Abstract
Objectives
To evaluate the prognostic value of preoperative radiological findings for nodal recurrence in clinically node-negative (cN0) patients with oral tongue squamous cell carcinoma (SCC).
Methods
The study population consisted of 52 patients with cT1-2N0 oral tongue SCC classified according to the 7th edition of the Union for International Cancer Control (UICC) staging system. The subjects had undergone preoperative radiological examinations, including magnetic resonance imaging (MRI) and 18F-fluorodeoxyglucose (FDG)-positron emission tomography (PET)/computed tomography. All patients were treated with local resection and watchful waiting for neck management. Using an unpaired t test, Pearson’s chi-squared test, and the Kaplan–Meier method, the MRI-derived depth of invasion (DOI), the standardized uptake value (SUV) on FDG-PET, and the T stage according to the 7th and 8th UICC were assessed as prognostic factors.
Results
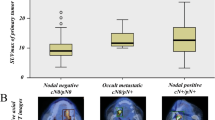
The MRI-derived DOI was recorded as ≤ 5 mm in 24 patients and > 5 mm in 28 patients. During the follow-up period, nine patients exhibited nodal recurrence, with the MRI-derived DOI being significantly higher in patients with positive than in those with negative (p = 0.011). The SUV was not significant. Five-year cumulative nodal recurrence probabilities were 4.5% for patients with an MRI-derived DOI ≤ 5 mm, while it was 32.1% for > 5 mm (p = 0.013). Although the T classifications were not significant, none of our patients whose T stage according to the 8th UICC was T1 suffered nodal recurrence.
Conclusions
MRI-derived DOI can predict nodal recurrence, while preoperative information may assist in treatment planning for oral tongue SCC.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Nodal recurrence is the most common cause of surgical treatment failure in oral cancer. Performing elective neck dissection (END) for cases with no clinical evidence of lymph node metastasis in the neck (cN0) in early stage T1-T2 oral squamous cell carcinoma (SCC) has been the subject of much debate, and continues to be controversial [1,2,3]. Historically, a 20% probability of occult lymph node metastases was considered the common threshold value for performing END [4].
18F-fluorodeoxyglucose (FDG)-positron emission tomography (PET)/ computed tomography (CT) fused imaging and its quantitative parameter, the standardized uptake value (SUV), have been widely used for deciding on which treatment strategy to use in patients with head-and-neck SCC [5,6,7]. The SUV of the primary tumor has been suggested to be related to aggressive tumor characteristics and poor clinical outcomes [8, 9]. Dual-phase FDG-PET/CT imaging has been found to improve diagnostic accuracy [10, 11]. However, many studies have found an incidence of false-negative lesions with this method [12, 13].
In the latest update (the 8th edition) of the Union for International Cancer Control (UICC) staging system, which was promulgated in 2017, the T category for oral cavity cancer now incorporates the depth of invasion (DOI) and no longer depends solely on greatest surface dimensions [14,15,16]. Tumors are classified as less- (≤ 5 mm), moderately- (from > 5 to ≤ 10 mm), and deeply invasive (> 10 mm). The DOI has been considered to be a risk factor for local recurrence and lymph node metastasis [16,17,18,19]. Although the DOI is defined by pathological findings after surgery, recent studies have demonstrated the reliability of magnetic resonance imaging (MRI)-derived DOI before surgery [20,21,22]. As MRI can clearly depict the configurations of the normal oral cavity and of oral cavity cancer, it is a useful modality for preoperative evaluation. The purpose of this study was to evaluate the prognostic value of preoperative radiological findings for predicting recurrence patterns in cN0 patients with oral tongue SCC.
Material and methods
Patient characteristics
Between April 2007 and December 2015, 59 patients with clinical T1N0 or T2N0 oral tongue SCC evaluated with dual-phase FDG-PET/CT imaging underwent curative surgery. Our inclusion criteria were as follows: (1) histologically verified SCC of the oral tongue, (2) clinical T1N0 or T2N0, (3) no previous malignancies, (4) no preoperative chemotherapy or radiotherapy, (5) availability of preoperative MRI within 1 month before surgery, and (6) availability of dual-phase FDG-PET/CT imaging within 1 month before surgery. We excluded four patients with a previous malignancy, and three who had missing MRI data. The clinical stage according to the 7th edition of the UICC staging system was identified at a routine preoperative conference of oral surgeons and radiologists who interpreted imaging data, with 23 were staged as T1N0 and 29 as T2N0. The radiological diagnosis of nodal involvement was based on accepted morphologic criteria and FDG uptake [5, 6]. There were 33 males and 19 females (median age, 70 years; age range, 29–86 years). All patients underwent transoral partial glossectomy alone as primary treatment and watchful waiting for neck management.
