Abstract
The aim of this study was to investigate the prevalence, antimicrobial susceptibility and resistant determinants of Elizabethkingia meningoseptica in a Beijing hospital. Four hundred and eighty-seven samples from medical devices, hospital surfaces and medical staff hands were collected. In total, 26 E. meningoseptica isolates were obtained. The sinks, faucets, and drains accounted for more than half of the total number of isolates recovered. Antimicrobial susceptibility testing revealed that 24 isolates were resistant to one or more antibiotics. All strains were susceptible to piperacillin/tazobactam and vancomycin. Although the trimethoprim/sulfamethoxazole has previously been shown to exhibit good activity against E. meningoseptica, in our study 15 strains were resistant to it. We detected trimethoprim/sulfamethoxazole resistance determinants using PCR; six isolates possessed the sulI gene and four possessed the sulII gene, whilst the dfrA12 gene was detected in only one of them. Pulsed-field gel electrophoresis (PFGE) analysis showed 9 distinct types and one dominant pattern with 12 strains was found. Our data indicate that antimicrobial resistant E. meningoseptica strains exist in the hospital environment and susceptibility testing revealed that vancomycin and piperacillin/tazobactam was the most effective antibiotics. These results have practical significance for treatment of E. meningoseptica infection.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Elizabethkingia meningoseptica (previously Flavobacterium meningosepticum, Chryseobacterium meningosepticum; Kim et al. 2005) is a Gram-negative, oxidase-positive, and non-glucose-fermenting bacterium which is widely distributed in soil, plants and water, but is not normally present in human microflora (Bernardet et al. 2005). E. meningoseptica is recognized as an occasional but serious opportunistic bacterial pathogen to humans, giving rise to meningitis, pneumonia, septic arthritis, endocarditis and conjunctivitis (Xie et al. 2009).
Environmental studies have revealed that this species can survive in water supplies, and often colonizes sinks, basins and taps, thus creating potential reservoirs of infection within hospitals (Hoque et al. 2001). E. meningoseptica can colonize patients via fluid contaminated medical devices (e.g., respirators, intubation tubes, mist tents, humidifiers, incubators for newborns and ice chests; Du Moulin 1979; Hoque et al. 2001). It can also be spread by wet and dry materials and surfaces, including hospital staff hands (Ceyhan et al. 2008). Nosocomial transmission of E. meningoseptica among immunocompromised adults in intensive care units (ICUs) has also been reported (Pokrywka et al. 1993).
Antimicrobial susceptibility data for E. meningoseptica is limited, since this pathogen has rarely been isolated from clinical specimens. Treatment of E. meningoseptica infections is challenging because of its increasing resistance to several classes of antimicrobial agents, for example, aminoglycosides, tetracyclines, chloramphenicol and erythromycin (Kirby et al. 2004; Lin et al. 2004). However, some fluoroquinolones have shown favourable results (Fraser and Jorgensen 1997; Kirby et al. 2004). Vancomycin has been successfully used for treating meningitis in infants in the past, but its efficacy against E. meningoseptica has been questioned (Fraser and Jorgensen 1997). Consequently, the lack of a broad spectrum agent against E. meningoseptica means that antimicrobial susceptibility testing is required to ensure effective treatment outcomes.
Trimethoprim and sulfamethoxazole share both a wide antibacterial spectrum including common urinary tract pathogens, respiratory tract pathogens, skin pathogens, as well as certain enteric pathogens (Huovinen et al. 1995). Because of the wide range of clinical indications, the combination of trimethoprim and sulfamethoxazole has been used extensively everywhere in the world. In previous studies trimethoprim/sulfamethoxazole has shown good activity against E. meningoseptica in vitro and has been considered as the potential therapy for E. meningoseptica infections (Lin et al. 2004, 2009). The survey of SENTRY Antimicrobial Surveillance Program indicated that the rate of resistance to trimethoprim/sulfamethoxazole was 20.8 % in E. meningoseptica isolates (Kirby et al. 2004). The lower resistance rate to trimethoprim/sulfamethoxazole (3.1 %) was observed in clinical isolates of E. meningoseptica from Taiwan (Lin et al. 2009). However, susceptibility to trimethoprim/sulfamethoxazole appears variable. Weaver et al. (2010) reported that among the 18 patient E. meningoseptica isolates 12 were resistant to trimethoprim/sulfamethoxazole. In addition, studies demonstrated the rate of trimethoprim/sulfamethoxazole resistance in E. meningoseptica isolates was high in some provinces of China, e.g., 80 % in Hebei, 43.9 % in Liaoning, 83.8 % in Zhejiang, and 49.1 % in Tianjin (Jia and Qin 2002; Wang et al. 2005; Yang et al. 2008; Zhang 2008). The usefulness of trimethoprim/sulfamethoxazole against E. meningoseptica infections has been questioned. To counter bacterial resistance it is essential to understand the molecular background of resistance mechanisms. The acquisition of trimethoprim/sulfamethoxazole resistance determinants by horizontal transfer, mediated by mobile elements, is thought to play an important role in the spread of resistance. However, very little is known about the capacity of E. meningoseptica to carry trimethoprim/sulfamethoxazole resistance determinants.
