Abstract
Pistacia atlantica Desf. (Beneh) is an important woody species that has been facing significant challenges to its natural regeneration and reforestation in Iran. This study investigates the interaction of soil moisture and shade on growth, chemical contents, and morphological and physiological characteristics of Beneh saplings. One-year-old Beneh saplings were treated with varying amounts of soil moisture (20, 50, and 100% of field capacity) and shade (0, 30, and 50% of full sunlight) in a split-plot experiment of a randomized complete block design in semiarid conditions of the Alborz Research Station of the Research Institute of Forests and Rangelands (RIFR) in Iran. The results indicate that soil moisture significantly affects the water content of the leaf, total chlorophyll, proline content, activity of catalase enzyme, leaf dry biomass, leaflet area, and dry stem biomass in the leaf. Shade significantly affected total chlorophyll, catalase enzyme activity, specific leaflet area, relative water content of the leaf, proline content, dry root biomass, and leaflet area. The interaction of shade and soil moisture significantly affected seedling height, catalase enzyme activity, specific leaflet area, and nitrogen and potassium content of the leaf. Shade moderates the stress of drought on Beneh saplings, but shading of Beneh saplings is not recommended in conditions where there is no concern about soil moisture. These conclusions can be used to improve the production of Beneh saplings in nurseries.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pistacia atlantica Desf. (Atlas mastic tree aka Beneh in Iran) is an ecologically adaptable woody species that is found in many environments of southern Europe, the Middle East, and northern Africa. It grows well in several vegetation zones in Iran, as well. It is found, for instance, in Arasbaran (northwestern Iran), Zagros (western Iran), Irano-Turanian (central Iran), and Khalijo-Ommani (southern Iran) (Owji and Hamzepour 2003). Three subspecies of Beneh have been identified in Iran (mutica, kurdica, and cabulica) (Mozaffarian 2010). Many studies have demonstrated its ecological flexibility and tolerance to difficult environmental conditions in Iran. This species forms both pure- and mixed-vegetation assemblages with other tree species depending upon the habitat (Emadi 1996). Therefore, natural forests of this species are found throughout Iran but are particularly common in the western and southern parts of the country (Zahedipour et al. 2005; Heydari 2016). Although it tolerates and adapts to diverse ecological conditions, the natural regeneration of Beneh has become difficult. Its seeds have a low germinative capacity, but there are other intensifying factors that make regeneration difficult as well. For instance, harvesting its fruit and resin, cutting its branches, destruction of protective shrubs in its understory, soil erosion, understory farming, grazing, and changing climates (increasing temperatures and decreasing precipitation), particularly during the growth season (Sohrabi 1995; Emadi 1996) have been reported to limit regeneration rates. As limited seed germination is also hampered by both their destruction (consumed by domesticated animals and rodents) and severely declining soil moisture levels in natural forests (Sabeti 1994; Negahdarsaber and Abbasi 2010), regeneration of Beneh depends on nurse species in the understory, specially wild almonds (particularly Amygdalus lycioides Spach). After Beneh saplings are established, the nurse species gradually weaken and are replaced by Beneh (Hamzepour et al. 2006). Many studies have shown that when Beneh seedlings are protected from herbivores, the Beneh seedlings established in the shade of nurse species are healthier (both quantitatively and qualitatively) than seedlings established in open (i.e., unprotected) areas (Hamzepour et al. 2006; Jahanpour et al. 2010; Negahdarsaber and Abbasi 2010). In fact, many species benefit from shade in arid and semiarid regions, but it is unclear whether improved performance under shade is due to protection against severe sunlight, due to moderation of the drought stress or both. Pilevar et al. (2012) reported that Quercus brantii Lindl. saplings grew better in full sunlight, when soil moisture wasn’t low or limited. Therefore, experimental and nursery studies are needed to better understand the interaction of shade and drought on woody plant species.
Some studies of the effect of drought stress on the characteristics of Beneh seedlings in greenhouses and laboratories have revealed that there are negative impacts from drought on the physiological and morphological characteristics of Beneh (Ranjbar Fordoei et al. 2000; Joulaei-Manesh 2011; Mirzaei 2011). Despite some studies of the effects of drought stress on various species (Aranda et al. 2005; Climent et al. 2006; Sofo et al. 2009; Li et al. 2011; Hernández et al. 2009; Markesteijn and Poorter 2009; Schall et al. 2012; Daniels et al. 2013; Maguire and Kobe 2015; Amissah et al. 2015; Kupers et al. 2019; Abbas et al. 2019), there has been no comprehensive study of the interactive effects of drought and shade on Beneh, particularly in Iran, where natural regeneration has diminished and reforestation has been unsuccessful in recent years.
