Abstract
Knowledge of the dynamics of plant nitrogen (N) uptake at varying irrigation water levels is critical for strategizing increased N recovery efficiency (NRE), water use efficiency (WUE), and maize yield. The N dynamics were studied under various irrigation regimes to evaluate NRE, WUE, and maize yield. A pot experiment was conducted using three irrigation water regimes (50, 75, and 100% field capacity (FC)) and four N fertilizer rates (0, 1.6, 3.2, and 4.8 g pot−1) applied with two fertilizer application methods including foliar and soil applications. The highest plant growth and grain yields were achieved by application of 4.8 g N pot−1 with 100% FC. Contrarily, the maximum WUE (7.0 g L−1) was observed by the lowest irrigation water (50% FC) with the highest N fertilizer rates (4.8 g pot−1). Nitrogen concentration in the stem and grain was linearly increased by increasing N fertilizer rates with irrigation water. However, in the root, N concentration was decreased when the crop was supplied with 100% FC. In plant, maximum N uptake (6.5 mg g−1) was observed when 4.8 g N pot−1 was applied with 100% FC. Nitrogen recovery efficiency was increased by increasing N rate up to 3.2 g pot−1 with 100% FC. Therefore, for achieving maximum WUE and NRE, the highest water and N applications, respectively, are not necessary.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Nitrogen (N) and water are the major nutrients that limit plant growth and are extensively used to enhance crop yields. All grain crops, such as maize (Zea mays L.), are frequently treated with large amounts of N fertilizers to obtain optimum yield. Globally, fertilizer nearly 1011 kg of N year−1 is applied in agricultural systems (Glass 2003). Nevertheless, agricultural crops are only able to use 30–40% of this applied N fertilizers (Raun and Johnson 1999; Ju et al. 2009) leaving the excess in the environment. High N fertilizer inputs and the extremely low crop recoveries of fertilizer nutrients (for N, <10%) have resulted in significant deterioration of soil and groundwater quality (Jia et al. 2014). Losses of N fertilizers raise the cost of crop production and magnify the environmental contaminations. The pollution of water resources by N and other nutrients is a consequence of agricultural crop management when nutrient input exceeds the quantity consumed by the crops (Zhu et al. 2005; Gheysari et al. 2009).
Recently, scientific and public concerns have increased and thus have emphasized on the protection of water bodies from pollution caused by N leaching from agricultural systems (Long and Sun 2012; Gholamhoseini et al. 2013). This adds to the alarming situation when the global agricultural production systems are rapidly expanding to meet the increasing demand of food, fiber, and shelter. In order to meet the increasing food needs of world population, the most of the farmers apply additional N fertilizers as a conventional non-scientific approach without considering environmental hazards (Wei et al. 2009) and also to combat several abiotic stress like salinity, drought, heat stress, etc. (Fahad and Bano 2012; Fahad et al. 2014; Fahad et al. 2015a,b,c; Fahad et al. 2016). Moreover, majority of the farming communities are unaware about the interaction of N fertilizer with other input sources under field conditions. Nitrogen use efficiency can be affected by many other factors including water availability. Nitrogen application rate response in terms of plant growth depends on the available irrigation water (Hammad et al. 2015). Research has shown the importance of N and irrigation water interactions to optimize maize productivity (Eghball et al. 1993). There is an increasing interest to enhance N productivity using optimum amount of irrigation water (Makurira et al. 2011; Ortega et al. 2004).
The plants absorb N through the roots if applied on soil or through the leaves in case of foliar application. Availability of N to the plant from soil depends on physiological capacity of the roots to uptake and assimilate N. The major factors that affect the capacity of the roots to take up N include soil moisture and texture. While N uptake of plants through foliar is influenced by the capacity of the leaves to absorb N. Foliar fertilizer application has the advantage over the soil application as the former is radially available to plant and is independent on soil conditions (Yildirim et al. 2007). However, higher doses of N fertilizer may have a damaging effect on leaf structure (Khan et al. 2002). Wang et al. (2009) proposed that two aspects may contribute in crop N uptake improvement under deficit irrigation supply to plants: an extensive plant root system for optimum N uptake and an improved N availability in the soil. Previous studies have shown that growth of the lateral roots of maize can be stimulated by partial root-zone drying (Kang et al. 1998) that, in turn, enhances the root surface area to facilitate enhanced WUE and N uptake. Earlier study also demonstrated that re-wetting of the dry soil enhances mineralization of soil organic N and improve N availability to plants (Birch 1958). This phenomenon is commonly known as “Birch effect,” which has been verified by several experimental studies (Jarvis et al. 2007; Pereira et al. 2007).
