Abstract
Purpose
The study aimed to investigate the change in the skin temperature after exercise-induced delayed onset muscle soreness (DOMS) and after cold water immersion during a recovery period.
Methods
Randomized controlled trial. Thirty-nine participants were randomly divided into four groups: group 1 (G1) which performed aerobic exercise; group 2 (G2) only performed plyometric exercise; group 3 (G3) was submitted to plyometric exercise and cold water immersion (CWI) protocol; and group 4 (G4) was the non-exercised control group. Skin temperature (TsK) was collected in two regions of interest (forehead and anterior thigh) at rest (T-rest), immediately after exercise (T0), 10, 20, and 30 min (T10, T20, and T30), and 24 and 48 h (T24 and T48) after exercise using an infrared camera. In addition, isometric knee extension strength and DOMS were assessed at times T-rest, T0, T24, and T48 for all groups.
Results
A higher skin temperature was indicated in the anterior thigh muscles that performed plyometric exercise (G2). This thermographic change was accomplished by a decrease in the knee extensor strength immediately after performing exercises. In addition, it seems clear from our data that CWI was effective in reducing Thigh’s TsK until 30 min after exercise, but had no effect on the Thigh’s TsK 24 and 48 h after exercise. CWI attenuated pain inducted by muscle compression 48 h after plyometric exercise.
Conclusion
This paper presents evidence that plyometric anaerobic exercise leads to a higher skin temperature on the exercised muscle groups than aerobic exercise and that the application of CWI has no influence on muscle temperature 24 h after exercise while minimizing DOMS and the deficit of isometric force.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Muscle blood flow suffers some physiological changes during exercise. There is acute skin vasoconstriction, and the blood flow is directed to active muscles and later a peripheral vasodilation as a thermoregulatory response to dissipate the heat produced by the muscle activity, thereby preventing an increase in body temperature [1]. According to Kenny et al. [2], tissue temperature is determined by the relative rate of heat production and loss of the measured areas at a certain moment. Thus, the process depends on the exercise type and duration as well environmental condition.
During short-term high-intensity exercise, such as plyometric exercise, the metabolic production of heat is high [3]. Moreover, eccentric exercise is associated with micro ruptures in the muscle fibers, pain, decreased neuromuscular excitability, and muscle weakness, which can inhibit neuromuscular response of the injured muscles [4]. The delayed onset muscle soreness (DOMS), also known as exercise-induced muscle damage (EIMD), is usually found in individuals performing high-intensity eccentric exercise after a long period of inactivity or in athletes who perform exercise beyond their normal limit [5]. In this way, the intensity of symptoms and increased discomfort arise within the first 24 h and may persist even 72 h after an intense plyometric exercise [6]. After high muscle tension caused by eccentric exercise, DOMS usually occurs due to destruction of contractile elements and the subsequent inflammatory processes [4].
Cold water immersion (CWI) is a technique widely used in post-exercise recovery promoting tissue cooling, local analgesia, and preventing DOMS [7]. In addition, it reduces blood flow, intramuscular metabolic rate, and nerve conduction while minimizing tissue damage caused by hypoxia [8]. In this aspect, Glasgow et al. [9] reported increased DOMs and decreased muscle strength 48 h after eccentric exercise; however, duration and temperature of CWI had no significant effects on indicators or measurements related to DOMs. Contrarily, Ingram, Dawson, Goodman, Wallman, and Beilby [10] reported decreased DOMs and isometric strength deficit in a group that performed CWI after exhaustive exercise compared to another group that did contrast water immersion and a control group. In addition, Peiffer et al. [11] reported that CWI decreases the skin and rectal temperature, but has a negative effect on neuromuscular function. These mentioned studies show a range of physiological results that could be led by CWI.