The questions to be answered by this research are as follows: (1) Does shade decrease drought stress in Beneh saplings? (2) Does full sunlight stress Beneh saplings? (3) Does shade moderate sunlight stress among well-watered saplings? This study investigates the interaction of soil moisture and shade on the growth, chemical contents, morphology, and physiology of Beneh saplings in semiarid regions of Iran. The results can help to better understand the responses of Beneh saplings to both shade and drought and may reveal the best conditions for natural regeneration and reforestation of this species in semiarid regions of the world.
Materials and methods
Study area
The site of this study is the Alborz Research Station of the Research Institute of Forests and Rangelands (RIFR) in Iran, located in Alborz Province at 35°48′ N and 50°54′ E (Fig. 1). The mean elevation of the site is 1300 m. Forty years of climate data indicate that the average annual precipitation is 250 mm, and the absolute minimum, absolute maximum, and average annual temperatures of this region are − 21.77 °C, 41 °C, and 13.7 °C. Based on the DeMarton classification, this region’s climate is semiarid (Calagari et al. 2018). The study was undertaken from June to October. Contemporaneous weather data were collected during that period (Table 1).
Research method
Thirty Beneh seedlings (originating from the Azgi Valley of Karaj) were cultivated at the Alborz Research Station and were transplanted to 30 cm × 35 cm pots in late February. The dimensions of the seedlings, about 10 cm in height and 5 mm in collar diameter, were approximately identical. The soil for the potting was acquired from the Azgi Valley and was added to the pots in equal amounts. Therefore, the soil used in the experiment was typical of the region. The soil characteristics were pre-determined (Table 2). The conditions under which the plants were kept in the nursery did not vary for three months to allow the saplings to be fully established.
In July, the pots were placed in plots of three contrasting soil-moisture levels (or stress) (no stress (100% of field capacity (FC)), moderate stress (50% FC), and severe stress (20% FC)) and three levels of shade (full sunlight (FS), moderate shade (50% FS), and low shade (30% FS). This design was replicated in three locations of ten saplings per plot and one sapling per pot (270 saplings in total). Artificial shades were installed at heights of 70 cm above the ground using two white polyethylene lace filters having different textures that reduce full sunlight to 50% and 30% of full sunlight (FS).
A pot was randomly selected from each plot every morning, and its weight (all pots were had the same weight) was used to calculate the amount of water needed to achieve a target weight (Zolfaghari 2008). In August, 5 g of the leaves of saplings in locations of the four cardinal edges of each plot was collected and transferred to the laboratory freezer (− 80 °C) for physiological measurements (Zolfaghari 2008). The physiological characteristics measured were RWC of the leaf (%) (Boyer 1968), proline content (μg/g of the wet weight of leaf) (Bates et al. 1973), TC (μg/g of the wet weight of leaf) (Lichtenthaler 1987), and catalase enzyme activity (unit/mL) (Beers and Sizer 1952).
Irrigation and moisture treatments continued until mid-November (just prior to the arrival of the first autumn rainfall). To measure the morphology of the leaves at this time, four terminal leaflets on the four sides of the saplings’ crowns were sampled (Fig. 2). Leaflet areas (LAs) were measured using a leaf-area meter (model: Gate House 4cht Aok). Given leaflet weights, the specific leaflet area (SLA) was calculated as an area-to-mass ratio in cm2/g (Xu et al. 2009).
The heights of the saplings were measured at the beginning of November using a digital caliper with 0.01-mm accuracy. All saplings were carefully removed from their containers. The main roots and sub-roots were washed, and the leaves of the saplings were collected. The roots and stems were cut at the root collar, and the biomass variables were estimated in the laboratory. Specific elements (nitrogen (N) and potassium (K)) were measured in the leaves. Root, stem, and leaf samples from saplings of the different plots were placed in an oven at 80 °C for 48 h. From these, the dry weights of the samples were determined. The percentage of N in the organs was measured by titration after distillation with the Kjeldahl device. K content was measured after dry burning and digestion using chloridric acid (Emami 1996).
The Anderson–Darling test was used to evaluate the normal distribution of data of each variable (Bayazidi et al. 2012). RWC of the leaf, LA, and N in the leaf were normalized with the Johnson conversion method (1949), because these variables were not normally distributed. The impact of soil moisture, shade, and their interaction were examined using analysis of variance (ANOVA) based on split plots with randomized complete blocks (Sadat Noori 2005). Post hoc tests using least significant difference (LSD) (Soltani 2010) in MSTAT-C 1.41 software were performed on the variables to determine which were significantly affected by soil moisture and shade.
