Abstract
Small headwater fens at high elevations exist in the dry climatic regime of western Colorado, despite increasing demands for water development since the 1800’s. Fens on Grand Mesa have accumulated plant material as peat for thousands of years due to cold temperatures and consistently saturated soils. The peatlands maintain unique plant communities, wildlife habitat, biodiversity, and carbon storage. We located and differentiated 88 fens from 15 wet meadows and 2 marshes on Grand Mesa. Field work included determining vegetation, soils, moisture regimes, and impacts from human activities. All fens were groundwater-supported systems that occurred in depressions and slopes within sedimentary landslide and volcanic glacial till landscapes. Fens occupied 400 ha or less than 1 % of the 46,845 ha research area and ranged in size from 1 to 46 ha. Peat water pH in undisturbed sites ranged from 4.3 to 7.1. Most fens had plant communities dominated by sedges (Carex) with an understory of brown mosses. Variation in vegetation was controlled by stand wetness, water table level, organic C, conductivity (EC), and temperature °C. Fen soils ranged from 13.6 to 44.1 % organic C with a mean of 30.3 %. Species diversity in fens was restricted by cold short growing seasons, stressful anaerobic conditions, and disturbance. Multivariate analysis was used to analyze relationships between vegetation, environmental, and impact variables. Stand wetness, water table level, OC, electrical conductivity (EC), and temperature were used to analyze vegetation variance in undisturbed fens, wet meadows, and marshes. Vegetation composition in impacted fens was influenced by flooding, sedimentation, stand wetness, water table level, OC, EC, and temperature. Hydrologically modified fens supported 58 plant species compared to 101 species in undisturbed fens. Analysis of historical 1936–2007 aerial photographs and condition scalars helped quantify impacts of human activities in fens as well as vegetation changes. Fourteen fens had evidence of peat subsidence, from organic soil collapse, blocks of peat in the margins, soil instability, and differences in surface peat height between the fen soil surface and the annually flooded soil surface. Of 374 ha of fens in the Grand Mesa study area, 294 ha (79 %) have been impacted by human activities such as ditching, drainage, flooding, or vehicular rutting. Many fens had little restoration potential due to severe hydrological and peat mass impacts, water rights, or the cost of restoration.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Peat accumulating wetlands, termed fens, have formed in many high elevation watersheds in the Rocky Mountains where groundwater discharges to the surface supporting perennially saturated soils (Cooper and Andrus 1994). Fens, wet meadows, and marshes are often addressed similarly by land management agencies yet they have distinctive vegetation, hydrologic regimes, and soils. Marshes and wet meadows differ from fens in having mineral soils, and from each other, marshes having seasonally deep standing water (Cooper et al. 2012). Fens support distinctive landforms created by peat accumulation, including floating mats and hummocks, and regionally critical biodiversity, including habitat for animals such as amphibians (Lemly 2007; Lamers et al. 2015). Many peatlands are repositories of paleoecological fossils preserving pre-historic plant and animal remains, including mastodons (Johnson and Miller 2012). Fens perform the key function of carbon storage (Cooper 1996; Chimner et al. 2010), and in the southern Rocky Mountains accumulate organic matter at a rate of 8–30 cm per 1000 years (Cooper 1990; Cooper and MacDonald 2000).
Peat accumulation and landform preservation requires perennial soil saturation that limits decomposition. Because fens are supported by nearly constant groundwater inflows from their watershed, they are highly sensitive to landscape changes that influence the quantity, chemistry or timing of inflows. Many fens in the southern Rocky Mountains have been accumulating peat for more than 10 millennia and natural disturbances such as fires and insect infestations that influence the surrounding high elevation forests have had little known impact on fen hydrologic regimes (Cooper et al. 2012; Schimelpfenig et al. 2014).
Since Caucasian settlement in the mid 1800’s human impacts to fens in western North America have been from localized gold and silver mining, roads, and large-scale water development for agriculture. In the San Juan Mountains of southwestern Colorado 10 % of fens have had severe hydrologic modifications from road construction, mining, housing development, and ditch drainage (Chimner et al. 2010). On Grand Mesa 478 ha of wetland and lake basins, many supporting fens, were hydrologically modified prior to 1995 to store water in support of lower elevation irrigated agricultural lands (USFWS 1995). The alteration of fen hydrologic regimes can reduce plant species diversity, disrupt soil carbon storage (Chimner and Cooper 2003a; Patterson and Cooper 2007), and lead to peat oxidation, decomposition, and subsidence (Armentano and Menges 1986; Holden et al. 2004; Siegel and Glaser 2006).
Many fens in the southern Rocky Mountains are geographically “isolated wetlands”, lacking direct surface connections to navigable waters. Therefore, US Army Corps of Engineers jurisdictional regulation, and state level regulations do not extend to all fens in the region (Leibowitz 2003). Information on the location of fens, their vegetation composition, condition, and the effects of hydrologic modifications is needed to manage fens in mountain watersheds. We conducted research to help provide guidelines for the management of fens in the Rocky Mountains. We determined the distribution of fens on Grand Mesa, Colorado, characterized floristic and soil differences between fens, marshes, and wet meadows, quantified environmental gradients controlling fen plant community composition, and characterized the type and extent of impacts to fens.
Methods
Study area
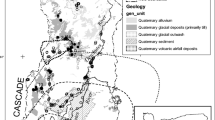
The 46,845 ha study area is located on Grand Mesa, a 1300 km2 plateau at 3100 m elevation in western Colorado (Fig. 1). Grand Mesa initially formed nearly 10 million years ago as a flat basalt plain underlain by calcareous sedimentary rocks (Fig. 2). Unlike most mountain regions in the southern Rocky Mountains, mass wasting and landslide activity have dominated its geologic history (Fig. 3). The weight of the basalt and glacial ice cap on weak underlying shale layers caused slump block movements that created hundreds of depressions (Retzer 1954; Yeend 1973; Drummond 1999). Most basins filled with water forming lakes. Where perennial groundwater, moving through landslide debris and glacial till, discharged to the surface, peat accumulated. Many peatlands occur on the upgradient side of lakes where hillslope ground water enters, maintaining shoreline water levels.
