Abstract
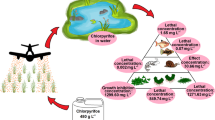
Diflubenzuron (DFB) is a larvicide widely used to control Aedes aegypti populations as an alternative to organophosphates (OPs), with a specific mechanism of action for insects by inhibiting their chitin synthesis. However, DFB is used extensively in urban and rural environments, having the aquatic environment as the major receptor. Thus, the present study aimed to investigate the toxicity of DFB-based formulation and compare it with the toxicity of the OP temephos (TMP)-based formulation, a larvicide still used to control A. aegypti, on freshwater fishes Oreochromis niloticus and Hyphessobrycon eques. Organisms were submitted to acute (48 h) and prolonged (7 days) exposures, in the presence and absence of organic sediment, seeking interactions between chemical and sediment. Histopathological analyses were performed on O. niloticus gills and liver. According to 48-h median lethal concentration (LC50), DFB- and TMP-based formulations were classified as harmful and toxic to fish, respectively, following the Globally Harmonized System of Classification (GHS). After prolonged exposure to sublethal concentrations, DFB-based formulation decreased H. eques body weight at concentrations 272-fold lower than its LC50. Ultrastructural responses of O. niloticus indicated edemas and aneurisms on gills, and hepatocyte hypertrophy and vascular congestion of the liver. TMP-based formulation also induced pyknotic nuclei, which may lead to irreversible necrosis. The addition of organic sediment did not alter the larvicide toxicity, suggesting that larvicides remained available to the organisms. Altogether, these results suggest that as an insect-specific pesticide, DFB still induces mortality and tissue damage in fishes; thus, both larvicides pose risks to fishes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Aedes aegypti (Diptera, Culicidae) is a mosquito widespread in the world, particularly in tropical and subtropical regions. This species plays an important role as the main vector for the transmission of dengue disease, in addition to the transmission of chikungunya, yellow fever, and Zika infection, and such epidemic diseases have rapidly spread worldwide. In a recent report, the World Health Organization estimated 390 million cases of dengue infection yearly (WHO 2018). The global concern has drastically increased the use of insecticides to control the A. aegypti population and interrupt the virus transmission. Brazil, the largest country in South America with a predominant tropical area, faces a dramatic incidence of dengue, with more than 265,000 probable cases of infection reported in 2018 (Brazil 2019).
The organophosphate (OP) insecticides temephos (TMP) and malathion, for instance, are widely used in urban and domestic areas for A. aegypti control; however, there is a decreasing trend in OP usage due to their mechanism of action on the cholinesterase system (USEPA 2000, 2017). It results in the loss of available acetylcholinesterase enzyme (AChE) so that the effector organ becomes overstimulated by the excess of the neurotransmitter acetylcholine (ACh) in the nerve ending, and consequently causing random synapses, muscle spasms, paralysis, and death (Fukuto 1990).
The non-specific mechanism of action of the OPs on insects (Lim and Bolstad, 2018), in addition to the increasing resistance of the vector to OPs (Grisales et al. 2013; Bellinato et al. 2016; Viana-Medeiros et al. 2017; Goindin et al. 2017), have stimulated the search for insecticide classes with mechanisms of action more selective to insects (Mulla 1995). Insect growth regulators (IGR) have been shown to be a promising alternative due to their specific mechanism of action on insects and their potential low toxicity to non-target organisms. Diflubenzuron (DFB) is an IGR belonging to the class of the benzoylphenylurea exhibiting the intoxication mechanism by inhibiting chitin synthesis, thus preventing insect molting (Sánchez-Bayo 2011). This pathway is present in insects and crustaceans, although some studies have shown that DFB can also affect fishes’ (Souza et al. 2011; Zaidi and Soltani 2011; Olsvik et al. 2013) and mammals’ health (Barros et al. 2014). However, studies designed to evaluate environmental concentrations of DFB and TMP in aquatic ecosystems are out of date and limited to aerial application in agriculture, marshes, and forest sites (Lores et al. 1985; Fischer and Hall 1992; Molina et al. 1995; Pierce et al. 2000).
Once the extensive use of larvicides becomes environmentally unsafe to aquatic ecosystems through runoff water from agricultural lands and urban areas or by direct application, these larvicides provide risks of environmental intoxication for several aquatic organisms (Hurst et al. 2007, Guimarães et al. 2007, Tilak and Kumari 2009, Ba-Omar et al. 2011, Singh 2013, Olsvik et al. 2013 and so on). Regarding the studies investigating the DFB and TMP effects to the aquatic environment, there is a lack of information of their toxicological potential. Considering the extensive use of these insecticides in public health and agriculture and the risk posed to non-target organisms, the present work aimed to investigate and compare the toxicity of DFB and TMP to the fishes Oreochromis niloticus and Hyphessobrycon eques after short (48 h) and prolonged (7 days) exposure. We also investigated whether the presence of organic sediment would change the toxicity of larvicides to the fishes. The endpoints assessed were lethality and sublethality, using histopathological analyses of gills, once the contact between contaminants and gills can alter their filaments and respiratory lamellae (Guimarães et al. 2007; Ba-Omar et al. 2011), and livers, as the main site of metabolization and biotransformation of xenobiotics (Singh 2013).