Results
The results of ANOVA (Table 3) indicate that soil moisture significantly affected RWC of leaves, TC, proline content, catalase enzyme activity, LDB, LA (p < 0.01), and stem dry biomass (SDB) (p < 0.05). Shade significantly impacted TC, catalase enzyme activity, SLA (p < 0.05), leaf RWC, proline content, RDB, and LA (p < 0.05). Shade and soil moisture acting together significantly affected only sapling height, catalase enzyme activity, SLA, and K and N in the leaves (p < 0.05).
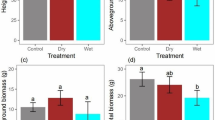
The results of post hoc tests for influences of drought stress on the variables showed that reduced soil moisture decreased the leaf RWC, TC, K, LDB, LA, SDB, and sapling height (Fig. 3). Drought-induced drought stress resistance in Beneh saplings, increasing the proline content and catalase enzyme activity in leaves. Post hoc tests for the effects of shade variation revealed that it moderated the drought stress, increased the leaf RWC, TC, LA, SLA, and decreased proline content, catalase enzyme activity, and RDB (Fig. 3).
Post hoc tests for shade and soil-moisture interaction revealed that together they account for catalase enzyme activity, SLA, and N and K in the levels. Increased moisture decreased the impact of shade on catalase enzyme activity. Despite severely decreased activity in drought-stressed (20% FC) saplings, increased shade caused a lowering of catalase enzyme activity compared to saplings in full sunlight. The highest level of activity of this enzyme was observed in the saplings treated with 20% FC moisture and no shade. The lowest activity of this enzyme was also observed in saplings in FC conditions with any level of shade (Fig. 4).
Increasing shade from FS to 30% generally increased SLA. The level of moisture was important to the effects of increased shade. The saplings in FS and stressed by drought had the greatest SLA, but 50% FS and 30% FS seemed to have no significant effect on SLA, even when the moisture levels changed.
Moisture-treatment impacts on K concentrations in leaves were dependent on shade levels. Increasing shade from FS to 30% FS did not affect K content in drought conditions (20% FC and 50% FC), but K decreased in 100% FC moisture level. Increased shade from 30 to 50% FS caused small changes in K at all soil-moisture conditions. The results were similar for N in leaves, but increasing shade had a greater impact on N in leaves than on K in drought-stressed saplings. The greatest concentrations of N and K were observed in the combination of FC moisture and FS, while the smallest concentrations were observed in 20% FC and FS.
Discussion
This study investigated the interactions of soil moisture and shade on the growth, chemical contents, and morphological and physiological characteristics of Beneh saplings in semiarid regions of Iran.
Effect of soil moisture and shade on physiological characteristics of Beneh saplings
RWC serves as a good index of the water content of a plant cell, and it is a useful guideline for selecting genotypes to survive specific drought conditions (Schonfeld et al. 1988). If water taken by roots is not equal to or greater than the water lost from leaves, stomata that open for photosynthesis will decrease the RWC of the leaves (Basra and Basra 1997). In this study, reduced soil moisture was observed to cause decreased RWC, reaching 68% in drought-stressed plants (20% FC) in FS. This is consistent with the findings of Joulaei-Manesh (2011) and Ben Hamed et al. (2016). Shade helps to maintain soil moisture by decreasing temperature beneath the canopy due to less insolation and lower soil temperature. Shade increased RWC in all experimental plots regardless of soil moisture levels. This was also observed during irrigation of the saplings, as those under shade needed less water to reach the desired target weight.
Reduced soil moisture-induced production of osmolytes, especially proline, to maintain cell osmosis potential and to prevent the cell-wall disjoint. Drought stress broke down and reduced the proteins in mature leaves, increasing free amino acids like proline. Accumulation of proline is compatible with osmosis potential, serving as a substrate for the reconstruction of secondary compositions (Basra and Basra 1997). Other studies (Joulaei-Manesh 2011; Mirzaei 2011) also arrived at this conclusion. Shade decreased proline content of Beneh leaves. This follows the reason as there is less need for osmosis adjustment as leaf RWC increases (Duan et al. 2005).
Reduced soil moisture decreased chlorophyll in Beneh leaves. This positive relationship has already been reported in other studies of Beneh (Ranjbar Fordoei et al. 2000; Mirzaei 2011). Drought stress breaks down chlorophyll and decreases it in the leaves (Anjum et al. 2011). Decreased chlorophyll in drought conditions is a non-stomata limiting factor (Behra et al. 2002). Shade increased chlorophyll in drought-stressed saplings. Increased chlorophyll levels in saplings in FC conditions were primarily due to greater sunlight.