Cross sectional view of the geology of Grand Mesa. Claystone layer is the Unnamed Formation. Adapted from Yeend (Yeend 1965)
Grand Mesa mean daily January −10.8 ° C and July temperature of 56 °C is similar to other high mountain regions including the San Juan Mountains of Colorado, Yellowstone National Park in Wyoming, and southern Canadian Rocky Mountains (High Plains Regional Climate Center 2015; Warner and Asada 2006; Western Regional Climate Center 2007). Mean annual precipitation on Grand Mesa is 83 cm compared to a range of 44–62 cm in these three other areas. Winter precipitation on Grand Mesa is largely from Pacific frontal storms, while summer rain is driven largely by the southwest monsoon (Yeend 1965).
Potential fen identification
A total of 111 potential fen sites were identified using 1:24,000 scale aerial photographs taken in 1988, National Wetland Inventory (NWI) topographic quads (1:24,000) and 1 m resolution color digital orthophoto quads from the National Agricultural Imagery Program (NAIP). Fens have features that can be identified on many air photographs, including saturated soil, reddish-brown colored mosses, and landforms such as strings, pools, and floating mats.
Field evaluations
Each potential fen site was visited to determine whether organic soil was present, using the definitions in Soil Taxonomy (NRCS 1999). In each site one soil sample was collected from 35 to 40 cm depth and analyzed for % organic carbon (C) with dry combustion at 900 C in a LECO TruSpec at Colorado State University’s Soil Laboratory in Fort Collins, Colorado. Soils with organic C content >18 % were considered organic, <12 % were considered mineral, while those containing 12 to 18 % were analyzed for clay content and the formula for organic soil determination was applied: % clay × 0.1 + 12 (smallest % of organic C) (NRCS 1999). For example, a soil sample with 13 % organic C and 25 % clay content produced a calculated value of 14.5 %, indicating a mineral soil. Sites with at least 40 cm of organic soils were considered fens (Chimner et al. 2010). For estimating site soil moisture, we used a modified six-point scalar (Cooper and Andrus 1994). Sites with consistently dry or moist soil conditions were ranked 1. Sites with seasonally high water tables and occasional soil saturation were ranked 2. Sites with consistently saturated surface soil conditions were ranked 3. Sites with water table levels 8–10 cm above the soil surface were ranked 4. Sites with earthen dams and artificial flooding were ranked 5. Sites with water levels >20 cm above the soil surface were ranked 6.
Each study site was visited three or more times between 2003 and 2007, homogenous stands of vegetation were identified in each, and one 4 m2 plot per stand was sampled using the relevé method (Mueller-Dombois and Ellenberg 1974). A total of 303 stands were sampled. All vascular plants were verified in the University of Colorado Herbarium (COLO), and vascular plant nomenclature follows Weber and Wittmann (2012). Vascular plant vouchers are stored at Western State Colorado University’s Herbarium (WSCU), Gunnison, Colorado. Bryophytes were identified by W. A. Weber and R. C. Wittmann and deposited at COLO. Sphagnum specimens were identified by R. C. Andrus and are vouchered at Binghamton University (BING), New York. Bryophyte nomenclature follows Anderson et al. (1990) and Weber and Wittmann (2007).
Ground water pH and conductivity were sampled in a 40 cm deep pit in each stand that was allowed to fill with fresh groundwater. Deep standing water was present in several highly modified sites, and in these sites water pH and EC were sampled from holes dug into large blocks of peat either floating or lodged on the pond edge. We did not sample the standing pond water. Water pH and temperature °C were measured using a YSI Environmental pH100 meter and conductance with a YSI EC300 conductivity meter. Conductivity was corrected for H+ ions (Sjors 1950).
Indicators of hydrologic modifications (dams, ditches, reservoir surface water) were identified using 1936, 1956, and 1978 USDA Forest Service aerial photographs, as well as 1 m resolution NAIP aerial photographs from 2005, and 2011 Earth Google aerial photographs. All disturbances were field verified. Other disturbances were identified during fieldwork. Ditches and dams had straight-line signatures while flooding produced by reservoirs created dark grey colors from mineral sediment overlying peat. A wetland condition scalar was used to evaluate disturbance from human activities on fen hydrologic regime, physical characteristics, and sedimentation in soil samples (Clarkson et al. 2004). We documented impacts to fens with photographs of flooding, floating peat, drowned plants, dams, ditches, vehicle ruts, peat collapse and subsidence, and bare soil. Many hydrologic modifications that occurred prior to 1995 are identified on National Wetland Inventory maps (USFWS USFWS 1995).
Data analysis
Plant communities were identified using agglomerative cluster analysis, with the Sorenson distance measure and flexible beta linkage using the computer program PC-ORD version 4.14 (van Tongeren 1995; McCune and Mefford 1999; McCune and Grace 2002). The input data to the cluster analysis was percent canopy cover for each plant species in the 303 stands sampled for this project. Detrended correspondence analysis (DCA), an indirect gradient analysis technique, was used to determine the overall variation in floristic composition in the same vegetation data set (Hill and Gauch 1980; McCune and Mefford 1999). Pearson and Kendall Correlation analysis was used to determine whether the DCA axes were related to the following environmental variables: percent soil OC, ground water pH and EC, soil temperature, slope, depth to water table, and the stand wetness scalar (McCune and Mefford 1999).
Vegetation composition was related to environmental variables with constrained ordination using canonical correspondence analysis (CCA) performed with the software program Canoco version 4.5 (ter Braak and Smilauer 2002). Constrained ordination complemented the indirect gradient analysis (DCA) with detailed analysis of the environmental and disturbance gradients (Økland 1996). Bi-plot scaling and inter-species distances were used for axis scaling. The significance of the species data set to environmental variables was tested with 499 Monte Carlo permutation tests. Only environmental variables that were significant at P < 0.05 are included in the final analysis.
The parameters of the DCA analysis (McCune and Mefford 1999) produced combinations of environmental variables including those indicative of impacts, stand wetness, slope, organic C, temperature °C, conductivity (EC), pH, water table depth, sedimentation, flooding, and drainage. Bi-plot scaling and inter-species distances were used for axis scaling (ter Braak 1986). The significance of the species to impact variable relationships was tested with 499 Monte Carlo permutation tests. The analysis included only environmental variables significant at P < 0.05.