Body temperature is recognized as a key indicator for human health [1, 12, 13]. Specifically, in DOMS, blood flow increases due to the inflammatory process resulting from muscle damage, and consequently causes an increased local temperature [14]. There are several techniques to assess and monitor skin temperature (Tsk) using, for example, mercury thermometers, contact thermosensors, or infrared thermography (IRT) [15, 16]. IRT has been highlighted at the clinical and experimental levels as a technology able to analyze organic tissues in various pathophysiological and physical stress situations, being used to investigate the relationship between pain and Tsk [17], to evaluate the Tsk response after incremental maximal test [18], to study Tsk response following breath training [19], and also for monitoring the Tsk during exercise and rehabilitation [20].
Although IRT has been widely used in the sport training area, few studies have focused on assessment by IRT of the thermal responses to eccentric exercise, in detecting DOMS [21, 22], and also in measuring the effects of CWI [23]. Studies with IRT have shown a decreased Tsk during aerobic exercise [24] and immediately after intense exercise [25, 26]. Tsk over the muscle is most likely influenced by the higher supply of blood flow to the muscle, being this mechanism dependent on the exercise modality applied [27]. However, there are no studies with IRT investigating the relationship between plyometric exercise and skin temperature. Thus, the aim of this study is to investigate the change in the skin temperature after exercise-induced DOMS and after cold water immersion during a recovery period. In addition, we hypothesized that DOMS and strength inhibition can be minimized by cold water immersion.
Materials and methods
Experimental design
This study was composed to 45 subjects. All subjects were within the following inclusion criteria: being considered physically active according to the IPAQ short version [28], aged between 18 and 25 years, BMI ≤25, without cardiovascular disease, musculoskeletal injuries, previous surgery and not have presented pain in the lower limbs in the last 3 months. In addition, volunteers should not present any other disease that could alter the body temperature. Seven subjects who lacked any assessment or those who failed to complete the experimental protocol were excluded from the study. The final sample (n = 38) was randomly divided into four groups: group 1 (G1) which performed aerobic exercise (n = 9, 8F/1 M, 22.6 ± 2.1 years, 59.4 ± 12.7 kg and 1.66 ± 0.07 m); group 2 (G2) who only performed plyometric exercise (n = 10, 8F/2 M, 21.2 ± 1.5 years, 59.1 ± 11.4 kg and 1.63 ± 0.07 m); group 3 (G3) who were submitted to plyometric exercise+cold water immersion protocol (n = 9, 8F/1 M, 22.0 ± 2.8 years, 58.5 ± 15.6 kg and 1.66 ± 0.08 m); and group 4 (G4) who were the non-exercised control group (n = 10, 9F/1 M, 22.4 ± 1.3 years, 57.7 ± 9.4 kg and 1.64 ± 0.07 m). The experimental design is presented in Fig. 1.
The sample size test for each group condition was determined in GPower (version 3.1.9.2; Franz Faul, Universität Kiel, Germany) [29]. The following design specifications were taken into account: α = 0.05; (1 − β) = 0.8; effect size f = 0.4; test family = F test and statistical test = ANOVA repeated measures, using within–between interaction. The sample size estimated according to these specifications was eight subjects for each group.
Exercise protocol
All subjects were previously instructed on the procedures of the exercise protocol and performed a warm-up session with active stretching (1 rep, 20 s) of the hip and knee flexors and extensors, plantar flexors, as well as their ankle dorsiflexors and paraspinals muscles. G1 group performed an aerobic exercise protocol consisting of 15 min running on a treadmill at 80% intensity of the age-predicted maximal heart rate (HRmax) = 220 − age [30]; G2 group performed an anaerobic exercise protocol consisting of 100 Squat-jumps without any external weight (5 sets of 20 repetitions) with 2 min of rest between sets [31], and G3 performed the same protocol as G2, but the subjects were immersed in a cold water barrel at 10 °C up to hip level after the exercise, and remained there for 15 min. Water temperature was continuously monitored using a model 62 infrared thermometer (FLUKE—USA) and adjusted its temperature when necessary by adding ice [32]. G4 was a non-exercised control group and remained at rest in the standing position during the entire trial period.
