Abstract
The invasion by exotic cordgrass (Spartina alterniflora) has become one of the most serious and challenging environmental and ecological problems in coastal China because it can have adverse effects on local native species, thereby changing ecosystem processes, functions, and services. In this study, 300 surface sediments were collected from 15 stations in the Jiulong River Estuary, southeast China, across four different seasons, in order to reveal the spatiotemporal variability of biogenic elements and their influencing factors in the subtropical coastal mangrove wetland. The biogenic elements including carbon, nitrogen, and sulfur (C, N, and S) were determined by an element analyzer, while the phosphorus (P) was determined by a flow injection analyzer. The concentrations of biogenic elements showed no significant differences among four seasons except total phosphorus (TP); however, our ANOVA analyses revealed a distinct spatial pattern which was closely related with the vegetation type and tidal level. Values of total carbon (TC) and total nitrogen (TN) in the surface sediment of mangrove vegetation zones were higher than those in the cordgrass and mudflat zones. The concentrations of TC, TN, TP, and total sulfur (TS) in the high tidal zones were higher than those in the middle and low tidal zones. Redundancy analysis (RDA) revealed that tidal level, vegetation type, and season had some significant influence on the distribution of biogenic elements in the Jiulong River Estuary, by explaining 18.2, 7.7, and 4.9 % of total variation in the four biogenic elements, respectively. In conclusion, S. alterniflora invasion had substantial effects on the distributions of biogenic elements in the subtropical coastal wetland. If regional changes in the Jiulong River Estuary are to persist and much of the mangrove vegetation was to be replaced by cordgrass, there would be significant decreases on the overall storage of C and N in this coastal zone. Therefore, the native mangrove reforestation and exotic cordgrass elimination should be a priority in mangrove sustainable management for coastal ecosystem health.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Coastal wetlands act as transition zone buffering the interactions between the marine and the terrestrial ecosystems (Bianchi et al. 2013; Chmura et al. 2003) and may function as carbon, nitrogen, and phosphorus sinks reducing the organic matter runoff into the sea and serving as source of marine nutrients (Cai 2011; Feller et al. 2010; Valiela and Teal 1979). Coastal wetlands are fragile and sensitive to human activities, and the physical, chemical, and biological factors of coastal wetlands change strongly at spatial and temporal scales. In recent years, coastal wetlands have been undergoing rapid economic development and population increase; thus, they will potentially face growing problems with environment quality because large amount of pollutants input to the estuaries. This can have a profound negative impact over primary productivity, biodiversity, and ecosystem function of the local wetland ecosystems (Vitousek et al. 1997). The biogeochemical cycles of carbon, nitrogen, phosphorus, and sulfur in coastal wetlands have been an important focus for research into global change for several decades (Adams et al. 2012). However, information on the biogenic elements of biogeochemical cycling in subtropical estuarine mangrove wetlands is still very limited.
Mangroves are composed of species of halophytic intertidal trees and shrubs derived from tropical genera and have unique ecological functions and important social economy values (Saintilan et al. 2014). They have a complex food web, high species diversity, and net primary productivity (Lin 1999; Lovelock et al. 2009). The Jiulong River Estuary wetland in Fujian Province is a typical mangrove wetland reserve in southeast coastal areas of China. The dominant species in this wetland was Kandelia obovata. The best pure K. obovata mangrove forests were only distributed in Taiwan and Jiulong River Estuary in Fujian globally (Yang 2004). However, due to intense aquaculture and agricultural activities, rapid urban development, and biological invasion, many mangroves have suffered disturbance and degradation (Holguin et al. 2006; Hopkinson et al. 2012). Over the last decades, the Spartina alterniflora has invaded the coastal wetlands. S. alterniflora, as an invasive C4 perennial grass, was first intentionally introduced to the coastal wetland of China from its native range, the USA in 1979 (Liao et al. 2007). S. alterniflora invasion has not only threatened biodiversity of native ecosystems, but also altered ecosystem process, function, and service (Ehrenfeld 2003; Mack et al. 2000), thereby resulting in changes of carbon, nitrogen, phosphorus, and sulfur cycles in the invaded ecosystem (Jackson et al. 2002; Liao et al. 2008; Zhou et al. 2007). Many studies have shown the effects of plant invasion on carbon and nitrogen cycles (Bianchi et al. 2013; Hibbard et al. 2001; Jackson et al. 2002; Johnson and Wedin 1997; Li et al. 2009; Liao et al. 2007); however, the results of these studies suggest that the response of ecosystem to plant invasion is variable due to different invasion species and diverse native ecosystems. Therefore, quantitative analysis from independent studies across species and ecosystems is necessary for evaluation of ecosystem response to plant invasion (Liao et al. 2008). Mangrove–salt marsh ecotones offer an exceptional window onto the changes in biogenic elements in such dynamic transitional regions, including responses to S. alterniflora invasion.
