Abstract
This study investigated the effect of three common quorum sensing signal molecules on anaerobic granular sludge (AnGS) in unbalanced nitrogen supply. Oligotrophic nitrogen supply transiently increased the level of N-butanoyl-homoserine lactone (C4-HSL), maintaining the stability of granules by encouraging synthesis of loosely bound extracellular polymeric substance (LB-EPS). Meanwhile, the decreasing autoinducer-2 (AI-2) content and increasing diffusible signal factor (DSF) content made a combined effect on AnGS, and then it recombined the regulation from N-acyl-homoserine lactones (AHLs). In addition, eutrophic nitrogen supply enhanced C4-HSL and N-hexanoyl-homoserine lactone (C6-HSL) secretion, suggesting that it increases loose bound protein (LB-PN) level to survive in the suppression of nitrogen supply. During the unbalanced nitrogen supply process, the weaker positive effect of AI-2 and enhanced negative effect of DSF reduced relative hydrophobicity (RH) and granular strength, which made a decline of granular diameter and a degeneration of granular sludge in nitrogen oligotrophic reactor (OR) and nitrogen eutrophic reactor (ER). Therefore, the effect of the interactions among multiple signal molecules should be further elucidated in future study.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Many microorganisms use quorum sensing (QS) signal molecules for cell density-dependent gene regulation. Generally, QS involves multiple regulatory systems (Galloway et al. 2011), including N-acyl-homoserine lactones (AHLs) family, autoinducer-2 (AI-2), and diffusible signal factor (DSF). Bacterial QS has been extensively studied as a regulator of bacterial growth (Xiong and Liu 2012), in biofouling control (Yeon et al. 2009), in sludge granulation (Ren et al. 2010, Tan et al. 2014), and in other aspects of environmental sciences. Previously, we have identified three types of signal molecules in anaerobic granular sludge (AnGS), namely, intraspecific signal molecules (AHLs), interspecific signal molecules (AI-2), and intra- and interspecific signal molecules (DSF) (Feng et al 2014). Our previous work and others have certified that AHLs and AI-2 promote microorganism granulation, while DSF exerts the opposite effect (Deng et al. 2011, March and Bentley 2004, Ding et al. 2015).
To maintain normal growth and metabolic processes in the sludge microorganisms of wastewater treatment processes, a stable operation of the sewage treatment unit and correct proportions of nutrients, especially the cell-sustaining elements carbon (C), nitrogen (N) and phosphorus (P), are required. The chemical oxygen demand (COD):N:P ratio of the influent substrate used in normal biochemical treatment of anaerobic sludge is 200:5:1. In actual industrial wastewater, the desired COD:N:P range is decided by the special production process and it is difficult to maintain ideal level for microorganisms. For instance, the nitrogen content in brewery, pharmaceutical, tanning, and dyeing wastewater is usually too low for bacterial metabolism, whereas that in the wastewater from fertiliser production or livestock breeding is excessively high. Previous studies have shown that the nutrients in influent substrate alter the activity, community and quantity of sludge (Hao et al. 2016). For example, nitrogen-poor substrate promotes the multiplication of filamentous bacteria with low nutritional requirements and sludge bulking, reducing the settleability of the sludge and increasing sludge washout. In the worst-case scenario, the sewage system collapses (Kapeler and Gujer 1994). In contrast, excessive nitrogen in the substrate, usually in the form of eutrophic ammonia, inhibits bacterial activity, leading to poor flocculation, settleability, and dewaterability of the sludge (Ye et al. 2011). These studies highlight the importance of the nutritional conditions on the metabolism and communities of biological sludge.
Because of the different ratios of C and N elements in metabolite and signal molecules, different substrate supplies will influence the microbial metabolic processes and the synthesis of signal molecules (Decho et al. 2011). Meanwhile, the varying environmental conditions, such as pH, temperature, ions, and other nutritions, affect the behaviors of signal molecules which have unstable molecular structures (Decho et al. 2009; Decho et al. 2011). Therefore, the way of nutrient supply may regulate QS mechanism by influencing the actual concentrations of signal molecules.