When free radicals, like superoxide, hydrogen peroxide, hydroxyl radical, and singlet oxygen, act more than antioxidants inside cells, they damage cells and textures. These radicals are found in several cell structures: mitochondria, chloroplast, microcosm, and apoplast (Kafi et al. 2012). Antioxidant enzymes fight oxidative stress in older cells and in stressful environmental conditions (Amini and Haddad 2013). Catalase is an antioxidant enzyme found in all living cells. It rapidly breaks down hydrogen peroxide to water and oxygen. Catalase, in fact, uses the hydrogen peroxide as substrate (Goel et al. 2003). In this study, drought stress was found to increase catalase enzyme activity in Beneh saplings. Ranjbar Fordoei et al. (2000) and Mirzaei (2011) also reported this finding in Beneh. The reason for increased enzyme activity is that saplings strive to limit the damages caused by oxygen radicals in a plant under photo-oxidative stress. Shade decreased catalase enzyme activity in the leaves, as it decreased photo-oxidation and the need to remove the oxygen radicals (Duan et al. 2005). This has been reported in other studies using other tree species (Huang et al. 2008; Li et al. 2011).
Effect of soil moisture and shade on growth and morphological characteristics of Beneh saplings
Drought stress severely decreases Beneh saplings’ LA. Decreasing LA is a morphological adaptation of plants exposed to drought stress. More LA enables greater photosynthesis potential, but more LA also increases transpiration. Therefore, to stabilize photosynthesis, there must be a balance between LA and transpiration. Decreased LA is a mechanism of adaptation. Drought stress not only reduces LA, but it also decreases transpiration and loss of water from stomata (Close et al. 2005). Many have reported this relationship between drought stress and LA in other species (Rad et al. 2011; Sapeta et al. 2013) as well as in Beneh (Mirzaei and Karamshahi 2015).
Shade yielded increased LA regardless of moisture treatment levels. Many others have shown that as LA increases, Mediterranean oaks absorb more light under shaded conditions (Aranda et al. 2005; Quero et al. 2006; Hernández et al. 2009). It seems that shade is important to enable cell swelling, and it also increases transpiration rates in drought-stressed saplings (Huang et al. 2009). Despite increasing LA as shade increases, leaflet biomass did not increase due to decreased leaflet thickness. This has been reported by others (Aranda et al. 2005; Quero et al. 2006; Hernández et al. 2009), as well. Increased leaflets allow for shaded leaves to get more sunlight per weight unit (Xu et al. 2009) and minimize the shading of chloroplasts (Quero et al. 2006). The interactive effects of shade and soil moisture were also observed. Saplings receiving moisture closer to FC produce greater SLA under increasing shade due to the production of thicker leaves in FS and greater LA in greater amounts of shade.
With these changes in Beneh saplings, biomass was also affected by drought stress. There was a significant reduction in height, LDB, and SDB of the saplings as soil moisture decreased. However, drought stress did not have a significant effect on RDB. The negative effects of drought stress on plant biomass production have been widely observed. The reason for this is the role of water in the development and division of plant cells and reduced photosynthesis due to the closing of stomata and decreasing of LA (Kafi et al. 2012). One study showed that the dry weight of the aerial organs decreases during drought (Siddique et al. 1993). Insufficient water severely affects leaf development. Reduced leaf development decreases carbon consumption and energy in aerial organs. More drought-assimilated materials are found in roots where the capability develops to absorb more water and more mineral material (Banwarie et al. 1994). This is consistent with the findings of Mirzaei and Karamshahi (2015) in a study of Beneh saplings. This may indicate that Beneh has a high capacity to respond to drought stress with less growth and less aerial organ biomass relative to the root biomass.
Shade decreases RDB and this has been observed previously (Pilevar et al. 2012; Smith and Huston 1989; Climent et al. 2006; Niinemets and Valladares 2006; Huang et al. 2008; Schall et al. 2012; Amissah et al. 2015). This is believed to be the main reason for shade stress. But the responses of the other characteristics seem to show that this change in biomass allocation does not severely affect drought stress in Beneh (at least at the age of these saplings). And, unlike the aforementioned studies, the reduction of root biomass did not cause a reduction of aerial organ biomass.
Effect of soil moisture and shade on chemical element contents of Beneh saplings
Drought stress significantly affected leaf N concentrations. N is either found in minerals or an organic from the plants. N, C, H, O, and, sometimes S, are combined to generate structures for amino acids, amino enzymes, nucleic acids, chlorophylls, alkaloids, and purine bases. Though mineral N can accumulate as nitrate in the stem and becomes a conductive texture of the plants, organic N is a part of most of the heavy protein molecules in plants (Engel 1997). In drought-stressed conditions, when light deterrence decreased photosynthesis in leaves, leaf N is transferred to the stem and roots (Castro-Díez et al. 2006). Because N is important to the structure of chlorophyll, leaves lacking N can turn yellow, and growth rates decrease to nil (Lambers et al. 2008).