In addition to root surface area, the plasticity of root architecture is also necessary to increase nutrient acquisition. Similar to mild water stress, low N availability promotes the root elongation in maize (Gaudin et al. 2011). While primary physiological mechanism is still unknown, root growth enhances the uptake of plant immobile nutrients, like phosphorus and zinc, in maize (Zhu and Lynch 2004; Liu et al. 2004). The effects of low level of N application on lateral root growth are controversial. Contradictory findings about role of N in root growth have been reported by Wang et al. (2004). Tian et al. (2008) noticed that N stress declined the plant root growth. However, a study with recombinant inbred lines (Liu et al. 2008) showed that N stress increased the root growth. In mature plants, Gaudin et al. (2011) determined that N stress results in a little increase of the crown roots (by 13%). In previous studies, it also looks that the effects of N deficiency on root development and growth are specific to the root types, severity of N stress, and root length (Zheng et al. 2016; Giehl and Von Wiren 2014; Linkohr et al. 2002).
Optimization of N recovery efficiency (NRE) via better irrigation and fertilizer management practices is therefore necessary to minimize environmental threats. Nevertheless, improving NRE will also require a well understanding of the interaction between irrigation regimes and N application methods. Nitrogen accumulation in maize ranges from 135 to 258 kg N ha−1 (Bundy and Andraski 2005) depending upon soil and environmental conditions. Maize N requirement is relatively high due to the higher aboveground dry matter production, which acts as a large N sink. Nitrogen uptake is primarily associated with root uptake ability and sink capacity of the shoots to metabolize the N, which is influenced by plant growth rate. Hence, maize N uptake is dependent on physiological mechanism occurring in the roots (Henry and Raper 1991) as well as on environmental conditions such as humidity, temperature, and water availability (Scholberg et al. 2002).
Although maize N requirement is high, studies have shown that NRE is less than 55% (Bundy and Andraski 2005), and it decreases with an increase in N application rates. In addition to this, method and timing of N application affects N uptake efficiency in maize (Subedi and Ma 2005). Nevertheless, overall NRE of maize seems to be influenced, to a large extent, by irrigation regimes (Kirda et al. 2005). Nitrogen uptake efficiency may be closely related to N uptake characteristics of the root that is dictated by irrigation practices (Scholberg et al. 2002). Various studies have outlined the effect of N application methods and rate on crop N response and yield; however, relatively few studies elucidated the uptake dynamics and NRE in maize production systems under different irrigation regimes (Bundy and Andraski 2005; Kristensen and Thorup-Kristensen 2004). Extensive literature research did not reveal an in-depth study to show the interactive effects of various irrigation water regimes on root growth and N uptake. Therefore, the present study was planned with the objectives to determine the effect of nitrogen application rates and methods with various irrigation regimes on nitrogen uptake, nitrogen recovery efficiency, and yield of maize.
Material and methods
Experimental design
To pursue the objective stated above, a pot experiment was carried out for 109 days at the College of Agriculture Layyah, Sub Campus Bahauddin Zakariya University Multan, during 2012. A rain shelter erected in a field to prevent the addition of rainwater to the pot soil and natural air temperature was maintained. Two fertilizer application methods, M1 and M2, (foliar and soil, respectively) and four N fertilizer rates (F1, F2, F3, and F4) 0, 1.6, 3.2, and 4.8 g pot−1 (equivalent to 0 control, 100, 200, and 300 kg ha−1, respectively) with three FC levels I1, I2, and I3 (50, 75, and 100% FC, respectively) were used. Therefore, the experiment was laid out in a completely randomized design with factorial arrangements, and each treatment was replicated five times. The pots (45-cm high with 25-cm radius from the neck and 20 cm from bottom, surface area 0.16 m2) were used having the capacity of 25.5 kg soil. The non-sterilized soil was sieved using a 4.5-mm sieve in order to remove plant roots and other debris. Each of the soil made pot was uniformly filled with 25.5 kg of dry soil equivalent to about 20-cm-deep soil layer. The soil was sandy loam and its properties are given in Table 1. In the bottom of each pot, a small hole was permitted for excess water to drain out. Three seeds of the maize hybrid Pioneer 31-R-88 were sown on July 30, 2012 in each pot. After three leaves emerged, one vigorous plant was established in each pot. The field capacity levels were maintained according to the treatments. In each pot, basal doses of phosphorus and potash at the rates of 2.0 and 1.6 g, respectively (equivalent to 125 and 100 kg ha−1, respectively), were applied at sowing time while N was applied according to the treatments. All other agronomic practices were kept same for all the treatments.