Recent papers have clarified the effects of nitrogen supply on granular sludge (Ye et al. 2011). Nitrogen concentration is correlated with the physicochemical properties of granular sludge, including extracellular polymeric substance (EPS) production and components, flocculation, and relative hydrophobicity which are affected by the C to N ratio of the influent (Ye et al. 2011). The producing process of metabolites needs the participation of basic nutritive elements. These investigations have revealed the effects of influent nutrient elements on the microbial and macroscopic properties of the sludge, but the underlying mechanisms remain too incomplete to develop a formal theory. In addition, QS signals are involved in restructuring the granular sludge community, including the granulation process (Xiong and Liu 2010; Zhang et al. 2011) and dispersion (Tan et al. 2014). The present study aims to clarify the variation of different signal molecules in unbalanced nitrogen supply and investigate the physicochemical properties of granular sludge mediated by QS. The results provide a new reference for further investigating the QS mechanisms of anaerobic sludge.
2 Materials and Methods
2.1 Experimental Configuration and Operating Conditions
The experiment was conducted in an expanded granular sludge bed with a total volume of 5 L. The reactor containing anaerobic granular sludge (20 g SS/L) was incubated at 30 °C in a greenhouse. The sludge was obtained from an expanded granular sludge bed of a paper-making factory (Jiaxing, China). The ratio of mixed liquor volatile suspended solids (MLVSS) to mixed liquor suspended solids ratio (MLSS) in the seeding sludge was 80 %. The buffer performance was determined by NaHCO3 addition, and the COD to alkalinity ratio (COD to Alk) was 3. The synthetic wastewater (COD:N:P = 200:5:1) consisted of nutrients (sucrose, NH4Cl, and KP2PO4) and trace elements, with an organic loading rate (OLR) of 8 kg COD/m3/day (COD = 4000 mg/L) and a COD removal efficiency of approximately 80 %. The hydraulic retention time (HRT) was 12 h.
The C to N ratios were controlled as follows: COD:N:P = 200:1:1 (nitrogen oligotrophic reactor, OR), COD:N:P = 200:5:1 (nitrogen moderate reactor, MR), and COD:N:P = 200:20:1 (nitrogen eutrophic reactor, ER). The C to N ratio was altered once the three reactors had reached stable state (26th HRT), and the nitrogen imbalance was continued for 40 HRT. The samples were determined in stable state (S0), 36th (S1), 46th (S2), and 66th (S3) HRT.
2.2 Signal Molecules Add-Back Studies
Many researchers have reported the regulation of the production of EPS by AHLs (Shrout and Nerenberg 2012; Tan et al. 2014; Wang et al. 2012), but roles of the definite position or component from AHLs and AI-2 were still not clear. Besides, the role of DSF had been confirmed in the microorganism, except for that in the sludge system. In this research, N-butanoyl-homoserine lactone (C4-HSL), AI-2 and DSF, three types of signal molecules in AnGS were used as exogenous additions for AnGS to investigate the potential effect of signal molecules. For example, the standards of C4-HSL and DSF (Sigma, USA) were added into reactors with synthetic wastewater and the final concentrations were 100 nM and 4.7 μM, respectively. And synthetic wastewater without signal molecules standard was regard as the negative control of AHLs and DSF. As for AI-2, the centrifugal supernatant of the V. haveyi BB170 culture (OD600 ≈ 1.0) was collected and replaced the added standards in AnGS after filtering through a 0.22-μm syringe filter. The culture containing AI-2 was used as the solvent of synthetic wastewater. And flesh culture mixed with synthetic wastewater was included as its medium control. The study lasted 6 days at 30 °C and at 130 rpm. The contents of protein (PN) and polysaccharide (PS) were measured at the 6th day.
2.3 Extraction of Signal Molecules
The distribution of signal molecules in anaerobic granular sludge was reported in our previous study (Feng et al. 2014). Referring to that study, the 100 mL sludge–water mixture was pretreated by first dividing into sludge and water. The sludge was crushed and resuspended in the water. The AHLs and DSF in the pretreated mixture were extracted for subsequent analysis by adding an equivalent volume of ethyl acetate, repeating extraction three times, then evaporating to dryness at 25 °C and redissolving the dried material in 2 mL of 50 % acetonitrile. The samples were stored at −70 °C before filtering to analyse. For AI-2 analysis, 1 mL of the supernatant from the sludge-water mixture was collected and filtered through a 0.22-μm syringe filter, then stored at −20 °C until required.