Thermographic evaluation
Before the test, subjects acclimatized for 15 min standing in a room with temperature between 22 and 24 °C and relative humidity <50%, with the room condition monitored by a thermohygrometer (Instrutemp, ITHT2250, China).
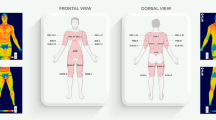
Two thermographic images were recorded at each time; one of the anterior thigh and other of the face (see Fig. 2). Subjects were in the orthostatic anterior anatomical position. The images were collected with a model T-360 thermographic camera (FLIR Systems, Sweden) with thermal sensitivity of 0.05 °C and a resolution of 320 × 240 pixels, spectral range of 7–13 µm and accuracy of ±2%. The camera was set on a tripod, positioned at 3 and 1 m of the subjects for the thigh and face views, respectively. The emissivity value adopted for human skin was 0.98 [33]. Both region of interests (ROI) (thigh and forehead) were delimited manually and absolute mean temperature of each ROI, which is the mean of temperature pixels within the ROI, was obtained using the thermography software Thermacam Reporter version 8.2 (FLIR Systems, Sweden). Therefore, the quadrangles configuration was determined as follows: (a) thigh: from 2 cm above the patella superior border to 5 cm bellow the inguinal line and (b) forehead: central region of the forehead equidistant from the sides. A visual example of the ROIs is shown in Fig. 2.
Subjects’ Tsk was assessed at seven different moments: after 15 min of acclimatization (T-rest), immediately after exercise (T0), 10, 20, and 30 min (T10, T20, and T30), and 24 and 48 h (T24 and T48) after exercise. For G3, T10 assessment was performed after 15 min of cold water immersion, while T20 and T30 were performed 10 and 20 min after T10.
To capture the thermographic images, subjects were instructed to abstain from eating or drinking in the 2 h before the evaluation. Subjects were allowed to drink only 500 ml of water at ambient temperature during and 30 min after the physical effort. Water ingestion was ad libitum in the 48 h before the experiment. Subjects did not use any stimulant medications, vasodilators, thermogenics, or practice any kind of strenuous exercise 24 h before evaluation, nor did they apply any moisturizing lotion or any other similar product on their body. All subjects used bathing suits during evaluation. G3 used a bath towel to dry the body after CWI, but avoided rubbing the region of interest. All these described procedures were followed to respect the infrared thermography guidelines [12, 34]. During the 48 h follow-up after the experiment, the participants were instructed not to perform any type of exercise and maintain their usual diet.
Other recorded variables
The level of pain in the quadriceps was evaluated at times T-rest, T0, T24, and T48 for all groups by measuring the compressive force that induces grade 5 pain in the Visual Analogue Scale (VAS) [35]. The measurement was performed in the middle third of the thigh using a dynamometer (DD-300, Instruthem®, Brazil). The subject verbally informed the researcher when the pain reached grade 5 of VAS and the generated pressure was recorded.
Isometric knee extension strength was assessed at times T-rest, T0, T24, and T48 for all groups using a chair, adapted and locked at a knee flexion angle of 120° with the hip positioned at 90°. The subject was positioned seated in the Bonnet chair with stabilization straps on the trunk and pelvis and was asked to perform two maximal isometric knee extensions for 6 s., with 1 min interval between contractions. The strength was measured by a dynamometer (DD-300, Instruthem®, Brazil).
Data analysis
The temperature of the thigh was measured to evaluate local responses, and the forehead temperature was measured to evaluate central response. The forehead was used as a reference, since it is an important place to determine the body temperature [36]. Each thigh temperature data was used separately as there was no difference between limbs.