Understanding of spatiotemporal variability of biogenic elements in the subtropical coastal mangrove wetland is crucial to explaining ecosystem processes and functions and modeling their responses to S. alterniflora invasion. Such an understanding will help us to better value the vulnerable ecosystems and to manage the mangrove wetland. Several studies have examined the biology and biogeochemistry in these mangrove wetlands (Alongi et al. 2005; Lin 1999; Yu et al. 2014), but little is known about the influence of vegetation, tide, and season on the distribution of biogenic elements. The aims of this study were to investigate the spatial and temporal variations of biogenic elements across four seasons, four vegetation types, and three tidal zones in the Jiulong River Estuary wetland and to reveal the effects of S. alterniflora invasion on the biogenic element distributions in the subtropical wetland.
Materials and methods
Study area and sampling
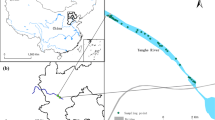
This study was conducted in a subtropical coastal wetland that is located in the Jiulong River Estuary (117° 53′–117° 55′ E, 24° 25′–24° 29′ N) in Fujian, southeast China. K. obovata is the dominant mangrove species, and S. alterniflora has invaded the large area over the last few decades (Yu et al. 2014). The climate in this area is subtropical maritime monsoon, the mean annual rainfall is 1,200 mm, and the mean annual mean temperature is 21 °C. The Jiulong River Estuary has a strong semidiurnal tide with an average tidal level of 2 m.
Three hundred surface sediment samples were collected from 15 stations during spring (April 2010), summer (August 2010), autumn (November 2010), and winter (January 2011) (Fig. 1). Samples were collected from 15 stations, which comprised four different habitat types, i.e., unvegetated bare mudflat, native mangrove zone, full S. alterniflora invaded zone, ecotone area with exotic S. alterniflora and native mangrove growing mixed together in the same area; three blocks were considered under different tidal levels, i.e., high-, middle-, low-level zones. The vegetation and tide type of 15 stations are presented in Table 1. Five parallel samples were collected from the four corners and center of a 1-m × 1-m quadrat at each site. All samples were collected from the top 0–10-cm layer in sediment using polyvinyl chloride (PVC) pipe (7 cm in diameter) and were packed in airtight plastic bags and transferred to the laboratory in a car refrigerator for further processing and analysis.
Chemical analysis
The sediment samples were freeze-dried at −80 °C, powdered with an agate mortar, and sieved through a standard sieve of 150 μm. The powered sediments of total carbon (TC), total nitrogen (TN), and total sulfur (TS) contents (as % dry weight of the sediment) were directly determined using Elemental Vario MAX CNS Analyzer (Germany), while the TP contents were measured by LACHAT QC 8500 Flow Injection Analyzer (USA) after digested with HClO4–H2SO4 and diluted with ultrapure (Mill-Q) water. Salinity of sediment pore water was measured in situ by an ATAGO digital salt meter (Japan). The pore water pH was measured using a Starter 2C pH meter (China). The grain size (D50) was analyzed by a Malvern 2000 laser granulometer (UK).
Due to the low inorganic carbon (<0.1 g kg−1) analyzed by SHIMADZU Total Organic Carbon Analyzer SSM-5000A (Japan) in the study area, TC can be considered roughly equivalent to total organic carbon (TOC) (Cheng et al. 2006).
Data analysis
We investigated the mean, standard error, and distribution of C, N, P, and S among 15 stations, 4 seasons, 4 vegetation types, and 3 tidal levels. One-way and three-way analyses of variance (ANOVA) were, respectively, performed on C, N, P, and S concentrations to test for differences among the different stations, seasons, vegetation types, and tidal levels after the data were Ln transformed to normal distribution. Where significant difference occurred, the Tamhane post hoc test was used to reveal the differences (P < 0.05). These statistical tests were performed by the SPSS 19.0.