Indicator species analysis was used to identify plant species with high fidelity (P < 0.01) to human-constructed structures, disturbance type, and water table level modification (Dufrene and Legendre 1997; McCune and Grace 2002). Indicator values were tested for statistical significance with a Monte Carlo method using 1000 iterations (McCune and Mefford 1999). To add strength to the analysis, we verified the relationships between impact variables and plants species using Statistix 7 software (Analytical Software 2007).
Results
Fen identification
Soil organic C content in sample stands ranged from 0.5 to 44.1 % with a mean of 27.7 %. Of the 111 sites, 88 had organic soils and are considered fens. Fen soils had a mean organic C of 30.3 %, and ranged from 13.6 to 44.1 %. Wet meadow and marsh stands had mean organic C of 8 %.
Fens had a mean mid-summer standing water depth of 28.9 cm. Wet meadow and marsh stands had standing water levels of −0.3 cm and +26.8 cm respectively (Table 1). Only thick organic soils supported floating mat communities dominated by species such as Carex limosa, Calliergon stramineum, Menyanthes trifoliata and Sphagnum teres (Fig. 4).
Site distribution and characteristics
Eighty-nine of the 111 potential fen study sites occurred on sedimentary landslide material, and 14 on basalt. Fens occurred in depressions, basins, the base of slopes, and on hillslopes. Vegetation stands were located in basins (n = 239) or gentle (1–4 %) slopes (n = 64). Fens occupied 374 ha, or 0.008 % of the 45,180 ha research area. The largest sampled fen covered 32 ha, the smallest 1 ha, and the mean size was 5 ha. Ground water chemistry varied little between the sampled fens (Table 1). Ground water pH ranged from 4.5 to 7.1 in intact fens (n = 72), 5.5 to 7.5 in disturbed fens (n = 16), and 5.3–6.8 in wet meadows and marshes (n = 15).
Vegetation classification
From the 303 relevés analyzed, 16 plant communities were identified using cluster analysis (Table 2). The first division of the cluster analysis separated open and treed stands on sloping or drier sites from those with consistently saturated soil conditions or high water tables. Vegetation composition in the data set was controlled by site wetness, water table level, organic C, water EC and temperature (Fig. 5). Species richness was highest in Carex simulata, Eleocharis quinqueflora, Carex illota, Salix planifolia, and Picea engelmannii dominated plant communities but the mean was only 10 species per relevé. Plant communities were grouped by dominant life forms.
Direct gradient analysis of fen (black dots), wet meadows, and marsh (white dots) stands (N = 303 stands). Plant variation is explained by environmental gradients (arrows) including stand wetness, water table depth, conductivity (EC), and organic C. The greater the distance between the species symbols, which represent plant community relevés, the more dissimilar their species composition. Species in the top left ¼ of the graph grow in drier more sloping sites with higher conductivity (EC). Species in the top right ¼ tolerate the deeper water levels often found in depressions and basins. The bottom left ¼ of the graft indicates drier slightly colder sites with poorer drainage. Plant species in the bottom right ¼ of the graph grow in sites with deep peat and higher organic C (i.e. floating peat mats). DCA eigenvalues for axes 1 and 2 were 0.869 and 0.572 respectively
Impact characteristics
The CCA produced combinations of environmental variables including those indicative of impacts, stand wetness, slope, organic C, temperature °C, conductivity (EC), pH, water table depth, sedimentation, flooding, and drainage. In impacted sites sedimentation, flooding, stand wetness, water table depth, organic C, conductivity (EC), and temperature controlled gradients of plant species composition. (Figure 6). Increased sedimentation, flooding, stand wetness, temperature, and conductivity resulted in the decline of fen plant species and an increase in disturbance triggered communities such as Eleocharis macrostachya—Potamogeton foliosus. As water table depth and organic C decreased with drainage, the presence of bare peat, and the disturbance indicators Rorippa teres and the community Eleocharis acicularis—Hippuris vulgaris became more common.
Canonical Correspondence Analysis (CCA) of 268 fen stands, including disturbed (filled triangles) and undisturbed (open circles) sites. Y-axis (R = 0.508, eigenvalue = 0.139, biplot score = 0.499) shows environmental variables related to greater impacts, including sedimentation and flooding (top right), and no disturbance (bottom). The X-axis (R = 0.697, eigenvalue = 0.346, biplot score = 0.697) shows increasing moisture from left to right. Arrows point in the direction of the sharpest increase in environmental and impact gradients with p values of < 0.05
Forty-two of the 88 fens sampled, covering a total of 294 ha, were influenced by disturbances ranging from minor impacts from earthen dams and elk wallows to severe impacts from dams that have inundated fens, as well as drainage ditches, and peat subsidence (Table 3). Fourteen fens had severe hydrological and peat mass modifications including subsidence, loss of the original soil surface elevation, and vegetation alterations (Fig. 7). These sites likely cannot be restored. Only 46 fens, with an area of 80 ha, had little or no disturbance.
Comparison of historic with 2007 aerial photography indicated increased anthropogenic disturbance at Kennecott Fen, reducing its size from 80 to approximately 15 acres, and triggering vegetation alteration, disintegration of the peat mass, and loss of the main peat body (Appendix 1). The site has been altered from a floating mat or large sedge community with abundant bryophytes to small sedge stands with bare peat, and an abundance of the disturbance tolerant species Rorippa teres and Eleocharis acicularis. Intact fens have irregular shapes, without straight lines from ditches or dams. Fens with impacts had lighter colored areas indicating dry soils, straight lines, and dark grey colored flooded areas (Fig. 8).
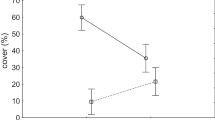
Hydrologically modified fens supported 58 species compared to 101 species in the undisturbed fens. Mean bryophyte canopy cover was 21.3 % in undisturbed fens (n = 211) compared with 8.2 % (n = 57) in impacted fen stands. Eleocharis macrostachya and Gnaphalium uliginosum were consistent indicators of flooding and drainage (Dufrene and Legendre 1997).
Discussion
Fens on Grand Mesa formed in sites where groundwater discharge and saturated soils have occurred for millennia allowing peat accumulation. Similar conditions support fens in mountain regions of Canada (Slack et al. 1980; Vitt and Wieder 2006), and the western U.S. (Cooper et al. 2002; Chimner and Cooper 2003b; Patterson and Cooper 2007). The majority of fens are on landslide deposits that formed suitable low gradient topography or basins. The largest fen is 46 ha, and the average fen is 5 ha, which is much larger than the 1.2 ha average reported for the San Juan Mountains (Chimner et al. 2010). The large fen size on Grand Mesa is likely due to the relatively level topography compared with the steeper San Juan Mountains.
