Abstract
The quantification of oxygen release by plants in different stages of wetland plant life cycle was made in this study. Results obtained from 1 year measurement in subsurface wetland microcosms demonstrated that oxygen release from Phragmites australis varied from 108.89 to 404.44 mg O2/m2/d during the different periods from budding to dormancy. Plant species, substrate types, and culture solutions had a significant effect on the capacity of oxygen release of wetland plants. Oxygen supply by wetland plants was estimated to potentially support a removal of 300.37 mg COD/m2/d or 55.87 mg NH4-N/m2/d. According to oxygen balance analysis, oxygen release by plants could provide 0.43–1.12 % of biochemical oxygen demand in typical subsurface-flow constructed wetlands (CWs). This demonstrates that oxygen release of plants may be a potential source for pollutants removal especially in low-loaded CWs. The results make it possible to quantify the role of plants in wastewater purification.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Increasing public awareness of global environmental issues is forcing the government to realize that ever more stringent environmental standards and more effective wastewater treatment technologies are needed for protecting the environment. However, due to lack of capital resources to implement more conventional wastewater treatment, systems/plants/facilities are becoming limiting and impractical especially in rural areas (Werker et al. 2002). Therefore, the development of alternative, innovative economical and ecologically-friendly wastewater treatment technologies is being stimulated (Erler et al. 2008).
Recently, constructed wetlands (CWs), which are recognized as a sustainable solution, have been applied successfully in pollutant removal from various wastewaters (Vymazal 2011; Hu et al. 2012). CW systems are generally comprised of wetland plants, media, microbes, and wastewater, functioning with a variety of physical, chemical, and microbial processes (Kadlec and Knight 1996; Vymazal 2011, 2013). Macrophytes are considered to be an essential component of CWs and play a very important role in wetland succession (Brix 1994). They not only directly take up pollutants, but also increase the environmental diversity in the rhizosphere by providing growth surface, carbon source, and oxygen source for organisms (Brix 1997; Kuschk et al. 2003).
As oxygen (supplied by plants, atmosphere, or artificial aeration) plays a key role in the pollutant removal of CW (Kadlec and Wallace 2009). Oxygen release from plants has been done from different aspects in micro and macro methods. Some researchers found that plants in treatment wetlands can transfer oxygen through the stems from the atmosphere to the roots and proved thorough anatomical and physiological studies (Brix 1989; Armstrong and Armstrong 1991; Armstrong et al. 1994; Crawford and Braendle 1996; Drew 1997; Allen 1997; Grosse and Frick 1999; Konnerup et al. 2011). Others measured rates of oxygen release from plants using different approaches such as oxygen-depleted method (Sorrell and Armstrong 1994), oxygen consumption model (Armstrong et al. 2000), microelectrode method (Bezbaruah and Zhang 2004), and the mass balance method (Ye et al. 2012). Typically, oxygen release rates from wetland plants are reported to range between 0.014 and 12 g/m2/d (Nivala et al. 2012). Due to the difference of experimental conditions and techniques in measurement of oxygen release, the amount of oxygen released by plants varies a lot. Furthermore, numerous studies have reported that quantification of oxygen release from plants could be affected by plant species, redox state, biomass, pH, oxygen concentration, chemical characteristics, temperature, light intensity, humidity, and wind velocity (Armstrong et al. 1990; Brix et al. 1996; Jespersen et al. 1998; Sorrell 1999; Stottmeister et al. 2003; Bezbaruah and Zhang 2004; Soda et al. 2007; Konnerup et al. 2011). Physiological activities, growth, and development of wetland plants at different growth stages tend to be different. However, no information has been reported on quantifying oxygen release by plants in different stages of wetland plant life cycles, which should be beneficial to improving design and operation of CW treatments. In addition, the capacity and variability of oxygen release by macrophytes and their influencing factors should be studied in more detail.
The main objectives of this study were (1) quantifying oxygen release by plants in different stages of wetland plant life cycle in the subsurface CW microcosm; (2) investigating the effects of plant species, substrate types, and culture solutions on capacity of oxygen release; and (3) estimating and quantifying the contribution of oxygen release from plants to biochemical oxygen demand for degradation of pollutants in typical subsurface-flow treatment wetlands.