Preliminary detrended correspondence analysis (DCA) on the C, N, P, and S data revealed that the longest gradient length was shorter than 3.0; thus, a redundancy analysis (RDA) was applied for exploring the variations in the contents of C, N, P, and S by constraining ordination axes to linear combinations of environmental factors. To explore the relationships between spatiotemporal variability of biogenic elements and environmental factors, RDA was performed using the CANOCO 4.5 (ter Braak and Smilauer 2002).Vegetation profiles were transformed into binary code, scoring each position as 1 (presence) or 0 (absence), i.e., mudflat (0, 0), cordgrass (1, 0), mangrove (0, 1), and mangrove-cordgrass ecotone (1, 1). Tidal profiles were transformed into gradient code, i.e., low tide (1), middle tide (2), and high tide (3). RDA with forward selection was initially employed as an iterative process to explore correlation within the environmental dataset, prior to final selection of significant variables. The forward selection procedure was first applied to all environmental variables. Only factors statistically significant at P < 0.05 confidence level were selected for the final analysis. The statistical significance of environmental factors was tested by using a Monte Carlo permutation test with 999 permutations. The final constrained model examined the response of TC, TN, TP, and TS contents to the 10 significant environmental variables defined by forward selection.
With three subsets of environmental data (season, vegetation, and tide), the total variation of TC, TN, TP, and TS contents was partitioned into seven components including covariance terms. The variation explained by these subsets was subtracted from the total variation (1.0 in case of RDA) to obtain the unexplained variation. The environmental variables are represented in the figures by arrows pointing in the direction of maximum change, and the arrow length is indicative of the importance of each environmental variable.
Results
General distributions of C, N, P, and S
The values of TC, TN, TP, and TS across the 15 stations (75 sites) from 4 seasons were significantly different (P < 0.001) (Fig. 2). The TC ranged from 8.40 to 23.87 g kg−1, with a mean ± standard deviation (SD) of 14.84 ± 3.42 g kg−1; the lowest and highest values were recorded in stations A1 (summer) and E3 (winter). The TN ranged from 0.85 to 2.57 g kg−1, with a mean ± SD of 1.50 ± 0.33 g kg−1; the lowest and highest values were recorded in stations A2 (summer) and E3 (summer). The TP ranged from 0.25 to 0.84 g kg−1, with a mean ± SD of 0.46 ± 0.09 g kg−1; the lowest and highest values were found in stations E2 (summer) and E3 (winter), respectively. TS ranged from 0.22 to 6.77 g kg−1, with a mean ± SD of 2.40 ± 1.07 g kg−1; the lowest and highest values were found in stations E3 (summer) and B3 (spring), respectively.
C, N, P, and S difference among four seasons, four vegetation types, and three tidal levels
The results of the three-way ANOVA indicated that vegetation, tide, and season had the most significant effects on TC, TN, TP, and TS contents. Further, there were significant effects on C, N, P, and S concentrations from interaction of vegetation, tide, and season with each other, except that tide that interacted with season had no significant effects on the distributions of TC and TN (Table 2).
Distributions of TC and TN concentrations varied significantly with vegetation and tidal level, and the Tamhane post hoc test revealed that mangrove > ecotone ≈ cordgrass > mudflat; high tidal level > middle tidal level > low tidal level (Fig. 3a, b). The TP differed significantly among vegetation types and tides: mangrove ≈ ecotone ≈ cordgrass > mudflat; high tide > middle tide > low tide (Fig. 3c). TS varied significantly with vegetation and tidal level, peaking in the mudflat and the high tidal zones, respectively (Fig. 3d). No significant difference was detected between four seasons for any of the biogenic elements except TP, which showed the lowest values in spring and the highest values in autumn.
Relationship between C, N, P, S, and environmental factors
The ten environmental variables identified by forward selection in RDA were all significant in explaining a portion of the variation in TC, TN, TP, and TS concentrations (P < 0.05). Eigenvalues for axes 1 and 2 were 0.338 and 0.029, respectively (Fig. 4). The first axis was significantly positively correlated with tide, mangrove, and cordgrass (P < 0.01) and showed a negative significant correlation with pH, D50, spring, and salinity (P < 0.01). The second axis was significantly positively correlated with tide and pH (P < 0.01) and showed a negative significant correlation with autumn (P < 0.05).
Tide was the primary factor that affected the distributions of TC, TN, TP, and TS. The unique variance explained by tidal level (18.2 %) was substantially higher than that explained by vegetation (7.7 %) or season (4.9 %) (Table 3).