Fens were distinguished from marshes and wet meadows using organic soil criteria, including % organic C content, thickness of organic layers, and water table levels (Driver 2010; Cooper et al. 2015; Chimner et al. 2010). The mean soil organic C of 30.3 %, is nearly identical to the 30.0 % organic C matter found in San Juan Mountain fens (Chimner et al. 2010) and 31.9 % organic C in Yellowstone National Park, Wyoming (Lemly and Cooper 2011). By contrast wet meadows had a mean organic C content of 8.2 %.
Water chemistry
Water chemistry and vascular and bryophyte plant species composition are often used to characterize fens along the rich to poor gradient (Vitt and Slack 1975; Cooper and Andrus 1994; Anderson and Davis 1997; Vitt 2000; Heidel and Jones 2006). Grand Mesa fens fit well into the concept of moderate-rich with soil water pH ranging from 5.7 to 7.0 and support an abundance of common rich-fen brown moss indicator species including Drepanocladus aduncus, Calliergon stramineum, Aulacomnium palustre and Tomentypnum nitens (Vitt and Chee 1990; Cooper and Andrus 1994; Vitt 2000). The treed fen community on Grand Mesa has high canopy shade and supports the mosses Climacium dendroides, Plagiomnium ellipticum and A. palustre similar to forested fens elsewhere in Colorado (Johnson 1997), northern Michigan (Vitt and Slack 1975), and Alberta, Canada (Whitehouse and Bayley 2005).
The three major bedrock types sampled on Grand Mesa (Table 1) created ground water with a chemical content similar to the basalt and rhyolite bedrock regions of Yellowstone National Park (Lemly 2007), as well as the volcanic substrates in the San Juan Mountains (Gage and Cooper 2006). Peat water EC and Ca2+ concentrations from sites on sedimentary landslide substrates were similar between Grand Mesa (28.5 μS/cm and 4.8 mg/L) and the San Juan Mountains (24.5 μS/cm and 4.0 mg/L).
Vegetation and environmental drivers
Water table depth, stand wetness, organic C, and soil water electrical conductivity were the gradients determining plant community distribution and site variation on Grand Mesa. This is distinct from mountain regions in Canada, Yellowstone National Park, the San Juan Mountains, South Park and other areas where the main drivers of fen vegetation composition are geology and source water pH (Zoltai and Vitt 1995; Cooper 1996; Lemly 2007; Chimner et al. 2010). The difference may be because relatively little variation in bedrock occurs on Grand Mesa, therefore little water chemistry variation occurs compared with other mountain regions. Carex species dominated the vegetation of most study fens and is likely the primary peat-forming species with brown mosses, as is typical in the western U.S. (Patterson and Cooper 2007; Chimner et al. 2010; Lemly and Cooper 2011; Lamers et al. 2015).
Impacts
Of the 374 ha of fens in the Grand Mesa study area, 294 ha (79 %) have been impacted by human activities. In addition, several novel plant communities have formed due to reservoir flooding, drainage, and bare peat formation. In the San Juan Mountains only 6 % (126 of 200) of fens are impacted by human activities (Chimner et al. 2010), while on Grand Mesa 47 % (42 of 88) of fens are impacted. In California impacts to fens is primarily from livestock grazing and trampling (Weixelman and Cooper 2009, Sikes et al. 2013, Wolf and Cooper 2015). The extent of hydrologic impacts on Grand Mesa is highly unusual for such a high elevation region (Bedford and Godwin 2003; Cooper and MacDonald 2000; Chimner et al. 2010; Johnston et al. 2012) and appears to represent one of the most highly impacted high mountain regions in the U.S.
Nearly complete alteration of the vegetation has occurred in 14 fens that are affected by annual deep flooding by the filling of reservoirs, drainage, or in one case, peat mining. The lack of fen vegetation or bryophytes and the presence of three species with affinities to disturbance, E. acicularis, E. macrostachya, and Gnaphalium uliginosum are excellent indicators of alteration. The lack of bryophytes in impacted fen communities is also an indicator of hydrologic modifications (Siegel 1968; Slack et al. 1980; Vitt 2014). Bare peat and the lack of fen vegetation can persist for decades after disturbances (Chimner 2011). In humid climates disturbed peatlands can support high plant species diversity (Warner and Asada 2006) and the recolonization of fens can occur within 5–10 years of shallow flooding or more slowly where peat has been removed (Asada et al. 2005; Wind-Mulder and Vitt 2000; Price et al. 2002). However, in Colorado’s more arid climate recolonization of fen vascular plant and bryophyte species occurs slowly, and the effects of disturbances can persist for decades after the original impacts (Cooper et al. 1998; Cooper and MacDonald 2000). The dewatering of peatlands using ditches is one of the most common human disturbances to peatlands in the world (Sherwood et al. 2013) and has occurred in the Rocky Mountains for more than a century (Loughlin 1918; Cooper et al. 1998; Schimelpfenig et al. 2014). Eleven fens on Grand Mesa with drainage ditches had a lowered water table and altered plant species composition.
After 56 years of flooding and draining, the peat body at Kennecott Fen, influenced by Kennecott Dam, persists although peat subsidence, slope instability, and vegetation alteration is ongoing. Peat blocks float to the surface due to air in plant roots, decay-resistant soil material, and methane accumulation (St. Louis et al. 2003). Research in the upper 15 cm of peat of four drained Colorado fens indicated losses of 14.7–91.0 tons of OM within a 20 year period. The ditches continued to compromise peatland functioning more than 20 years after being restored (Schimelpfenig et al. 2014). Known peat soil subsidence rates in North America range from 1.0 to 9.7 cm/year, depending on water levels, peat thickness, and site management (Ewing and Vepraskas 2006). However, once a peat soil is drained, shrinkage continues from the initial compression depth (Holden et al. 2004; Price et al. 2005).