Material and methods
Plant material
All plants used in this study were collected from Xinxue River CW in Weishan County, Shandong province, China (34° 44′ 30″ N, 117° 08′ 32″ E). The CW has been operating successfully for treating polluted river water since 2008, and plants such as Phragmites australis (P. australis), Typha orientalis (T. orientalis), Cyperus rotundus (C. rotundus), and Zizania aquatica (Z. aquatica) had grown for four full growing seasons. For measuring oxygen release of wetland plants in different stages of wetland plant life cycle (budding, seedling, elongation, blooming, maturation, senescence, and dormancy), the oxygen release of P. australis, the most common plant in CW systems, was determined in April, May, July, August, October, November, and December of the Year 2012. T. orientalis, C. rotundus, and Z. aquatica were also collected in June 2012 for comparing the difference among plant species. All plants were collected manually without damage to their roots (Robbins et al. 1964; Keddy 2010). The plants were washed manually in running tap water to remove soil and dead plant tissue (Wu et al. 2011a), and then plants were transferred into a container with a 10 % modified Hoagland’s solution (Hoagland and Arnon 1950) to cultivate in hydroponic culture until they were used in the experiments. Filtered air was also bubbled into each container to provide aeration. Experiments were carried out within 3 days from plant collection.
Experimental reactor and design
A modified design and method was used in this experiment to evaluate oxygen release by plants in CWs (Armstrong et al. 2000; Bezbaruah and Zhang 2004; Soda et al. 2007). The experiment was conducted in the greenhouse laboratory (similar to the outdoor conditions) of the Shandong University in Jinan, China. The reactor (25 cm high, diameter 15 cm, and made of polyethylene tub, Fig. 1) was designed as a subsurface CW. Gravel (20 cm depth, particle size 0.5–1.0 cm, and porosity 45 %) was filled as the wetland substrate, water level was kept 2 cm below the sediment surface, which gives an effective volume of 2 L. Tap water, which has necessary nutrients for the normal growth of plants, was used as test solution. The components of test solution mainly consisted of nutrients and micronutrients for the normal growth of plants (mg/L); 5 N, 0.2 P, 10 Ca, 5 Mg, 7 S, 0.3 Fe, 0.03 Zn, 0.01 Cu, 0 0.03 Mn, 0.03 B, and 0.002 Mo. Perforated pipes (16 mm in diameter) were placed on the side of the reactor and allowed that electrodes could be inserted to the proper depths (near the rhizosphere of roots). The cover (diameter 15 cm, polyethylene plate) was designed with an opening (diameter 20 mm) in the center. This opening was necessary for plants insertion and positioning. In order to prevented oxygen from entering the system by diffusing from the atmosphere, two perforated pipes were placed on the two opposite side, which allowed the N2 gas to continuously stream over the surface of the sediment in the reactor during measurements so that root oxygen release was the only possible source of oxygen in the system. A drainage pipe was also set at the bottom of the outlet side. To prevent light penetration, a piece of black plastic was stuck on the side of the reactor. For studying the effect of substrate types on oxygen release by plants, the reactors with the river sand (particle size 1–2 mm, Porosity 35 %) as the substrate were used. In addition, a 10 % modified Hoagland’s solution (Hoagland and Arnon 1950) and the simulated wastewater (Wu et al. 2011a) were used for comparing the difference of oxygen release by plants in different culture solutions.
Measurement of oxygen release
Oxygen release from plants was measured using the modified electrode method in the current experiment (Armstrong et al. 2000; Bezbaruah and Zhang 2004; Soda et al. 2007). Before every measurement, oxygen electrodes (HQ30d 53LEDTM, HACH, USA) were positioned in the reactor. Oxygen release from plants was investigated using individual plants in this study. The prepared plant was carefully placed into the reactor and sealed with Vaseline to prevent atmospheric oxygen transfer into the reactor. Subsequently, the test solution was fed into the reactor with an initial oxygen concentration corresponding to air-saturation, and then nitrogen gas was supplied to prevent atmospheric oxygen transfer into the reactor. Immediately, oxygen electrodes were connected to the measuring equipment and the computer running special data-processing software.
The experimental data (oxygen concentration, oxygen saturation, and temperature) were recorded at intervals of 5 min for 24 h. To ensure that a constant value of oxygen release by plants had been attained, three successive measurements were made on each plant. At the end of the experiment, plants were collected from each reactor, and then the above- and below-ground portions were weighed after above- and below ground portions were separated.