K is the most fluent cation in plant cells. It is a factor in water relationships in plants, it adjusts cell acidity, and it influences enzyme activity, protein construction, and photosynthesis (Kafi et al. 2012). A lack of K in leaves can disrupt osmosis, affecting the opening and closing of stomata, disrupting protein construction, reducing enzyme activities, causing yellowing and burning of leaf margins, and decreasing plant growth (Kafi et al. 2012). There was no significant, observable effect of soil-moisture variation on leaf K concentrations. The ability of Beneh saplings to transport K ions from roots to leaves has been reported by Fayyaz et al. (2013). There may be specific channels within which K ions are transported in Beneh (Latorre et al. 2003). Specific proteins like CIPKs adjust the opening and closing of the stomata and absorb the K ions when drought stressed. These proteins may be very important (Cheong et al. 2007) and should be examined in more detail.
Effect of shade-soil moisture interaction on Beneh saplings
Shade-soil moisture interaction had a statistically significant effect on leaf N content. Changes to the amount of shade seemed to not affect the leaf N of saplings treated with 20% FC moisture, but leaf N content did diminish with FC moisture. There was a slight increase of N, when receiving 50% FC from 30% FS to FS. Although increasing shade increased chlorophyll, of which N is a component (Kafi et al. 2012), there was a reduction of photosynthesis capacity of the leaves due to reduced content and activity of rubisco (Lambers et al. 2008) and reduced leaf demand even at FC. Huang et al. (2009) also reported that medium shade (67% FS) severely decreased the maximum photosynthesis rate and N content of two poplar populations in conditions close to FC. These increased in saplings that were experiencing drought stress. Similar effects from shade-soil moisture interaction were observed on leaf K content. Shade negatively affects leaf K content without moisture-stress, producing less K and osmotic pressure adjustment of the stomata (Kafi et al. 2012) and less active stomata in FS. The depressive effects of shade on stomata in light-demanding trees have been reported elsewhere (Castro-Díez et al. 2006; Amissah et al. 2015).
Shade had different effects on sapling height at different soil-moisture levels. Increased shading on saplings in 20% FC treatments increased the height of saplings. This effect diminished as soil moisture increased. Shade actually decreased the height of saplings at FC. The positive effect of shade on height of saplings under stress demonstrates the benefits of shade for decreasing photo-oxidative stress and for increasing photosynthesis in saplings under stress (Abrams et al. 1992; Holmgren 2000; Quero et al. 2006; Sofo et al. 2009; Li et al. 2011). Despite shade’s positive influence on chlorophyll and LA of the FC-treated saplings, photosynthesis decreased because of the lower light conditions, rubisco activity, and rubisco activase enzymes (which are activated by light) (Lambers et al. 2008), and sapling growth decreased. Even in medium shade (50% FS), the height of saplings treated to FC was greater than in lower moisture conditions. Saplings treated with 50% FC were of intermediate height as low shade benefited the plants, but increasing shade did not cause remarkable changes in height.
Low and medium shade moderated the negative impacts of drought stress in Beneh saplings at 20% FC conditions. Low shade increased the growth and production in saplings grown at 50% FC, but shade decreased the growth of saplings that were not drought stressed.
Conclusions
The results of this study showed that shade can help to mitigate the damages caused by drought stress on Beneh saplings. But full sunlight is not suitable for well-watered saplings. This may partly explain the behavior of Beneh saplings when sheltered in the shade of nurse plants. Due to the problems of seedling production in arid and semiarid regions, the use of artificial shade in warm seasons can greatly reduce irrigation costs and increase the quality of seedlings in nurseries. In order to comment more precisely on the effect of shade on the natural regeneration of Beneh, it is necessary to conduct additional in situ studies of the natural habitats of this species.
Availability of data and materials
The datasets generated and/or analyzed during the current study are not publicly available [because this data is the results of the author’s efforts and studies], but are available from the corresponding author on reasonable request.