2.4 Analytical Methods
2.4.1 Ultra performance liquid chromatography–tandem mass spectrometry (UPLC–MS/MS) determination of signal molecules
AHLs were analysed using a Waters UPLC–MS/MS (Xevo TQD, Waters, USA) system with a waters ACQUITY UPLC BEH C-18 column (1.7 μm d, 2.1 × 50 mm, Waters, Ireland). The nebuliser gas was N2; the flow rate of the dryer gas was set to 800 L/h. The heated capillary and voltage were maintained at 500 °C and 0.5 kV, respectively (Maurera et al. 2013). The samples were then passed through a 0.22-μm filter and eluted with ammonium acetate buffer (0.05 M) and acetonitrile.
2.4.2 High-Performance Liquid Chromatography (HPLC) Determination of Signal Molecules
DSF was analysed using a Waters HPLC system with an X Bridge C-18 column (5 μm day, 4.6 × 250 mm). The samples were then passed through a 0.22-μm filter in readiness for HPLC analysis. More precisely, the DSF was eluted with 80 % acetonitrile in water at a flow rate of 0.7 mL min−1 with a separation injection volume of 10 μL. The wavelength was set to 210 nm (Feng et al. 2014).
2.4.3 AI-2 Determination
The AI-2 activity was measured by an indicator strain, Vibrio harveyi BB170, which produces light in the presence of AI-2. The bioluminescence was measured by a multifunctional microplate reader (SpectraMax M5, USA), as described in our previous studies (Feng et al. 2014).
2.4.4 Other Methods
COD was measured by potassium dichromate fast digestion-spectrophotometric method using a DR2800 spectrophotometer (Hach Company, Loveland, CO, USA) and the granular diameter was measured by wet sieving method. Volatile fatty acids (VFAs) were measured by gas chromatography (GC 7890 II, Shanghai Tianmei Science Instrument Co., Ltd., Shanghai, China) equipped with a flame ionization detector (Huang et al. 2013). Relative hydrophobicity (RH) was measured by bacterial adhesion to hydrocarbons (Chang and Lee 1998).
The easily extractable EPS were isolated by a two-step heating EPS extraction procedure (Yang and Li 2009). Easily extractable EPS consists of loosely bound EPS (LB-EPS) and tightly bound EPS (TB-EPS). The EPS extraction solutions, including protein (PN) and polysaccharide (PS), were analysed by the phenol/sulfuric acid method (Dubois et al. 1956) and the Lowry method (Lowry et al. 1951), respectively.
The strength of the AnGS was measured by a modified Ghangrekar method (Ghangrekar et al. 1996). The results were expressed in terms of the integrity coefficient (IC), defined as the ratio of residual solids to the total weight of the granular sludge.
All data are expressed as mean ± standard error from three replicates. Statistical analyses (one-way analysis of variance) were conducted using SPSS software (version 17.0).
3 Results and Discussion
3.1 Bioreactor Performance
The COD removal efficiencies of the three reactors in first 26 HRTs (prior to nitrogen imbalance) were over 75 % (Fig. 1a). Nitrogen imbalance reduced the removal efficiencies in the OR to approximately 65 % and accumulated acetic acid, propionic acid, and n-butyric acid in the end of experiment, while those in the MR and ER remained stable at approximately 80 and 75 %, respectively. According to the NH4 +-N concentration in the OR (Fig. 1b), nitrogen limitation restrained the microbial metabolism, reducing the C assimilation. In the ER, NH4 +-N accumulated to a maximum of 283 mg/L, and its toxicity to bacteria reduced substrate assimilation and other microbial activities (Sile et al. 2010).