In this study, we collected E. meningoseptica isolates from the hospital environment and investigated their susceptibility to various antimicrobial agents. We detected the presence of class 1 and 2 integrons and the genetic determinants responsible for trimethoprim/sulfamethoxazole resistance in this pathogen.
Materials and methods
Sampling
Sampling was performed over a 5-month period (March through July 2011) at a Beijing hospital. In this study, ICUs, neonatal department, obstetric department, operating rooms, catheterization rooms, hemodialysis rooms, and wards were selected for testing. Specimens were collected from various hospital surfaces, and inanimate objects, such as bed frames, bed sheets, night tables, door handles, lockers, light switches, tables, chairs, sinks, faucets, drains, stands for infusion apparatus, infant incubators, medicine cabinets, instruments, respirators and mobile instrument tables. The samples were taken with sterile cotton swabs moistened with sterile brain heart infusion broth (BHI; Huankai Ltd., Guangzhou, Guangdong, China). The samples were transported to the research laboratory immediately and cultures were then spread on BHI agar plates and incubated at 37 °C for 24–48 h.
Species identification
After incubation, colonies were streaked for purity and subcultured on BHI plates. All oxidase-positive Gram-negative isolates were selected for identification. Initially the isolates were typed by PCR amplification of 16S rRNA gene. The primers of 16S rRNA gene and PCR program were performed as described previously (Moreno et al. 2002). The isolates were PCR typed for identity and then confirmed using the VITEK 2 bacterial identification system (BioMérieux, Marcy l’Etoile, France).
Antimicrobial susceptibility testing
E. meningoseptica susceptibility to antimicrobials was determined using the broth microdilution method (Srinivasan et al. 2008). The Clinical and Laboratory Standards Institute (CLSI) MIC breakpoint for Staphylococcus spp. was applied to vancomycin (Lin et al. 2009). The CLSI MIC breakpoints for non-Enterobacteriaceae were applied to the other antimicrobials tested here.
PCR and gene sequence analysis
The isolates were screened for presence of the sulI, sulII and dfrA1–dfrA12 genes which are responsible for trimethoprim/sulfamethoxazole resistance, using the PCR primers and conditions as described previously (Chen et al. 2004). In addition, isolates were screened for the presence of class 1 and class 2 integrons using previously described PCR methods (Yaqoob et al. 2011). The PCR products were sequenced at Invitrogen Biotechnology Co., Ltd. (Beijing, China). The resulting DNA sequence data were compared to GenBank database sequences using the BLAST algorithm available at the National Center for Biotechnology Information web site (www.ncbi.nlm.nih.gov).
Pulsed-field gel electrophoresis (PFGE)
PFGE of all isolates was performed as described previously (Weaver et al. 2010). E. meningoseptica DNA plugs were lysed, washed and digested with 50 U of the restriction enzyme ApaI (CHIMERx, Madison, Wisconsin, USA) at 30 °C overnight. The PFGE was performed for 22 h at 14 °C with the CHEF MAPPER apparatus (Bio-Rad, Hercules, CA, USA) at 6 V/cm with switch time ranging from 5 to 35 s. The gel was stained in ethidium bromide solution and photographed using a Bio-Rad Gel Doc. PFGE patterns were compared using Quantity One software (Bio-Rad).
Results
Bacterial isolation
A total of 32 nursing staff hand samples and 455 samples from hospital surfaces and medical devices were screened for E. meningoseptica. Twenty-six isolates were obtained from the hospital environment and hand specimens. The overall isolation rate of E. meningoseptica was 5.3 %. The distribution of the isolates from various samples is presented in Table 1.