Data were analyzed using the Statistical Package for Social Sciences (SPSS—20.0) by means of descriptive and inferential statistics. Initially, data normality (Shapiro–Wilk test) and homogeneity of variances (Levene’s test) were observed, followed by applying the ANOVA for repeated measures (times × groups, with repeated measures on time) to compare the temperature variables, isometric strength of knee extension, and compressive force during pain induction. When a significant F value was found, a Post Hoc Tukey test was used for localizing the significant differences between groups. A significance level of 5% was considered for all analyses.
Results
Skin temperature results
Table 1 presents the results of skin temperature between groups in each moment of the experiment.
In the overall analysis of the subject’s anterior thigh, significant differences for times F (2.9, 210.2) = 35.751, p = 0.001 and interaction times × groups F (8.7, 210.2) = 38.721, p = 0.001 were verified for Tsk with between group effects F (3, 72) = 24.039, p = 0.001. A significant intergroup difference was verified for G3 compared with all other groups. In the intragroup analysis of the subject’s anterior thigh, a significant decrease for Tsk was observed at T0 for groups G1 (p = 0.001) and G2 (p = 0.004), and returned to baseline level at T20 and T10 (p = 0.72 and p = 0.056), respectively. In G3, Tsk decreased at T15 immediately after cold water immersion (p = 0.001) and only recovered partially at T20 and T30 (p = 0.001), with full recovery at T24 and T48 (p = 0.21 and p = 0.67), respectively. No intragroup differences were observed in G4 (p > 0.05).
In the overall analysis of the subject’s forehead, significant differences for Tsk were verified for times F (3.7, 128.6) = 19.117, p = 0.001 and interaction times × groups F (11.3, 128.6) = 4.997, p = 0.001 with between group effects F (3, 34) = 6.002, p = 0.002. A significant intergroup difference was verified for G2 compared with G1 (p = 0.011) and G4 (p = 0.002), but not for G3 (p = 0.345). In the intragroup analysis, there was a significant decrease in forehead TsK immediately after exercise (T0) for G1 (p = p = 0.005), which recovered at T10 (p = 0.09) when compared to T-rest. For G2 and G3, a decreased Tsk was observed at T0 (p = 0.001 and p = 0.029) which recovered at T20 and T10 (p = 0.143 and p = 0.312), respectively, when compared to T-rest. Control group (G4) presented no difference during the experimental period.
Dynamometry results
In the overall analysis of the subject’s isometric strength, significant differences were verified for times F (2.4, 83.2) = 4.824, p = 0.007 and interaction times × groups F (7.3, 83.2) = 2.433, p = 0.024, but no between group effects F (3, 34) = 0.137, p = 0.937. In the intragroup analysis, G2 group’s strength decreased at T0 when compared to T-rest (p = 0.028). Despite G3 group presenting no change between T-rest and T0 (p > 0.05), a significant increase in isometric strength was observed at T24 compared to T0 (p = 0.016). Dynamometry results did not significantly change in G1 or G4 during the whole experimental period (Table 2).
Compressive force for pain induction
In the overall analysis of pain, significant differences were verified for times F (3, 102) = 3.127, p = 0.029 but not interaction for times × groups F (9, 102) = 1.685, p = 0.102) or between group effects F (3, 34) = 0.884, p = 0.428. It was observed that the compressive force for grade 5 pain induction (VAS) had a significant difference at T48 compared to T-rest (p = 0.012), but a decrease in the intragroup analysis was only observed for G2 when T24 or T48 and T0 were compared (p = 0.038 and 0.019), respectively. However, there was no significant difference between groups (p > 0.05) (Table 3).
Discussion
This study provides important findings which point to a higher skin temperature in the anterior thigh muscles which performed plyometric exercise. This thermographic change was accomplished by a decrease in the knee extensor strength immediately after exercises. In addition, it seems clear from our data that CWI was effective in reducing the thigh’s TsK until 30 min after exercise, but had no effect on the thigh’s TsK 24 and 48 h after exercise. CWI attenuated pain inducted by muscle compression 48 h after plyometric exercise.