The COD removal efficiencies (a) and NH4 +-N concentration (b) of three reactors in unbalanced nitrogen supply. The NH4 +-N concentrations in synthetic wastewater were 20 mg/L (OR, C:N:P = 1000:5:5), 100 mg/L (MR, C:N:P = 1000:25:5), and 400 mg/L (ER, C:N:P = 1000:100:5). S1, S2, and S3 represent 36th, 46th, and HRT in unbalanced nitrogen supply
3.2 Signal Molecules in Unbalanced Nitrogen Supply
3.2.1 AHL Content in Unbalanced Nitrogen Supply
Table 1 showed that the different trends of various AHLs were an increasing process in the early stages of unbalanced nitrogen supply and a decreasing process in the late of unbalanced nitrogen supply. The content of C4-HSL in the OR was significantly higher than that in the control (p < 0.05). Meanwhile, in the ER, the content of C4-HSL dropped to the level of the control in S3, but C6-HSL was higher than that in the control and OR (p < 0.05). As for 3-oxo-C6-HSL, the contents in the OR and ER after 10 HRT in unbalanced nitrogen supply were always lower than that in the control (p < 0.05).
Because of the better water solubility, diffusivity, and more simple molecular structure (Feng et al. 2014), C4-HSL became the most sensitive kind of signal molecule in unbalanced nitrogen supply conditions, especially its relative high content in inadequate nitrogen supply. As for other signal molecules with longer acyl chain or substituents, C6-HSL and 3-oxo-C6-HSL tended to play a more important role in eutrophic nitrogen environment. The above results indicated that the instantaneous variation of environment forced sludge to secrete more AHLs in the early of unbalanced nitrogen supply. These AHLs were used to adjust microbial behavior and adapt to a new surroundings (Decho et al. 2011; Shrout and Nerenberg 2012). In addition, a long-time of inadequate or excessive nitrogen supply would make a selective change of AHLs family, and then form a new AHLs-QS regulating system. It would screen the surviving bacteria that were suited to the environment of unbalanced nitrogen supply (Magdalena et al. 2012).
3.2.2 Relative AI-2 Content in Unbalanced Nitrogen Supply
As shown in Fig. 2, in the oligotrophic nitrogen supply, the secretion of AI-2 content was significantly declined (p < 0.05), and it was lower than that in the control. Interestingly, the environment of eutrophic nitrogen supply made a rapid increase of AI-2 content in S1, and the content was higher than that in the control (p < 0.05). In the next stage, the secretion of AI-2 content reduced to 0.40 ± 0.05 and it significantly lower than that in the control and OR (p < 0.05).
The above processes suggested that the communication and effect of interspecific signal molecules would decrease to a lower, but more stable level in the unbalanced nitrogen supply. In other words, lower AI-2 content meant lower activity of microbial communication or fewer kinds of bacteria. Besides, the increasing content of AI-2 might be a stress response to the impact of short-term high concentration of nitrogen. During the oligotrophic nitrogen supply, the competition for limited nutrients among microorganism would make a decline on community diversity (Wu et al. 2012). Meanwhile, the accumulation of high concentration of nitrogen would also form a unidirectional filtering for microbial strains by altering metabolic pathway of anaerobic sludge fermentation (Fu et al. 2012).
3.2.3 DSF Content in Unbalanced Nitrogen Supply
Figure 3 showed that the DSF content in MR has a slight decrease as the steady and suitable environment. The DSF content under the eutrophic nitrogen supply had a continuous growth, and it was 159.42 ± 4.60 μg/L in S3 stage, which was four times as great as that of the control content. And it might be the forced behavior of microorganism to the changed environment. The DSF content in the oligotrophic nitrogen supply also increased up to the same level of ER in S2 and then decreased in S3 stage, but it was still higher than that in the control. Therefore, the effect of DSF on granular sludge was enhanced at the stage of the changed nitrogen supply. In addition, a more lasting and effective role of DSF seemed to be obviously under eutrophic nitrogen supply condition. It is not to say that ammonia toxicity would result in a higher DSF effect in ER than that in OR, and its relationship needed to have a further study.
3.3 The Role of Signal Molecules in the Properties of AnGS
3.3.1 EPS
The roles of the added signal molecules to EPS in AnGS were shown in Table 2. It indicated that the addition of C4-HSL only had a positive effect on LB-PN production. On the other hand, DSF played a negative role in the synthesis of LB-PN, TB-PN, and LB-PS, the role of which in AnGS was opposite to that of AI-2.