Conclusions
Interest in maintaining fen biodiversity and complexity, carbon storage, and other ecosystem services has increased around the world in recent decades (Lamers et al. 2015). Fens are highly vulnerable to even minor alterations of their groundwater connections (Winters et al. 2006; Chimner et al. 2010; Lamers et al. 2015). A large proportion of the fens on Grand Mesa have been impacted by minor impacts, such as elk wallows, to severe impacts from draining and flooding for reservoir use since the late 1800’s. It may be difficult to determine the thresholds below which irreversible impacts occur (Aldous and Bach 2014). Fens with severe disturbances to their peat body or hydrologic regime may not be restored by public land managers because of the high costs and lack of expertise (Schimelpfenig et al. 2014). Sites with private water rights could still be developed as water supply reservoirs causing additional impacts. Ditches and vehicle ruts could be filled to restore fen hydrologic regimes and peat accumulation functions (Chimner et al. 2010; Schimelpfenig et al. 2014). Buffers of 100 m or less are inadequate for protecting the hydrologic processes and water quality of rich fens (Jones 2003). Buffer effectiveness depends on surrounding slope, groundwater flow regimes, OC, and soil type (Hruby 2013). Travel management guidelines should be updated to restrict ATV and snowmobile travel across fens. Reservoir maintenance permits should include guidelines for avoiding fens with mechanized equipment. Fen reference sites, such as Coyote Fen on Grand Mesa, should be preserved, and the remaining unimpacted ~80 ha of fen on Grand Mesa protected.
References
Aiken SG, Dallwitz MJ, Consaul LL, McJannet CL, Gillespie LJ, Boles RL, Argus GW, Gillett JM, Scott PJ, Elven R, LeBlanc MC, Brysting AK, Solstad H (1999) Flora of the Canadian Arctic Archipelagod, Illustrations, identification, and information retrieval. Version: 29, April 2003. http://www.mun.ca/biology/delta/arcticf/’. Accessed 27 Nov 2007
Aldous AR, Bach LB (2014) Hydro-ecology of groundwater-dependent ecosystems: applying basic science to groundwater management. Hydrol Sci J 59:530–544
Analytical Software (2007) Statistix 7 software program. Tallahassee. www.statistix
Anderson DS, Davis RB (1997) The vegetation and its environments in Maine peatlands. Can J Bot 75:1785–1805
Anderson LE, Crum HA, Buck WR (1990) List of the mosses of North America north of Mexico. Bryologist 93(4):448–499
Armentano TV, Menges ES (1986) Patterns of change in the carbon balance of organic-soil wetlands of the temperate zone. J Ecol 74:755–774
Asada T, Warner BG, Schiff SL (2005) Effects of shallow flooding on vegetation and carbon pools in boreal peatlands. Appl Veg Sci 8:199–2008
Bedford BL, Godwin KS (2003) Fens of the United States: distribution, characteristics, and scientific connection versus legal isolation. Wetlands 23(3):608–629
Carsey K, Kittel G, Decker K, Cooper DJ, Culver D (2003) Field guide to the wetland and riparian plant associations of Colorado. Fort Collins, Colorado Natural Heritage Program
Chimner RA (2011) Restoring sedges and mosses into frost heaving iron fens, San Juan Mountains, Colorado. Mires Peat 8:1–9
Chimner RA, Cooper DJ (2003a) Carbon balances of pristine and hydrologically modified fens in the southern Rocky Mountains. Can J Bot 81:477–491
Chimner RA, Cooper DJ (2003b) Influence of water table levels on CO2 emissions in a Colorado subalpine fen: an in situ microcosm study. Soil Biol Biochem 35:345–351
Chimner RA, Lemly JM, Cooper DJ (2010) Mountain fen distribution, types and restoration priorities, San Juan Mountains, Colorado, USA. Wetlands 30:763–771
Clarkson BR, Sorrell BK, Reeves PN, Champion PD, Partridge TR, Clarkson BD (2004) Handbook for monitoring wetland condition. Coordinated Monitoring of New Zealand Wetlands. National Institute of Water and Atmospheric Research. Christchurch
Cooper DJ (1990) Ecology of wetlands in Big Meadows, Rocky Mountain National Park, Colorado. Biol Rep 90:15
Cooper DJ (1991) Additions to the peatland flora of the southern Rocky Mountains: habitat descriptions and water chemistry. Madroño 38:139–143
Cooper DJ (1996) Water and soil chemistry, floristics, and phytosociology of the extreme rich High Creek fen, in South Park, Colorado, U.S.A. Can J Bot 74:1801–1811
Cooper DJ, Andrus R (1994) Patterns of vegetation and water chemistry in peatlands of the west-central Wind River Range, Wyoming. Can J Bot 72:1586–1597
Cooper SV, Jones WM (2004) A plant community classification for Kootenai National Forest peatlands. Unpublished report. Montana Natural Heritage Program, Montana State Library, Helena
Cooper DJ, MacDonald LH (2000) Restoring the vegetation of mined peatlands in the southern Rocky Mountains of Colorado, U.S.A. Restor Ecol 8:103–111
Cooper DJ, MacDonald LH, Wenger SK, Woods S (1998) Hydrologic restoration of a fen in Rocky Mt. National Park, Colorado. Wetlands 18:335–345
Cooper DJ, Andrus R, Arp CD (2002) Sphagnum balticum in a southern Rocky Mountain iron fen. Madrono 49:186–188
Cooper DJ, Chimner RA, Merritt DM (2012) Mountain wetlands of North America. In: Batzer D, Balswin A (eds) Wetland habitats of North America: ecology and conservation concerns. University of California Press, Berkeley
Cooper DJ, Wolf EC, Ronayne MJ, Roche JW (2015) Effects of groundwater pumping on the sustainability of a mountain wetland complex, Yosemite National Park, California. J Hydro: Reg Stud 3:87–105
Dachnowski-Stokes AP (1941) Peat resources of Alaska. US Dept Agri Tech Bull 769
Driver KM (2010) Distinguishing the hydrologic regimes and vegetation of fens and wet meadows in the Rocky Mountains. Thesis Colorado State University, Ft. Collins
Drummond MA (1999) A multi-proxy study of late-quaternary climate history, Grand Mesa, western Colorado. Thesis University of Denver, Denver