In terms of skin temperature response after exercise, the significantly lower Tsk on the thigh area immediately after plyometric exercise in group G2 (−0.5 °C; p < 0.05) agrees with previously published studies with resistance exercise [25, 37] that reported Tsk reductions on the agonist muscle areas after effort; however, the significant TsK reductions obtained in group G1 (1.3 °C; p < 0.05) were not obtained in the study by Fernández-Cuevas et al. [25] immediately after aerobic exercise.
The blood flow in short duration and high intensity exercises, as the plyometric exercise protocol used in the present study, is directed predominantly to the contracted muscles (i.e., quadriceps) reducing the peripheral blood supply of the skin on this area [13]. However, an aerobic exercise normally maintains Tsk in the areas corresponding to the exercised areas [25]; these results are not the same as obtained in our study. Perhaps due to some characteristics of the sample (14 males in the Fernández-Cuevas et al. [25] vs. one male and eight females in our sample) and the main exercise performed (15 min at 80% in our study vs. 40 min at 60–75% of the maximum heart rate) could lead to a different response by the subjects to the exercise. The environmental conditions were quite similar and should not have influenced the results.
Resting skin temperature showed a variation between groups. There are various factors that interfere in individual skin temperature at rest, which include anthropometry, metabolic rate, and skin blood flow [12]. Regarding the temperature of the thigh, although it presented a difference of 0.8 °C in T-rest (G4 vs. G3), this variation can be considered as normal, since skin temperature adjusts dynamically to preserve the internal temperature [38]. In addition, considering the temperature of the forehead, some studies have shown that the emotional status influences its regulation [39], which can be one of the causes of the variation of 1 °C (G1 vs. G2) presented in the present study.
Various authors have studied the temperature (Tsk) on exercising muscles using infrared thermography [1, 26]. These studies have shown no change or a decrease [26] in skin temperature immediately after exercise [1]. Adamczyk et al. [26] carried out a comparative study using three types of warm-ups (jogtrot, jogtrot+stretching, and dynamic exercises with an elastic band) at moderate intensity (10 min, 75% HRmax) on Tsk of the quadriceps muscle. The authors showed a significant decrease in Tsk only in groups that performed the protocol of doing specific exercises with the elastic band. On the other hand, studies using thermosensors implanted in the muscle indicate that there is an increased tissue temperature [2, 27]. These authors hypothesized that there may be a link between muscle temperature and sweating during exercise [40]. According to Petrofsky [5], the maintenance of increased blood supply to the muscles after exercise can keep the muscle and the overlying skin heated, since the core blood is warmer.
In this study, no changes in skin temperature could be associated to DOMS. Temperature increased after plyometric exercise and returned to baseline level after 24 h. These results are in contrast with the study published by Bandeira et al. [41], who found an increase in temperature of the quadriceps in a group of adolescent athletes who underwent an eccentric/concentric high-intensity quadriceps training (80% RM) compared to controls trained at moderate intensity (running at 50–60% of HR max). In the same direction, Al-Nakhli et al. [14] also reported increased skin temperature on the Biceps Brachial muscle for subjects 24 h after an exercise protocol to induce DOMS. However, these authors used exercise protocols directed to a specific muscle group, while exercise protocols in this study were carried out involving several muscle groups to simulate real condition training. In addition, the intensity of the plyometric exercise protocol applied may have been higher compared to the resistance exercise of the cited studies. In the present study, the squat-jumps were performed with fixed repetitions and using only the body weight, which can lead to high intensities depending on the weight and physical capacity of the participant.
The results of this study show that the implemented CWI protocol was effective for cooling anterior thigh muscles. Only the group submitted to CWI after plyometric exercise had a decreased skin temperature that was maintained until 30 min after CWI; however, this effect did not remain at 24 or 48 h after exercise. These findings are in agreement with other studies [11, 16] which demonstrate the effectiveness of CWI for acute temperature decrease, although the effect on muscle temperature after 24 or 48 h of CWI procedure has not been tested.
