Abstract
Background, aim, and scope
The aim of the present study was to evaluate the distribution of petroleum hydrocarbons in ten commercial fish species and water samples in three estuaries along Tamilnadu coast, Bay of Bengal, India.
Materials and methods
Fish and water samples collected from Tamilnadu coast, India, were extracted and analyzed for petroleum hydrocarbons by ultraviolet fluorescence (UVF) spectroscopy.
Results
The petroleum hydrocarbon concentration (PHC) in coastal waters and fish species varied between 2.28 and 14.02 μg/l and 0.52 and 2.05 μg/g, respectively. The highest PHC concentration was obtained in Uppanar estuarine waters (14.02 ± 0.83) and the lowest was observed in Vellar estuarine waters (2.28 ± 0.25).
Discussion
Among the ten fish species, Sardinella longiceps have high PHC concentration from all the locations. This study suggests that S. longiceps can be used as a good biological indicator for petroleum hydrocarbon pollution in water.
Conclusions
The concentration of petroleum hydrocarbons in coastal waters along Tamilnadu coast is markedly higher than that in the background, but there is no evidence for its increase in fish of this region. From a public health point, petroleum hydrocarbon residue levels in all fish samples analyzed in this study are considerably lower than the hazardous levels.
Recommendations and perspectives
At present, as Tamilnadu coastal area is in a rapid development stage of new harbour, chemical industries, power plants, oil exploration and other large-scale industries, further assessment of petroleum hydrocarbons and the various hydrodynamic conditions acting in the region are to be studied in detail and continuous pollution monitoring studies should be conducted for improving the aquatic environment. The results will also be useful for pollution monitoring program along the coastal region and also to check the levels of petroleum hydrocarbons.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
The problem of oil pollution in various marine and estuarine environments has received considerable scientific attention with respect to the effects of petroleum spills, as well as inherent toxicities to specific biological ecosystem components and individual species. Petroleum products are carcinogens and affect a variety of biological processes and potent cell mutagens. Estimates revealed that about 6.1 million metric tons of petroleum products were being released to global oceans annually (Capone and Bauer 1992). Hydrocarbons are quantitatively the most important constituents of petroleum, and arise from natural as well as anthropogenic sources (Law and Biscaya 1994; Medeiros et al. 2005). Human-mediated sources of petroleum hydrocarbons include offshore oil production, marine transportation, atmospheric or aerial depositions from combustion of coal and gas flaring, direct ocean dumping, coastal, municipal and industrial wastes, and runoff (NRC 2000). However, among the anthropogenic sources, point discharges, contamination by urban runoffs, refineries and other coastal effluents are in aggregate substantial and are important in causing local, chronic pollution in the vicinity of estuaries, creeks, harbours and coastal settlements (Abu-Hilal and Khordagui 1994; Venkatachalapathy et al. 2010a). Petroleum hydrocarbons as priority organic contaminants are extremely complex assemblage of chemicals coming from various sources. Although a considerable fraction of petroleum hydrocarbons entering the marine environment is removed by evaporation, some of them get dispersed in water, accumulate in sediment and transfer to biota (Chouksey et al. 2004).
Compared with other marine ecosystems, coastal habitats are particularly exposed to anthropogenic pollutants and especially to petroleum hydrocarbons (Halpern et al. 2008). Petroleum hydrocarbons are an important organic component and have received much attention because most of them are toxic, mutagenic and carcinogenic (Yunker et al. 1993; Hostettler et al. 1999; Venkatachalapathy et al. 2010b). In an aquatic environment, because of the low water solubility and hydrophobic properties, hydrocarbons are commonly found to be transported to the marine biota (Chouksey et al. 2004; Venkatachalapathy et al. 2011). Oil, which does not dissolve in water, floats on the sea surface as a thin layer when spilled or otherwise discharged because it is less dense than water and insoluble in it. This thin surface layer tends to break into droplets by wave action and these droplets become dispersed in the volume of sea water beneath the slick. Photochemical and biological oxidation of the slicks and of the dispersed oil droplets takes place producing oxygenated derivatives of the original oil components and these may form true solutions in water (Kennish 1992, 1997; Blanke and Wibbe 1999; Kahru et al. 2002).