MRI
Preoperative MRI was performed on a 3 T superconducting imager (Philips Achieva 3.0 T TX, Philips Healthcare, Best, the Netherlands). TSE sequences were used to obtain T1WI (500–600/9 [TR msec/TE msec]; axial), T2WI (4000–5000/80; axial, coronal), STIR (5000/70/230 [TR msec/TE msec/TI msec]; axial, coronal), and gadolinium contrast-enhanced T1WI with fat suppression (axial, coronal). The parameters used for all images were as follows: 220 mm FOV, 512 matrix, 4 mm thickness, and 0.4 mm slice gap.
FDG-PET/CT imaging
FDG-PET/CT imaging was performed using a whole-body three-dimensional (3D) PET/CT scanner (Gemini GXL; Philips Medical Systems, Cleveland, OH, USA), which permits the acquisition of coregistered CT and PET images in the same session. The patients had fasted for more than 5 h before receiving the FDG injection and were confirmed to have blood sugar levels of less than 120 mg/dl. To minimize normal muscle uptake, patients laid in a quiet, dark room. The scan was performed twice: an early whole-body scan acquired at approximately 60 min after the intravenous administration of 5–10 mCi (185–370 MBq) of FDG and a delayed scan at approximately 120 min. Unenhanced CT images (150 mA, 120 kV, 600 mm FOV, 512 matrix, and 5 mm thickness) were acquired. Attenuation correction was performed on the CT transmission data, while PET images were reconstructed using the line of response-row-action maximum likelihood algorithm (LOR-RAMLA). The parameters used for reconstruction of the images were as follows: 576 mm FOV, 144 matrix, and 4 mm thickness.
Image interpretations
All images were transferred to a workstation (ViewR, version 1.24.02; Yokogawa Medical Solutions Corporation, Tokyo, Japan) by using the DICOM format. One of the authors (R.M.) who had 24 years of experience in MRI diagnoses of head-and-neck cancer analyzed the location and the direction of tumor invasion and determined the MRI-derived DOI based on both axial and coronal planes (Fig. 1) [21]. T staging was performed according to the 8th edition of the UICC staging system. Two of the authors consensually assessed tumor findings on early and delayed FDG-PET/CT images. SUV is the ratio between the measured and expected uptake if FDG was distributed evenly throughout the body. Thus, SUV = FDG uptake in each voxel/(injected dose/body weight). The maximum value of SUV (SUVmax) in the primary tumor was measured on early and delayed images and recorded as E-SUV and D-SUV, respectively. For tumors which were not detected, we recorded the MRI-derived DOI as 0 mm and SUVmax as 1.
Example of the measurement of depth of invasion (DOI) on MRI. MRI sequences are chosen on a case-by-case basis: (a) axial T1WI, (b) Coronal STIR. The reference line (oblique line) is determined as the line connecting the junctions of the tumor and the normal mucosa on both sides, depending on the location and the direction of tumor invasion. MRI-derived DOI (arrows) is measured by drawing a perpendicular line from the reference line to the deepest point of invasion. The protruding portion (arrowhead) is ignored
Follow-up evaluation
All patients were followed up to evaluate tumor control on an outpatient basis at every 2–4 weeks during the first year after surgery and at 1–6 month intervals thereafter. The patients also underwent follow-up ultrasound, CT, and/or MRI every 3–6 months. During the follow-up period, the initial sites of recurrence were classified as local, nodal, and distant. Patients with poor tumor control underwent salvage surgery, radiotherapy, chemotherapy, and/or best supportive care.
Statistical analyses
The difference between the E-SUV and the D-SUV for each tumor was compared using a paired t test. The relationships between preoperative radiological findings (the MRI-derived DOI, the E-SUV, and the D-SUV) and recurrence patterns were evaluated using an unpaired t test. Being prognostic factors for nodal recurrence, the T classifications based on the 7th and 8th editions of the UICC staging system, as well as the MRI-derived DOIs, were assessed using a Pearson’s chi-squared test. Cumulative probabilities of nodal recurrence according to MRI-derived DOIs were also calculated using the Kaplan–Meier method and the log-rank test. All statistical computations were performed using commercial software (IBM SPSS Statistics, version 27, IBM Japan, Tokyo). Probabilities of less than 0.05 were accepted as significant.