2 Materials and Methods
2.1 Chemicals
The commercial formulations of DFB (CAS no. 35367-38-5, wettable powder formulation Champion® 25% active ingredient (a.i.), Champion Farmoquímico Ltda, Brazil) and TMP (CAS no. 3383-96-8, sand granule formulation Fersol® 1% a.i., Fersol Indústria e Comércio S/A, Brazil), were kindly provided by the Ministry of Health of Brazil. Stock solutions of diflubenzuron were prepared in distilled water followed by successive dilutions in dechlorinated tap water for nominal exposure concentrations. Temephos was directly weighted and diluted in dechlorinated tap water for each nominal exposure concentration. Additional property information of the active ingredients are shown in Table 1.
2.2 Organic Sediment
Organic sediment was collected from the topsoil in a natural area that had never received pesticide application and had not been subject of residues from human activities. The sediment was sieved and the chemical and textural compositions are shown in Table 2.
2.3 Test Organisms
Organisms used in the subsequent tests were O. niloticus fingerlings (average weight of 2 g ± 0.5) and H. eques adults (average weight of 0.9 g ± 0.1), obtained from the Aquaculture Center of the São Paulo State University (CAUNESP) (Jaboticabal, Brazil). Homogeneous batches of fishes were transferred to the Laboratory of Ecotoxicology and Occupational Health of the São Paulo State University (UNESP) (Jaboticabal, Brazil) and acclimated for 10 days in a 250-L water tank. Fishes were fed daily with commercial feed (Polinutri®, crude protein 32%). The oxygenation and water renewal were kept constant and debris were removed daily. Acclimated laboratory conditions were maintained constant along all tests (27 ± 2 °C, 16/8 h of light/dark photoperiod).
2.4 Toxicity Tests
Tests were performed according to the OECD guideline 203 ( 1992) to fish acute toxicity test, with adaptations to acute (48-h) and prolonged (7-day) exposure. Based on preliminary tests (data not shown), we exposed O. niloticus fingerlings and H. eques adults to a gradient of concentrations of DFB and TMP formulations (Table 3). Dechlorinated tap water was used as negative control.
In short, after a 15-d acclimation period in 500-L tanks, fishes were randomly selected for larvicidal exposures in the presence and absence of sediment. Fishes were distributed into glass aquariums filled with 2 L dechlorinated tap water, and let them re-acclimate during 24 h. Thereafter, larvicides were carefully added into the aquariums to 3 L final volume of test solutions. In tests with sediment, before the tests begin, 900 g of dry sediment were added into aquariums filled with 2 L dechlorinated tap water and allowed to decant during 24 h. Thereafter, tests were carried out as described above. Tests were performed in a static system (no solutions renewal).
The 48-h exposures of H. eques and O. niloticus were carried out without feeding and mortality was recorded daily. At the end of the exposure period, at least three surviving O. niloticus per treatment and control were euthanized by immersion bath with benzocaine (Sigma-Aldrich) and gills and livers were collected for histopathological analysis. Tests were performed in three independent replicates, each replicate consisting of 3 glass aquariums containing 3 fishes each (n = 9 organisms × 3 replicates).
The 7-day exposures of H. eques were carried out with constant oxygenation; fishes were fed every 2 days, followed by debris siphoning, and mortality recorded. The organisms were weighed before starting the tests and at the end, and fish body weight was evaluated by subtracting the final weight from the initial weight. Tests were performed in three independent replicates, each replicate consisting of 5 glass aquariums containing 5 fishes each for the 7-day exposure (n = 15 organisms × 3 replicates).
During the experiments, the abiotic parameters (temperature, conductivity, dissolved oxygen, and pH) were continuously monitored (data not shown).
2.5 Tissue Collection and Processing
After collection, gills and livers of O. niloticus were immediately fixed in formaldehyde 10% buffered (PBS buffer, 0.1 M, pH 7.2). After 24 h, tissue samples were washed in PBS buffer and submitted to dehydration in graded ethanol series, diaphanization, and inclusion in Histosec® (Merck, Germany). Semi-thin sections (4.0 μm) were obtained by automatic microtome (Leica, RM-2155) and stained with hematoxylin-eosin (H.E.), following the method described by Carraschi et al. (2012). Slides with gill and liver tissues were analyzed and photographed under a light microscope (Leica 5000MB coupled with camera DP10).