Abbreviations
- ANOVA:
-
Analysis of variance
- FC:
-
Field capacity
- FS:
-
Full sunlight
- LA:
-
Leaflet area
- LDB:
-
Leaf dry biomass
- RDB:
-
Root dry biomass
- RWC:
-
Relative water content
- SDB:
-
Stem dry biomass
- SLA:
-
Specific leaflet area
References
Abbas AM, Rubio-Casal AE, De Cires A, Grewell BJ, Castillo JM (2019) Differential tolerance of native and invasive tree seedlings from arid African deserts to drought and shade. S Afr J Bot 123:228–240
Abrams MD, Kloeppel BD, Kubiske ME (1992) Ecophysiological and morphological responses to shade and drought in two contrasting ecotypes of Prunus serotina. Tree Physiol 10:343–355
Amini Z, Haddad R (2013) Role of photosynthetic pigments and antioxidant enzymes against oxidative stress. J Cell Mol Res 26(3):251–265
Amissah L, Mohren GMJ, Kyereh B, Poorter L (2015) The effects of drought and shade on the performance, morphology and physiology of Ghanaian tree species. PLoS ONE 10(4):e0121004. https://doi.org/10.1371/journal.pone.0121004
Anjum SA, Xie X, Wang LC, Saleem MF, Man C, Lei W (2011) Morphological, physiological and biochemical responses of plants to drought stress. Afr J Agric Res 6:2026–2032
Aranda I, Castro L, Pardos M, Gil L, Pardos JA (2005) Effects of the interaction between drought and shade on water relations, gas exchange and morphological traits in cork oak (Quercus ruber L.) seedlings. For Ecol Manage 210:117–129
Banwarie L, Kaushik SK, Guatam RC (1994) Effects of soil moisture regime, kaolin spray and phosphorous fertilizer on nodulation, P uptake and water use of lentil (Lense culinaris). Indian J Agron 39:241–245
Basra AS, Basra RK (1997) mechanism of environmental stress resistance in plants. Harward Academic Publishers, Harward, p 407
Bates IS, Waldern RP, Teare ID (1973) Rapid determination of free Prolin for water stress studies. Plant Soil 39:205–207
Bayazidi A, Oladi B, Abbasi N, Afaridoun Kh (2012) Statistical analysis using Minitab16. Abed Press, Tehran, p 308
Beers RF Jr, Sizer IW (1952) A spectrophotometric method for measuring the breakdown of hydrogen peroxide by catalase. J Biol Chem 195:133–140
Behra RK, Mishra PC, Choudhur NK (2002) High irradiance and water stress induce alteration in pigment composition and chloroplast activities of primary wheat leaves. J Plant Physiol 159:967–973
Ben Hamed S, Lefi E, Chaieb M (2016) Physiological responses of Pistacia vera L. versus Pistacia atlantica Desf. to water stress conditions under arid bio climate in Tunisia. Sci Hortic 203:224–230
Boyer JS (1968) Measurement of the water status of plants. Annu Rev Plant Physiol 9:351–363
Calagari M, Ghasemi R, Asadi F, Baghery R (2018) Promotion of wood production of some poplar clones using sprouts management in Karaj. Iran J For 10(1):79–88
Castro-Díez P, Navarro J, Pintado A, Sancho LG, Maestro M (2006) Interactive effects of shade and watering on the performance of seedlings of three Mediterranean Quercus species. Tree Physiol 26:389–400
Cheong YH, Pandey GK, Grant JJ, Batistic O, Li L, Kim BG, Lee SC, Kudla J, Luan S (2007) Two calcineurin B-like calcium sensors, interacting with protein kinase CIPK23, regulate leaf transpiration and root potassium uptake in Arabidopsis. Plant J 52:223–239
Climent JM, Aranda I, Alonso J, Pardos JA, Gil L (2006) Developmental constraints limit the response of Canary Island pine seedlings to combined shade and drought. For Ecol Manage 231:164–168
Close DC, Beadle CL, Brown PH (2005) The physiological basis of containerised tree seedling ‘transplant shock’: a review. Aust For 68(2):112–120
Daniels CW, Mabusela WT, Marnewick JL, Valentine AJ (2013) Photosynthetic adaptation of two semi-arid species of Gethyllis (Kukumakranka) to drought-and-shade stress. S Afr J Bot 88:36–41
Duan B, Lu Y, Yin C, Junttila O, Li C (2005) Physiological responses to drought and shade in two contrasting Picea asperata populations. Physiol Plant 124:476–484
Emadi MH (1996) study plan on Beneh. Rural Research Institute, Tehran, p 186
Emami A (1996) Methods of Analyzing Plants (Vol. 1). Report No. 982, Soil & Water Research Institute, Karaj
Engel R (1997) Response of Oat to Water and Nitrogen, Fertilizer Facts. Report No. 15, Montana State University, Agricultural Extension Service, Bozeman
Fayyaz P, Etemadi E, Julaiee-Manesh N, Zolfaghari R (2013) Sodium and potassium allocation under drought stress in Atlas mastic tree (Pistacia atlantica subsp. Mutica). iForest 6:90–94