Antimicrobial susceptibility
The results of the sensitivity tests for the isolates to 17 different antibiotics are shown in Table 2 and Fig. 1. Out of 26 isolates tested, 24 (92.3 %) were resistant to one or more antibiotics, while only two (7.7 %) were fully susceptible. All strains were susceptible to piperacillin/tazobactam and vancomycin. Resistance to trimethoprim/sulfamethoxazole (57.7 % of the isolates were resistant), resistance to aztreonam (53.8 %), and resistance to amikacin (53.8 %) were observed most often, whereas resistance to piperacillin, levofloxacin, and ciprofloxacin were observed less frequently.
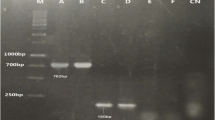
PFGE patterns and antimicrobial resistance profiles of E. meningoseptica isolates. Black boxes indicate resistance to a particular antimicrobial. PIP piperacillin; TZP piperacillin/tazobactam; CAZ ceftazidime; CRO ceftriaxone; FEP cefepime; AZT aztreonam; IPM imipenem; MEM meropenem; AMK amikacin; GEN gentamycin; TOB tobramycin; CIP ciprofloxacin; LEV levofloxacin; SXT trimethoprim/sulfamethoxazole; VAN vancomycin; TET tetracycline; CHL chloramphenicol
Detection of integrons and trimethoprim/sulfamethoxazole resistance determinants
Class 1 and class 2 integrons bearing antibiotic resistance determinants were not detected in the E. meningoseptica strains. Of the 15 trimethoprim/sulfamethoxazole resistant isolates, six isolates possessed the sulI gene and four possessed the sulII gene. None of the susceptible isolates yielded positive sul PCR products. Among the four sulII positive isolates, one harboured the dfrA12 gene which was responsible for trimethoprim resistance.
PFGE
PFGE analysis revealed 9 different pulsotypes among the 26 E. meningoseptica tested (Fig. 1). The dominant pattern I included 12 isolates, while the majority of remaining types identified (n = 7) had 2 or 1 strains. There was no complete correlation between resistance pattern and PFGE.
Discussion
Hospitals contain a reservoir of microorganisms, many of which are multiply resistant to antimicrobials (Dancer 1999). Hospital staff and patients are considered the most important sources of nosocomial microorganisms, however there is growing evidence that the hospital environment itself is also of substantial importance (Dancer 1999). The aim of this short-term study, therefore, was to provide insight into the prevalence of E. meningoseptica in a hospital environment in China.
In present study, 487 samples were collected for environmental surveillance, and twenty-six E. meningoseptica isolates were obtained. Especially the sinks, faucets, and drains, accounted for 57.7 % of the total number of isolates recovered, which was consistent with the previous report that E. meningoseptica often colonizes in humid environments (Du Moulin 1979; Hoque et al. 2001). Three isolates were recovered from the surfaces of tables in ICUs and one from hand specimens of a nurse. Although there were no reported cases of infection associated with E. meningoseptica in the hospital during our sampling periods, the potential risk of E. meningoseptica infection in hospital patients still existed. Since E. meningoseptica was isolated from medical personnel hand samples, it is possible that transmission of the bacterium could occur between nursing staff and patients. Environmental surfaces and shared medical devices may also place patients at risk of E. meningoseptica infection. Our study indicates that surveillance of potential reservoirs of E. meningoseptica is important, as is maintenance of strict infection control measures in the operating rooms and ICUs (Lin et al. 2009).
According to previous studies, quinolones, rifampin, and trimethoprim/sulfamethoxazole were most active against E. meningoseptica in vitro, whilst the β-lactams, aminoglycosides and carbapenems exhibited poor activity (Kirby et al. 2004; Lin et al. 2004; Hung et al. 2008). Most E. meningoseptica clinical isolates have been reported as resistant or even multiresistant to antibiotics (Xie et al. 2009). Since more than 90 % of the E. meningoseptica strains tested here were resistant to antimicrobials, it is clear that the isolates originating within this hospital displayed a similarly high level of resistance as known clinical isolates of this bacterium (Lin et al. 2004, 2009). We found that among the β-lactams, the most active agents overall were the piperacillin/tazobactam combination, whilst cephalosporins and carbapenems exhibited poor activity against these isolates. Moreover, the strains showed substantially high rates of resistance to aminoglycosides. The quinolones were once considered the treatment of choice for E. meningoseptica infections (Hung et al. 2008). In our study, ciprofloxacin and levofloxacin also showed the higher potency of activity against the overall collection of E. meningoseptica. In addition, some reports have documented the efficacy of vancomycin for treating E. meningoseptica infections, hence this antimicrobial agent was recommended as the therapeutic choice (Di Pentima et al. 1998). Our study confirmed the effectiveness of vancomycin against all E. meningoseptica strains, which is consistent with previous results (Di Pentima et al. 1998; Xie et al. 2009). We noted that the MICs for the E. meningoseptica strains tested were slightly different from other recent studies (Kirby et al. 2004; Lin et al. 2009); this is most likely due to our small sample size. What is clear is that there is no absolutely optimal regimen to treat E. meningoseptica infections, hence antimicrobial therapy should be based on the MIC data from standardized susceptibility tests.
