Abstract
The influence of stand age (5, 10, 15, 20, 30 and 40 years) on the decomposition of litter fractions, nutrient and energy release of mixtures of N2-fixing alder (Alnus nepalensis) and non-N2-fixing large cardamom (Amomum subulatum) systems was compared. Seasonal decomposition rates were distinct with the highest rate in the first 6 months followed by subsequent seasons. The decomposition rate was substantially high in younger stands (10- to 15-years) and declined in the older stands. Heat sink from the stand floor litter increased from 171 × 106 kJ year−1 in 5 years to 299 × 106 kJ year−1 at 15 years and then considerably decreased with advancing age. However, energy and nutrient releases were slow at a high initial lignin-to-initial N ratio and C-to-N ratio, and there was an inverse relationship between the k-value of ash-free-mass and N expressed as a function of the C-to-N ratio. Quantities of nutrient release and energy loss per unit area in 24 months of decomposition were highest in 15 years and subsequently they lowered with advancing age. Nutrient loss indicated approximately uniform absolute and relative rates. Absolute energy consistently decreased by 81–88% in 24 months. Ash-free mass of decomposing litter remaining at different retrieval dates was associated with a narrowing of the C-to-N ratio. The relative loss rate of ash-free mass, nutrients and energy content was strongly related to the C-to-N ratio, litter temperature and litter moisture. The influence of Alnus in the younger stands on nutrient and energy releases were rapid, indicating accelerated nutrient cycling and energy dynamics. The intensity of the processes was highly phenomenal and considerably high in younger stands up to 20 years. Thus, an appropriate management cycle of the Alnus-cardamom system for sustainability is 15–20 years.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Large cardamom (Amomum subulatum) is the most important perennial cash crop of the eastern Himalayan region, with 30,000 ha cultivated in Sikkim and Darjeeling between elevations of 600 and 2,000 m (Spices Board 2001). In recent years, the cultivation of large cardamom has been extended in eastern Nepal and southern Bhutan in the eastern Himalayas. Its capsule (fruit) is used as a spice/condiment and contains about 3% essential oil rich in cineole (Gupta et al. 1984). It is cultivated as an understorey crop with Himalayan alder (Alnus nepalensis) as a shade tree; both are native to the region. The increased productivity of plants growing near N2-fixing species has long been recognized.
Studies on litter production and decomposition dynamics of managed agroforestry systems are limited. There has been a growing effort to include N2-fixing species in agroforestry systems of tropic and temperate regions, which is a new intervention for influencing the soil fertility and quick nutrient dynamics. There are reports of much greater litter production in mixed stands of tree plantations with N2-fixing associates than in stands containing only non-N2-fixing trees (Tarrant et al. 1969; Binkley et al. 1992a). Litter of N2-fixing species generally decomposes faster, and the addition of N2-fixing tree litter may accelerate the decomposition of non-N2-fixing litter types (Taylor et al. 1989). The ratio of initial lignin-to-N and C-to-N predicts litter decomposition well in temperate forests (Berg and McClaugherty 1987; Osono and Takeda 2004). The decomposition of N2-fixing litter is typically much higher than those of other species, although decomposition rates can vary substantially among N2-fixing trees (Sankaran et al. 1993; Mwiinga et al. 1994).
Many litter decomposition studies have been carried out in the tropics (Mugundi and Nair 1996; Loranger et al. 2002; Kwabiah et al. 1999; Seneviratne 2000), but the literature is sparser for the Himalayan region. Nitrogen and P are essential elements limiting litter decomposition (Berg 1986; Melillo et al. 1989; Aber et al. 1990; Bargali et al. 1993). Some studies have analyzed the dynamics of N and P based on the relationship with lignin (Aber and Melillo 1982; Berg and McClaugherty 1989; Osono and Takeda 2005) and have suggested that lignin is an indicator for N and P dynamics.