Statistical analyses
The rates of oxygen release by plants can be approximated by summation and calculated in the following equation:
Where OCR is oxygen consumption rate in reactors, mgO2/g fresh weight (FW)/d and mgO2/m2/d. OCR were obtained as follows:
Where C n and C n + 1 are the consecutive concentrations of oxygen in reactors before and after the moment of recording, mg/L; t n and t n + 1 are the time before and after the moment of recording, and the time interval is 30 min; m is the fresh weight of plants, g; r is 0.075 m.
Results
Growth characteristics of wetland plants
During the whole study period, all of the plants in different stages grew well without obvious symptoms of toxicity or nutrient deficiency. Height and fresh weights of the plants during the growing phase are shown in Table 1. It was observed that the heights of P. australis from budding to blooming period rose rapidly from 16.6 to 150.6 cm with the increasing air temperature. In the later days, P. australis grew slowly and reached the maximum (157.6 cm) in the senescence phase. When entering into the dormancy period, the heights of plants declined (157.1 cm) because plant metabolism was slowing down.
On the whole, above-ground and below-ground biomasses of P. australis showed the different growing trends during the life cycle of plants from budding to dormancy. Below-ground biomass was larger than above-ground biomass in different stages. Specifically, below-ground biomass increased from 5.9 to 16.4 g FW, and above-ground biomass varying between 2.7 and 7.6 g FW and reached the maximum in the stage of senescence. Biomass of T. orientalis, C. rotundus, and Z. aquatica used in this study was also determined with 7.2 ∼ 13.4 g FW (below-ground) and 4.3 ∼ 5.6 g FW (above-ground), respectively.
Oxygen release during different growth stages
Oxygen release from P. australis in different stages of plant life cycle is shown in Fig. 2. In this study, the rates of oxygen release by P. australis were significantly (p < 0.01) different during the growing phase, and no measurable oxygen release from plants without shoots was detected compared with variable oxygen release in the other growing periods. As is shown in Fig. 2, oxygen release rates of P. australis were 0.11 ∼ 0.78 mg O2/g FW/d or 108.89 ∼ 404.44 mg O2/m2/d during the life cycle of plants from budding to dormancy. The wavy fluctuations on oxygen release from plants in the plant life cycle were also observed, but oxygen release rates calculated by fresh biomass weight varied slightly from the rates in terms of unit area. On the whole, oxygen release rates in the budding, elongation, maturation, and dormancy phases were about three times higher than the values obtained in other stages of plant growing. The maximum rates of oxygen release were 0.78 mg O2/g FW/d (in budding) or 404.44 mg O2/m2/d (in dormancy), and the minimum rates of oxygen release were 0.11 mg O2/g FW/d (in senescence) or 108.89 mg O2/m2/d (in blooming).
The diurnal variations of OCR, water temperature, and DOS in different stages of wetland plant life cycle were represented graphically in Fig. 3. It shows that OCR was higher during daytime than in darkness when shoots of plants had not begun to grow (Fig. 3a). Dissolved oxygen saturation was decreasing gradually, which demonstrated that no available and ample oxygen was released from the plants. When the plants entered into the period of budding (Fig. 3b) and elongation (Fig. 3c), the timing of maximum OCR was at 1100 h with the increasing temperature and light intensities. More importantly, negative OCR emerged before and after midday due to the moderate temperatures and light intensities for photosynthesis, and the curve of diurnal variation of DOS showed a bimodal distribution pattern, which was homologous with the variation of plant photosynthesis during the growing season. During the elongation (Fig. 3d) and blooming (Fig. 3e) stages, the results revealed a difference in the variation of OCR and DOS during day and night. The peak value of OCR was observed in the midmorning (before 1100 h), following a negative unimodal distribution pattern of DOS, and average OCR was also lower in these stages because of increasing biomass of plants. The OCR and DOS curves in the maturation (Fig. 3f) and senescence (Fig. 3g) periods had a similar shape, although the maximum OCR in maturation was higher than in senescence as a result of lower temperature and decreasing plant metabolism. Entering the dormancy (Fig. 3h) stage, the variation of OCR and DOS was similar to the results during the budding period. The peak value of OCR was achieved in the midafternoon, and a rising in OCR and DOS was also observed during the dark period. The results revealed a difference in the diel variation of OCR and DOS under the varying growing phases. There were negative OCR values in the daytime, which indicated that a certain amount of oxygen might still be released by dormant plants.