2.6 Data Analysis
GraphPad Prism 5.01 software (GraphPad Software, San Diego, California, USA) was used for statistical analysis. One-way ANOVA was applied with a significant level of 5% and Dunnett’s test was used to compare differences between controls and treatments and determined the lowest observed effect concentration (LOEC) values. The median lethal concentration (LC50) and confidence intervals (upper and lower limits, respectively UL and LL) were calculated by the trimmed Spearman-Karber method (Hamilton et al. 1977). In addition, the potential aquatic toxicity classification of DFB- and TMP-based formulations was determined according to the criteria of the Globally Harmonized System of Classification guidance on hazards to the aquatic environment (GHS 2017).
3 Results and Discussion
3.1 Toxicity Tests
In the present study, the larvicide DFB- and TMP-based formulations were individually evaluated on two freshwater fishes H. eques and O. niloticus in the presence and absence of sediment. Both larvicides induced fish mortality after acute exposure in a dose-dependent manner (suppl. data), and the TMP-based formulation was slightly more toxic than the DFB-based formulation to both fish species. The LC50 of the TMP-based formulation was in the range of 5–7 mg a.i./L, and the LC50 of the DFB-based formulation was at 10–12 mg a.i./L (Table 4).
The presence of organic sediment did not significantly alter the lethal toxicity of both larvicide formulations to the fishes (Table 4). Despite the hydrophobicity of DFB and TMP molecules, which would allow them to be easily adsorbed in sediment due to their high adsorption coefficient (Swann et al. 1983), our results indicated that DFB- and TMP-based formulations were not significantly absorbed in order to decrease the toxicity to the tested organisms, suggesting a similar bioavailability between the toxicity tests with and without sediment.
Comparing our results of acute toxicity with data from literature, we found several ranges of toxicity of inhibitors of chitin synthesis-based pesticides (Souza et al. 2011; Medeiros et al. 2013) and OP-based pesticides (Selvi et al. 2005, Zhang et al. 2010, Uner et al. 2010, Ba-Omar et al. 2011, Kavitha and Rao 2009, Patil and David 2010, Singh et al. 2018) to fishes (Table 5). Data summarized by Fischer and Hall (1992) showed that DFB 96-h LC50 for fish species ranged from 40 to 660 mg/L after exposure to 25%-WP formulation. Souza et al. (2011) also reported the 96-h LC50 of DFB-based formulation Dimilin® of 151.98 mg/L to fish Poecilia reticulata. Aquatic invertebrates showed to be hundred times more susceptible to DFB than fishes during acute exposures. The 48-h LC50 DFB reported for D. magna ranges from 0.06 to 16 μg/L, categorizing the larvicide as extremely toxic to the species due to the mechanism of DFB toxicity of disruption of chitin formation (Fischer and Hall 1992; EPA 1997; Souza et al. 2011; Abe et al. 2014). Based on our results of the acute toxicity for fish, TMP-based formulation is classified as toxic to aquatic life (1 < LC50 < 10 mg/L) and DFB-based formulation as harmful to aquatic life (10 < LC50 < 100 mg/L), according to the GHS guidance on hazards to the aquatic environment (GHS 2017).
The variety in the toxicity of larvicides reported in the literature to fish species has also been linked to different formulations of pesticides, which plays a role for the toxicity characterization (Mesnage et al. 2015; Cruz et al. 2016). The presence of adjuvants, for example, would enable and enhance the pesticide activity, despite that they are considered inert diluents (Mesnage et al. 2015). Thus, the knowledge about the formulation toxicity is fundamental for decision-making, concerning the monitoring of possible environmental effects. Our experiments were conducted to estimate the effects of formulated product as encountered in reality, and not the technical-grade larvicides.
DFB-based formulation also decreased the body weight of H. eques after 7-day exposure (Fig. 1). The LOEC values for body weight loss were in the range of 40–90 μg a.i./L (Tables 3 and 4), weighing about 60 up to 80% less than fishes from the control group. This concentration range is up to 6.25-fold lower than the maximum concentration of DFB recommended by the Ministry of Health of Brazil to control A. aegypti larvae in urban areas. To date, there is a lack of information on the residual amount of DFB reaching the aquatic environment. Once the intensive use DFB-based formulations to control A. aegypti populations has extensively increased, it might reach the aquatic environment mainly through runoff water from urban areas or direct application on water, which becomes environmentally unsafe to aquatic ecosystems. According to the GHS guidance of classification of hazards to the aquatic environment (GHS 2017), DFB-based formulation is very toxic to H. eques (LOEC ≤0.1 mg/L) with long-lasting effects (Table 3). Fishes Gambusia affinis exposed during 28 days to lower concentrations of DFB-based formulation (16–72 ng a.i./L, WP formulation Dimilin® 25% a.i.) did not change length and body weight (Zaidi and Soltani 2011), showing that lower concentrations of DFB could not pose a concern to the fish species.