Inclusion of mixtures of N2-fixing Alnus in large cardamom agroforestry in the eastern Himalayas is a good example for understanding the influence of stand age and performance. Few reports on accelerated cycling of N and P in the mixed stands of N2-fixing Alnus and Albizia are available (Binkley et al. 1992b; Sharma et al. 1994). Sharma et al. (2002a, b) carried out a detailed study to see the impact of age on the performance of Alnus-Cardamom agroforestry in the eastern Himalayas with respect to productivity, energetics, efficiency and nutrient dynamics. The present study quantifies the influence of stand age on nutrient cycling of mixed N2-fixing tree- and non-N2-fixing cash crop-based agroforestry and on the decomposition and dynamics of energy and nutrient release from the litter fraction. It also deals with the initial lignin-to-N ratio as a determinant of N and P dynamics along the agroforestry age series for understanding the interaction between the litter lignin and relative changes in decomposition with time.
Methods
Study area
Three experimental sites selected for the study are located at Kabi (north district), Thekabong (east district) and Sumik (east district) of Sikkim in the eastern Himalayas. These sites extend within 27°15′ 05′′ to 24°17′ 36.6′′ N latitude and 88°28′ 6.9′′ to 88°39′ 27.1′′ E longitude with an elevation distribution between 1,350 and 1,600 m asl. These study sites are in the Indian monsoon region with a temperate climate. Mean monthly maximum temperature ranged from 14.3 to 23.3°C, mean monthly minimum temperature from 5.4 to 15.8°C and rainfall from 2,500 to 3,500 mm at the study sites. Relative humidity varied between 80 and 95% during the rainy season and decreased to about 45% in spring.
All three experimental sites have several pure agroforestry stands of large cardamom grown under A. nepalensis shade trees. These Alnus-cardamom agroforestry stands at each of the sites represented an age series of 5, 10, 15, 20, 30 and 40 years numbering 18 plots altogether. Age designation refers to the same age of both large cardamom and Alnus as they are planted at the same time. The study sites at three locations were closely comparable; the structural and functional differences were attributed to the age of the agroforestry systems. The soils are in general acidic in reduction due to heavy rainfall and leaching of bases from surface soil to represent Typhic Hapludolls and Dystric Eutrochrepts largely under cardamom plantations. Red and yellowish podzolic soils are found in the agroforestry stands of Sikkim (Mukhopadhyaya 1998). The soil was acidic (pH 3.8–5.6), and pH varied widely with depth; the variation in pH of surface soils was small between stands (17%). The soil was sandy loam in composition ranging from 11 to 30% clay, 15 to 40% silt and 34 to 65% sand.
Litter decomposition
Sample plots of 30 × 40 m were marked at each of the six Alnus-cardamom agroforestry stands (5, 10, 15, 20, 30 and 40 year stands), with all three sites giving a total of 18 plots (0.36 ha per age group, totaling 2.16 ha). Litter production and decomposition were estimated at each of the plots. Litter decomposition studies were carried out by using the litterbag technique (Witkamp and Olson 1963). Freshly fallen litter from Alnus and slashed cardamom pseudo-stem and leaves was collected in December 1997. Dried litter was placed in nylon litterbags (20 × 20 cm) with a mesh size of 1 mm to permit only the movement of microarthropods and other microbial invasion. At the time of cardamom harvest fruited pseudo-stems with leaves were harvested as part of management practice. Monthly litter production estimations were carried out (1998–2000) using five litter traps of 1 m2 collecting area in each sample plot and pooled to annual values. The equivalent of 10 g dry weight litter from air-dried stock was placed in each litterbag. This litter weight per unit area of litterbags falls within the range of annual litter production weights per unit area on the floor.
Four major litter fractions comprised of Alnus leaf (AL), Alnus twig (AT), cardamom leaf (CL) and cardamom pseudo-stem (CPS) were sampled for decomposition. A total of 864 litterbags were placed within a plot size of 4 × 4 m in 18 plots (288 m2) at different sample sites in the age series of Alnus-cardamom agroforestry stands in January 1998. Litterbags containing litter fractions were retrieved in replicates of three at each collection time of 3, 6, 12 and 24 months from the date of placement for analysis.