Goel A, Coel AK, Sheran JF (2003) Changes in oxidative stress enzymes during artificial aging in cotton seeds. J Plant Physiol 160:1093–1100
Hamzepour M, Bordbar SK, Joukar L, Abbasi AR (2006) The potential of rehabilitation of Beneh forests through straight seed sowing and seedling planting. Iran J For Poplar Res 14(3):207–220
Hernández EI, Vilagrosa A, Luis VC, Llorca M, Chirino E, Vallejo VR (2009) Root hydraulic conductance, gas exchange and leaf water potential in seedlings of Pistacia lentiscus L. and Quercus suber L. grown under different fertilization and light regimes. Environ Exp Bot 67:269–276
Heydari M (2016) Autecology and phenology of Pistacia atlantica in relation to edaphic and physiographic factors in Kabirkoh forests of Darreh Shahr county, Ilam province. J Plant Res 29(1):80–95
Holmgren M (2000) Combined effects of shade and drought on tulip poplar seedlings: trade-off in tolerance or facilitation. Oikos 90:67–78
Huang X, Xiao X, Zhang S, Korpelainen H, Li C (2009) Leaf morphological and physiological responses to drought and shade in two Populus cathayana populations. Biol Plant 53(3):588–592
Huang X, Yin Ch, Duan B, Li Ch (2008) Interactions between drought and shade on growth and physiological traits in two Populus cathayana populations. Can J for Res 38(7):1877–1887
Jahanpour FA, Fatahi M, Karamian R (2010) Studying the influence of light on surviving of pistachio saplings in Lorestan province. Iran J For 3(2):91–98
Johnson NL (1949) Systems of frequency curves generated by methods of translation. Biometrika 36(1/2):149–176
Joulaei-Manesh N (2011) Study on anatomical, physiological and biochemical responses of Atlas mastic tree (Pistacia atlantica subsp. mutica) seedlings under drought stress. M.Sc. thesis, Faculty of Natural Resources, Yasouj University, Yasouj, p 64
Kafi M, Borzoee A, Salehi M, Kamandi A, Masoumi A, Nabati J (2012) Physiology of environmental stress in plants. Jihade Daneshgahi Press, Mashhad, p 502
Kupers SJ, Wirth Ch, Engelbrecht BMJ, Hernández A, Condit R, Wright SJ, Rüger N (2019) Performance of tropical forest seedlings under shade and drought: an interspecific trade-off in demographic responses. Sci Rep 9:18784. https://doi.org/10.1038/s41598-019-55256-x
Lambers H, Chapin FS, Pons ThL (2008) Plant physiological ecology. Springer, New York, p 604
Latorre R, Muñoz F, González C, Cosmelli D (2003) Structure and function of potassium channels in plants: some inferences about the molecular origin of inward rectification in KAT1 channels. Mol Membr Biol 20(1):19–25
Li Y, Zhao H, Duan B, Korpelainen H, Li Ch (2011) Effect of drought and ABA on growth, photosynthesis and antioxidant system of Cotinus coggygria seedlings under two different light conditions. Environ Exp Bot 71(1):107–113
Lichtenthaler HK (1987) Chlorophylls and carotenoids-pigments of photosynthetic bio membranes. In: Packer, L., Douce, R. (Eds.), Methods in Enzymology, Vol. 28, Plant Cell Membranes. Academic Press, London, pp. 350–382, pp 762
Maguire AJ, Kobe RK (2015) Drought and shade deplete nonstructural carbohydrate reserves in seedlings of five temperate tree species. Ecol Evol 5(23):5711–5721
Markesteijn L, Poorter L (2009) Seedling root morphology and biomass allocation of 62 tropical tree species in relation to drought- and shade-tolerance. J Ecol 97:311–325
- Mirzaei J (2011) Identification of Arbuscular mycorrhizal fungi associated with Pistacia atlantica and P. khinjuk in Ilam province and their effects on seedlings growth under drought stress. Ph.D. thesis, Faculty of Natural Resources and Marine Sciences, Tarbiat Modares University, Noor, pp 157
Mirzaei J, Karamshahi A (2015) Effects of drought stress on growth and physiological characteristics of Pistacia atlantica seedlings. J Wood For Sci Technol 22(1):31–43
Mozaffarian V (2010) Trees and shurbs of Iran. Farhang Moaser publication, Tehran, 1054 p
Negahdarsaber MR, Abbasi AR (2010) Impacts of ground cover vegetation on natural regeneration of Beneh (Pistacia atlantica) (Case study: Beneh experimental forest, Fars province). Iran J For Poplar Res 18(4):630–638
Niinemets Ü, Valladares F (2006) Tolerance to shade, drought, and waterlogging of temperate Northern Hemisphere trees and shrubs. Ecol Monogr 76:521–547