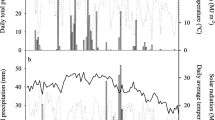
Daily meteorological information (rainfall, maximum and minimum temperatures) of experimental site during growing seasons is presented in Fig. 1.
Field capacity
Field capacity levels of each pot were kept as per treatments. The moisture percentage of soil in each pot was calculated on daily basis with the help of soil moisture meter (SM 150). When moisture contents were decreased to 30% of the treatment, then pots were irrigated to maintain FC level according to the respective treatments. This procedure was continued up to appraisal of seedlings.
Shoot harvesting
The plants were cut to ground level. Fresh shoot biomass of each fraction (stem, leaves, and cob) from the each pot was measured, and representative samples were oven-dried at 70°C until constant weight (Sharkey 1970). The oven-dried samples were ground and passed through 1-mm sieve and processed for N content through Kjeldahl method as explained by Houba et al. (1989). Subsequently, the shoot N uptake was calculated by multiplying the shoot dry matter yield with its N concentration. Thereafter, NRE was calculated as described by Shah et al. 2013.
where TNUn is total N uptake (g plant−1) by the plant from fertilized pot, TNUc is total N uptake (g plant−1) by the plant from unfertilized pot, and TNa is total N applied.
Root harvesting and nitrogen uptake
The roots of each plant were separated from soil in order to determine dry matter yield and N uptake at each 25-day intervals. There were four harvests throughout the growing period, from the seedling to physiological maturity stage (for details, see Fig. 2a, b). Two replications were used for root analysis. After separation from the soil, the root material of each plant was oven-dried at 70°C until constant weight (Sharkey 1970). The dried plant material was weighed and ground to pass through 1-mm sieve and analyzed for total N content by Kjeldahl method (Houba et al. 1989). Finally, the root N uptake of each plant was calculated by multiplying the root dry matter yield with its N concentration. At maturity, the plants were harvested for observation of the yields. The samples were preceded for determining grain yield, and the obtained yields were converted in t ha−1.
Water use efficiency
Water use efficiency (g L−1) was determined by dividing the grain yield (g pot−1) with the total water (L) consumed by the crop (i.e., evapotranspiration) during the season (Latiri-Souki et al. 1998).
The plant’s water consumption in terms of evapotranspiration (ET) of the crop over a growing season was determined from a water balance formula by calculating the difference in weight of the pots with their plants and soil, and the mass of water added to them.
Statistical analysis
The effects of N fertilizer rate, fertilizer application methods, and deficit irrigation on the studied variables were analyzed by ANOVA using the SAS statistical software (SAS Institute 2004). The least significant difference (LSD) test was used for comparing treatment means when the F-values were significant.
Results
The fertilizer application methods and rates significantly affected the production of plant’s above ground biomass. The plant attained the highest leaf and stem weight (17.5 and 143 g plant−1, respectively) by the application of soil N fertilizer (Table 2). A significant interactive effect of irrigation regimes and N fertilizer rate was recorded on leaf and stem weight. Table 2 shows that leaf and stem biomass were significantly (P < 0.0002 and P < 0.001, respectively) affected by the irrigation regimes and by the N fertilizer rates. Normal irrigation regime (I3) with maximum N fertilizer application rate (F4) yielded the highest leaf and stem weight (23.8 and 179 g plant−1, respectively).
Significant difference in grain weight was found across both the foliar and soil N fertilizer application methods. The soil N application methods resulted in higher grain weight (5.8%) than foliar N application. Irrigation regimes and N fertilizer application rates showed significant interactions for grain weight. The maximum grain weight (98.9 g plant−1) was achieved by normal irrigation regime (I3) and the highest N fertilizer rate (F4). The grain weight was significantly associated with plant root weight and total dry matter production with R 2 = 0.95 and 0.98, respectively (Fig. 3a, b).