Samples were cleaned carefully and oven-dried at 80°C for 48 h, weighed and ground to pass through a 2-mm sieve. Ground samples were analyzed for total nitrogen by modified Kjeldahl method and phosphorus following ascorbic acid method (Anderson and Ingram 1993). Lignin of the decomposing litter fraction was estimated by using the Tecator Fibertec system. Carbon contents of the plant samples were estimated by assuming 48% carbon at different stages of decomposition (Sharma and Ambasht 1987).
Energy value on an ash-free mass basis was estimated by using an oxygen bomb calorimeter (Lieth 1975). The mean energy content of floor litter was calculated by analyzing the calorific content of different litter layers estimated during different intervals of decomposition for all age groups of agroforestry stands. The sum of the total heat release values of different stages of decomposing litter samples per year gave the total heat release from the floor litter (Sharma et al. 1997). The sum of the heat release values from different stages of decomposing litter samples per year represented the total heat release from the floor litter. Rainfall data were recorded using an automatic weather station (Campbell Scientific Inc., UT). Temperature measurements were made using a soil thermometer placed within the litter–soil interface at every stand. Statistical analysis was done using Systat version 6.0. Loss of ash-free mass, N, P and energy contents from decomposing litter fractions was described by a single exponential decay function. The half-life (T 1/2) was calculated using the decomposition constant (k) as 0.693/k (Olson 1963).
Results
Litter composition and initial chemical properties
Among the litter fractions, the concentrations of nutrients were highest in leaves of both Alnus (N 2.63–3.64%; P 0.15–0.21%) and understorey cardamom (N 1.97–2.16%; P 0.09–0.17%). Concentrations were comparatively low in Alnus twig (N 0.78–0.95%; P 0.02–0.05%) and cardamom pseudo-stems (N 0.33–0.61%; P 0.03–0.08%).
Ratios of dry litter composition through production by both N2-fixing Alnus and non-fixing understorey cardamom and their chemical content were highly variable in the age sequence. The production of litter by the 5-year stand was 5.88 t ha−1 (Alnus 64%, cardamom 36%). This subsequently increased to 10.25 t ha−1 (Alnus 60%, cardamom 40%) at age 15 and consistently declined to >50% by age 40 years. Litter production increase was attributed to an increase in tree and understorey cardamom bush density from 5-year (347 trees ha−1, 6,786 bush ha−1) to 15-year (553 trees ha−1, 25,316 bush ha−1) stands. After 15-year stands, tree density decreased with advancing age due to thinning as a management practice, while cardamom bush also decreased in aging agroforestry stands. The stand dry mass contribution of Alnus was 56–69%, while cardamom contributed between 31 and 44%. Similarly, stand nutrient contribution by Alnus (66–81% N, 52–79% P) was higher than cardamom (20–34% N, 22–48% P). The initial N content was significantly correlated to the initial P content of the litter fractions of all stands (r 2 = 0.94, P < 0.001). The mean ratio of contents of N and P was 19:01.
The percent initial lignin and initial lignin-to-N ratio in litter fractions were variable. Percent lignin was highest in Alnus leaf (32.63 ± 2.31) followed by Alnus twig (26.45 ± 1.32), and comparatively less in cardamom leaf (21.31 ± 1.89) and cardamom pseudo-stem (22.37 ± 1.17). Percent initial lignin-to-N ratio was highest in cardamom pseudo-stem (34.54 ± 1.62) followed by Alnus twig (25.21 ± 1.78), cardamom leaf (15.66 ± 2.41) and Alnus leaf (13.15 ± 1.48). Lignin concentration remained relatively constant or fairly increased during the first 3 months, and subsequently decreased thereafter during the decomposition period.
Mass loss rate of litter and nutrient and energy release during decomposition
The percent ash content ranged from 3.05 to 16.30 in the litter fractions with the highest value in Alnus leaf and lowest in cardamom pseudo-stem at different stages of decomposition. As the decomposition progressed, percent ash increased consistently with time, whereas ash-free mass remaining in decomposing litter fractions during each sampling date decreased significantly (P < 0.0001) among stand age, retrieval date and litter fraction. The ash-free mass remaining in the Alnus leaf ranged within 15–32%, Alnus twig 28–47%, cardamom leaf 17–38% and pseudo-stem 18–41%, during the 24 months of decomposition.