The world’s fishing industries have depended historically on the availability of various kinds of high quality and uncontaminated fish from offshore and inshore fisheries (Burt et al. 1992). Oil pollution in coastal waters represents a serious threat to recreational and commercial fisheries. Many coastal regions sustain valuable fisheries resources that may be at risk during large oil spills. Juvenile and adult fish are generally able to avoid oil slicks in open seas. However, the rapid advection of large volumes of oil into estuary and embayment can trap fish populations, culminating in substantial fish mortality (Kennish 1997). Studies of the accidental and intentional releases of petroleum-based products to the aquatic environment indicate that aquatic organisms are able to bio-accumulate petroleum hydrocarbon fractions (Benson et al. 2008).
Egg, larvae and early juvenile stages of fish are most sensitive to oil. The contact of egg masses with surface oil can be lethal. Apart from the lethal effects of oil on embryos and larvae, sub-lethal effects on early life stages commonly include abnormal development, reduced growth, premature and delayed hatching of eggs, and cellular abnormalities (Kennish 1997). Adult fish tolerate much higher concentrations of petroleum hydrocarbons than eggs and larvae. Sub-lethal effects of oil typically manifested in adult fish are changes in heart and respiratory rates, gill hyperplasia, enlarged liver, reduce growth, fin erosion, impaired endocrine system, behavioral modification, as well as alternations in feeding, migration, reproduction, swinging activity, schooling and burrowing behavior. A variety of biochemical, blood, and cellular changes may also arise (Kennish 1992, 1997). Connell (1997) suggested that the pattern of distribution for lipophilic chemicals in aquatic system is transferred from low level tropic biota to higher level tropic biota to aquatic birds and marine mammal. Bioavailability and organic physiology are the two important variables that have a major effect on contaminant body burden. Physiological factors, including lipid levels and the rate of uptake and elimination (metabolism, diffusion and excretion), also have an effect on the contaminant body burden (Clark 1989; Connell 1997).
Oil may enter fish through the skin or gills. In addition, contaminants such as tar balls may ingress through the intestine by water gulped in the physiological process of desalination. Although human health had not been considered to be at risk from concentrations of petroleum hydrocarbons in fish, the possible consequences of bioaccumulation cannot be ignored especially in communities consuming large quantities of fish (Shriadah 2001).
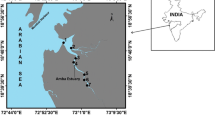
Tamilnadu is the southernmost state on the east coast of India with a coastline of nearly 1,000 km, having extensive areas of estuaries, mangroves, brackish water lagoons. The environmental chemistry of minor river basins in India has received less attention, despite several environmental studies on major rivers (Subramanian et al. 1987; Ramesh et al. 1989). This study reports the level of PHC contamination in ten commercial fish species and water samples along Tamilnadu coast. Cuddalore Town is a significant coastal city in Tamilnadu state that hosts a number of large-scale industries. The Uppanar River runs parallel to the coast south of Cuddalore Town having number of small streams of domestic, treated and untreated effluent discharges from industries. Uppanar estuary is constituted by the confluence of the Gadilam and Paravanar rivers that flow into the Bay of Bengal. The main industries along the western bank of Uppanar River include chemicals, beverage manufacturing, tanneries, oil, soap, paint production, paper, and metal processing plants. Cuddalore harbour, located in the estuarine region of the Uppanar River, is occupied by a fleet of mechanized fishing boats, which operates in the coastal zone. This open estuary has an average depth of 2.5 m and a width of 30 m near the mouth and 20 m upstream. The tidal influence extends to 6 km upstream. The Vellar river originates at the Kalvarayan hills in the Salem district, runs for 90 km and flows through the borders of Villupuram and Perambalur districts for a few kilometers. In its stretch, it enters the Cuddalore district, flowing for another 105 km, and ends its journey into the Bay of Bengal at Parangipettai. The Vellar estuary always remains open and is called a “true estuary.” The maximum amplitude of the tide reaches about 1 m. The tidal influence felt up to 10–15 km upstream. The average depth of the estuary is 2.5 m. The Coleroon River is located in the southern part of the study area.