Dufrene M, Legendre P (1997) Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecol Monogr 67:345–366
Earth Google (2011) Online satellite images of the earth. Google Earth Version 1.3.21.69. Mountain View, CA. http://earth.google.com. Accessed 23 Feb 2011
Ewing JM, Vepraskas MJ (2006) Estimating primary and secondary subsidence in an organic soil 15, 20, and 30 years after drainage. Wetlands 26:119–130
Gage E, Cooper DJ (2006) Carex limosa L. (mud sedge): a technical assessment. Unpublished peer-reviewed document prepared for the USDA Forest Service. Colorado State University, Fort Collins
Gignac LD, Gauthier R, Rochefort L, Bubier J (2004) Distribution and habitat niches of 37 peatland Cyperaceae species across a broad geographic range in Canada. Can J Bot 82:1292–1313
Glaser PH, Wheeler GA, Gorham E, Wright HE Jr (1981) The patterned mires of the Red Lake Peatland, Northern Minnesota: vegetation, water chemistry and landforms. J Ecol 69:575–599
Heidel B, Jones G (2006) Botanical and ecological characteristics of fens in the Medicine Bow Mountains, Medicine Bow National Forest: Albany and Carbon Counties, Wyoming. Unpublished document. Wyoming Natural Heritage Database, U of Wyoming, Laramie
Heidel B, Laursen S (2003) Botanical and ecological inventory of peatland sites on the Medicine Bow National Forest. Unpublished document. Wyoming Natural Diversity Database, U of Wyoming. Laramie
High Plains Regional Climate Center (2015) National climate services support program. High Plains Regional Climate Center, Lincoln. http://www.hprcc.unl.edu/cgi-bin/cli_perl_lib/cliMAIN.pl?co7656. Accessed 19 April 2015
Hill MO, Gauch HG (1980) Detrended correspondence analysis: an improved ordination technique. Vegetatio 42:47–58
Holden J, Chapman PJ, Labadz JC (2004) Artificial drainage of peatlands: hydrological and hydrochemical process and wetland restoration. Prog Phys Geogr 28:95–123
Hruby, T (2013) Update on wetland buffers: the state of the science, final report. Washington State Department of Ecology Publication #13-06-11
Johnson JB (1997) Stand structure and vegetation dynamics of a subalpine treed fen in Rocky Mountain National Park, Colorado. J Veg Sci 8(3):337–342
Johnson K, Miller I (2012) Digging Snowmastodon: discovering an ice age world in the Colorado Rockies. Denver Museum of Nature and Science, Denver
Johnson JB, Steingraeber DA (2003) The vegetation and ecological gradients of calcareous mires in the South Park valley, Colorado. Can J Bot 81:201–219
Johnston BC (1987) Plant associations of Region Two, Edition 4. USDA Forest Service, Rocky Mountain Regional Office. Publication R2-ECOL-87-2. Lakewood
Johnston BC, Stratton BT, Young WR, Mattson LL, Almy JM, Austin G (2012) Inventory of fens in a large landscape of west-central Colorado: Grand Mesa, Uncompahgre, and Gunnison National Forests. USDA Forest Service
Jones WM (2003) Kootenai National Forest peatlands: description and effects of forest management. Montana Natural Heritage Program, Natural Resource Information System, Montana State Library. Helena, MT
Kartesz JT (1999) A synonymized checklist and atlas with biological attributes for the vascular flora of the United States, Canada, and Greenland. First edition. In: Kartesz, J.T., and C.A. Meacham. Synthesis of the North American Flora, Version 1.0. North Carolina Botanical Garden, Chapel Hill
Lamers LPM, Vile MA, Grootjans AP, Acreman MC, van Diggelen R, Evans MG, Richardson CJ, Rochefort L, Kooijman AM, Roelofs JGM, Smolders AJP (2015) Ecological restoration of rich fens in Europe and North America: from trial and error to an evidence-based approach. Biol Rev 90:182–203
Leibowitz S (2003) Isolated wetlands and their functions: an ecological perspective. Wetlands 23:517–531
Lemly JM (2007) Fens of Yellowstone National Park, USA: regional and local controls over plant species distribution. Thesis Colorado State University, Fort Collins
Lemly JM, Cooper DJ (2011) Multiscale factors control community and species distribution in mountain peatlands. Botany 89:689–713
Loughlin GF (1918) Mineral resources of the United States, part II—nonmetals. Department of the Interior, United States Geological Survey. Government Printing Office, Washington, D.C. 331–356
McCune B, Grace JB (2002) Analysis of ecological communities. MjM Software Design, Gleneden Beach
McCune B, Mefford MJ (1999) PC-ORD. Multivariate analysis of ecological data. Version 4.0. MjM Software, Gleneden Beach
Mueller-Dombois D, Ellenberg H (1974) Aims and methods of vegetation ecology. Wiley, New York
NatureServe (2008) An online encyclopedia of life. Version 6.3. Arlington. http://www.NatureServe.org/explorer. Accessed 3 Jan 2008
NRCS (Natural Resource Conservation Service) (1999) Soil taxonomy, a basic system of soil classification for making and interpreting soil surveys, 2nd edn. United States Department of Agriculture, Natural Resources Conservation Service, Washington
Økland RH (1996) Are ordination and constrained ordination alternative or complementary strategies in general ecological studies? J Veg Sci 7:289–292
Patterson L, Cooper DJ (2007) The hydrologic and ecological indicators for the restoration of drainage ditches and water diversions in a mountain fen, Cascade Range, California. Wetlands 27:290–304
Price JS, Rochefort L, Campeau S (2002) Use of shallow basins to restore cutover peatlands: hydrology. Restor Ecol 10(2):259–266
Price JS, Cagampan J, Kellner E (2005) Assessment of peat compressibility: is there an easy way? Hydrol Process 19:3469–3475
Retzer JL (1954) Glacial advances and soil development, Grand Mesa, Colorado. Am J Sci 252:26–37
Rocchio J (2004) Wetland classification of Blanca Wetlands, San Luis Valley, Colorado. Unpublished document. Colorado Natural Heritage Program. Ft. Collins