PFGE analysis revealed 9 distinct types among E. meningoseptica strains. The dominant pattern I accounted for 46.2 % of all isolates. Such clear predominance of one clone indicates that E. meningoseptica may be capable of permanent existence within the hospital environment. One the whole, there was no complete correlation between resistance pattern and PFGE. Some isolates belonging to the same PFGE patterns had variable resistance profiles. On the other hand, some isolates with the different PFGE types shared the common resistance profiles.
Interestingly, studies have shown that trimethoprim/sulfamethoxazole exhibited good activity against E. meningoseptica (Kirby et al. 2004); however, more than 50.0 % of the isolates were resistant to this agent in our study. Of the 26 isolates, six isolates possessed the sulI gene and four possessed the sulII gene. Among the four sulII positive isolates, one harboured the dfrA12 gene. None of the corresponding resistance genes evaluated were detected in other resistant isolates, suggesting that the possibility of other resistance mechanisms are responsible for trimethoprim/sulfamethoxazole resistance in these isolates. Since the sulI gene was commonly found to be associated with integron, we investigated whether the E. meningoseptica isolates carried integrons. Unfortunately no class 1 or class 2 integron bearing antibiotic resistance determinants were detected in all of the isolates. Given that the sulfamethoxazole resistance gene sulII is predominantly located on plasmids (Enne et al. 2001), we speculate that these resistant genes might be located on resistance plasmids in the resistant E. meningoseptica strains. The acquisition of drug resistance genes by horizontal transfer, mediated by plasmids, is currently thought to play an important role in the development of multi-drug resistance. However, additional longer-term studies conducted in more locations of these resistance genes are needed to validate this hypothesis.
In summary, we collected twenty-six E. meningoseptica isolates from various hospital environmental samples and hand specimens, thus indicating a need for greater infection control surveillance. The PFGE typing results presented indicate that some E. meningoseptica strains may be capable of adaptation to hospital environment conditions and continuous existence in this environment. Our results show that vancomycin and piperacillin/tazobactam are most active against E. meningoseptica, however, trimethoprim/sulfamethoxazole combination may not represent effective option. Nevertheless, the use of antimicrobial agents to treat infections caused by this bacterium needs further clinical evaluation.
References
Bernardet JF, Vancanneyt M, Matte-Tailliez O, Grisez L, Tailliez P, Bizet C, Nowakowski M, Kerouault B, Swings J (2005) Polyphasic study of Chryseobacterium strains isolated from diseased aquatic animals. Syst Appl Microbiol 28:640–660
Ceyhan M, Yildirim I, Tekeli A, Yurdakok M, Us E, Altun B, Kutluk T, Cengiz AB, Gurbuz V, Barin C, Bagdat A, Cetinkaya D, Gur D, Tuncel O (2008) A C. meningosepticum outbreak observed in 3 clusters involving both neonatal and nonneonatal pediatric patients. Am J Infect Control 36:453–457
Chen S, Zhao S, White DG, Schroeder CM, Lu R, Yang H, McDermott PF, Ayers S, Meng J (2004) Characterization of multiple-antimicrobial-resistant salmonella serovars isolated from retail meats. Appl Environ Microbiol 70:1–7
Dancer SJ (1999) Mopping up hospital infection. J Hosp Infect 43:85–100
Di Pentima MC, Mason EO, Kaplan SL (1998) In vitro antibiotic synergy against F. meningosepticum: implications for therapeutic options. Clin Infect Dis 26:1169–1176
Du Moulin GC (1979) Airway colonization by Flavobacterium in an intensive care unit. J Clin Microbiol 10:155–160
Enne VI, Livermore DM, Stephens P, Hall LMC (2001) Persistence of sulphonamide resistance in Escherichia coli in the UK despite national prescribing restriction. Lancet 357:1325–1328