2 Materials and methods
Ten fish species (Terapon jarbua, Caranx para, Stolephorus indicus, Sardinella longiceps, Kathala axillaris, Arius arius, Rastrilleger kanagurta, Mugil cephalus, Siganus javus and Etroplus suratensis) along with water samples were collected from Uppanar (S1), Vellar (S2) and Coleroon (S3) estuaries in April 2010 (Fig. 1).
Five samples from each fish species and water samples were collected from all three estuaries. Collected fish species were dissected; the soft tissue portion was wrapped in an aluminium foil, sealed in thick polythene bag and preserved at −20°C until analysis (Food and Agricultural Organization [FAO] 1983). The thawed tissue was homogenized, saponified using KOH–methyl alcohol mixture, centrifuged and filtered. The filtrate was extracted with n-hexane, and the organic layer was washed with distilled water, dried, evaporated to a small volume, chromatographed on alumina and fluorescence measured (IOC-UNESCO 1984). Petroleum hydrocarbon concentrations (PHC) in fish species were determined using ultraviolet fluorescence (UVF) spectroscopy (Varian make Cary Eclipse). The fluorescence of the samples was measured at an emission wavelength of 360 nm (excitation wavelength, 310 nm). Water samples were collected using pre-cleaned amber-glass bottles of 2 l capacity. The bottles were cleaned with n-hexane, and dried and rinsed with the ambient seawater before sampling. Seawater was extracted with n-hexane to transfer PHC in the organic phase and the organic extract was concentrated after drying. Fluorescence of the extract was measured using UVF spectroscopy (IOC-UNESCO 1984). All blanks, standards and samples were measured in a Teflon-capped 1-cm silica fluorescence cell under identical instrumental settings and conditions. The methods for estimation of petroleum hydrocarbons in water and fish were calibrated using replicate recovery tests on a sample spiked with known concentrations of the selected standard (Hunt et al. 1986; Abdullah et al. 1996; Ehrhardt and Burns, 1999; Chouksey et al. 2004). Appropriate blanks were analyzed with each set of samples, together with standard reference material (Chrysene) for quality assurance/quality control. Percentage recovery for spiked samples ranged from 96% to 99%, whereas precision agreed within 5%. All experiments were conducted in five replicates and the averages of the values were reported along with standard deviations. The results were treated statistically using the Student’s t-test (p < 0.05).
3 Results and discussion
3.1 Petroleum hydrocarbons in water
The distribution of petroleum hydrocarbons in water samples along Tamilnadu coast varies in a wide range of 2.28–14.02 μg/l (Fig. 2). The highest concentration of petroleum hydrocarbons (14.02 ± 0.83) were obtained in Uppanar estuary and the PHC values in Vellar and Coleroon estuaries are 2.28 ± 0.25 and 2.82 ± 0.44, respectively. The lowest PHC were found in Vellar (S2) and are considered the background level for this study. Considerable deterioration in water quality of the Uppanar estuary with respect to pH, dissolved oxygen (DO), salinity and dissolved trace metals due to anthropogenic perturbations has been well documented (Jonathan et al. 2008). The low DO content occurs when industrial effluents pollute the waters; the high chemical oxygen demand of the effluents result in reduced DO, which occur very close to the river bank where the combined effect of temperature, photosynthetic action, and biochemical degradation of the waters enter the water column (Jonathan et al. 2008). On the basis of profiles of petroleum hydrocarbons in surface sediments from the same study area (Veerasingam et al. 2010), it has been concluded that the highest PHC level in Uppanar estuarine waters might be found in land-based sources, such as industrial effluents originating from nearby industries, municipal wastes and atmospheric fallout, and marine-based sources, such as Cuddalore harbour and fishing activities. Other sources include illegal discharges of water from oil tank washing and machine oil residues from ships. But in Vellar and Coleroon estuarine waters, the PHC levels are nearly 7 times lower than that found in the Uppanar estuary. In the Bay of Bengal region, no natural oil seeps have been reported and accidental spills from ships are infrequent. Therefore, it may be inferred that discharges from ships and land-based sources contribute more to the present level of PHC in this region.