Owji MGh, Hamzepour M (2003) Comprehensive study on Baneh (Pistacia atlantica) Experimental Forest of Fars province of Iran. Iran J For Poplar Res 10(1):283–308
Pilevar B, Kakavand M, Akbari H, Ismaeili A, Soosani J, Mirazadi Z (2012) Growth and morphological responses of Manna oak (Quercus brantii) seedlings to different light levels at nursery in the first growing year. Iran J For Poplar Res 20(1):74–83
Quero JL, Villar R, Marañón T, Zamora R (2006) Interactions of drought and shade effects on seedlings of four Quercus species: physiological and structural leaf responses. New Phytol 170:819–834
Rad MH, Assareh MH, Meshkat MA, Soltani M (2011) Effects of drought stress on biomass, several growth parameters and water use efficiency of eucalyptus (Eucalyptus camaldulensis Dehnh) in response to drought stress. Iran J Range For Plant Breed Gen Res 19(1):13–27
Ranjbar Fordoei A, Samson R, Van Damme P, Lemeur R (2000) Effect of drought stress induced by polyethylene glycol on pigment content and photosynthetic gas exchange of Pistacia khinjuk and P. mutica. Photosynthetica 38(3):443–447
Sabeti H (1994) Forests, Trees and Shrubs of Iran. Yazd University, Yazd, p 874
Sadat Noori SA (2005) Applied statistics and experimental designs in agricultural sciences (vol. 2). University of Tehran, Tehran
Sapeta H, Costa JM, Lourenço T, Maroco J, van der Linde P, Oliveira MM (2013) Drought stress response in Jatropha curcas: Growth and physiology. Environ Exp Bot 85:76–84
Schall P, Lödige C, Beck M, Ammer C (2012) Biomass allocation to roots and shoots is more sensitive to shade and drought in European beech than in Norway spruce seedlings. For Ecol Manage 266:246–253
Schonfeld MA, Johnson RC, Carver BF, Mornhinweg DW (1988) Water relations in winter wheat as drought resistance indicators. Crop Sci 28:526–531
Siddique KHM, Walton GH, Seymour M (1993) A comparison of seed yields of winter grain legumes in Western Australia. Aust J Exp Agric 33:915–922
Smith T, Huston M (1989) A theory of the spatial and temporal dynamics of plant communities. Vegetatio 83:49–69
Sofo A, Dichio B, Montanaro G, Xiloyannis C (2009) Shade effect on photosynthesis and photoinhibition in olive during drought and rewatering. Agric Water Manag 96:1201–1206
Sohrabi A (1995) Impacts of harvesting on natural regeneration of Beneh in Lorestan province. In: Proceedings of the First National Symposium on Pistacia atlantica. 3–4 November, Ilam, pp 337–340
Soltani A (2010) Review in application of statistical methods, 2nd edn. Jihade Daneshgahi Press, Mashhad, p 74
Xu F, Guo W, Xu W, Wei Y, Wang R (2009) Leaf morphology correlates with water and light availability: what consequences for simple and compound leaves? Prog Nat Sci 19(12):1789–1798
Zahedipour H, Fattahi M, Mirdavoodi Akhavan HR, Godarzi G, Azdoo Z (2005) Distribution of different species of Pistachio in Markazi Province of Iran. Iran J For Poplar Res 13(1):33–78
Zolfaghari R (2008) Investigation of resistance to drought of Quescus brantii seedlings using morphological, physiological, biochemical and molecular markers. Ph.D. thesis, Tarbiat Modarres University, Tehran, pp 137
Author information
Authors and Affiliations
Contributions
Mohammad Hosein Sadeghzadeh Hallaj conducted the project, designed the experiment, measured the variables, performed the experimental works, and analyzed the data. He edited the paper, as well.
Davoud Azadfar supervised the project.
Hossein Mirzaei Nodoushan supervised the project.
Saeedeh Eskandari wrote and edited the paper. She prepared the geographic and thematic figures, as well. She was the corresponding author.
John P. Tiefenbacher edited the English, grammar, and writing elements.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All authors approve the ethics and consent to participate in this research.
Consent for publication
All authors have consented to publish this paper.
This manuscript doesn’t contain data from any individual person: “Not applicable”.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Philippe Garrigues
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sadeghzadeh Hallaj, M.H., Azadfar, D., Mirzaei Nodoushan, H. et al. Shade moderates the drought stress on saplings of Beneh (Pistacia atlantica Desf. subsp. mutica) in semiarid areas of Iran. Environ Sci Pollut Res 29, 55201–55212 (2022). https://doi.org/10.1007/s11356-022-19635-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-19635-8