Effects of diflubenzuron (DFB) and temephos (TMP)-based formulations on Hyphessobrycon eques after a 7-day exposure in the presence (red bars) and absence (black bars) of sediment. a Body weight loss (%) of H. eques exposed to DFB-based formulation. b Mortality (%) of H. eques exposed to TMP-based formulation. Values represented per mean ± SEM (n = 45 organisms per treatment). The asterisk denotes Dunnett’s test, p < 0.05, for differences between exposed and control groups within each treatment
In contrast, some fishes exposed to low concentrations of TMP-based formulation during 7 days died until the end of the test, precluding the body weight measurement (Fig. 1), and the GHS classification for prolonged exposure to aquatic life. The LOEC values to mortality were in the range of 1.3–1.6 mg a.i./L for H. eques (Tables 3 and 4), up to 30% higher than the maximum concentration of TMP recommended by the Ministry of Health of Brazil to control A. aegypti larvae in urban areas. Other studies have reported that low concentrations of different TMP-based formulations (Abate 100E 10% a.i. and Abate® 50 EC 50% a.i.) also showed sublethal effects, such as lamellar damage (1 mg a.i./L) (Ba-Omar et al. 2011) and erratic swimming (0.33 mg a.i./L) (Hurst et al. 2007).
3.2 Histopathological Analyses
The effects of DFB- and TMP-based larvicide formulations were evaluated on gills and livers of O. niloticus after acute exposure in the absence of sediment, in order to investigate lesions or alterations at the tissue level. Both larvicides induced alterations in gill and liver cells, but TMP were more aggressive to the liver tissue, leading to cell necrosis.
3.3 Gill Histopathology
Light micrographs of O. niloticus gills tissue are described in Fig. 2. The histopathological analysis from fishes of the control group showed an organized gill tissue in arches supporting the primary lamellae. The secondary lamellae originated from the primary showed mucous and pillar cells supporting it, with interlamellar spaces between them comprising chlorite and lining cells. In fishes exposed to treatments from 3 mg a.i./L of DFB and 10 mg a.i./L of TMP-based formulations, gill tissue showed a breakdown of the epithelial cell system, with edemas in the epithelial cells and blood congestion (aneurism) inside the lamellae (Fig. 2 b–c). It is worth pointing out that TMP-based formulation caused significant mortality at this concentration (10 mg a.i./L), whereas the DFB-based formulation induced mortality at concentrations 3-fold higher (10 mg a.i./L) than concentrations able to induce such histopathological changes in gills, showing an early warning effect of its lethal toxicity.
Cross-sections of Oreochromis noliticus gills. a Control group, CVS: central venous sinus, PL: primary lamellae, SL: secondary lamellae, EC: epithelial cells, PC: pillar cells, BC: basal cells. b and c Diflubenzuron (3 mg a.i./L), ED: edema, AN: aneurism. D: temephos (10 mg a.i./L), ED: edema (400× amplification, color HE)
Such histopathological events have previously been reported in fish Aphanius dispar exposed to the commercial TMP 50 EC (Abate® 4-E, 50% a.i.) at 1 mg a.i./L, aggravating at 5 mg a.i./L with hypertrophy and lamellar fusion and at 25 mg a.i./L with cartilage destruction (Ba-Omar et al. 2011). Guimarães et al. (2007) also reported edema in the secondary lamellae, lamellar fusion, cell hypertrophy, and vascular congestion in O. niloticus exposed to 0.25 mg a.i./L of the OP trichlorfon. In the particular case of edemas, these events might be associated with a physiological process in response to toxicants that result in an enlargement of the distance between blood and water, and may lead to insufficient oxygen supply of the blood (Barillet et al. 2010). Tilak and Kumari (2009) have found a relationship between the toxicity of the OP dichlorvos to fish Ctenopharyngodon idella and the decrease of the oxygen consumption, due to damages in the epithelium of gill tissue and the depletion of gas exchange, causing a disorder in the respiratory system.
Breakdown of pillar cells was also observed in fishes exposed to 3 mg a.i./L of DFB-based formulation, altering the direction of the lamellae in relation to the water flow, which decreases the oxygen absorption (Fanta et al. 2003). As a result of this event, aneurisms were also observed (Fig. 2c). Gill aneurysm is a severe pathology resulting from the collapse of the pillar cell system and it can impair the vascular integrity and cause disruptions of the lamellar epithelium and subsequent hemorrhage (Hinton and Lauren 1990). A lamellar aneurysm is related to a serious and irreversible pathology in fish gills (Nascimento et al. 2012).