Results
The mean (± standard deviation) value of the MRI-derived DOI was 5.0 ± 3.1 mm. The MRI-derived DOI was recorded as ≤ 5 mm, 5–10 mm, and > 10 mm in 24, 25, and 3 patients, respectively. Upon classification using the 8th edition of the UICC staging system, 12 and 2 patients, respectively, were up-staged from T1 to T2 and from T2 to T3, with 11, 39, and 2 tumors being staged as T1, T2, and T3, respectively. The mean value of the D-SUV (5.0 ± 3.1) was significantly higher than that of the E-SUV (4.2 ± 2.5) (p < 0.001). During the median follow-up period of 42 months (range, 2–134 months), 6 and 9 patients, respectively, exhibited local and nodal recurrence even in our cN0 patients who had undergone preoperative radiological examinations including both MRI and FDG-PET/CT imaging. The local recurrences were observed a maximum of 48 months (range, 2–48 months) after surgery. The nodal recurrences appeared within 16 months (range, 2–16 months). There were no patients with distant recurrence. Of the 52 patients in this series, 5 succumbed to primary (n = 4) or intercurrent disease (n = 1). The 5-year disease-free survival (DFS) and overall survival (OS) rates were 64.7% and 88.7%, respectively, while the 5‑year cumulative local recurrence and nodal recurrence probabilities were 8.9% and 19.0%, respectively.
Figure 2 shows the relationship between the MRI-derived DOI and recurrence patterns. For nodal recurrence, the mean value of the MRI-derived DOI was significantly higher in patients with positive (7.3 ± 2.1 mm) than in those with negative (4.5 ± 3.0 mm) (p = 0.011). The E-SUV and the D-SUV were not significant (Figs. 3, 4). No radiological findings predicted the local recurrence.
For nodal recurrence, the MRI-derived DOI classified the groups with significantly different risks, with the incidence being 4.2% for patients with an MRI-derived DOI ≤ 5 mm and 28.6% for patients with values > 5 mm (p = 0.020) (Table 1). Prognostic values were obtained using the Kaplan–Meier method, with the 5‑year cumulative nodal recurrence probability being 4.5% for patients with an MRI-derived DOI ≤ 5 mm and 32.1% for patients with values > 5 mm (p = 0.013) (Fig. 5). Although the T classifications were not significant for predicting nodal recurrence, none of our patients whose T stage according to the 8th edition UICC was T1 suffered nodal recurrence (Table 1).
Discussion
Preoperative information on nodal involvement strongly influences the treatment and prognosis of oral tongue cancer. We evaluated the prognostic value of the preoperative radiological findings for recurrence patterns in the said condition. Of the 52 patients included in the study having cN0 oral tongue SCC, nine suffered nodal recurrence, and their 5-year cumulative nodal recurrence probability was 19.0%. The MRI-derived DOI classified groups based on their significantly different risks: the 5-year cumulative nodal recurrence probability was 4.5% for patients with an MRI-derived DOI ≤ 5 mm and 32.1% for patients with values > 5 mm (p = 0.013). When patients who possess an expected risk of nodal involvement exceeding 20% undergo END, MRI-derived DOI can provide the useful preoperative information for deciding on which treatment strategy to use [4].
In the 8th edition of the UICC staging system, the T classification for oral cavity cancer incorporates the DOI [14,15,16]. The DOI, which is originally defined by pathological findings after surgery, is a risk factor for local recurrence and lymph node metastasis [16,17,18,19]. Recent studies have demonstrated the reliability of the MRI-derived DOI as a preoperative parameter [20,21,22]. Mao et al. [20] who evaluated patients not only with early but also with advanced oral tongue SCC confirmed nodal metastasis based on either pathological examination on immediate neck dissection or follow-up evaluation. In this study, we identified nodal recurrence during the follow-up period. Our results demonstrated the prognostic value of the MRI-derived DOI for nodal recurrence in patients with early oral tongue SCC treated with local resection and watchful waiting for neck management. However, overestimations on MRI and atrophy of postoperative pathological sections have been also revealed, with the MRI-derived DOI being potentially 2–3 mm greater than the pathological DOI [20,21,22]. We must contemplate differences in the DOI between the MRI and pathological sections. Although we classified the MRI-derived DOI according to the 8th edition of the UICC staging system (≤ 5 mm vs. > 5 mm), further investigations are necessary to elucidate the optimal cutoff value.