Factors influencing oxygen release
The oxygen release rate is usually affected by many factors including light intensity, air and water temperature, wind velocity, air pressure, humidity, redox state, oxygen concentration, plant species, substrate types, culture solutions, and biomass (Armstrong et al. 1990; Brix et al. 1996; Jespersen et al. 1998; Sorrell 1999; Stottmeister et al. 2003; Bezbaruah and Zhang 2004; Soda et al. 2007; Konnerup et al. 2011). Among those, we specifically examined the effect of plant species, substrate types, and culture solutions on the oxygen release rate because these factors are crucial to design and management of CWs in practice.
Figure 4a shows the variations of oxygen release rates for different plants measured in the seeding period, and rates of oxygen release were in the range of 0.24 ∼ 0.37 mg O2/g FW/d or 126.67 ∼ 373.33 mg O2/m2/d. The rate of oxygen release from T. orientalis was highest, and Z. aquatica had a lower oxygen release rate, following by C. rotundus and P. australis. The results indicated that the different anatomical and growing characteristics of plants may affect the capacity of radial oxygen release and diffusion to the rhizosphere. Moreover, the capacity of oxygen release of different plants appeared to be associated with biomass of plants (Table 1). Oxygen release from P. australis in different culture solution is shown in Fig. 4b, and rates of oxygen release were 0.05 ∼ 0.44 mg O2/g FW/d or 26.67 ∼ 228.89 mg O2/m2/d. The rate of oxygen release from P. australis cultured in Hoagland’s solution was higher than that in tap water, and the lowest oxygen release was measured in wastewater. This result suggested that due to oxygen consumption for degradation of the organic matters and nitrification of ammonia, very little oxygen was detected in wastewater using the electrodes in this study. Figure 4c shows that oxygen release rates in the gravel reactors (0.24 mg O2/g FW/d or 126.67 mg O2/m2/d) were lower than that in river sand reactors (0.76 mg O2/g FW/d or 514.04 mg O2/m2/d). The difference of oxygen release rate in two substrate types revealed that oxygen release capacity may be affected by porosity which could influence oxygen transfer efficiency and diffusion processes in experimental systems.
The relationship between water temperature and oxygen release rates during the entire growing period in the study was analyzed (Figure S1a). Water temperature ranged from 15 to 30 °C. Oxygen release rates increased with increasing temperature, and then decreased as the temperature increased to higher levels. The results revealed that oxygen release from wetland plants was influenced by temperature. Figure S1b presents the relationship between biomass and oxygen release rates of P. australis in different stages of plant life cycle. As is shown, oxygen release rates decreased with the increasing of biomass as plants grow, which was contrary to the results obtained from different plant species (Fig. 4a and Table 1).
Discussion
Evaluation on capacity of oxygen release by wetland plants
The plant in subsurface wetland systems has the ability to pass oxygen, from its leaves through stems and rhizomes into the rhizosphere (Brix 1994). In this study, average rate of oxygen release from P. australis during the life cycle of plants was 0.26 g O2/m2/d. Armstrong et al. (1990) estimated oxygen release by Phragmites to be in the range of 5 ∼ 12 g O2/m2/d, whereas Brix and Schierup (1990) recorded oxygen release from Phragmites to be approximately 0.02 g O2/m2/d. Bezbaruah and Zhang (2004) reported an oxygen release of 0.8 g O2/m2/d for P. australis by measuring oxygen consumption for pollutant removal. The work by Ye et al. (2012) using the mass balance method reported the oxygen release rate of 0.014 ∼ 0.015 g O2/m2/d for P. australis in vertical-flow constructed wetlands fed with wastewater; however, based on this same approach, Dong et al. (2011) showed that oxygen release rate was ranged from 20.3 to 58.3 g O2/m2/d. In addition, estimates on oxygen release rates of P. australis were also made to be 4.1 g O2/m2/d using an isotope analysis method (Wu et al. 2011b). The US EPA (2000) also summarized work by others and reported plant oxygen release as 0 ∼ 3 g O2/m2/d.