According to FAO (1982), seawater containing hydrocarbon levels of less than 2.5 μg/l are considered to be the natural background level in this region. The overall PHC in the Tamilnadu coastal waters, although higher than the background level, do not indicate the overall levels of contamination compared with values reported for selected costal areas in India and around the world (Table 1).
3.2 Petroleum hydrocarbons in fish tissue
The PHC found in ten fish species are represented in Fig. 2.
PHC residues in ten fish species along Tamilnadu coast varied between 0.52 and 2.05 μg/g (wet wt). The highest PHC concentration was found in Uppanar estuarine fish species and the lowest in Vellar estuary. Concentrations of PHC in Uppanar estuary fish species decreased in the order of S. longiceps (2.05 ± 0.14) > A. arius (2.02 ± 0.24) > T. Jarbua (1.95 ± 0.12) > S. javus (1.74 ± 0.25) > C. para (1.63 ± 0.33) > R. kanagurta (1.50 ± 0.15) > S. indicus (1.21 ± 0.24) > M. cephalus (0.91 ± 0.17) > E. suratensis (0.83 ± 0.24) > K. axillaris (0.72 ± 0.33). Concentrations of PHC in Vellar estuary fish species decreased in the following order: S. longiceps (1.35 ± 0.24) > R. kanagurta (1.05 ± 0.44) > C. para (1.01 ± 0.51) > S. javus (0.97 ± 0.21) > A. arius (0.84 ± 0.35) > T. Jarbua (0.83 ± 0.53) > S. indicus (0.78 ± 0.42) > M. cephalus (0.64 ± 0.32) > K. axillaris (0.59 ± 0.26) > E. suratensis (0.52 ± 0.34). Concentrations of PHC in Coleroon estuary fish species decreased in the order of S. longiceps (1.42 ± 0.27) > C. para (1.10 ± 0.33) > A. arius (1.06 ± 0.54) > S. javus (1.04 ± 0.37) > T. Jarbua (0.94 ± 0.24) > R. kanagurta (0.93 ± 0.22) > S. indicus (0.82 ± 0.21) > M. cephalus (0.74 ± 0.36) > E. surstensis (0.66 ± 0.28) > K. axillaris (0.54 ± 0.41). Generally, the lowest PHC levels found in Vellar (S2) estuarine fish species are considered as the background level for the present study. Among the three estuaries, the concentrations of petroleum hydrocarbons in all fish species in Uppanar estuary are high. This higher concentration might be caused by discharges from industrial and domestic effluents, boating and fishing activities from Cuddalore harbour. The uptake and release patterns have been particularly remarkable especially when the organisms are exposed to petroleum hydrocarbons. In areas where persistent discharges occur, organisms may be subjected to physiological stress, gill and skin infections and subsequent death (Asuquo et al. 2004). Among the ten fish species, S. longiceps has a high PHC concentration from all locations. This study suggests that S. longiceps can be used as a good biological indicator for petroleum hydrocarbon pollution in water. PHC in the range 0.1–10 ppm (wet wt) commonly occur in marine fish, while fish from areas polluted by petroleum can have PHC concentrations of 10–1000 times higher (Hellou et al. 1990). Though the concentration of petroleum hydrocarbons in water is markedly higher than that in the background, there is no evidence for its increase in fish of Uppanar estuary. Moreover, petroleum hydrocarbon residue levels in all fish samples analyzed in this study are considerably lower than hazardous levels (GESAMP 1977, 2007; Hellou et al. 1990). Although the number of analyzed samples was perhaps too small to provide definitive conclusions, the low PHC concentrations here suggest that human consumption of fish should be of very little concern for public health.