3.4 Liver Histopathology
Light micrographs of O. niloticus liver tissues are described in Fig. 3. The histopathological analysis from fishes of the control group showed central veins that branch out and originate from the blood vessels, responsible for the irrigation of the whole organ, and capillary sinusoids originated from the irrigation vessels. The hepatocytes had a slightly hexagonal shape, with central basophil nucleus and acidophil cytoplasm, organized through the capillary sinusoids with cordonal aspect. Within the hepatic tissue, hepatopancreatic exocrine was also observed, bordering the central veins (Fig. 3a).
Cross sections of Oreochromis noliticus liver tissues. a Control group, CV: central vein, arrow: hepatocytes. b Diflubenzuron (10 mg a.i./L), asterisk: hypertrophy of the hepatocytes. c Temephos (15 mg a.i./L), asterisk: hypertrophy of the hepatocytes, PN: pyknotic nucleus, VC: vascular congestion (400× amplification, Color HE)
Histopathological changes in liver tissue of O. niloticus exposed to 3 mg a.i./L of DFB and 5 mg a.i./L of TMP-based formulations included hepatocyte hypertrophy with displacement of the nucleus to the periphery of the cell, which occurs due to the increase in the number of organelles, such as the smooth endoplasmic reticulum and Golgi complex that are responsible for the metabolism of toxins (Ba-Omar et al. 2011; Carraschi et al. 2012). Fishes exposed to 10 mg a.i./L of DFB and 15 mg a.i./L of TMP-based formulations also showed vascular congestion inside the sinusoid capillaries (Fig. 3 b, c), when fish mortality was also significant.
These events, therefore, can be associated with defense mechanisms against surrounding toxicants, since the liver is the main site of detoxification process. The alterations occurred in attempt to eliminate the insecticide during the detoxification process. The capacity of the fish liver to detoxify DFB has been previously reported (Olsvik et al. 2013). Zaidi and Soltani (2011) indeed reported that fishes Gambusia affinis exposed to the commercial formulation of DFB Dimilin (25% a.i. wettable powder) increased the activity of the detoxification enzyme glutathione s-transferase in the liver at 78 ng a.i./L after 28 days of exposure, indicating that DFB is metabolized and detoxified by liver cells. The OP profenofos also increases the glutathione s-transferase activity in Oreochromis mossambicus fish, and induces the lipid peroxidation, suggesting that reactive oxygen species-induced oxidative damage could be one of the main toxic effects of the OP (Kavitha and Rao 2009).
In contrast to the reversible effects observed, TMP-based formulation at 30 mg a.i./L induced pyknotic nuclei, an extreme irreversible situation that organelles do not support the amount of toxins, condensing chromatins and causing necrosis of the tissue (Fig. 3c). Such events (hepatocytes hypertrophy, displacement of the nucleus to the periphery of the cell, vascular congestion, and pyknotic nuclei) have been previously reported in fishes Cyprinus carpio after 24 and 72 h of exposure to 0.4 mg a.i./L of the OP dimethoate (Singh 2013).
Therefore, the health effects of IGR and organophosphates deserve careful consideration. The ultra-structural changes in gill and liver tissues of O. niloticus exposed to DFB- and TMP-based formulations showed that those larvicides induce effects on tissue structure addressing physiological stress conditions, such as the detoxification process in the liver cells, and damage in the gill tissue hampering the oxygen consumption. DFB and TMP-based formulations showed to be acutely harmful and toxic, respectively, to the aquatic life. In an ecological point of view, the larvicides can compromise the fish interactions in the environment and impair survival at concentrations likely to be found in an environmentally realistic scenario, mainly after prolonged exposure to sublethal doses. These results might be useful to guide monitoring and mitigation programs to protect human health and the environment from the adverse effects of hazardous substances.
4 Conclusions
The present work provides additional insights into the toxic potential of DFB- and TMP-based larvicide formulations in fish H. eques and O. niloticus. DFB- and TMP-based formulations are acutely harmful and toxic to the fish species, respectively. After prolonged exposure to low concentrations (μg/L order), the larvicides can compromise fish health, and consequently interactions into the environment are threatened. DFB- and TMP-based formulations also induce stress conditions at the cellular level linked to physiological changes, such as an increase of detoxification processes in liver cells, and damage in the gill tissue hampering oxygen consumption. This combined approach affords an integrative analysis of DFB- and TMP-based larvicides on freshwater fish, useful for assessing environmental risk to fish species and in monitoring and mitigation programs to protect human and environmental health.
References
Abe, F.R., Coleone, A.C., Machado, A.P., Machado-Neto, J.G. (2014). Ecotoxicity and environmental risk assessment of larvicides used in the control of Aedes aegypti to Daphnia magna (Crustacea, Cladocera). Journal of Toxicology and Environmental Health, Part A: Current Issues, 77(1–3), 37–45. https://doi.org/10.1080/15287394.2014.865581.