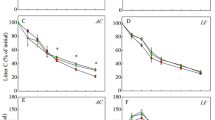
Decomposition and ash-free mass loss were not conspicuous during the first 3 months, while in the first 6 months, about 30–40% of initial mass loss was recorded. The environmental factors relating to decomposition were favorable during the warm rainy season and extremely slow during dry winter. Patterns of ash-free mass loss in different litter fractions in 24 months was in the order of Alnus leaf > cardamom leaf > cardamom pseudo-stem > Alnus twig, while between stand ages it was in the order 15 > 10 > 5 > 20 > 30 > 40 year stand. The T 1/2 values of ash-free mass among the litter fractions was lowest in Alnus leaf (8.84–14.85 months) and highest in Alnus twig (13.41–17.69 months), indicating that the mass loss was much more rapid in leaf litter of N2-fixing Alnus. The half-life and turnover time of ash-free mass were lowest (8.84–13.41; 12.76–19.35) in the 15-year stand with a high decomposition constant (0.62–0.94). The decomposition constant (k) was lowest (0.43–0.57) in the 40-year stand with a corresponding high value of half life and turnover time of the litter fractions. The k value was recorded in the order 15 > 20 > 10 > 5 > 30 > 40 years in the age series of agroforestry stands (Table 1). The single exponential decay functions show that the k values decreased with stand maturity, indicating that loss was faster in younger stands. Relationships between ash-free mass remaining as a function of the C-to-N ratio of litter fractions were highly significant for all the agroforestry stand ages (r 2 = 0.76–0.91, P < 0.0005).
The dynamics of N, P and energy showed a similar pattern in all the agroforestry age sequence. Nitrogen and P remaining in decomposing litter fractions decreased significantly (P < 0.001) with advancing retrieval time, whereas the concentration increased as the decomposition progressed. An inverse relationship was recorded between the percent weight remaining and N concentration in the residual litter fraction in the age series (r 2 = 0.62–0.88, P < 0.005–0.0001). The 15-year-old stand consistently lost more N and P from the litterbags than other stands, while the 40-year-old stand showed the least. Within the litter fractions, nutrient loss was comparatively more in Alnus leaf followed by cardamom leaf. The nutrient turnover time and half-life in decomposing litter fractions were highly variable within the agroforestry stands in the order 15 > 20 > 10 > 5 > 30 > 40 years stand. Within the litter fractions, the half life and turnover time of N was highest in Alnus twig.
The quantities of nutrient release per unit area were highest in 15-year (N 36.56 g m−2, P 5.45 g m−2), about 1.6–2.5 times more for N and 2.5–3.6 more for P than that of 5- and 40-year-old stands (Table 2). The absolute release of nutrients in the age sequence was 15 > 20 > 10 > 5 > 30 > 40 years. The release of nutrients (N 6.02–10.84 g m−2, P 0.48–1.59 g m−2) was highest in the first 6 months and declined gradually with time. The nutrient loss and release pattern were the same for all agroforestry stand ages, and the relative mobility of nutrients was P > N; the absolute release of N in comparison to P was 10–15 times higher. The initial percentage of labile fraction of N was more than the P. The k value and turnover time of P ranged between 0.64 and 0.88 (highest) in Alnus leaf and 0.41–0.81 (lowest) in Alnus twig. The k value was comparatively higher for P than N in the litter fractions with the consequence that T 1/2 values were short for P. The T 1/2 values for both N and P leaf litter were lower, indicating faster decomposition (Table 1). Stand total nutrient release increased with age and peaked at intermediate 15-year-stands and decreased thereafter with a relatively very low value in 40 years. The contribution of nutrient release by Alnus litter (N 8.26–23.81 g m−2, P 0.87–3.17 g m−2) was fairly high compared to cardamom (N 6.27–12.72 g m−2, P 0.71–2.28 g m−2).
Energy content of the litter fraction decreased significantly at every retrieval date from the initial values throughout 24 months. The mean caloric content of Alnus leaf (20.92 ± 0.82%) was highest followed by Alnus twig (20.44 ± 0.16%), cardamom leaf (19.93 ± 0.22%) and cardamom pseudo-stem (15.02 ± 0.08%). The absolute energy content in decomposing litter fractions decreased significantly (P < 0.0001) with incubation time in the analysis of variance. Interactions between the stand ages and retrieval of litter fractions were also significant.