The TNM staging system has been updated several times and is being used in clinical trials to predict the prognosis, tailor treatment, and compare outcomes [14,15,16]. Although the T classifications were not significant for the nodal recurrence, none of our patients whose T stage according to the 8th edition UICC staging system was T1 (size ≤ 2 cm and MRI-derived DOI ≤ 5 mm) suffered nodal recurrence. The results thus suggest that performing END is unnecessary on patients staged as T1 according to the 8th edition UICC staging system.
The delayed phase of FDG-PET/CT imaging can increase tumor detectability due to the higher FDG uptake compared to the early phase of imaging [10, 11]. In this study, the mean value of the D-SUV (5.0 ± 3.1) was found to be significantly higher than that of the E-SUV (4.2 ± 2.5) (p < 0.001). Although FDG-PET imaging has high diagnostic performance for nodal staging, it can also lead to false-negative findings [12, 13]. Nodal recurrence appeared during the follow-up period even in our cN0 patients who had undergone preoperative FDG-PET/CT imaging. From the results of the histopathologic analyses of END samples, it could be concluded that FDG-PET imaging is not yet accurate enough compared to neck dissection in identifying occult lymph node metastasis in patients with cN0 [12].
The SUVs of primary tumors have been suggested to be related to aggressive tumor characteristics and poor clinical outcomes [8, 9]. Our hypothesis was that primary tumors with high metabolic activity might have preponderance for local and nodal recurrence. However, in the patients included in this study who had cN0 oral tongue SCC, the E-SUV and the D-SUV were not able to predict local or nodal recurrence. The staging of the patients had been classified as cN0 based on FDG-PET/CT imaging. Therefore, the results could reflect selection bias, with primary tumors with high FDG uptake possibly excluded.
Our study had substantial limitations. It was retrospective, included only a small number of patients, and was a single-center study. The preoperative radiological findings used to predict recurrence pattern were not addressed with respect to other stages because the study population included only patients staged as cT1-2N0 according to the 7th edition of the UICC staging system. However, MRI-derived DOI can be used to predict nodal recurrence in early clinical stages. We assessed not postoperative histopathological findings but preoperative radiological findings for nodal recurrence. Postoperative findings should provide the useful additional information on prognosis [16,17,18,19].
Conclusions
The current study evaluated the prognostic value of preoperative radiological findings in oral tongue SCC. Nodal recurrence appeared during the follow-up period even in our cN0 patients who had undergone preoperative radiological examinations including both MRI and FDG-PET/CT imaging. Our results suggest that the MRI-derived DOI can predict nodal recurrence, while preoperative information may assist in treatment planning for oral tongue SCC. Prospective studies having larger study populations are needed to validate the use of MRI-derived DOIs for predicting nodal metastasis.
References
Abu-Ghanem S, Yehuda M, Carmel NN, Leshno M, Abergel A, Gutfeld O, et al. Elective neck dissection vs observation in early-stage squamous cell carcinoma of the oral tongue with no clinically apparent lymph node metastasis in the neck: a systematic review and meta-analysis. JAMA Otolaryngol Head Neck Surg. 2016;142:857–65.
D’Cruz AK, Vaish R, Kapre N, Dandekar M, Gupta S, Hawaldar R, et al. Elective versus therapeutic neck dissection in node-negative oral cancer. N Engl J Med. 2015;373:521–9.
McDonald C, Lowe D, Bekiroglu F, Schache A, Shaw R, Rogers SN. Health-related quality of life in patients with T1N0 oral squamous cell carcinoma: selective neck dissection compared with wait and watch surveillance. Br J Oral Maxillofac Surg. 2019;57:649–54.
Weiss MH, Harrison LB, Isaacs RS. Use of decision analysis in planning a management strategy for the stage N0 neck. Arch Otolaryngol Head Neck Surg. 1994;120:699–702.
Ng SH, Yen TC, Liao CT, Chang JT, Chan SC, Ko SF, et al. 18F-FDG PET and CT/MRI in oral cavity squamous cell carcinoma: a prospective study of 124 patients with histologic correlation. J Nucl Med. 2005;46:1136–43.
Murakami R, Uozumi H, Hirai T, Nishimura R, Shiraishi S, Ota K, et al. Impact of FDG-PET/CT imaging on nodal staging for head-and-neck squamous cell carcinoma. Int J Radiat Oncol Biol Phys. 2007;68:377–82.
Murakami R, Uozumi H, Hirai T, Nishimura R, Katsuragawa S, Shiraishi S, et al. Impact of FDG-PET/CT fused imaging on tumor volume assessment of head-and-neck squamous cell carcinoma: intermethod and interobserver variations. Acta Radiol. 2008;49:693–9.