4 Conclusion
This study presents a baseline distribution assessment of petroleum hydrocarbons in coastal waters and fish species along Tamilnadu coast, Bay of Bengal, India. The highest PHC value that was obtained at Uppanar estuary might be attributed to land- and marine-based anthropogenic sources. Although the concentration of PHC in coastal waters of the Uppanar region is markedly higher than that in the background, there is no evidence for its increase in fish found in the region. From a public health viewpoint, petroleum hydrocarbon residue levels in all analyzed fish samples in this study are considerably lower than the hazardous levels. At present, as the Tamilnadu coastal area is in a rapid development stage of a new harbour, chemical industries, power plants, oil exploration and other large-scale industries, there is a need for further detailed assessment of petroleum hydrocarbons and the various hydrodynamic conditions impacting the region, and continuous pollution monitoring studies should be conducted to improve the aquatic environment. The results will also be useful for pollution monitoring programs along the river and coastal regions, and for the purpose of checking the levels of petroleum hydrocarbons.
References
Abdullah AR, Woon WC, Bakar RA (1996) Distribution of oil and grease and petroleum hydrocarbons in the Straits of Johor, Peninsular Malaysia. Bull Environ Contam Toxicol 55:155–162
Abu-Hilal AH, Khordagui HK (1994) Petroleum hydrocarbons in the nearshore marine sediments of the United Arab Emirates. Environ Pollut 85:315–319
Asuquo FE, Ewa-Oboho I, Asuquo EF, Udo PJ (2004) Fish species used as biomarker for heavy metal and hydrocarbon contamination for Cross River, Nigeria. Environmentalist 24:29–37
Barbier M, Joly D, Saliot A, Tourres D (1973) Hydrocarbons from sea water. Deep Sea Res 20:305–314
Benson NU, Essien JP, Ebong GA, Williams AB (2008) Total petroleum hydrocarbons in Macura reptantia, Procambarus clarkia and benthic sediments from the Qua Iboe, Estuary Nigeria. Environmentalist 28:275–282
Bidleman TF, Castleberry AA, Foreman WT, Zaranski MT, Wall DW (1990) Petroleum hydrocarbons in the surface water of two estuaries in the southeastern United States. Estuar Coast Shelf Sci 30:91–109
Blanke M, Wibbe ML (1999) Aliphatic hydrocarbons in an oil-contaminated soil carbon economy during microbiological decontamination. Environ Sci Pollut Res 6:2–6
Burt JR, Hardy R, Whittle KJ (1992) Pelagic fish: the resource and its exploitation. Fishing News Books, Oxford
Capone DG, Bauer JE (1992) Environmental microbiology. Clarendon, Oxford
Chouksey MK, Kadam AN, Zingde MD (2004) Petroleum hydrocarbon residues in the marine environment of Bassein–Mumbai. Mar Pollut Bull 49:637–647
Clark RB (1989) Marine pollution, 2nd edn. Clarendon, Oxford
Connell DW (1997) Basic concepts on environmental chemistry. Lewis Publishers, New York
DouAbul AAZ (1984) Petroleum residues in the waters of Shatt Al-Arab River and the North-West region of the Arabian Gulf. Environ Int 10:265–267
Ehrhardt M, Burns KA (1999) Determination of petroleum residues dissolved and/or finely dispersed in surface seawater. In: Grosshoff K, Kremling K, Ehrhardt M (eds) Methods of seawater analysis. Wiley-VCH, Weinheim, pp 467–477
El-Samra MI, Emara HI, Shunbo E (1986) Dissolved petroleum hydrocarbons in the North-Western Arabian Gulf. Mar Pollut Bull 17:65–68