Plant root weight was significantly (P < 0.0001) affected by N fertilizer application method. The plant attained higher root weight (11.1 g plant−1) when N fertilizer was applied by soil application method. Significant interactive effects of irrigation regimes and N fertilizer rates were found. The treatment I1 × F3 statistically attained the maximum root weight (16.5 g plant−1). A linear increase in root weight was attained by increasing N fertilizer; however, it was decreased when the plants were subjected to normal irrigation (I3). The same trend was observed for WUE. The maximum WUE (7.3 g L−1) was obtained at 50% field capacity (I1) with application of maximum N fertilizer. The WUE was decreased by increasing the field capacity level.
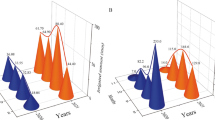
The concentration of N in the root was significantly affected by N fertilizer rates (Fig. 2a) and irrigation regimes (Fig. 2b). Generally, N concentration in the root was higher at early growth stages and decreased at maturity. Similarly, N concentration in the roots was decreased with increases in irrigation water/FC levels. However, it was significantly enhanced by increasing fertilizer rates equivalent to 300 kg N ha−1 (Fig. 2b). Nitrogen concentration in the stem (including the leaves and cob husk), grain, and root at maturity was significantly higher (15.9, 15.7, and 10.7 mg g−1, respectively) in the treatments of soil N application. Neither irrigation regimes nor N fertilization rate had a significant effect on N concentration in the stem (Table 3).
Irrigation regimes and N fertilizer application significantly influenced N concentration in the grains. It was linearly correlated in the grain by increasing both nutrients. Contrary, in the root, N concentration was significantly decreased by increasing FC level. However, its concentration was linearly increased in the roots with increases in N fertilizer dose, and the maximum N concentration (14.1 mg g−1) in the roots was achieved in the treatment (I1 × F4). Table 3 shows the effect of N on total N uptake by maize plant. The plant accumulated significantly higher N (3.9 mg g−1) when N fertilizer was applied by granular method. Total N uptake increased by increasing FC from 50 to 100% and N fertilizer level. The plant N uptake was maximum (6.5 mg g−1) by the treatment I1 × F4, and it was decreased at lower FC and lower N fertilizer rate. Total plant N uptake was significantly and positively correlated (R 2 = 0.96) with grain weight.
Nitrogen recovery efficiency was significantly influenced by N fertilizer application methods. The soil N application method resulted in maximum NRE (55.3%). The FC level significantly increased NRE; however, NRE beyond 200 kg ha−1 of the plant was significantly decreased. The treatment I3 × F3 has the maximum NRE (83.4%) in the study.
Discussion
Plant growth rate is measured by its ability to capture nutrients and other resources and convert these into biomass (Hammer et al. 2009). Plant leaves are determinate organs, which work as the main photosynthetic structure of land plants (Piazza et al. 2005). In the present experiment, the plant leaf weight was significantly influenced by irrigation regimes and N fertilizer application levels. Similar trend was observed in the plant stem and grain weights as both are driven by leaf growth rate as leaf response quickly to changing input and environmental conditions (Wahid et al. 2007). Similarly, maize root and shoot ratio is a character often used to estimate root biomass when shoot biomass is determined or estimated (Amos and Walters 2006). The results showed that maize root growth and N uptake synchronized with shoot development. These evidences support the fact that the maize plants that have higher total leaf weight do produce higher root biomass because the N fertilizer presence in soil results in growth of plant dry matter controlled by the plant’s leaf area index (Hirel et al. 2007).