The energy loss per unit area almost doubled in the 15-year-old stand (55,025 kJ m−2) compared to the premature young and oldest stands (Table 2). The T 1/2 values (7.09–9.45 months) and turnover time (11.43–13.64 months) of energy in decomposing litter fractions were lower at the 15-year stand (Table 2). Half life and turnover time of energy in Alnus leaf were lowest (7.09–9.56; 11.43–13.39 months) among the litter fractions. The energy loss pattern was similar in all the stand ages and the sequence of absolute energy loss was 15 > 20 > 10 > 5 >30 > 40 year stand. The sampling interval and cumulative loss from the decomposing litter fraction showed that a considerable amount of energy was lost during 24 months, with the highest loss (30–40%) during the first 6 months. Energy loss was more from Alnus leaf and least from Alnus twig among the litter fractions. Loss of energy from the litter fractions after 6 months of decomposition was relatively uniform and closely resembled ash-free mass losses in all the agroforestry stand age sequences.
Relationships between decomposition rates, litter quality and environmental factors
Multiple regression analysis was carried out to understand the relationships of ash-free mass with the C-to-N, C-to-P and N-to-P ratios remaining in litter for the retrieval dates of each age, but the ash-free mass appeared to be strongly related to the C-to-N ratio only (Table 3). Thus, relationships between the ash-free mass and C-to-N ratio were observed in the simple linear regression analysis. It showed that the reduction in the ash-free mass remaining for all the litter fractions was associated with narrowing of the C-to-N ratio in the agroforestry stands. Coefficients of determination, variance ratio and intercepted slopes of ash-free mass as a function of the C-to-N ratio of retrieved decomposing litter fraction in the age series of agroforestry stands were highly significant (r 2 = 0.76–0.91, P < 0.0005).
The relative loss rate of ash-free mass, nutrient and energy content was strongly related to the C-to-N ratio, litter temperature and litter moisture (Table 3). The relative loss rate of ash-free mass and nutrients of the litter fractions depended more strongly on litter quality followed by litter temperature and then litter moisture, whereas in the case of energy, litter temperature had a greater effect than the C-to-N ratio and litter moisture.
Discussion
The maximum litter decomposition was reported at the time of canopy closure (20 to 30 years of age) using the production to accumulation ratio in Douglas fir stands (Turner and Long 1975). It was supported by k values based on the litterbag technique after 12 months decomposition in the same stands (Edmonds 1979). Sharma and Ambasht (1987) reported the maximum decomposition at the time of canopy closure in a 30-year Alnus nepalensis stand. In the present study, using both the above methods maximum decomposition and heat release were recorded in the younger stands (10 to 15 year stands) with full canopy closure that declined with a decrease in tree density and crown cover. As a management practice, a substantial reduction of tree densities after heavy thinning has been found in the older agroforestry stands after 15 years. The decomposition constant (k) in alder-cardamom stands was high (0.59–1.05) at low half life (7.09–14.09 months) and turnover time (11.43–20.34 months). The lowest k value, half-life and turnover time at the 40-year stand were attributed to the low canopy closure and low tree density factor. The loss of initial ash-free mass after 12-month decomposition was 46–67%. This rate is comparable to 46–61% loss of ash-free mass per year in A. nepalensis (Sharma and Ambasht 1987) and 55% decomposition per year in A. rubra (Edmonds 1980) and A. crispa (Van Cleve 1971).
Decomposition rates of all the litter fractions are predicted from lignin or nutrient (N and P) content and the C-to-N ratio and lignin:N ratio. An inverse curvilinear relationship between the decomposition constants of ash-free mass and N expressed as a function of the ratio of initial lignin-to-N ratio was observed in the regression analysis, indicating that high levels of lignin may slow decomposition rates, which was also reported by Aber and Melillo (1982). A significant non-linear relationship was reported between relative initial proportions of labile to recalcitrant fraction of both initial lignin content and initial C-to-N ratio (Wieder and Lang 1982). Initial lignin content or initial C-to-N ratio of litter determines the decomposition rate to a considerable extent (Meentemeyer 1978; Melillo et al. 1982). Our study recorded slow decomposition at high initial lignin-to-N and C-to-N ratio. The relationship suggests that the higher the lignin content of the litter fraction, the larger the change in the N concentration of the material per unit carbon respired during decomposition (Aber and Melillo 1982).