On the whole, the results of the present study are in agreement with those reported in the past. However, there is also a clear distinction between various results on oxygen release from wetland plants. As indicated in earlier literatures, many factors might affect plant oxygen release, and they mainly include: plant species, growth conditions (e.g., light intensity, temperature, availability of nutrients, degradable materials in culturing solution, and media in systems), and the conditions (e.g., reactor design, oxygen concentration, HRT, day, oxygen concentration, and wind and air pressure) under which the measurements are made (Armstrong et al. 1990; Brix et al. 1996; Bezbaruah and Zhang 2004; Soda et al. 2007). It has been also proved that plant growing stage, plant species, substrate types, and culture solutions made a great impact on the oxygen release by wetland plants. In addition, it should be noted that different methods of the oxygen release measurement used in different studies (electrode method, gas tracer method, respirometry method, mass balance method, etc.) might be another key factor (Kadlec and Knight 1996; Nivala et al. 2012).
Role of oxygen release by plants in CWs
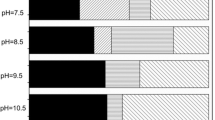
Wetland species such as P. australis could transport and release oxygen to facilitates the aeration of belowground organs and respiration of microbe in the rhizosphere (Brix and Sorrell 1992). The results of the present study indicated that oxygen from release of plants, according to oxygen consumption models (Kadlec and Knight 1996), could potentially supply enough oxygen to remove 300.37 mg COD/m2/d through aerobic degradation or 55.87 mg NH4-N/m2/d through nitrification. In order to fully understood the role of oxygen release by wetland plants in CW treatments, oxygen supply and biochemical oxygen demand in typical subsurface-flow CWs in the previously studies were also estimated (Nivala et al. 2012). Figure 5 shows that average oxygen demands for treating farm effluents with the low loads (average areal loading of 36.3 g COD/m2/d and 0.61 g NH4-N/m2/d) and tannery wastewater with the high loads (average areal loading of 92.3 g COD/m2/d and 3.75 g NH4-N/m2/d) in subsurface-flow CWs (Schulz et al. 2003; Calheiros et al. 2007) were 24.08 and 57.71 g O2/m2/d during the experiment period. Based on oxygen balance analysis, oxygen supply by plants was estimated to satisfy 1.12 % of biochemical oxygen demand in low-loaded CWs, while plant supply merely satisfied 0.43 % of biochemical oxygen demand in highly-loaded CWs. A comparison between supply and demand of oxygen in these estimates indicates that oxygen release of plants may be a potentially significant source in low-loaded CWs, but oxygen release is of minor quantitative importance in highly-loaded CWs. This is coincident with some previous conclusions that plants do not release enough oxygen into the rhizosphere for removing pollutants especially in subsurface-flow CWs (Bezbaruah and Zhang 2004). However, according to Kadlec and Wallace (2009), oxygen supply (mainly from plants, atmosphere, or artificial aeration) plays a key role in the pollutant removal of CWs.
Although the capacity and importance of oxygen release from wetland plants in CWs have been in debate, it should be noted that the role of plants in CWs is not merely oxygen release to rhizosphere, but wetland plants can influence the wetland treatment performance by other processes (Brix 1997). Firstly, wetland plants enhance abundance and diversity of microorganisms in the rhizosphere by increasing available surface area for bacterial attachment and growth. Secondly, the roots of wetland plants exude a range of degradable organic compounds, including sugars, organic acids, and amino acids, which can especially provide a continuing supply of carbon for denitrification bacteria in wetland systems. Thirdly, wetland plants can take up nutrients for their growth and reproduction, and other contaminants such as heavy metals and micro-pollutants (Teuchies et al. 2012; Huang et al. 2012). Finally, the existence of plants is thought to increase and stabilize the hydraulic conductivity in CWs (Brix 1994).
Conclusions
The present results indicated that the ability of oxygen release from wetland plants in CWs varied in different growing stages of plant life cycle, and oxygen release rates in the budding, elongation, maturation, and dormancy phases were about three times higher than values obtained in other stages of plant growing. The capacity of oxygen release of wetland plants could be influenced by plant species, substrate types, and culture solutions. Oxygen supply by plants was a potential source, but might be limited for the oxygen demand of wastewater in CWs. An efficient oxygen supply to CWs would be expected for possible improvement in the design and operation of subsurface flow constructed wetlands treating wastewater.