Fraser SL, Jorgensen JH (1997) Reappraisal of the antimicrobial susceptibilities of Chryseobacterium and Flavobacterium species and methods for reliable susceptibility testing. Antimicrob Agents Chemother 41:2738–2741
Hoque SN, Graham J, Kaufmann ME, Tabaqchali S (2001) Chryseobacterium (Flavobacterium) meningosepticum outbreak associated with colonization of water taps in a neonatal intensive care unit. J Hosp Infect 47:188–192
Hung PP, Lin YH, Lin CF, Liu MF, Shi ZY (2008) Chryseobacterium meningosepticum infection: antibiotic susceptibility and risk factors for mortality. J Microbiol Immunol Infect 41:137–144
Huovinen P, Sundström L, Swedberg G, Sköld O (1995) Trimethoprim and sulfonamide resistance. Antimicrob Agents Chemother 39:279–289
Jia HL, Qin JL (2002) Analysis of nosocomial infection by Flavobacterium. Chin J Nosocomiol 12:75–76 in Chinese
Kim KK, Kim MK, Lim JH, Park HY, Lee S (2005) Transfer of C. meningosepticum and Chryseobacterium miricola to Elizabethkingia gen. nov. as E. meningoseptica comb. nov. and Elizabethkingia miricola comb. nov. Int J Syst Evol Microbiol 55:1287–1293
Kirby JT, Sader HS, Walsh TR, Jones RN (2004) Antimicrobial susceptibility and epidemiology of a worldwide collection of Chryseobacterium spp.: report from the SENTRY antimicrobial surveillance program (1997–2001). J Clin Microbiol 42:445–448
Lin P, Chu C, Su L, Huang C, Chang W, Chiu C (2004) Clinical and microbiological analysis of bloodstream infections caused by C. meningosepticum in nonneonatal patients. J Clin Microbiol 42:3353–3355
Lin Y, Chiu C, Chan Y, Lin M, Yu K, Wang F, Liu C (2009) Clinical and microbiological analysis of E. meningoseptica bacteremia in adult patients in Taiwan. Scand J Infect Dis 41:628–634
Moreno C, Romero J, Espejo RT (2002) Polymorphism in repeated 16S rRNA genes is a common property of type strains and environmental isolates of the genus Vibrio. Microbiology 148:1233–1239
Pokrywka M, Viazanko K, Medvick J (1993) A F. meningosepticum outbreak among intensive care patients. Am J Infect Control 21:139–145
Srinivasan V, Nam H, Sawant AA, Headrick SI, Nguyen LT, Oliver SP (2008) Distribution of tetracycline and streptomycin resistance genes and class 1 integrons in Enterobacteriaceae isolated from dairy and nondairy farm soils. Microb Ecol 55:184–193
Wang Q, Guo Y, Deng XY, Chu YZ (2005) Clinical distribution and resistance of 41 strains of F. meningosepticum. China J Mod Med 15:873–874 (in Chinese)
Weaver KN, Jones RC, Albright R, Thomas Y, Zambrano CH, Costello M, Havel J, Price J, Gerber SI (2010) Acute emergence of E. meningoseptica infection among mechanically ventilated patients in a long-term acute care facility. Infect Control Hosp Epidemiol 31:54–58
Xie Z, Zhou Y, Wang S, Mei B, Xu X, Wen W, Feng Y (2009) First isolation and identification of E. meningoseptica from cultured tiger frog, Rana tigerina rugulosa. Vet Microbiol 138:140–144
Yang HW, Yang JH, Yu LL (2008) Prevalence and drug resistance of nosocomial infection by C. meningosepticum. J Pract Med 24:3960–3961 (in Chinese)
Yaqoob M, Wang LP, Fang T, Lu CP (2011) Occurrence and transmission of class 1 and 2 integrons among phenotypic highly ampicillin-resistant avian E. coli isolates from Pakistan. World J Microbiol Biotechnol 27:2041–2050
Zhang L (2008) Analysis of drug resistance of C. meningosepticum from neurosurgery ICU patients. Chin J Med Lab Technol 9:261–262 (in Chinese)
Acknowledgments
This work was supported by the Major State Basic Research Development Program of China (973 Program) (no. 2010CB35704; 2012CB723705), Guangzhou technological planning projects (11C12080718), the Fundamental Research Funds for the Central Universities, SCUT (2012ZZ0083), and the Food Safety Key Lab of Liaoning Province Open Project Funding (LNSAKF2011002).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jiang, X., Wang, D., Wang, Y. et al. Occurrence of antimicrobial resistance genes sul and dfrA12 in hospital environmental isolates of Elizabethkingia meningoseptica . World J Microbiol Biotechnol 28, 3097–3102 (2012). https://doi.org/10.1007/s11274-012-1119-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-012-1119-x