chem Phys, 2011, 13: 21600–21607
Wu G, More KL, Johnston CM, Zelenay P. High-performance electrocatalysts for oxygen reduction derived from polyaniline, iron, and cobalt. Science, 2011, 332: 443–447
Subramanian NP, Kumaraguru SP, Colon-Mercado H, Kim H, Popov BN, Black T, Chen DA. Studies on Co-based catalysts supported on modified carbon substrates for PEMFC cathodes. J Power Sources, 2006, 157: 56–63
Lalande G, Côté R, Guay D, Dodelet JP, Weng LT, Bertrand P. Is nitrogen important in the formulation of Fe-based catalysts for oxygen reduction in solid polymer fuel cells? Electrochim Acta, 1997, 42: 1379–1388
Gojkovic SLJ, Gupta S, Savinell RF. Heat-treated iron (III) tetramethoxyphenyl porphyrin chloride supported on high-area carbon as an electrocatalyst for oxygen reduction. Part II. Kinetics of oxygen reduction. J Electroanal Chem, 1999, 462: 63–72
Faubert G, Côté R, Guay D, Dodelet JP, Dénès G, Bertrandc P. Iron catalysts prepared by high-temperature pyrolysis of tetraphenylporphyrins adsorbed on carbon black for oxygen reduction in polymer electrolyte fuel cells. Electrochim Acta, 1998, 43: 341–353
Lefevre M, Dodelet JP. O2 reduction in PEM fuel cells: Activity and active site structural information for catalysts otained by the pyrolysis at high temperature of Fe precursors. J Phys Chem B, 2000, 104: 11238–11247
Wu G, Nelson M, Ma S, Meng H, Cui G, Shen PK. Synthesis of nitrogen-doped onion-like carbon and its use in carbon-based CoFe binary non-precious-metal catalysts for oxygen-reduction. Carbon, 2011, 49: 3972–3982
Nallathambi V, Lee J-W, Kumaraguru SP, Wu G, Popov BN. Development of high performance carbon composite catalyst for oxygen reduction reaction in PEM proton exchange membrane fuel cells. J Power Sources, 2008, 183: 34–42
Lefèvre M, Proietti E, Jaouen F, Dodelet JP. Iron-based catalysts with improved oxygen reduction activity in polymer electrolyte fuel cells. Science, 2009, 324: 71–74
Pylypenko S, Mukherjee S, Olson TS, Atanassov P. Non-platinum oxygen reduction electrocatalysts based on pyrolyzed transition metal macrocycles. Electrochim Acta, 2008, 53: 7875–7883
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Yi, Q., Zhang, Y., Liu, X. et al. Carbon-supported Fe/Co-N electrocatalysts synthesized through heat treatment of Fe/Co-doped polypyrrole-polyaniline composites for oxygen reduction reaction. Sci. China Chem. 57, 739–747 (2014). https://doi.org/10.1007/s11426-013-5027-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-013-5027-1