In this work, isometric muscular strength decreased immediately after exercise in subjects that performed plyometric exercise in both groups G2 and G3 (39.6 Kgf before and 26.0 Kgf immediately after exercise in G2, and 41.2 Kgf before and 31.9 Kgf immediately after exercise in G3); however, only the group submitted to post-exercise CWI recovered completely after 24 h. This fact shows a possible attenuation of strength deficit in the group submitted to Plyo+CWI (G3). In this same direction, our findings of induced pain correlate with this data, since only G2 presented a decrease of compressive force for pain induction 48 h after plyometric exercise. These results can be interpreted as one of the long-term effects of DOMS.
CWI was apparently effective in mitigating the loss of strength due to DOMS in G3. Several authors [10, 32, 42] have reported attenuation of muscle damage and DOMS in a group that performed CWI compared to the control. These authors used isometric strength and the visual analogue scale of pain among other variables indicative of DOMS, but they did not examine skin temperature. In contrast, other authors [9, 43] did not report a significant intergroup difference for muscle damage marker or DOMS when comparing CWI with different intensities, short contrast immersion, or the control group. In this aspect, Goodall and Howatson [31] studied two groups of subjects submitted to a protocol of 100 drop jumps with and without CWI application immediately, 24 and 48 h after exercise. The authors also did not observe an enhancement in muscle damage variables, including isometric strength, in subjects submitted to CWI.
Despite the significant findings, interpretation of these data should considerer certain attention. This study did not control the sweat rate of subjects in each group, which may represent a limitation as this can influence the temperature measured on the skin. Furthermore, the study population was limited to healthy adults from both sexes, and the thermal response may be different for other age groups, between sexes, and unhealthy populations. In future studies, it is believed that a longer aerobic exercise using the same gender athletes and sweat rate control can make the power of comparing anaerobic exercises more sensitive. Another limitation of our study is that the number of repetitions of plyometric protocol was fixed for all participants which can lead different workloads. Future studies have to considerer the relative intensity of plyometric exercise to obtain a better relationship between exercise intensity and skin temperature response.
Conclusion
This paper presents evidence that plyometric anaerobic exercise leads to a higher skin temperature on the exercised muscle groups than aerobic exercise and that CWI application has no influence on muscle temperature after 24 h of exercise, while it minimizes DOMS and the deficit of isometric force. Infrared thermography was more sensitive in evaluating exercises with different loads when expressed as a relative rate to forehead temperature. These results support the use of CWI protocols to minimize the deleterious effects of heavy exercise on muscle function.
References
Ferreira JJ, Mendonca LC, Nunes LA, Andrade Filho AC, Rebelatto JR, Salvini TF (2008) Exercise-associated thermographic changes in young and elderly subjects. Ann Biomed Eng 36(8):1420–1427
Kenny GP, Reardon FD, Zaleski W, Reardon ML, Haman F, Ducharme MB (2003) Muscle temperature transients before, during, and after exercise measured using an intramuscular multisensor probe. J Appl Physiol 94(6):2350–2357
Racinais S, Oksa J (2010) Temperature and neuromuscular function. Scand J Med Sci Sports 20(s3):1–18
Proske U, Morgan D (2001) Muscle damage from eccentric exercise: mechanism, mechanical signs, adaptation and clinical applications. J Physiol 537(2):333–345