Studies have demonstrated that a low level of field capacity can potentially increase WUE (Dodd 2009). In agreement with this, the results of the present study (Table 3) also revealed that WUE was higher for plant grown at low FC level. The possible reason behind the increased WUE may be the mycorrhizal association that increases plant water uptake (Kaya et al. 2003). Similarly, Liu et al. (2015) concluded that popular plants in association with mycorrhizae showed better water uptake and WUE under drought conditions. Besides improving crop WUE, studies on other crops have demonstrated that lower field capacity level can also significantly increase plant N nutrition (Wang et al. 2009, 2010). Our results illustrate that across the four N fertilization rates, N acquisition in the plant was higher at lower FC level that was further enhanced by increasing N fertilizer rate. When the plants were subjected to water deficit, the root weight was significantly increased, which resulted in a large amount of N accumulation in plant roots. Gao et al. (2015) reported that when maize plant received low N fertilizer with deficit irrigation, N uptake by the plant was significantly decreased and shoot N concentration was reduced. The reduced shoot N concentration inhibits the shoot growth, while the root dry weight increases as it has been reported that root shoot ratio is controlled by shoot N concentration (Andrews et al. 2006). Contrary to this, low N application reduces the root length in plants (Gaudin et al. 2011). The results of present study also showed that low N application reduced the root dry weight. These results are in agreement with the findings of Tian et al. (2008). Under low N application, the decline in the root weight and increase in root N concentration suggest that the roots had sufficient N (Gao et al. 2015); therefore, it is hypothesized that root response to exterior N fertilizer is controlled by a long-distance shoot-to-root signal through phloem showing shoot N status (Forde and Lorenzo 2001; Zhang et al. 2007).
In the present study, a low N treatment with optimum irrigation water reduced the root dry weight; hence, water and N fertilizer showed antagonistic effects. The results are supported by the previous findings that under water-limited conditions, the amount of N required to obtain optimum plant growth fluctuates and it could be adjusted accordingly (Hammad et al. 2015). Similarly, It has been reported that plants grown at optimum water availability have larger root surface area and amplified rooting depth, which are critical for N uptake from the wider and deeper root zone (Kang and Zhang 2004; Wang et al. 2009). Hence, crop N uptake is facilitated through optimum irrigation so enhanced N use efficiency and WUE may be attained simultaneously (Wang et al. 2010).
Changes in grain’s N concentration were controlled by the N fertilization rates and irrigation regimes. To achieve the maximum grain yield, optimum N fertilizer with normal irrigation water is required. It was observed that grain yield was strongly correlated with total dry matter production but that the increases in plant total dry matter did not correlate with N uptake for the maize (Ciampitti and Vyn 2011). Similarly, many researchers reported increased N concentration in the grains where plants were grown with non-limiting N fertilizer supply (Uhart and Andrade 1995; Gooding et al. 2007). Nitrogen recovery efficiency might be improved with optimum irrigation regimes; however, it did not increase linearly by enhancing N rates. In the study reported here, the low level of N was resulted in higher NRE. Ciampitti and Vyn (2011) anticipated that high levels of NRE are closely associated with the plant root weight and activity. In the study reported by Ladha et al. (2005), the NRE was around 70% with lower dose of N applied with overall average of 45%. In our research, NRE was 83.4% with medium N application rate with optimum irrigation water. In general, the maximum values for NRE might be achieved with the use of low N rates warranting the amount of irrigation water supplied and the environmental conditions.
Maize shoot and root growth is highly dependent on irrigation water and N fertilizer application. Nitrogen concentration in plant is also driven by irrigation water and N fertilizer application method and rates. Accordingly, the irrigation water and N fertilizer are the major inputs for improving crop productivity. Application of low field capacity irrigation and low nitrogen fertilizer rates improves water use efficiency and NRE simultaneously. By optimizing irrigation water, N could be used more efficiently.