Irrespective of agroforestry age and site, litter exhibited an increase in N concentration during decomposition, but the absolute N content decreased from the beginning, which is also reported in pure A. glutinosa and A. nepalensis stands (Bocock 1964; Sharma and Ambasht 1987). The reason would be due to the microbial invasion in litter fractions, mineralization of nutrients in soils and microbial immobilization during the process of decomposition (Osono and Takeda 2004).
In A. rubra (Edmonds 1980) and A. nepalensis (Sharma and Ambasht 1987) stands, P was found to be rapidly lost in the first 3 months, and absolute weights never exceeded the initial weights. Phosphorus loss in this study was relatively high during the first 6 months of decomposition. Phosphorus release per unit area increased from 5 years onwards and decreased sharply after 20 years showing maximum availability in the younger stands. The initial C-to-P ratio (300–686) of the litter fractions in all the plantation stands was comparable to A. rubra (574) and A. nepalensis (637) stands (Edmonds 1980; Sharma and Ambasht 1987). High rainfall during 3–6 months of decomposition may be the reason for rapid P loss. The pattern of release of P from a unit area of floor was similar in all the agroforestry stands.
The proportion of ash-free mass and energy content of decomposing litter fractions in litterbags within a 24-month period declined at different absolute and relative rates. The loss of nutrients indicated approximately uniform absolute and relative rates in all stands across the sampling dates. Energy release in the form of heat was substantial in the 15-year stand. The energy value per unit weight of the residual ash-free mass of decomposing litter decreased across the entire decomposition period. Absolute energy content also decreased considerably with 81–88% loss during the 24-month decomposition.
The relative decomposition rates of ash-free mass, N and P were significantly related to the C-to-N ratio, litter temperature and moisture in the multiple regression analysis. Linear regression of ash-free mass remaining as a function of the C-to-N ratio of retrieved litter during decomposition showed a strong positive relationship. The ash-free mass release and nutrient mineralization were also related to the C-to-N ratio, with high partial regression coefficients of the C-to-N ratio for nutrient mineralization.
The ash-free mass, nutrient and energy release during the first 6 months of decomposition were highest, which is attributable to the high rate of loss of labile fractions and most favorable environmental conditions for decomposition. Each fraction of litter decomposes at a specific rate and is variably dependent on resource quality (C-to-N ratio, initial lignin-to-N ratio) and largely contributed by environmental factors. We conclude that the C-to-N ratio was better predicted to nutrient N release throughout the decomposition period, which can be compared to the report made by Seneviratne (2000). The initial lignin-to-N ratio and C-to-N ratio of the litter fractions played a more important role in determining the rate of litter decomposition than litter moisture and temperature. Osono and Takeda (2004) have also reported that the lignin-to-N ratio and lignin-to-P ratio are the indicators of N and P dynamics during decomposition. The relative loss rate of 60–90% of the ash-free mass, nutrients and energy in the present study were related to the lignin-to-N ratio, litter quality and environmental factors. In the growing age series until 20-year stand, N availability also increases through N2-fixation by Alnus; during such situation high N concentrations may have an accelerating effect on lignin degradation and thus faster decomposition.