Discussion
Spatiotemporal variability of biogenic elements
The distributions of TC, TN, TP, and TS in surface sediments from coastal areas are key issues in the circle of Land-Ocean Interactions in the Coastal Zone (LOICZ) program (Marchand et al. 2003; Yu et al. 2012). In Jiulong River Estuary, the concentrations of biogenic elements are intermediate on a global scale compared with previously reported data (Alongi et al. 2005; Yu et al. 2012). The TC and TN values in this study were similar to previous data from the Zhangjiangkou mangrove wetland in Fujian province of China, but they were slightly lower than mangrove coast in French Guiana (Marchand et al. 2003; Zhang et al. 2008). The TP in the study was similar to that reported for mangrove wetland in Zhangjiangkou, while the TS values were lower than Guiana mangrove coast (Marchand et al. 2003; Zhang et al. 2008). Although range of TOC, TN, and TS contents in French Guiana Mangrove were high, the young mangrove forest in Guiana was similar with that of Jiulong River Estuary wetland because TOC fluctuated from 1 to 2 %; total N values ranged from 0.1 to 0.2 % (Marchand et al. 2003).
There was no significant difference in the season for TC, TN, and TS concentrations, but TP showed higher values in autumn and lower values in spring. This distribution pattern of TP was probably due to different forms of phosphorus in the sediments (Zhou et al. 2007). Recently, Pan et al. (2010) found that organic P was the main part of TP in this area, which accounted for 50–60 % of TP. So, the concentration of TP was higher in autumn than that in spring, because the amount of litter contributing organic P was abundant in autumn.
Vegetation and tidal control of organic matter
The spatial and temporal variations of organic matter were found to vary significantly across different sedimentation environments, vegetation types, tidal levels, and industrial discharge (Wang et al. 2003; Zhou et al. 2007). However, this issue has received little integrative attention. Wang et al. (2003) found that mash vegetation contributed significant amounts of organic matter to the sediment. The organic matter in surface sediments of the Yangtze Estuary marsh was mainly controlled by the chemical composition of the suspended particulate matter (Zhou et al. 2007). In the present study, factors including tidal level, vegetation type, and season on the distribution of biogenic elements were all considered.
Interestingly, we found that values of TC and TN in the mangrove vegetation zones were higher than those in invasive plant zones and mudflat zones. These results were similar with previous reports in the Zhangjiangkou and the Jiulong River Estuary mangrove wetland in Fujian province of China (Pan et al. 2010; Zhang et al. 2008) and the Gulf of Mexico (Bianchi et al. 2013). However, the results were inconsistent with the Mornington Peninsula temperate mangrove and salt marsh systems in the southern hemisphere, because mangrove sediments from a cooler and drier temperate latitude may store less C than mangroves in warmer and wetter tropical latitudes (Livesley and Andrusiak 2012). Normally, mangroves have high productive, high decomposition rate, and short return time (Lin 1999). According to Clark et al. (2001) and Alongi (2009), the mean of net primary productivity (NPP) for mangrove ranged from 2 to 50 Mg C ha−1 year−1, rivaling some of the most productive old growth tropical forests (Reef et al. 2010). Mangroves can fix and store more carbon than marshes on regional and global scale, and the contents of organic matter in mangroves are higher than mudflat; thus, they are consistent with the recent reports on blue carbon sinks (Cai 2011; Hopkinson et al. 2012; Bianchi et al. 2013).
Our results clearly indicated that all biogenic elements increased significantly from the low tidal zones to the high tidal zones in the Jiulong River Estuary. In fact, the grain size distribution can influence the spatial characteristics of C, N, P, and S, which were significant negative correlation with D50. There are a number of reasons for this. First, the sediment particles tended to become finer from the low-tide level to the high-tide level, while the highest concentration of biogenic elements was closely associated with the finer fractions (Ashagrie et al. 2005; Zhou et al. 2007). Second, the middle- and high-tide regions have more vegetation and litter which are rich in organic matter. Third, TOC and N stocks in soils of a marine and a brackish marsh increased with decreasing inundation frequency (Spohn and Giani 2012). High tidal zones have shorter duration of tidal inundation, which affects salinity, oxidation state, and nutrient availability, thereby resulting in less loss of organic matter. Finally, coastal wetlands receive significant organic matter inputs as a direct result of intense human activity, and the impact of land-based pollution in high tidal zone is stronger (Alongi et al. 2005; Pan et al. 2011).