References
Amos B, Walters DT (2006) Maize root biomass and net rhizodeposited carbon: an analysis of the literature. Soil Sci Soc Am J 70:1489–1503
Andrews M, Raven JA, Lea P, Sprent JI (2006) A role for shoot protein in shoot root dry matter allocation in higher plants. Annals of Bot 97:3–10
Birch HF (1958) The effect of soil drying on humus decomposition and nitrogen availability. Plant Soil 10:9–31
Bundy LG, Andraski TW (2005) Recovery of fertilizer nitrogen in crop residues and cover crops on an irrigated sandy soil. Soil Sci Soc Am J 69:640–648
Ciampitti IA, Vyn TJ (2011) A comprehensive study of plant density consequences on nitrogen uptake dynamics of maize plants from vegetative to reproductive stages. Field Crops Res 121:2–18
Dodd IC (2009) Rhizposphere manipulations to maximize ‘crop per drop’ during deficit irrigation. J Exp Bot 60:1–6
Eghball B, Settimi JR, Maranville JW, Parkurst AM (1993) Fractal analysis for morphological description on corn roots under nitrogen stress. Agron J 85:287–289
Fahad S, Bano A (2012) Effect of salicylic acid on physiological and biochemical characterization of maize grown in saline area. Pak J Bot 44:1433–1438
Fahad S, Hussain S, Bano A, Saud S, Hassan S, Shan D, Khan FA, Khan F, Chen Y, Wu C, Tabassum MA, Chun MX, Afzal M, Jan A, Jan MT, Huang J (2014) Potential role of phytohormones and plant growth-promoting rhizobacteria in abiotic stresses: consequences for changing environment. Environ Sci Pollut Res. doi:10.1007/s11356-014-3754-2
Fahad S, Hussain S, Saud S, Hassan S, Tanveer M, Ihsan MZ, Shah AN, Ullah A, Nasrullah KF, Ullah S, Alharby H, Nasim W, Wu C, Huang J (2016) A combined application of biochar and phosphorus alleviates heat-induced adversities on physiological, agronomical and quality attributes of rice. Plant Physiol Biochem 103:191–198
Fahad S, Hussain S, Saud S, Khan F, Hassan S, Amanullah W, Arif M, Wang F, Huang J (2015b) Exogenously applied plant growth regulators affect heat-stressed rice pollens. J Agron Crop Sci. doi:10.1111/jac.12148
Fahad S, Hussain S, Saud S, Tanveer M, Bajwa AA, Hassan S, Shah AN, Ullah A, Wu C, Khan FA, Shah F, Ullah S, Chen Y, Huang J (2015a) A biochar application protects rice pollen from high-temperature stress. Plant Physiol Biochem 96:281–287
Fahad S, Nie L, Chen Y, Wu C, Xiong D, Saud S, Hongyan L, Cui K, Huang J (2015c) Crop plant hormones and environmental stress. Sustain Agric Rev 15:371–400
Forde BG, Lorenzo H (2001) The nutritional control of root development. Plant Soil 232:51–68
Gao J, Zybailov BL, Byrd AK, Griffin WC, Chib S, Mackintosh SG, Tackett AJ, Raney KD (2015) Yeast transcription co-activator Sub1 and its human homolog PC4 preferentially bind to G-quadruplex DNA. Chem Commun Cam 51:7242–7244
Gaudin ACM, McClymont SA, Holmes BM, Lyons E, Raizada MN (2011) Novel temporal, fine-scale and growth variation phenotypes in roots of adult-stage maize (Zea mays L.) in response to low nitrogen stress. Plant Cell Enviro 34:2122–2137
Gheysari M, Mirlati SM, Bannayan M, Homaee M, Hoogenboom G (2009) Interaction of water and nitrogen on maize grown for silage. Agric Water Manag 96:809–821
Gholamhoseini M, AghaAlikhani M, Modarres SA, Sanavy M, Mirlatifi SM (2013) Interactions of irrigation, weed and nitrogen on corn yield, nitrogen use efficiency and nitrate leaching. Agric Water Manag 126:9–18
Giehl RFH, Von Wiren N (2014) Root nutrient foraging. Plant Physiol 166:509–517
Glass ADM (2003) Nitrogen use efficiency of crop plants: physiological constraints upon nitrogen absorption. Criti Reviews Plant Sci 22:453–470
Gooding MJ, Gregory PJ, Ford KE, Ruske RE (2007) Recovery of nitrogen from different sources following applications to winter wheat at and after anthesis. Field Crops Res 100:143–154
Hammad HM, Ahmad A, Abbas F, Farhad W, Cordoba BC, Hoogenboom G (2015) Water and nitrogen productivity of maize under semi-arid environments. Crop Sci 55:877–888
Hammer GL, Dong ZS, McLean G, Doherty A, Messina C, Schussler J, Zinselmeier C, Paszkiewicz S, Cooper M (2009) Can changes in canopy and/or root system architecture explain historical maize yield trends in the U.S. corn belt? Crop Sci 49:299–312