The release of nutrients due to rapid decomposition from nutrient-rich litter of Alnus makes more nutrients available for uptake by associate cardamom, and with time, the nutrient cycle is expected to be accelerated, even in the non-N2-fixing associate. The percent contribution of nutrient release by the shade tree Alnus was substantial (N 57–63, P 54–58) compared to the understorey cardamom crop (N 35–43, P 42–46). This provides better production potential for associate crops in the stands with nitrogen fixers (Sharma et al. 1997; Sharma et al. 2002b). The magnitude of release of nutrients and energy in the younger plantations was rapid, indicating accelerated nutrient cycling through litter production, decomposition and heat sink compared to the older stands beyond 20 years age. This is explained by high tree density, productivity, energetics and efficiencies, litter production, N2-fixation and efficiency, resource quality, cardamom crop management and environmental factors in these stands (Sharma et al. 2002a, b). The intensity of these processes was phenomenal and observed to be considerably high in younger plantations up to 20 years of age. Thus, the appropriate management cycle of the Alnus-cardamom system for its sustainability is 15–20 years. The system also provides additional income at the end of the rotational cycle by harvesting Alnus.
References
Aber JD, Melillo JM (1982) Nitrogen immobilization in decaying hardwood leaf litter as a function of initial nitrogen and lignin content. Can J Bot 60:2263–2269
Aber JD, Mellizo JM, McClaugherty CA (1990) Predicting long-term pattern of mass loss, nitrogen dynamics, and soil organic matter formation from initial fine litter chemistry in temperate forest ecosystems. Can J Bot 68:2201–2208
Anderson JM, Ingram JSI (1993) Tropical soil biology and fertility: a handbook of methods. CAB International, Wallingford
Bargali SS, Singh SP, Singh RP (1993) Pattern of weight loss and nutrient release from decomposing leaf litter in an age series of Himalayan eucalypt plantations. Soil Biol Biochem 25:1731–1738
Berg B (1986) Nutrient release from litter and humus in coniferous forest soils: a mini review. Can J For Res 1:359–369
Berg B, McClaugherty CA (1987) Nitrogen release from litter in relation to the disappearance of lignin. Biogeochemistry 4:219–224
Berg B, McClaugherty CA (1989) Nitrogen and phosphorus release from decomposing litter in relation to the disappearance of lignin. Can J Bot 67:1148–1156
Binkley D, Sollins P, Bell R, Sachs D, Myrold D (1992a) Biogeochemistry of adjacent conifer and alder/conifer stands. Ecology 73:2022–2033
Binkley D, Dukin K, DeBell D, Ryan MG (1992b) Production and nutrient cycling in mixed plantations of Eucalyptus and Albizia in Hawaii. For Sci 38:393–408
Bocock KL (1964) Changes in the amounts of dry matter, nitrogen, carbon and energy in decomposing woodland leaf litter in relation to the activities of the soil fauna. J Ecol 61:481–485
Edmonds RL (1979) Decomposition and nutrient release in Douglas-fir needle litter in relation to stand development. Can J For Res 9:132–140
Edmonds RL (1980) Litter decomposition and nutrient release in Douglas-fir, red alder, western hemlock, and Pacific silver fir ecosystems in western Washington. Can J For Res 10:327–337
Gupta PN, Naqvi AN, Mishra LN, Sen T, Nigam MC (1984) Gas chromatographic evaluation of the essential oils of different strains of Amomum subulatum Roxb. growing wild in Sikkim. Sonderdruck aus Parfumeric und kodmetik 65:528–529
Kwabiah A B, Voroney RP, Palm CA, Stoskopf NC (1999) Inorganic fertilizer enrichment of soil: effect on decomposition of plant litter under sub-humid tropical conditions. Biol Fertil Soils 30:224–231
Lieth H (1975) Measurement of caloric values. In: Lieth H, Whittaker RH (eds) Primary productivity of the biosphere. Springer, Heidelberg, pp 119–129
Loranger G, Jean-Francois P, Daniel I, Patric L (2002) Leaf decomposition in two semi-evergreen tropical forests: influence of litter quality. Biol Fertil Soil 35:247–252
Meentemeyer V (1978) Microclimate and lignin control of litter decomposition rates. Ecology 59:465–472