Petrofsky JS, Laymon M, Berk DRL, Al-Nakhli HH, Banh A, Eisentrout A, Tokar A, Valentine M, Batt J (2012) A pilot study using blood biomarkers and physiological parameters to assess thermacare heat wraps for efficacy and timing of application to reduce delayed onset muscle soreness from exercise. J Appl Res 11(2):84–96
Hedayatpour N, Hassanlouei H, Arendt-Nielsen L, Kersting UG, Falla D (2011) Delayed-onset muscle soreness alters the response to postural perturbations. Med Sci Sports Exerc 43(6):1010–1016
Barnett A (2006) Using recovery modalities between training sessions in elite athletes. Does it help? Sports Med 36(9):781–796
Costello JT, Donnelly AE, Karki A, Selfe J (2014) Effects of whole body cryotherapy and cold water immersion on knee skin temperature. Int J Sports Med 35(1):35–40
Glasgow PD, Ferris R, Bleakley CM (2014) Cold water immersion in the management of delayed-onset muscle soreness: is dose important? A randomised controlled trial. Phys Ther Sports 15(4):228–233
Ingram J, Dawson B, Goodman C, Wallman K, Beilby J (2009) Effect of water immersion methods on post-exercise recovery from simulated team sport exercise. J Sci Med Sport 12(3):417–421
Peiffer JJ, Abbiss CR, Nosaka K, Peake JM, Laursen PB (2009) Effect of cold water immersion after exercise in the heat on muscle function, body temperatures, and vessel diameter. J Sci Med Sport 12(1):91–96
Fernández-Cuevas I, Marins JCB, Lastras JA, Carmona PMG, Cano SP, García-Concepción MÁ, Sillero-Quintana M (2015) Classification of factors influencing the use of infrared thermography in humans: a review. Infrared Phys Tech 71:28–55
Vainer BG (2005) FPA-based infrared thermography as applied to the study of cutaneous perspiration and stimulated vascular response in humans. Phys Med Biol 50(23):R63–R94
Al-Nakhli HH, Petrofsky JS, Laymon MS, Berk LS (2012) The use of thermal infra-red imaging to detect delayed onset muscle soreness. J Vis Exp 59:e3551
Byrne C, Lim CL (2007) The ingestible telemetric body core temperature sensor: a review of validity and exercise applications. Br J Sports Med 41(3):126–133
Costello JT, McInerney CD, Bleakley CM, Selfe J, Donnelly AE (2012) The use of thermal imaging in assessing skin temperature following cryotherapy: a review. J Therm Biol 37(2):103–110
Rossignoli I, Fernández-Cuevas I, Benito PJ, Herrero AJ (2016) Relationship between shoulder pain and skin temperature measured by infrared thermography in a wheelchair propulsion test. Infrared Phys Technol 76:251–258
Ludwig N, Trecroci A, Gargano M, Formenti D, Bosio A, Rampinini E, Alberti G (2016) Thermography for skin temperature evaluation during dynamic exercise: a case study on an incremental maximal test in elite male cyclists. Appl Opt 55(34):D126–D130
Ludwig N, Gargano M, Formenti D, Bruno D, Ongaro L, Alberti G (2012) Breathing training characterization by thermal imaging: a case study. Acta Bioeng Biomech 14(3):42–47
Hildebrandt C, Raschner C, Ammer K (2010) An overview of recent application of medical infrared thermography in sports medicine in Austria. Sensors 10(5):4700–4715
Al-Nakhli HH, Petrofsky JS, Laymon MS, Berk LS (2012) The use of thermal infra-red imaging to detect delayed onset muscle soreness. J Vis Exp 59:e3551–e3551
Al-Nakhli HH, Petrofsky JS, Laymon MS, Arai D, Holland K, Berk LS (2012) The use of thermal infrared imaging to assess the efficacy of a therapeutic exercise program in individuals with diabetes. Diabetes Technol Ther 14(2):159–167
Costello JT, McInerney CD, Bleakley CM, Selfe J, Donnelly AE (2012) The use of thermal imaging in assessing skin temperature following cryotherapy: a review. J Therm Biol 37(2):245–274
Fernandes AA, Amorim PRS, Brito CJ, Moura AG, Moreira DG, Costa CMA, Sillero-Quintana M, Marins JCB (2014) Measuring skin temperature before, during and after exercise: a comparison of thermocouples and infrared thermography. Physiol Meas 35(2):189