Schimelpfenig DW, Cooper DJ, Chimner RA (2014) Effectiveness of ditch blockage for restoring hydrologic and soil processes in mountain peatlands. Restor Ecol 22:257–265
Sherwood JH, Kettridge N, Thompson DK, Morris PJ, Silins U, Waddington JM (2013) Effect of drainage and wildlife on peat hydrophysical properties. Hydrol Process, John Wiley and Sons, Ltd., Wiley. Online. doi:10.1002/hyp.9820
Siegel DI (1968) Evaluating cumulative effects of disturbance on the hydrologic function of bogs, fens, and mires. Environ Manag 12:621–626
Siegel DI, Glaser P (2006) The hydrology of peatlands. In: Wieder RK, Vitt DH (eds) Boreal Peatland Ecosystems. Springer, Berlin Heidelberg
Sikes K, Cooper DJ, Weis S, Keeler-Wolf T, Barbour M, Ikeda D, Stout D, Evens J (2013) Fen conservation and vegetation assessment in the National Forests of the Sierra Nevada and adjacent mountains, California. Unpublished document. California Native Plant Society Vegetation Program, Colorado State University, US Forest Service, California Department of Fish and Wildlife, University of California Davis. Revised public version 2
Sjors H (1950) On the relation between vegetation and electrolytes in northern Swedish mire waters. Oikos 2:241–258
Slack NG, Vitt DH, Horton DG (1980) Vegetation gradients of minerotrophically rich fens in western Alberta. Can J Bot 58:330–350
St. Louis VL, Partridge AD, Kelly CA, Rudd JWM (2003) Mineralization rates of peat from eroding peat islands in reservoirs. Biogeochemistry 64:97–100
ter Braak CJF (1986) Canonical correspondence analysis: a new eigenvector technique for multivariate direct gradient analysis. Ecology 67:1167–1179
ter Braak CJF, Smilauer P (2002) CANOCO reference manual and CanoDraw for Windows user’s guide: software for canonical community ordination (version 4.5). Microcomputer Power, Ithaca
USFWS (US Fish and Wildlife Service) (1995) National wetland inventory maps. US Dept of the Interior, Fish and Wildlife Service, Washington
van Tongeren OFR (1995) Cluster analysis. In: Jongman RHG et al (eds) Data Analysis in Community and Landscape Ecology. Cambridge University Press, New York
Viereck LA, Dyrness CT, Batten AR, Wenzlick KJ (1992) The Alaska vegetation classification. US Dept of Agriculture, Forest Service, Pacific Northwest Research Station. Gen Tech Rep, PNWGTR-286. Portland
Vitt DH (2000) Peatlands: ecosystems dominated by bryophytes. In: Shaw AJ, Goffinet B (eds) Bryophyte Biology. Cambridge University Press, Cambridge
Vitt DH (2014) A key and review of bryophytes common in North American peatlands. In: S. LaGreca (Ed.). Evansia 31:121-156
Vitt DH, Chee WL (1990) The relationships of vegetation to surface-water chemistry and peat chemistry in fens of Alberta, Canada. Vegetatio 89:87–106
Vitt DH, Slack NG (1975) An analysis of the vegetation of Sphagnumdominated kettle-hole bogs in relation to environmental gradients. Can J Bot 53:332–359
Vitt DH, Wieder RK (2006) Boreal peatland ecosystems: our carbon heritage. In: R.K. Wieder and D.H. Vitt (Eds.), Ecol Studies
Warner BG, Asada T (2006) Biological diversity of peatlands in Canada. Aquat Sci 68:240–253
Weber WA, Wittmann RC (2012) Colorado flora: western slope. University Press of Colorado, Boulder
Weber WA, Wittmann RC (2007) Bryophytes of Colorado: mosses, liverworts, and hornworts. Pilgrims Process, Inc, USA
Weixelman DA, Cooper DJ (2009) Assessing proper functioning condition for fen areas in the Sierra Nevada and Southern Cascade Ranges in California, a user guide. Vallejo, CA: US Dept of Agriculture, Forest Service, Pacific Southwest Region, Gen Tech Rep R5-TP-028
Western Regional Climate Center (2007) Bonham Reservoir (050825) period of record monthly climate summary, 7/1/1963 to 6/30/2007. Online data: wrcc@dri.edu. Accessed 27 Nov 2007
Whitehouse HE, Bayley SE (2005) Vegetation patterns and biodiversity of peatland plant communities surrounding mid-boreal wetland ponds in Alberta, Canada. Can J Bot 83:621–637
Wind-Mulder HL, Vitt DH (2000) Comparisons of water and peat chemistries of a post-harvested and undisturbed peatland with relevance to restoration. Wetlands 20(4):616–628
Winters DS, Bohn B, Cooper DJ, Eaglin G, Hamerlinck JD, Hirsch C, Poff NL, Quimby CM, Rahel FJ, Rau P, Scaife D, Staley DM, Wohl EE (2006) Conceptual framework and protocols for conducting multiple scale aquatic, riparian, and wetland ecological assessments for the USDA Forest Service, Rocky Mountain Region. Species Conservation Project, Lakewood
Wolf EC, Cooper DJ (2015) Fens of the Sierra Nevada, California, USA: patterns of distribution and vegetation. Mires and Peat 15: in press
Yeend WE (1965) Quaternary geology of the Grand Mesa area western Colorado. Doctoral thesis. U of Wisconsin, Ann Arbor
Yeend WE (1973) Slow-sliding slumps, Grand Mesa, Colorado. Mt Geol 10:25–28
Zoltai SC, Vitt DH (1995) Canadian wetlands: environmental gradients and classification. Vegetatio 118:131–137
Acknowledgments
The research was supported in part by the USDA Forest Service (USFS), Gunnison Ranger District. We appreciated the statistical assistance from Joanna Lemly. Special thanks to Drs. Bill Weber, Ron Wittmann and Richard Andrus for bryophyte identification. We also thank Mark Hatcher, Linda Bledsoe, and Mike Brown, USFS, for logistical support.
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix 1
Historic aerial photographs of impacts in Kennecott Fen on Grand Mesa. Top left clockwise, 1936, 1956, 1978, 2011. The 1936 and 1956 photographs indicate that the primary peat mass is still intact. The 1978 photograph shows flooding (dark color) and the peat mass breaking apart. In the Earth Google (2011) photograph the peat mass has sunken to the bottom of the reservoir and any remaining plant communities are drowning. 
Appendix 2
Descriptions of the 16 fen, marsh, wet meadow, and modified fen plant communities.