Henry LT, Raper CD (1991) Soluble carbohydrate allocation to roots, photosynthetic rate of leaves, and nitrate assimilation as affected by nitrogen stress and irradiance. Bot Gaz 152:23–33
Hirel B, Gouis JL, Ney B, Gallais A (2007) The challenge of improving nitrogen use efficiency in crop plants: towards a more central role for genetic variability and quantitative genetics within integrated approaches. J Exp Bot 58:2369–2387
Houba VJG, Van Der Lee JJ, Novozamsky I, Walinga I (1989) Soil and plant analysis, a series of syllabi, part 5. Wageningen University, the Netherlands
Jarvis P, Rey A, Petsikos C, Wingate L, Rayment M, Pereira J, Banza J, David J, Miglietta F, Borghetti M, Manca G, Valentini R (2007) Drying and wetting of Mediterranean soils stimulates decomposition and carbon dioxide emission: the “Birch effect”. Tree Physiol 17:929–940
Jia X, Shao L, Liu P, Zhao B, Gu L, Dong S, Bing SH, Zhang J, Zhao B (2014) Effect of different nitrogen and irrigation treatments on yield and nitrate leaching of summer maize (Zea mays L.) under lysimeter conditions. Agric Water Manag 137:92–103
Ju XT, Xing GX, Chen XP, Zhang SL, Zhang LJ, Liu XJ, Cui ZL, Yin B, Christie P, Zhu ZL, Zhang FS (2009) Reducing environmental risk by improving N management in intensive Chinese agricultural systems. Proc Natl Acad Sci U S A 106:3041–3046
Kang S, Liang Z, Hu W, Zhang J (1998) Water use efficiency of controlled alternate irrigation on root-divided maize plants. Agric Water Manag 38:69–76
Kang S, Zhang J (2004) Controlled alternate partial root-zone irrigation: its physiological consequences and impact on water use efficiency. J Exp Bot 55:2437–2446
Kaya C, Higgs D, Kirnak H, Tas I (2003) Mycorrhizal colonisation improves fruit yield and water use efficiency in watermelon (Citrullus lanatus Thunb.) grown under well-watered and water-stressed conditions. Plant Soil 253:287–292
Khan DF, Peoples MB, Herridge DF (2002) Quantifying below-ground nitrogen of legumes. 1. Optimising procedures for 15N shoot-labeling. Plant Soil 245:327–334
Kirda C, Topcu S, Kaman H, Ulger AC, Yazici A, Cetin M, Derici MR (2005) Grain yield response and N-fertiliser recovery of maize under deficit irrigation. Field Crops Res 93:132–141
Kristensen HL, Thorup-Kristensen K (2004) Uptake of N-15 labeled nitrate by root systems of sweet corn, carrot, and white cabbage from 0.2–2.5 meters depth. Plant Soil 265:93–100
Ladha JK, Pathak H, Krupnik J, Six J, Kessel C (2005) Efficiency of fertilizer nitrogen in cereal production: retrospect and prospects. In: Donald LS (ed) Advances in agronomy. Academic Press, San Diego, CA, pp. 85–156
Latiri-Souki K, Nortcliff S, Lawlor DW (1998) Nitrogen fertilizer can increase dry matter, grain production and radiation and water use efficiencies for durum wheat under semi-arid conditions. Eur J Agron 9:21–34
Linkohr BI, Williamson LC, Fitter AH, Leyser HMO (2002) Nitrate and phosphate availability and distribution have different effects on root system architecture of Arabidopsis. The Plant J 29:751–760
Liu J, Li JSH, Chen FJ, Zhang FS, Ren TH, Zhang ZJ, Mi GH (2008) Mapping QTLs for roots traits under different nitrate levels at the seedling stage in maize (Zea mays L). Plant Soil 305:253–265
Liu T, Sheng M, Wang CY, Chen H, Li Z, Tang M (2015) Impact of arbuscular mycorrhizal fungi on the growth, water status, and photosynthesis of hybrid poplar under drought stress and recovery. Photosynthetica 53:250–258
Liu Y, Mi GH, Chen FJ, Zhang ZJ, Zhang FS (2004) Rhizosphere effect and root growth of two maize (Zea mays L.) genotypes with contrasting P efficiency at low P availability. Plant Sci 167:217–223
Long GQ, Sun B (2012) Nitrogen leaching under corn cultivation stabilized afterfour years application of pig manure to red soil in subtropical China. Agri Ecosyst Environ 146:73–80