Applications for mangrove ecosystem management and conservation
Plant invasion is recognized as a serious threat to native ecosystems, which can alter their biogenic element cycles, biodiversity, and ecosystem services (Lövei 1997). However, the high variability among the experimental results could stem from the differences in life forms of invasive and native plants. Liao et al. (2007) showed that Spartina invasion resulted in changes of net primary production and litter decomposition and enhanced C and N stocks in the Scirpus and Phragmites-dominated ecosystems. Bianchi et al. (2013) found significantly higher carbon sequestration in mangrove compared to marsh sites on Mud Island. Song et al. (2014) suggested that conversion from cropland to woodland could lead to significantly greater soil organic carbon accumulation than would the conversion of cropland to grassland. However, other studies have indicated that C and N can be lost from native ecosystem invaded by exotic plants (Jackson et al. 2002; Johnson and Wedin 1997). Jackson et al. (2002) reported that the C stock decreased with the invasion of woody plant into grassland ecosystem, while Johnson and Wedin (1997) observed that the N stock loss from a dry tropical forest invaded by grass. Our study focused on the invasion of a C4 grass into woody plant community, e.g., mangrove wetland invaded by S. alterniflora, and attempted to evaluate the ecosystem response to plant invasion. Values of TC and TN in mangrove vegetation zone were higher than those in the S. alterniflora in the top 30-cm layer, indicating that the S. alterniflora invasion into mangrove wetland caused ecosystem organic matter loss (Yu et al. 2012). If regional changes in the Jiulong River Estuary are to persist and much of the mangrove vegetation was to be replaced by cordgrass, there would be significant decreases in the overall storage of C and N in the coastal zone.
On the other hand, plant cover has notably effect on the local sediment heterogeneity. However, Prusty et al. (2009) showed that the distribution of some plant species across the different habitats could not result in significant differences of the major nutrients (i.e., C, N, P, and S). More importantly, our study found that TC, TN, TP, and TS concentrations in the mangrove-cordgrass ecotone zone were similar with cordgrass zone, due to S. alterniflora invasion reduced sediment biogenic elements heterogeneity in the mangrove wetland.
Significantly higher carbon sequestration in the mangrove zone was found compared to the cordgrass and mudflat zones, indicating that mangroves store more carbon than marshes. Higher carbon sequestration will stabilize the rates of organic carbon turnover in the coastal wetlands, may prove to have an ameliorating effect on regional atmospheric CO2 increases (Bianchi et al. 2013). In summary, these studies have all let to the conclusion that S. alterniflora invasion into mangrove wetland can affect soil biochemical processes by means of decreased storage of C and N and reduced biogenic element heterogeneity. It appears that preserving and restoring mangrove are important and urgent, which will help to mitigate local climate change in the near future. Future work on changes in the sequestration of CO2 on this regional scale is needed to further verify that mangrove conservation has an ameliorating effect on local atmospheric CO2 increases.
Conclusion
Our study describes and evaluates the spatiotemporal patterns of biogenic elements in surface sediments from Jiulong River Estuary mangrove wetland. The values of TC, TN, TP, and TS across the 15 stations from 4 seasons were significantly different. However, no significant difference was detected between four seasons for any of the elements except for TP. Values of TC and TN in mangrove vegetation zones were higher than those in mudflat and cordgrass. The concentrations of TC, TN, TP, and TS in the high tidal level regions were higher than those in the middle and low tidal levels.
Tidal level, vegetation type, and seasonality significantly affected the distribution of four biogenic elements in surface sediments of the Jiulong River Estuary, by explaining 18.2, 7.7, and 4.9 % of total variation, respectively. Further, the invasion of S. alterniflora in mangrove wetlands caused ecosystem C and N stocks loss and affected on the local sediment biogenic element heterogeneity. This suggests that it is essential to protect and restore mangrove wetland and to prevent and control the spread of S. alterniflora.