Semi-aquatic communities
-
(1)
Nuphar lutea ssp. polysepala—Potamogeton nodosus a non-peat forming floating aquatic plant community, occurred in fen pools (n = 10) and marshes (n = 1). Similar communities occur in Wyoming (Cooper and Andrus 1994; Heidel and Laursen 2003; Lemly 2007), Alaska (Viereck et al. 1992), Idaho, Montana, Oregon, Washington, California, and British Columbia (NatureServe 2008).
-
(2)
Eleocharis macrostachya—Potamogeton foliosus; a semi-aquatic non-peat-forming plant community, occurred primarily in the pools of basins that had been modified for reservoir use. Similar communities (A2) occur primarily in hydrologically modified fens on Grand Mesa and marshes with fluctuating water levels across North America (Kartesz 1999).
Large sedge communities
-
(3)
Carex vesicaria dominated a peat-forming plant community that was distributed widely across the study area, primarily in fens and wet meadow basins. Standing water occurred during measurement periods. Similar communities occur in Colorado (Carsey et al. 2003), Wyoming (Heidel and Laursen 2003; Heidel and Jones 2006), Montana, Idaho, Washington, Oregon, and California (NatureServe 2008).
-
(4)
Carex utriculata dominated a peat-forming plant community that was widespread in the study area. Carex utriculata dominated vegetation occurs widely in Alaska (Viereck et al. 1992), Colorado (Cooper 1990; Carsey et al. 2003), and Wyoming (Cooper and Andrus 1994; Heidel and Laursen 2003).
-
(5)
Carex saxatilis—Drepanocladus aduncus was the most widespread peat-forming plant community in fens (n = 54) and wet meadows (n = 9) in the study area. Similar communities occur in Colorado (Carsey et al. 2003), Alaska (Aiken et al. 1999), western Canada (Gignac et al. 2004), Utah, Montana, and Washington (Natureserve 2008).
Floating mat and bryophyte communities
-
(6)
Menyanthes trifoliata dominated a peat-forming plant community found only in seasonal to perennial pools in basins and small depressions. It is rare in the study area and occurred only in undisturbed sites. Similar communities occur in Alaska (Dachnowski-Stokes 1941; Viereck et al. 1992).
-
(7)
Carex limosa—Sphagnum teres—Calliergon cordifolium is a peat-forming plant community that occurred only in basins with consistent soil saturation and mean water table depth of 6 cm. Similar communities are found in Wyoming (Cooper and Andrus 1994; Heidel and Laursen 2003; Heidel and Jones 2006; Lemly 2007), Montana (Cooper and Jones 2004), Colorado (Johnson and Steingraeber 2003), Minnesota (Glaser et al. 1981), California (Wolf and Cooper 2015), and Canada (Gignac et al. 2004).
-
(8)
Calliergon stramineum—Carex limosa—Menyanthes trifoliata is a peat-forming plant community found in basins with open water.
-
(9)
Sphagnum teres—Calliergon cordifolium—Carex canescens, a peat-forming plant community found in basin fens with floating peat mats. This community is similar to C. canescens communities in Wyoming (Cooper and Andrus 1994; Heidel and Laursen 2003) and Canada (Gignac et al. 2004).
Small sedge communities
-
(10)
Carex aquatilis—Drepanocladus aduncus is the second most widespread peat-forming plant community found in fens (n = 37) and wet meadows (n = 3). Stands occur along wetter margins of fens or as the dominant community in wet meadows. Similar communities are common in Colorado (Johnston 1987; Cooper 1990; Carsey et al. 2003), Wyoming (Cooper and Andrus 1994; Heidel and Laursen 2003; Heidel and Jones 2006), Montana (Cooper and Jones 2004), and Canada (Gignac et al. 2004).
-
(11)
Carex simulata—Drepanocladus aduncus, a peat producing community in fens (n = 16) and wet meadows (n = 2) occurred on gently sloping hillsides and small depressions. These stands had higher EC and high canopy cover of D. aduncus. Similar communities occur in mid to high elevations in Colorado (Cooper 1990; Carsey et al. 2003; Rocchio 2004), Wyoming (Heidel and Laursen 2003; Lemly 2007), Montana (Cooper and Jones 2004), California (Wolf and Cooper 2015), Nevada, Utah, Oregon, and Idaho (NatureServe 2008).
-
(12)
Eleocharis quinqueflora—Drepanocladus aduncus—Carex simulata, a widespread peat-forming plant community found in fens (n = 24) and wet meadows (n = 7) on slopes and in depressions. Similar communities occur across the western United States (NatureServe 2008), including Colorado (Cooper 1990; Carsey et al. 2003), Wyoming (Heidel and Laursen 2003; Lemly 2007), and Montana (Cooper and Jones 2004).
-
(13)
Carex illota—Aulacomnium palustre—Pedicularis groenlandica, a peat-forming community found along the margins of seven basin fens and one wet meadow. Similar communities occur in Wyoming (Cooper and Andrus 1994), California (Wolf and Cooper 2015), Colorado (Carsey et al. 2003), and Oregon (NatureServe 2008).
-
(14)
Eleocharis acicularis—Hippuris vulgaris, a non-peat forming plant community found only in hydrologically modified fens with flooding, drainage, fluctuating water tables, and bare peat. Similar communities occur in Colorado, Wyoming, and California and typically has little plant diversity (NatureServe 2008).
Shrub and Forest communities
-
(15)
Salix planifolia—Calamagrostis canadensis occurred on the margins of fens and wet meadows. This type is common on Grand Mesa and in Colorado (Cooper 1990; Carsey et al. 2003; Johnson and Steingraeber 2003), Alaska (Viereck et al. 1992), Utah, and Wyoming (NatureServe 2008).
-
(16)
Picea engelmannii—Salix planifolia—Climacium dendroides occurred on the margins of fens and wet meadows. Of 13 stands, 11 were fens. Johnston (1987) classified a Picea engelmannii community in Colorado which is possibly related to a similar community (F1) on Grand Mesa.
Rights and permissions
About this article
Cite this article
Austin, G., Cooper, D.J. Persistence of high elevation fens in the Southern Rocky Mountains, on Grand Mesa, Colorado, U.S.A.. Wetlands Ecol Manage 24, 317–334 (2016). https://doi.org/10.1007/s11273-015-9458-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11273-015-9458-7