Halfpenny W, Hain SF, Biassoni L, Maisey MN, Sherman JA, McGurk M. FDG-PET. A possible prognostic factor in head and neck cancer. Br J Cancer. 2002;86:512–6.
Zhang B, Li X, Lu X. Standardized uptake value is of prognostic value for outcome in head and neck squamous cell carcinoma. Acta Otolaryngol. 2010;130:756–62.
Hustinx R, Smith RJ, Benard F, Rosenthal DI, Machtay M, Farber LA, et al. Dual time point fluorine-18 fluorodeoxyglucose positron emission tomography: a potential method to differentiate malignancy from inflammation and normal tissue in the head and neck. Eur J Nucl Med. 1999;26:1345–8.
Kubota K, Itoh M, Ozaki K, Ono S, Tashiro M, Yamaguchi K, et al. Advantage of delayed whole-body FDG-PET imaging for tumour detection. Eur J Nucl Med. 2001;28:696–703.
Cacicedo J, Navarro A, Del Hoyo O, Gomez-Iturriaga A, Alongi F, Medina JA, et al. Role of fluorine-18 fluorodeoxyglucose PET/CT in head and neck oncology: the point of view of the radiation oncologist. Br J Radiol. 2016;89:20160217.
Lowe VJ, Duan F, Subramaniam RM, Sicks JD, Romanoff J, Bartel T, et al. Multicenter trial of [(18)F]fluorodeoxyglucose positron emission tomography/computed tomography staging of head and neck cancer and negative predictive value and surgical impact in the N0 neck: results from ACRIN 6685. J Clin Oncol. 2019;37:1704–12.
Brierley JD, Gospodarowicz MK, Wittekind C. TNM classification of malignant tumours. 8th ed. Chichester, West Sussex, UK: John Wiley & Sons, Inc.; 2017.
Huang SH, Sullivan B. Overview of the 8th Edition TNM classification for head and neck cancer. Curr Treat Options Oncol. 2017;18:40.
Vuity D, McMahon J, Takhiuddin S, Slinger C, McLellan D, Wales C, et al. Is the 8th edition of the Union for International Cancer Control staging of oral cancer good enough? Br J Oral Maxillofac Surg. 2018;56:272–7.
Huang SH, Hwang D, Lockwood G, Goldstein DP, O’Sullivan B. Predictive value of tumor thickness for cervical lymph-node involvement in squamous cell carcinoma of the oral cavity: a meta-analysis of reported studies. Cancer. 2009;115:1489–97.
Ebrahimi A, Gil Z, Amit M, Yen TC, Liao CT, Chaturvedi P, et al. Primary tumor staging for oral cancer and a proposed modification incorporating depth of invasion: an international multicenter retrospective study. JAMA Otolaryngol Head Neck Surg. 2014;140:1138–48.
Sakata J, Yamana K, Yoshida R, Matsuoka Y, Kawahara K, Arita H, et al. Tumor budding as a novel predictor of occult metastasis in cT2N0 tongue squamous cell carcinoma. Hum Pathol. 2018;76:1–8.
Mao MH, Wang S, Feng ZE, Li JZ, Li H, Qin LZ, et al. Accuracy of magnetic resonance imaging in evaluating the depth of invasion of tongue cancer: a prospective cohort study. Oral Oncol. 2019;91:79–84.
Murakami R, Shiraishi S, Yoshida R, Sakata J, Yamana K, Hirosue A, et al. Reliability of MRI-derived depth of invasion of oral tongue cancer. Acad Radiol. 2019;26:e180–6.
Baba A, Hashimoto K, Kayama R, Yamauchi H, Ikeda K, Ojiri H. Radiological approach for the newly incorporated T staging factor, depth of invasion (DOI), of the oral tongue cancer in the 8th edition of American Joint Committee on Cancer (AJCC) staging manual: assessment of the necessity for elective neck dissection. Jpn J Radiol. 2020;38:821–32.
Acknowledgements
We would like to thank Editage (www.editage.com) for English language editing. This work was supported by JSPS KAKENHI Grant Number 18K07757.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by RM, FS, RY, JS, AH, KK, and KY. The first draft of the manuscript was written by AM and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Human and animal rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008.
Informed consent
The institutional review board of our institute approved this retrospective study (RINRI Number 1427). Informed consent was waived due to the retrospective nature of this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Minamitake, A., Murakami, R., Sakamoto, F. et al. Can MRI-derived depth of invasion predict nodal recurrence in oral tongue cancer?. Oral Radiol 37, 641–646 (2021). https://doi.org/10.1007/s11282-020-00505-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11282-020-00505-3