References
Allen JLH (1997) Mechanisms and rates of O2 transfer to and through submerged rhizomes and roots via aerenchyma. Soil Crop Sci Soc Fla Proc 56:41–54
Armstrong J, Armstrong W (1991) A convective through-flow of gases in Phragmites australis (Cav.) Trin. ex Steud. Aquat Bot 39:75–88
Armstrong W, Armstrong J, Beckett PM (1990) Measurement and modelling of oxygen release from roots of Phragmites australis. In: Cooper PF, Findlater BC (eds) The use of constructed wetlands in water pollution control. Pergamon, Oxford, pp p41–p51
Armstrong W, Braendle R, Jackson MB (1994) Mechanisms of flood tolerance in plants. Acta Bot Neerl 43:307–358
Armstrong W, Cousins D, Armstrong J, Turner DW, Beckett PM (2000) Oxygen distribution in wetland plant roots and permeability barriers to gas-exchange with the rhizosphere: a microelectrode and modeling study with Phragmites australis. Ann Bot 86:687–703
Bezbaruah AN, Zhang TC (2004) pH, Redox, and oxygen microprofiles in rhizosphere of bulrush (Scirpus validus) in a constructed wetland treating municipal wastewater. Biotechnol Bioeng 88(1):60–70
Brix H (1989) Gas exchange through dead culms of reed, Phragmites australis (Cav.) Trin. ex Steudel. Aquat Bot 35(1):81–89
Brix H (1994) Functions of macrophytes in constructed wetlands. Wat Sci Tech 29(4):71–78
Brix H (1997) Do macrophytes play a role in constructed treatment wetlands. Wat Sci Tech 35(5):11–17
Brix H, Schierup H (1990) Soil oxygenation in constructed reed beds: The role of macrophyte and soil-atmosphere interface oxygen transport. In: Constructed Wetlands in Water Pollution Control. Pergamon Press, Oxford, p.53–66
Brix H, Sorrell BK (1992) Internal pressurization and convective gas flow in some emergent freshwater macrophytes. Limnol Oceanogr 37:1420–1433
Brix H, Sorrell BK, Schierup HH (1996) Gas fluxes achieved by in situ convective flow in Phragmites australis. Aquat Bot 54:151–163
Calheiros CS, Rangel AO, Castro PM (2007) Constructed wetland systems vegetated with different plants applied to the treatment of tannery wastewater. Water Res 41(8):1790–1798
Crawford RMM, Braendle R (1996) Oxygen deprivation stress in a changing environment. J Exp Bot 47:145–159
Dong C, Zhu W, Zhao YQ, Gao M (2011) Diurnal fluctuations in root oxygen release rate and dissolved oxygen budget in wetland mesocosm. Desalination 272:254–258
Drew MC (1997) Oxygen deficiency and root metabolism: Injury and acclimation under hypoxia and anoxia. Annu Rev Plant Physiol Plant Mol Biol 48:223–250
Erler DV, Eyre BD, Davison L (2008) The contribution of anammox and denitrification to sediment N2 production in a surface flow constructed wetland. Environ Sci Technol 42(24):9144–9150
Grosse W, Frick HJ (1999) Gas transfer in wetland plants controlled by Graham’s law of diffusion. Hydrobiologia 415:55–58
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil. Calif. Agr. Exp. Sta. Circ. p347
Hu Y, Zhao Y, Zhao X, Kumar JLG (2012) High rate nitrogen removal in an alum sludge-based intermittent aeration constructed wetland. Environ Sci Technol 46:4583–4590
Huang JC, Passeport E, Terry N (2012) Development of a constructed wetland water treatment system for selenium removal: use of mesocosms to evaluate design parameters. Environ Sci Technol 46(21):12021–12029
Jespersen DN, Sorrell BK, Brix H (1998) Growth and root oxygen release by Typha latifolia and its effects on sediment methanogenesis. Aquat Bot 61:165–180
Kadlec RH, Knight RL (1996) Treatment Wetlands. CRC Press LLC, Boca Raton, FL 33431, USA
Kadlec RH, Wallace SD (2009) Treatment Wetlands, 2nd edn. CRC Press, Boca Raton, USA
Keddy PA (2010) Wetland ecology: Principles and conservation (2nd edition). Cambridge University Press, Cambridge, p497