Ba-Omar, T. A., Al-Jardani, S., & Victor, R. (2011). Effects of pesticide temephos on the gills of Aphanius dispar (Pisces: Cyprinodontidae). Tissue & Cell, 43, 29–38. https://doi.org/10.1016/j.tice.2010.11.002.
Barillet, S., Larno, V., Floriani, M., Devaux, A., & Adam-Guillermin, C. (2010). Ultrastructural effects on gill, muscle, and gonadal tissues induced in zebrafish (Danio rerio) by a waterborne uranium exposure. Aquatic Toxicology, 100, 295–302. https://doi.org/10.1016/j.aquatox.2010.08.002.
Barros, A. L., Cavalheiro, G. F., Souza, A. V. M., Traesel, G. K., Anselmo-Franci, J. A., Kassuya, C. A. L., & Arena, A. C. (2014). Subacute toxicity assessment of diflubenzuron, an insect growth regulator, in adult male rats. Environmental Toxicology, 31(4), 407–414. https://doi.org/10.1002/tox.22054.
Bellinato DF, Viana-Medeiros PF, Araújo SC, Martins AJ, Lima JBP, Valle D (2016) Resistance status to the insecticides temephos, deltamethrin, and diflubenzuron in Brazilian Aedes aegypti populations. BioMed Res Int. ID 8603263. doi https://doi.org/10.1155/2016/8603263.
Brazil. Ministry of Health. Secretariat of Health Surveillance (2019). Monitoramento dos casos de dengue, febre de chikungunya e doença aguda pelo vírus Zika até a Semana Epidemiológica 52 de 2018. Epidemiological Bulletin, 50(04). Available in: http://portalarquivos2.saude.gov.br/images/pdf/2019/janeiro/28/2019-002.pdf. Accessed in: 4 Feb. 2019.
Carraschi, S. P., Cruz, C., Machado-Neto, J. G., Ignácio, N. F., Barbuio, R., & Machado, M. R. F. (2012). Histopathological biomarkers in pacu (Piaractus mesopotamicus) infected with aeromonas hydrophila and treated with antibiotics. Ecotoxicology and Environmental Safety, 83, 115–120. https://doi.org/10.1016/j.ecoenv.2012.06.016.
Cruz, C., Carraschi, S. P., Shiogiri, N. S., Silva, A. F., Pitelli, R. A., & Machado, M. R. F. (2016). Sensitivity, ecotoxicity and histopathological effects on neotropical fish exposed to glyphosate alone and associated to surfactant. Journal of Environmental Chemistry and Ecotoxicology, 8(3), 25–33. https://doi.org/10.5897/JECE2015.0362.
EPA, (1997). Reregistration Eligibility Decision (RED) – Diflubenzuron. United States Environmental Protection Agency, Washington, DC. Available in: https://www3.epa.gov/pesticides/chem_search/reg_actions/reregistration/red_PC-108201_1-Jun-97.pdf. Accessed in: 4 Feb. 2019.
Fanta, E., Rios, F. S., Romão, S., Vianna, A. C. C., & Freiberger, S. (2003). Histopathology of the fish Corydoras paleatus contaminated with sublethal levels of organophosphorus in water and food. Ecotoxicology and Environmental Safety, 54, 119–130. https://doi.org/10.1016/S0147-6513(02)00044-1.
Fischer, S. A., & Hall, L. W. (1992). Environmental concentrations and aquatic toxicity data on diflubenzuron (Dimilin). Critical Reviews in Toxicology, 22(1), 45–79.
Fukuto, T. R. (1990). Mechanism of action of organophosphorus and carbamate insecticides. Environmental Health Perspectives, 87, 245–254. https://doi.org/10.1289/ehp.9087245.
GHS (2017). Globally Harmonized System of Classification and Labelling of Chemicals (GHS). Globally Harmonized System, GHS, New York and Geneva, United Nations, ST/SG/AC.10/30/Rev.7.
Goindin, D., Delannay, C., Gelasse, A., Ramdini, C., Gaude, T., Faucon, F., David, J. P., Gustave, J., Vega-Rua, A., Fouque, F. (2017). Levels of insecticide resistance to deltamethrin, malathion, and temephos, and associated mechanisms in Aedes aegypti mosquitoes from the Guadeloupe and Saint Martin islands (French West Indies). Infect Dis Poverty 6(38). doi: https://doi.org/10.1186/s40249-017-0254-x.
Grisales, N., Poupardin, R., Gomez, S., Fonseca-Gonzalez, I., Ranson, H., & Lenhart, A. (2013). Temephos resistance in Aedes aegypti in Colombia compromises dengue vector control. PLoS Neglected Tropical Diseases, 7(9), e2438. https://doi.org/10.1371/journal.pntd.0002438.