FAO (Food and Agriculture Organisation) (1982) The review of the health of the oceans. FAO/IMCO/UNESCO/WMO/WHO/IAEA/UNEP Joint Group of Experts on Scientific Aspects of Marine Pollution (GESAMP) Rep Stud Gesamp 15, 108 pp
FAO (1983) Manual of methods in aquatic environment research: Part 9. Analyses of Methods and Organochlorines in Fish. FAO Fishery Technical Paper No. 212, 33 pp
GESAMP (1977) Joint group of experts on the scientific aspects of marine pollution. Impact of oil on the marine environment, GESAMP Reports and studies, No.6, 250 pp
GESAMP (2007) Estimates of oil entering the marine environment from sea based activities. International Maritime Organization, London
Halpern BS, Walbridge S, Selkoe KA, Kappel CV, Micheli F, D'Agrosa C, Bruno J, Casey KS, Ebert C, Fox HE, Fujita R, Heinemann D, Lenihan HS, Madin EMP, Perry M, Selig ER, Spalding M, Steneck R, Watson R (2008) A global map of human impact on marine ecosystems. Science 319:948–952
Hellou J, Stenson G, Payne JF (1990) Polycyclic aromatic hydrocarbons in muscle tissue of marine mammals from the Northwest Atlantic. Mar Pollut Bull 21:469–473
Hostettler FD, Pereira WE, Kvenvolden KA, Van Green A, Luoma SN, Fuller CC, Anima R (1999) A record of hydrocarbon input to San Francisco Bay as traced by biomarker profiles in surface sediment and sediment core. Mar Chem 64:115–127
Hunt DTE, Wilson AL, Hunt DTE, Wilson AL (1986) The chemical analysis of water. General principles and techniques, 2nd edn. The Royal Society of Chemistry, London, p 683 pp
IOC-UNESCO (1984) Manual for monitoring oil and dissolved dispersed petroleum hydrocarbons in marine waters and on beaches. Manual and Guides No. 13, 35 pp
Jonathan MP, Srinivasalu S, Thangadurai N, Ayyamperumal T, Armstrong-Altrin JS, Ram-Mohan V (2008) Contamination of Uppanar River and coastal waters off Cuddalore, Southeast coast of India. Environ Geol 53:1391–1404
Kahru A, Maloverjan A, Sillak H, Põllumaa L (2002) The toxicity and fate of phenolic pollutants in the contaminated soil with the oil-shale industry. Environ Sci Pollut Res 9:27–33
Kennish MJ (1992) Ecology of estuaries: anthropogenic effects. Press Raton, Boca CRC
Kennish MJ (1997) Practical handbook of estuarine and marine pollution. CRC Press, Boca Raton
Law RJ (1981) Hydrocarbon concentrations in water and sediments from UK marine waters, determined by fluorescence spectroscopy. Mar Pollut Bull 12:153–157
Law R, Andrulewicz E (1983) Hydrocarbons in water, sediment and mussels from the southern Baltic Sea. Mar Pollut Bull 14:289–293
Law RJ, Biscaya JL (1994) Polycyclic aromatic hydrocarbons (PAH) — Problems and progress in sampling, analysis and interpretation. Mar Pollut Bull 29:235–241
Marchand M, Caprais JC (1985) Hydrocarbons and halogenated hydrocarbons in coastal waters of the English Channel and the North Sea. Mar Pollut Bull 16:78–81
Marchand M, Caprais JC, Pignet P (1988) Hydrocarbons and halogenated hydrocarbons in coastal waters of the western Mediterranean (France). Mar Environ Res 25:131–159
Medeiros PM, Bicego MC, Castelao RM, Rosso CD, Fillmann G, Zamboni AJ (2005) Natural and anthropogenic hydrocarbon inputs to sediments of Patos Lagoon Estuary, Brazil. Environ Int 31:77–87
Natarajan M, Balasubramanian T, Ponnusamy G, Raja P (2006) Distribution of dissolved and dispersed petroleum hydrocarbons in coastal waters of Tamilnadu and Andhrapradesh. J Annamalai Univ Sci 42:63–70