References
Adams CA, Andrews JE, Jickells T (2012) Nitrous oxide and methane fluxes vs. carbon, nitrogen and phosphorous burial in new intertidal and saltmarsh sediments. Sci Total Environ 434:240–251
Alongi DM (2009) The energetics of mangrove forests. Springer, Dordrecht
Alongi DM, Pfitzner J, Trott LA, Tirendi F, Dixon P, Klumpp DW (2005) Rapid sediment accumulation and microbial mineralization in forests of the mangrove Kandelia candel in the Jiulongjiang Estuary, China. Estuar Coast Shelf Sci 63:605–618
Ashagrie Y, Zech W, Guggenberger G (2005) Transformation of a Podocarpus falcatus dominated natural forest into a monoculture Eucalyptus globulus plantation at Munesa, Ethiopia: soil organic C, N and S dynamics in primary particle and aggregate-size fractions. Agric Ecosyst Environ 106:89–98
Bianchi TS, Allison MA, Zhao J, Li X, Comeaux RS, Feagin RA, Kulawardhana RW (2013) Historical reconstruction of mangrove expansion in the Gulf of Mexico: linking climate change with carbon sequestration in coastal wetlands. Estuar Coast Shelf Sci 119:7–16
Cai WJ (2011) Estuarine and coastal ocean carbon paradox: CO2 sinks or sites of terrestrial carbon incineration? Annu Rev Mar Sci 3:123–145
Cheng X, Luo Y, Chen J, Lin G, Chen J, Li B (2006) Short-term C4 plan Spartina alterniflora invasions change the soil carbon in C3 plant-dominated tidal wetlands on a growing estuarine Island. Soil Biol Biochem 38:3380–3386
Chmura GL, Anisfeld SC, Cahoon DR, Lynch JC (2003) Global carbon sequestration in tidal, saline wetland soils. Global Biogeochem Cycles 17:1111–1123
Clark DA, Brown S, Kicklighter DW, Chambers JQ, Thomlinson JR, Ni J, Holland EA (2001) Net primary production in tropical forests: an evaluation and synthesis of existing field data. Ecol Appl 11:371–384
Ehrenfeld JG (2003) Effects of exotic plant invasions on soil nutrient cycling processes. Ecosystems 6:503–523
Feller IC, Lovelock CE, Berger U, McKee KL, Joye SB, Ball MC (2010) Biocomplexity in mangrove ecosystems. Annu Rev Mar Sci 2:395–417
Hibbard KA, Archer S, Schimel DS, Valentine DW (2001) Biogeochemical changes accompanying woody plant encroachment in a subtropical savanna. Ecology 82:1999–2011
Holguin G, Gonzalez-Zamorano P, De-Bashan LE, Mendoza R, Amador E, Bashan Y (2006) Mangrove health in an arid environment encroached by urban development-a case study. Sci Total Environ 363:260–274
Hopkinson CS, Cai WJ, Hu X (2012) Carbon sequestration in wetland dominated coastal systems—a global sink of rapidly diminishing magnitude. Curr Opin Environ Sustain 4:186–194
Jackson RB, Banner JL, Jobbágy EG, Pockman WT, Wall DH (2002) Ecosystem carbon loss with woody plant invasion of grasslands. Nature 418:623–626
Johnson NC, Wedin DA (1997) Soil carbon, nutrients, and mycorrhizae during conversion of dry tropical forest to grassland. Ecol Appl 7:171–182
Li B, Liao CZ, Zhang XD, Chen HL, Wang Q, Chen ZY, Gan XJ, Wu JH, Zhao B, Ma ZJ, Cheng XL, Jiang LF, Chen JK (2009) Spartina alterniflora invasions in the Yangtze River estuary, China: an overview of current status and ecosystem effects. Ecol Eng 35:511–520
Liao CZ, Luo YQ, Jiang LF, Zhou XH, Wu XW, Fang CM, Chen JK, Li B (2007) Invasion of Spartina alterniflora enhanced ecosystem carbon and nitrogen stocks in the Yangtze Estuary, China. Ecosystems 10:1351–1361
Liao CZ, Peng RH, Luo YQ, Zhou XH, Wu XW, Fang CM, Chen JK, Li B (2008) Altered ecosystem carbon and nitrogen cycles by plant invasion: a meta-analysis. New Phytol 177:706–714
Lin P (1999) Mangrove ecosystems in China. Science Press, Beijing (in Chinese)
Livesley SJ, Andrusiak SM (2012) Temperate mangrove and salt marsh sediments are a small methane and nitrous oxide source but important carbon store. Estuar Coast Shelf Sci 97:19–27
Lövei GL (1997) Biodiversity: global change through invasion. Nature 388:627–628
Lovelock CE, Ball MC, Martin KC, Feller IC (2009) Nutrient enrichment increases mortality of mangroves. PLoS ONE 4:e5600