Makurira H, Savenije HHG, Uhlenbrook S, Rockstrom J, Senzanje A (2011) The effect of system innovations on water productivity in subsistence rainfed agricultural systems in semi-arid Tanzania. Agric Water Manag 98:1696–1703
Ortega JF, Juan JA, Tarjuelo JM (2004) Evaluation of the water cost effect on water resources management: application to typical crops in a semiarid region. Agric Water Manag 66:125–144
Pereira JS, Mateus JA, Aires LM, Pita G, Pio C, David JS, Andrade V, Banza J, David TS, Paço TA, Rodrigues A (2007) Net ecosystem carbon exchange in three contrasting Mediterranean ecosystems ? The effect of drought. Biogeoscience 4:791–802
Piazza P, Jasinski S, Tsiantis M (2005) Evolution of leaf developmental mechanisms. New Phytol 167:693–710
Raun WR, Johnson GV (1999) Improving nitrogen use efficiency for cereal production. Agron J 91:357–363
SAS Institute (2004) SAS/STAT 9.1 user’s guide. SAS Inst., Cary, NC
Scholberg JMS, Parsons LR, Wheaton TA, McNeal BL, Morgan KT (2002) Soil temperature, nitrogen concentration, and residence time affect nitrogen uptake efficiency in citrus. J Environ Qual 31:759–768
Shah GM, Rashid MI, Shah GA, Groot JCJ, Lantinga EA (2013) Mineralization and herbage recovery of animal manure nitrogen after application to various soil types. Plant Soil 365:69–79
Sharkey MJ (1970) Errors in measuring nitrogen and dry matter content of plant and faeces material. J Brit Grassl Soc 25:289–294
Subedi KD, Ma BL (2005) Effects of N-deficiency and timing of N supply on the recovery and distribution of labeled 15 N in contrasting maize hybrids. Plant Soil 273:189–202
Tian QY, Chen FJ, Liu JX, Zhang FS, Mi GH (2008) Inhibition of maize root growth by high nitrate supply is correlated with reduced IAA levels in roots. Plant Physio 165:942–951
Uhart SA, Andrade FH (1995) Nitrogen deficiency in maize: I. Effects on crop growth, development, dry matter partitioning, and kernel set. Crop Sci 35:1376–1383
Wahid A, Gelani S, Ashraf M, Foolad MR (2007) Heat tolerance in plants: an overview. Envir Experi Bot 61:199–223
Wang H, Liu F, Andersen MN, Jensen CR (2009) Comparative effects of partial root-zone drying and deficit irrigation on nitrogen uptake in potatoes (Solanum tuberosum L.). Irrig Sci 27:443–448
Wang Y, Liu F, Andersen MN, Jensen CR (2010) Improved plant nitrogen nutrition contributes to higher water use efficiency in tomatoes under alternate partial root-zone irrigation. Funct Plant Biol 37:175–182
Wang Y, Mi GH, Chen FJ, Zhang JH, Zhang FS (2004) Response of root morphology to nitrate supply and its contribution to nitrogen accumulation in maize. J Plant Nutri 27:2189–2202
Wei YP, Chen DL, Hu KL, Willett IR, Langford J (2009) Policy incentives forreducing nitrate leaching from intensive agriculture in desert oases of Alxa, InnerMongolia, China. Agric Water Manag 96:1114–1119
Yildirim E, Guvenc I, Turan M, Karatas A (2007) Effect of foliar urea application on quality, growth, mineral uptake and yield of broccoli (Brassica oleracea L., var. italica). Plant Soil Envir 53:120–128
Zhang H, Rong H, Pilbeam D (2007) Signalling mechanisms underlying the morphological responses of the root system to nitrogen in Arabidopsis thaliana. J Experi Bot 58:2329–2338
Zheng H, Pan X, Deng Y, Wu H, Liu P, Li X (2016) AtOPR3 specifically inhibits primary root growth in Arabidopsis under phosphate deficiency. Scientific Rep. doi:10.1038/srep24778
Zhu J, Kaeppler SM, Lynch JP (2005) Mapping of QTL controlling root hair length in maize (Zea mays L.) under phosphorus deficiency. Plant Soil 270:299–310
Zhu J, Lynch JP (2004) The contribution of lateral rooting to phosphorus acquisition efficiency in maize (Zea mays L.) seedlings. Functional Plant Bio 31:949–958
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible Editor: Yi-ping Chen
Rights and permissions
About this article
Cite this article
Hammad, H.M., Farhad, W., Abbas, F. et al. Maize plant nitrogen uptake dynamics at limited irrigation water and nitrogen. Environ Sci Pollut Res 24, 2549–2557 (2017). https://doi.org/10.1007/s11356-016-8031-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-016-8031-0