NRC (2000) Oil in the sea: inputs, fates and effects. National Academy Press, National Research Council, Washington DC, p 806
Ramesh R, Subramanian V, Van Grieken R, Van’t Dack L (1989) The elemental chemistry of sediments in the Krishna river basin, India. Chem Geol 74:331–341
Readman JW, Mantoura RFC, Rhead MM, Brown L (1982) Aquatic distribution and heterotrophic degradation of polycyclic aromatic hydrocarbons (PAH) in the Tamar Estuary. Estuar Coast Shelf Sci 14:369–389
Selvaraj K, Jonathan MP, Mohan VR, Thangaraj GS, Pugalendhi M, Jayaraman B (1999) Observations of petroleum hydrocarbons and some water quality parameters during oil spill, near madras harbour. Indian J Mar Sci 28:245–248
Sen Gupta R, Qasim SZ, Fondekar SP, Topgi RS (1980) Dissolved petroleum hydrocarbons in some regions of the northern Indian Ocean. Mar Pollut Bull 11:65–68
Sen Gupta R, Fondekar SP, Alagarsamy R (1993) State of oil pollution in the northern Arabian Sea after the 1991 Gulf oil spill. Mar Pollut Bull 27:85–91
Shriadah MA (2001) Petroleum hydrocarbon concentrations in Arabian Gulf tissues. Bull Environ Contam Toxicol 67:560–567
Subramanian V, Van Grieken R, Van’t Dack L (1987) Heavy metals distributions in the sediments of Ganges and Brahmaputra rivers. Environ Geol 9:93–103
Tahir NM, Abdullah AR, Shanmugam S (1997) Determination of total hydrocarbon concentration in coastal waters and sediments off the east coast of Peninsular Malaysia. Environ Geochem Health 19:67–71
Topgi RS, Noronha RJ, Fondekar SP, Gupta S (1982) Dissolved petroleum hydrocarbons along the oil tanker route in southern Bay of Bengal. Indian J Mar Sci 11:175–176
Veerasingam S, Raja P, Venkatachalapathy R, Mohan R, Sutharsan P (2010) Distribution of petroleum hydrocarbon concentrations in coastal sediments along Tamilnadu coast, India. Carpathian J earth environ Sci 5:5–8
Venkatachalapathy R, Veerasingam S, Basavaiah N, Ramkumar T (2010a) Comparison between petroleum hydrocarbon concentrations and Magnetic properties in Chennai coastal sediments, Bay of Bengal, India. Mar Petrol Geol 27:1927–1935
Venkatachalapathy R, Veerasingam S, Ramkumar T (2010b) Petroleum hydrocarbon concentrations in marine sediments along Chennai Coast, Bay of Bengal, India. Bull Environ Contam Toxicol 85:397–401
Venkatachalapathy R, Veerasingam S, Basavaiah N, Ramkumar T, Deenadayalan K (2011) Environmental magnetic and petroleum hydrocarbons records in sediment cores from the north east coast of Tamilnadu, Bay of Bengal, India. Mar Pollut Bull (in press)
Yunker MB, Macdonald RW, Cretney WJ, Fowler BR, McLaughlin FA (1993) Alkane, terpene and polycyclic aromatic hydrocarbon geochemistry of the Mackenzie River and Mackenzie Shelf: riverine contributions to Beaufort Sea coastal sediment. Geochim Cosmochim Acta 57:3041–3061
Acknowledgement
We thank Prof. T. Balasubramanian, Dean, faculty of Marine Sciences, and Prof. AN. Kannappan, Dean, faculty of science, Annamalai University, for providing all the necessary facilities. We would like to acknowledge the contribution of two anonymous reviewers who provided important and constructive comments.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Veerasingam, S., Venkatachalapathy, R., Raja, P. et al. Petroleum hydrocarbon concentrations in ten commercial fish species along Tamilnadu coast, Bay of Bengal, India. Environ Sci Pollut Res 18, 687–693 (2011). https://doi.org/10.1007/s11356-011-0466-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-011-0466-8