Melillo JM, Aber JD, Muratore JF (1982) Nitrogen and lignin control of hardwood leaf litter decomposition dynamics. Ecology 63:621–626
Mellilo JM, Aber JD, Linkins AE, Ricca A, Fry B, Nadelhoffer KJ (1989) Carbon and nitrogen dynamics along the decay continuum: plant litter to soil organic matter. Plant Soil 115:189–198
Mugundi DN, Nair PKR (1996) Predicting the decomposition patterns of tree biomass in tropical highland microregions of Kenya. Agrofor Sys 35(2):187–201
Mukhopadhyaya SC (1998) Geomorphology of the Sikkim Himalaya. In: Rai SC, Sundriyal RC, Sharma E (eds) Sikkim perspective for planning and Development. Bhisen Singh and Mahendra Pal Singh, Dehra Dun, India
Mwiinga RD, Kwesiga FR, Kamara CS (1994) Decomposition of leaves of six multipurpose tree species in Chipata, Zambia. For Ecol Manag 64:209–216
Olson JS (1963) Energy storage and the balance of producers and decomposers in ecological systems. Ecology 44:322–331
Osono T, Takeda H (2004) Accumulation and release of nitrogen and phosphorus in relation to lignin decomposition in leaf litter of 14 tree species. Ecol Res 19:593–598
Osono T, Takeda H (2005) Decomposition of organic chemical components in relation to nitrogen dynamics in leaf litter of 14 tree species in a cool temperate forest. Ecol Res 20:41–49
Sankaran KV, Balasudaran M, Thomas TP, Sujatha MP (1993) Litter dynamics, microbial associations and soil studies in Acacia auriculiformis plantations in Kerala. Kerala Forest Research Institute Research Report No. 91, Kerala, India
Seneviratne G (2000) Litter quality and nitrogen release in tropical agriculture: a synthesis. Biol Fertil Soil 31:60–64
Sharma E, Ambasht RS (1987) Litterfall, decomposition and nutrient release in an age sequence of Alnus nepalensis plantation stands in the eastern Himalaya. J Ecol 75:997–1010
Sharma R, Sharma E, Purohit AN (1994) Dry matter production and nutrient cycling in agroforestry systems of cardamom grown under Alnus and natural forest. Agrofor Sys 27:293–306
Sharma R, Sharma E, Purohit AN (1997) Cardamom, mandarin and nitrogen-fixing trees in agroforestry systems in India’s Himalayan region I. Litterfall and decomposition. Agrofor Sys 35:239–253
Sharma G, Sharma E, Sharma R, Singh KK (2002a) Performance of an age series of Alnus-cardamom plantations in the Sikkim Himalaya: productivity, energetics and efficiencies. Ann Bot 89:261–272
Sharma G, Sharma E, Sharma R, Singh KK (2002b) Performance of an age series of Alnus-cardamom plantations in the Sikkim Himalaya: nutrient dynamics. Ann Bot 89:273–282
Spices Board (2001) Area under large cardamom in Sikkim and West Bengal-survey reg. No. SKM/T-2000–2001. Ministry of Commerce, Government of India
Tarrant RF, Lu KC, Bollen W, Franklin JF (1969) Nitrogen enrichment of two forest ecosystems by red alder. United States Service Research Paper PNW–76
Taylor BR, Parsons WFJ, Perkinson D (1989) Decomposition of Populus trimuloides leaf litter accelerated by Alnus crispa litter. Can J For Res 19:674–679
Turner J, Long JN (1975) Accumulation of organic matter in a series of Douglas-fir stands. Can J For Res 5:681–690
Van Cleve K (1971) Energy and weight loss functions for decomposing foliage in birch and aspen forests in interior Alaska. Ecology 52:720–723
Wieder RK, Lang GE (1982) A critique of the analytical methods in examining decomposition data obtained from litterbags. Ecology 63:1636–1642
Witkamp M, Olson JS (1963) Breakdown of confined and non-confined oak litter. Oikos 14:138–147
Acknowledgments
The authors are thankful to the Director of the G. B. Pant Institute of Himalayan Environment and Development for providing facilities and ICAR New Delhi for funding the study. Facilities provided by ICIMOD and UNU during the preparation of the manuscript are highly appreciated.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Sharma, G., Sharma, R. & Sharma, E. Influence of stand age on nutrient and energy release through decomposition in alder-cardamom agroforestry systems of the eastern Himalayas. Ecol Res 23, 99–106 (2008). https://doi.org/10.1007/s11284-007-0377-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11284-007-0377-9