Mack RN, Simberloff D, Mark Lonsdale W, Evans H, Clout M, Bazzaz FA (2000) Biotic invasions: causes, epidemiology, global consequences, and control. Ecol Appl 10:689–710
Marchand C, Lallier-Vergès E, Baltzer F (2003) The composition of sedimentary organic matter in relation to the dynamic features of a mangrove-fringed coast in French Guiana. Estuar Coast Shelf Sci 56:119–130
Pan QK, Luo ZX, Yan CZ, Zhang DD (2010) Phosphorus fractions and theirs correlation analysis in surface sediment of urban coastal wetland. Ecol Environ Sci 19:2117–2122 (in Chinese with English abstract)
Pan QK, Luo ZX, Qiu ZZ, Yan CZ (2011) Distribution characteristics of nitrogen forms in surface sediments of Jiulongjiang Estuary wetland. Res Environ Sci 24:673–678 (in Chinese with English abstract)
Prusty BAK, Chandra R, Azeez P (2009) Distribution of carbon, nitrogen, phosphorus, and sulfur in the soil in a multiple habitat system in India. Soil Res 47:177–189
Reef R, Feller IC, Lovelock CE (2010) Nutrition of mangroves. Tree Physiol 30:1148–1160
Saintilan N, Wilson NC, Rogers K, Rajkaran A, Krauss KW (2014) Mangrove expansion and salt marsh decline at mangrove poleward limits. Glob Chang Biol 20:147–157
Song X, Peng C, Zhou G, Jiang H, Wang W (2014) Chinese Grain for Green Program led to highly increased soil organic carbon levels: a meta-analysis. Sci Rep 4:4460
Spohn M, Giani L (2012) Carbohydrates, carbon and nitrogen in soils of a marine and a brackish marsh as influenced by inundation frequency. Estuar Coast Shelf Sci 107:89–96
ter Braak CJF, Smilauer P (2002) CANOCO reference manual and CanoDraw for windows User’s guide: software for canonical community ordination (version 4.5). Microcomputer Power, Ithaca
Valiela I, Teal JM (1979) The nitrogen budget of a salt marsh ecosystem. Nature 280:652–656
Vitousek PM, Mooney HA, Lubchenco J, Melillo JM (1997) Human domination of Earth’s ecosystems. Science 277:494–499
Wang X, Chen R, Berry A (2003) Sources and preservation of organic matter in Plum Island salt marsh sediments (MA, USA): long-chain n-alkanes and stable carbon isotope compositions. Estuar Coast Shelf Sci 58:917–928
Yang Z (2004) Evaluation and protection countermeasure of wetlands in Jiulong river estuary. Cen South For Invent Plan 23:40–43 (in Chinese with English abstract)
Yu XQ, Yang J, Liu LM, Tian Y, Yu Z, Wang CF (2012) Spatial variations of biogenic elements in coastal wetland sediments of the Jiulong River Estuary. Environ Sci 33:3739–3747 (in Chinese with English abstract)
Yu Z, Yang J, Yu XQ, Liu LM, Tian Y (2014) Aboveground vegetation influences belowground microeukaryotic community in a mangrove nature reserve. Wetlands 34:393–401
Zhang X, Shi S, Pan G, Li L, Zhang X, Li Z (2008) Changes in eco-chemical properties of a mangrove wetland under Spartina Invasion from Zhangjiangkou, Fujian, China. Adv Earth Sci 23:974–981 (in Chinese with English abstract)
Zhou J, Wu Y, Kang Q, Zhang J (2007) Spatial variations of carbon, nitrogen, phosphorous and sulfur in the salt marsh sediments of the Yangtze Estuary in China. Estuar Coast Shelf Sci 71:47–59
Acknowledgments
We thank Drs. Shen Yu, Changzhou Yan, Zhuanxi Luo, and Xiaoru Yang for field sampling. We thank Dr. David M. Wilkinson for language polishing. This research was supported by the Knowledge Innovation Program of the Chinese Academy of Sciences (KZCX2-YW-Q02-04), the Science and Technology Planning Project of Xiamen, China (3502Z20120012), and the International Science and Technology Cooperation Program of China (2011DFB91710).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Yu, X., Yang, J., Liu, L. et al. Effects of Spartina alterniflora invasion on biogenic elements in a subtropical coastal mangrove wetland. Environ Sci Pollut Res 22, 3107–3115 (2015). https://doi.org/10.1007/s11356-014-3568-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-014-3568-2