Fernández-Cuevas I, Sillero-Quintana M, Garcia-Concepcion MA, Serrano JR, Gómez-Carmona P, Marins JB (2014) Monitoring Skin Thermal Response to Training with Infrared Thermography. New Stud Athl 29:57–71
Adamczyk JG, Boguszewski D, Siewierski M (2012) Physical effort ability in counter movement jump depending on the kind of warm-up and surface temperature of the quadriceps. Balt J Health Phys Act 4(3):164–171
Todd G, Gordon CJ, Groeller H, Taylor NA (2014) Does intramuscular thermal feedback modulate eccrine sweating in exercising humans? Acta Physiol 212(1):86–96
Silva GSF, Bergamaschine R, Rosa M, Melo C, Miranda R, Bara-Filho M (2007) Evaluation of the physical activity level of undergraduation students of health/biology fields. Rev Bras Med Esporte 13(1):39–42
Faul F, Erdfelder E, Lang AG, Buchner A (2007) G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 39(2):175–191
Tanaka H, Monahan KD, Seals DR (2001) Age-predicted maximal heart rate revisited. J Am Coll Cardiol 37(1):153–156
Goodall S, Howatson G (2008) The effects of multiple cold water immersions on indices of muscle damage. J Sports Sci Med 7(2):235–241
Bailey DM, Erith SJ, Griffin PJ, Dowson A, Brewer DS, Gant N, Williams C (2007) Influence of cold-water immersion on indices of muscle damage following prolonged intermittent shuttle running. J Sports Sci 25(11):1163–1170
Ammer K (2008) The Glamorgan Protocol for recording and evaluation of thermal images of the human body. Thermol Int 18(4):125–129
Ring EFJ, Ammer K (2000) The Technique of Infra red Imaging in Medicine. Thermol Int 10(1):7–14
Price DD, McGrath PA, Rafii A, Buckingham B (1983) The validation of visual analogue scales as ratio scale measures for chronic and experimental pain. Pain 17(1):45–56
Machado-Moreira CA, Wilmink F, Meijer A, Mekjavic IB, Taylor NA (2008) Local differences in sweat secretion from the head during rest and exercise in the heat. Eur J Appl Physiol 104(2):257–264
Formenti D, Ludwig N, Trecroci A, Gargano M, Michielon G, Caumo A, Alberti G (2016) Dynamics of thermographic skin temperature response during squat exercise at two different speeds. J Therm Biol 59:58–63
Kenny GP, Journeay WS (2010) Human thermoregulation: separating thermal and nonthermal effects on heat loss. Front Biosci 15:259–290
Paolini D, Alparone FR, Cardone D, van Beest I, Merla A (2016) “The face of ostracism”: the impact of the social categorization on the thermal facial responses of the target and the observer. Acta Psychol 163:65–73
Kenny GP (2014) Muscle temperature and sweating during exercise: a new link? Acta Physiol 212(1):11–13
Bandeira F, Muniz de Moura MA, Abreu de Souza M, Nohama P, Borba Neves E (2012) Can thermography aid in the diagnosis of muscle injuries in soccer athletes? Rev Bras Med Esporte 18(4):234–239
Ascensao A, Leite M, Rebelo AN, Magalhaes S, Magalhaes J (2011) Effects of cold water immersion on the recovery of physical performance and muscle damage following a one-off soccer match. J Sports Sci 29(3):217–225
Sellwood KL, Brukner P, Williams D, Nicol A, Hinman R (2007) Ice-water immersion and delayed-onset muscle soreness: a randomised controlled trial. Br J Sports Med 41(6):392–397
Acknowledgements
The National Research Council—CNPq (Brazil), which funded this research project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical Standards of the institutional or national research committee and with the 1964 Helsinki declaration and its later amendments or compatible ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in this study.
Rights and permissions
About this article
Cite this article
Silva, Y.A., Santos, B.H., Andrade, P.R. et al. Skin temperature changes after exercise and cold water immersion. Sport Sci Health 13, 195–202 (2017). https://doi.org/10.1007/s11332-017-0353-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11332-017-0353-x