Guimarães, A. T. B., Silva de Assis, H. C., & Boeger, W. (2007). The effect of trichlorfon on acetylcholinesterase activity and histopathology of cultivated fish Oreochromis niloticus. Ecotoxicology and Environmental Safety, 68, 57–62. https://doi.org/10.1016/j.ecoenv.2006.08.005.
Hamilton, M. A., Russo, R. C., & Thurston, R. V. (1977). Trimmed Spearman-Karber method for estimating median lethal concentrations in toxicity bioassays. Environmental Science & Technology, 11, 714–719. https://doi.org/10.1021/es60130a004.
Hinton, D. E., & Lauren, D. J. (1990). Integrative histopathological approaches to detecting effects of environmental stressors on fishes. In R. Adams & R. Lloyd (Eds.), Biological indicators of stress in fish. Bethesda: American Fisheries Society.
Hurst, T. P., Kay, B. H., Ryan, P. A., & Brown, M. D. (2007). Sublethal effects of mosquito larvicides on swimming performance of larvivorous fish Melanotaenia duboulayi (Atheriniformes: Melanotaeniidae). Journal of Economic Entomology, 100(1), 61–65. https://doi.org/10.1093/jee/100.1.61.
Kavitha, P., & Rao, J. V. (2009). Sub-lethal effects of profenofos on tissue-specific antioxidative responses in a Euryhyaline fish, Oreochromis mossambicus. Ecotoxicol Environ Saf., 72, 1727–1733. https://doi.org/10.1016/j.ecoenv.2009.05.010.
Lewis, K. A., Tzilivakis, J., Warner, D., & Green, A. (2016). An international database for pesticide risk assessments and management. Human and Ecological Risk Assessment: An International Journal, 22(4), 1050–1064. https://doi.org/10.1080/10807039.2015.1133242.
Lores, E. M., Moore, J. C., Moody, P., Clark, J., Forester, J., & Knight, J. (1985). Temephos residues in stagnant ponds after mosquito larvicide applications by helicopter. Bulletin of Environmental Contamination and Toxicology, 35, 308–313.
Lim, L., Bolstad, H. M. (2018). Organophosphate insecticides: Neurodevelopmental effects. In: Reference Module in Earth Systems and Environmental Sciences. https://doi.org/10.1016/B978-0-12-409548-9.10930-3
Medeiros, L. S., Souza, J. P., Winkaler, E. U., Silvia, P. C., Cruz, C., Souza-Júnior, S. C., & Machado-Neto, J. G. (2013). Acute toxicity and environmental risk of teflubenzuron to Daphnia magna, Poecilia reticulata and Lemna minor in the absence and presence of sediment. Journal of Environmental Science and Health. Part. B, 48(7), 600–606. https://doi.org/10.1080/03601234.2013.775000.
Mesnage, R., Defarge, N., Spiroux de Vendômois, J., & Séralini, G. E. (2015). Potential toxic effects of glyphosate and its commercial formulations below regulatory limits. Food and Chemical Toxicology, 84, 133–153. https://doi.org/10.1016/j.fct.2015.08.012.
Molina, C., Durand, G., & Barceló, D. (1995). Trace determination of herbicides in estuarine waters by liquid chromatography-high-flow pneumatically assisted electrospray mass spectrometry. Journal of Chromatography. A, 712, 113–122.
Mulla, M. S. (1995). The future of insect growth regulators in vector control. Journal of the American Mosquito Control Association, 11(2), 269–273.
Nascimento, A. A., Araújo, F. G., Gomes, I. D., Mendes, R. M. M., & Sales, A. (2012). Fish gills alterations as potential biomarkers of environmental quality in a eutrophized Tropical River in South-Eastern Brazil. Anatomia Histologia Embryologia, 41, 209–216. https://doi.org/10.1111/j.1439-0264.2011.01125.x.
OECD. (1992). Test No. 203: Fish, Acute Toxicity Test, OECD Guidelines for the Testing of Chemicals, Section 2. Paris: OECD Publishing. https://doi.org/10.1787/9789264069961-en.
Olsvik, P. A., Samuelsen, O. B., Erdal, A., Holmelid, B., & Lunestad, B. T. (2013). Toxicological assessment of the anti-salmon lice drug diflubenzuron on Atlantic cod Gadus morhua. Diseases of Aquatic Organisms, 105, 27–43. https://doi.org/10.3354/dao02613.
Patil, V. K., & David, M. (2010). Behavioral and morphological endpoints: as an early response to sublethal malathion intoxication in the freshwater fish, Labeo rohita. Drug and Chemical Toxicology, 33(2), 160–165. https://doi.org/10.3109/01480540903196816.
Pierce, R. H., Henry, M. S., Kelly, D., & Kozlowski, W. (2000). Hazard assessment of temephos applied to a southwest Florida, USA, salt marsh community. Environmental Toxicology and Chemistry, 19(2), 501–507.