Konnerup D, Sorrell BK, Brix H (2011) Do tropical wetland plants possess convective gas flow mechanisms. New Phytol 190:379–386
Kuschk P, Wiessner A, Kappelmeyer U, Weissbrodt E, Kästner M, Stottmeister U (2003) Annual cycle of nitrogen removal by a pilot-scale subsurface horizontal flow in a constructed wetland under moderate climate. Water Res 37(17):4236–4242
Nivala J, Wallace S, Headley T, Kassa K, Bri H, Afferden M, Müller R (2012) Oxygen transfer and consumption in subsurface flow treatment wetlands. Ecol Eng http://dx.doi.org/10.1016/j.ecoleng.2012.08.028
Robbins WW, Weier TE, Stocking CR (1964) Botany: an introduction to plant science, 3dth edn. John Wiley and Sons, New York
Schulz C, Gelbrecht J, Rennert B (2003) Treatment of rainbow trout farm effluents in constructed wetland with emergent plants and subsurface horizontal water flow. Aquaculture 217(1/4):207–221
Soda S, Ike M, Ogasawara Y, Yoshinaka M, Mishima D, Fujita M (2007) Effects of light intensity and water temperature on oxygen release from roots into water lettuce rhizosphere. Water Res 41(2):487–491
Sorrell BK (1999) Effect of external oxygen demand on radial oxygen loss by Juncus roots in titanium citrate solutions. Plant Cell Environ 22:1587–1593
Sorrell BK, Armstrong W (1994) On the difficulties of measuring oxygen release by root systems of wetland plants. J Ecol 82:177–183
Stottmeister U, Wiessner A, Kuschk P, Kappelmeyer U, Kästner M, Bederski O, Müller RA, Moormann H (2003) Effects of plants and microorganisms in constructed wetlands for wastewater treatment. Biotechnol Adv 22(1–2):93–117
Teuchies J, De Jonge M, Meire P, Blust R, Bervoets L (2012) Can acid volatile sulfides (AVS) influence metal concentrations in the macrophyte Myriophyllum aquaticum? Environ Sci Technol 46(16):9129–3917
United States Environmental Protection Agency.(2000) Constructed wetlands treatment of municipal wastewaters (Manual; Report #EPA/625/R-99/010. Cincinnati, OH: Office of Research and Development, U.S. EPA
Vymazal J (2011) Constructed wetlands for wastewater treatment: Five decades of experience. Environ Sci Technol 45:61–69
Vymazal J (2013) Emergent plants used in free water surface constructed wetlands: a review. Ecol Eng 61:582–592
Werker AG, Dougherty JM, McHenry JL, Van Loon WA (2002) Treatment variability for wetland wastewater treatment design in cold climates. Ecol Eng 19:1–11
Wu H, Zhang J, Li P, Zhang J, Xie H, Zhang B (2011a) Nutrient removal in constructed microcosm wetlands for treating polluted river water in northern China. Ecol Eng 37(4):560–568
Wu S, Jeschke C, Dong R, Paschke H, Kuschk P, Knoller K (2011b) Sulfur transformations in pilot-scale constructed wetland treating high sulfate-containing contaminated groundwater: a stable isotope assessment. Water Res 45(20):6688–6698
Ye J, Wang L, Li D, Han W, Ye C (2012) Vertical oxygen distribution trend and oxygen source analysis for vertical-flow constructed wetlands treating domestic wastewater. Ecol Eng 41:8–12
Acknowledgments
This work was supported by the Independent Innovation Foundation of Shandong University (2012JC029), Natural Science Foundation for Distinguished Young Scholars of Shandong Province (JQ201216), the National Water Special Project (2012ZX07203-004)
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Hailong Wang
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 286 kb)
Rights and permissions
About this article
Cite this article
Zhang, J., Wu, H., Hu, Z. et al. Examination of oxygen release from plants in constructed wetlands in different stages of wetland plant life cycle. Environ Sci Pollut Res 21, 9709–9716 (2014). https://doi.org/10.1007/s11356-014-2905-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-014-2905-9