Sánchez-Bayo, F. (2011). Insecticides mode of action in relation to their toxicity to non-target organisms. Journal of Environmental & Analytical Toxicology, S4, 002. https://doi.org/10.4172/2161-0525.S4-002.
Selvi, M., Sarikaya, R., Erkoç, F., & Koçak, O. (2005). Investigation of acute toxicity of chlorpyrifos-methyl on guppy Poecilia reticulata. Chemosphere, 60, 93–96. https://doi.org/10.1016/j.chemosphere.2004.11.093.
Singh, R. N. (2013). Effects of dimethoate (30% EC), an organophosphate pesticide on liver of common carp, Cyprinus carpio. Journal of Environmental Biology, 34, 657–661.
Singh, S., Tiwari, R. K., & Pandey, R. S. (2018). Evaluation of acute toxicity of triazophos and deltamethrin and their inhibitory effect on AChE activity in Channa punctatus. Toxicol Reports, 5, 85–89. https://doi.org/10.1016/j.toxrep.2017.12.006.
Souza, J. P., Medeiros, L. S., Winkaler, E. U., & Machado-Neto, J. G. (2011). Acute toxicity and environmental risk of diflubenzuron to Daphnia magna, Poecilia reticulata and Lemna minor in the absence and presence of sediment. Pesticidas: R Ecotoxicol Meio Ambiente, 21, 1–12.
Swann, R. L., Laskowski, D. A., Mccall, P. J., Kuy, K. V., & Dishburger, H. J. (1983). A rapid method for the estimation of the environmental parameters octanol/water partition coefficient, soil sorption constant, water to air ratio and water solubility. Residue Reviews, 85, 17–28.
Tilak, K. S., & Kumari, R. S. (2009). Acute toxicity of Nuvan®, an organophosphate to freshwater fish Ctenopharyngodon idella and its effect on oxygen consumption. Journal of Environmental Biology, 30(6), 1031–1033.
Uner, N., Sevgiler, Y., & Piner, P. (2010). Tissue-specific in vivo inhibition of cholinesterases by the organophosphate fenthion in Oreochromis niloticus. Environmental Toxicology, 25(4), 391–399. https://doi.org/10.1002/tox.20514.
USEPA. U.S. Environmental Protection Agency (2000). The use of data on cholinesterase inhibition for risk assessments of organophosphorous and carbamate pesticides. Washington, DC. Available in: https://www.epa.gov/sites/production/files/2015-07/documents/cholin.pdf. Accessed in: 18 Apr. 2018.
USEPA. U.S. Environmental Protection Agency (2017). Pesticides Industry Sales and Usage: 2008–2012 Market Estimates. Washington, DC. Available in: https://www.epa.gov/sites/production/files/2017-01/documents/pesticides-industry-sales-usage-2016_0.pdf. Accessed in: 18 Apr. 2018.
Viana-Medeiros, P. F., Bellinato, D. F., Martins, A. J., & Valle, D. (2017). Insecticide resistance, associated mechanisms and fitness aspects in two Brazilian Stegomyia aegypti (=Aedes aegypti) populations. Medical and Veterinary Entomology, 31, 340–350. https://doi.org/10.1111/mve.12241.
World Health Organization. (2018). Dengue and Severe Dengue. Factsheet on dengue. Available in: http://www.who.int/news-room/fact-sheets/detail/dengue-and-severe-dengue. Accessed in: 13 Sep. 2018.
Zaidi, N., & Soltani, N. (2011). Environmental risks of two chitin synthesis inhibitors on Gambusia affinis: chronic effects on growth and recovery of biological responses. Biological Control, 59, 106–113. https://doi.org/10.1016/j.biocontrol.2011.04.001.
Zhang, Z. Y., Yu, X. Y., Wang, D. L., Yan, H. J., & Liu, X. J. (2010). Acute toxicity to zebrafish of two organophosphates and four pyrethroids and their binary mixtures. Pest Management Science, 66, 84–89. https://doi.org/10.1002/ps.1834.
Acknowledgments
We thank the Ministry of health of Brazil for providing the formulations of the larvicides, and the Aquaculture Center of the São Paulo State University (CAUNESP) and its employees, especially Márcio Alves, for providing the fishes for this work.
Funding
This study was financially supported by the Brazilian National Council for Scientific and Technological Development (CNPq grant number: 152513/2010-8).
Author information
Authors and Affiliations
Contributions
All authors contributed equally.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 293 kb)
Rights and permissions
About this article
Cite this article
Abe, F.R., Machado, A.A., Coleone, A.C. et al. Toxicity of Diflubenzuron and Temephos on Freshwater Fishes: Ecotoxicological Assays with Oreochromis niloticus and Hyphessobrycon eques. Water Air Soil Pollut 230, 77 (2019). https://doi.org/10.1007/s11270-019-4128-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-019-4128-7