Table 3 listed the EPS contents under different nitrogen supply conditions. The LB-EPS (PN and PS) contents increased throughout the incubation in OR, and they were higher than that in the control and ER (p < 0.05). However, the TB-EPS (PN and PS) contents in OR and ER under the unbalanced nitrogen supply did not have a significant difference with that in the control (p < 0.05). Comparing the EPS contents in the three reactors, the LB- and TB-EPS contents in the OR were higher than that in the ER after a certain effective period of unbalanced nitrogen supply.
As a well-studied representative metabolite, EPS alters the resilience of sludge granules and is regulated by QS (Wang et al, 2012; Shrout and Nerenberg 2012), including AHLs, AI-2, and DSF (Shrout and Nerenberg 2012; Tan et al. 2014; Wang et al. 2012). The add-back test results have summarized the explicit role of signal molecules to the synthesis of EPS. According to signal molecule contents in three reactors, the variation of LB- and TB-EPS contents in ER could only suggest that C4-HSL and C6-HSL would promote the synthesis of LB-PN in the eutrophic nitrogen environment. And the EPS would be used to resist the toxicity inhibition of high concentration of ammonia nitrogen and maintain the stability of granular sludge. Besides, the increase of LB-EPS (PN and PS) contents in OR indicated that C4-HSL and AI-2 regulated the synthesis of EPS synchronously. However, the stabilization of TB-EPS content indicated that the promotion to the synthesis of TB-PN from AI-2 was restrained by the existence of DSF. In addition, the effect of C6-HSL to EPS in oligotrophic nitrogen supply was still ambiguous.
The above results indicated that one or several types of signal molecules coordinated the production of EPS in AnGS under unbalanced nitrogen supply, mainly to increase the closely bound one. As a result, the EPS would be used to maintain the structure stability of granules.
3.3.2 Relative Hydrophobicity
The aggregation ability of sludge microorganisms is assayed by RH. Figure 4 showed that the RH in ER and OR decreased and they were only 50 % in the control. In addition, the RHs were similar between the unbalanced nitrogen supply conditions (p > 0.05). The variation of RH content in Fig. 4 showed a positive correlation between RH and the relative AI-2 content, but an opposite change between RH and the DSF content.
The results indicated that the decrease of AI-2 content and the increase of DSF content played a combined role on granular sludge under unbalanced nitrogen supply and that made a decline of RH and degeneration of granules. In addition, the lower content of AI-2 and higher content of DSF in ER suggested that the higher content of C6-HSL might be a reason for maintaining the relative considerable level of RH with that in OR.
3.3.3 Granular Strength
Granular strength is indicated by IC. A higher IC indicates a higher granular strength and a more stable granular structure. As shown in Fig. 5, the IC remained around 88 ∼ 90 % in MR. Besides, in the OR and ER, the variations of granular strength were consistent with that of the corresponding RH. And the IC in OR and ER fell to 80.25 ± 0.98 % and 79.68 ± 1.23 %, respectively, which were significantly lower than that in the control (p < 0.05). The change of IC in three reactors indicated that the weaker positive effect of AI-2 and stronger negative effect of DSF was the major reason of the granular sludge degradation.
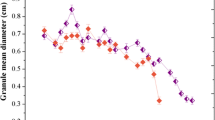
3.3.4 Granular Diameter
Table 4 showed that the granular diameters in OR and ER were markedly reduced, and they decreased by 10.19 and 13.56 %, respectively. The degeneration of AnGS in granular diameter was followed by the decrease of RH and granular strength in unbalanced nitrogen supply.
The above results demonstrated that few AHLs under unbalanced nitrogen supply would tend to improve the stability of granular sludge by increasing the production of LB-EPS, but that was not enough to make up for the degradation of granules by the combination effect of AI-2 and DSF. As a result, the AnGS became to be degraded and disintegrated under the coordination of several types of signal molecules.
4 Conclusion
As granular sludge is a complex system of microbial aggregates, it must contain a variety of signal molecules that simultaneously affect the formation and maintenance of the anaerobic sludge (Deng et al. 2010). The present study suggests that an unknown, multi-molecular signal system regulates the distribution and effect of QS in sludge microorganism communities.
This research suggested that with unbalanced nitrogen supply of carbon to nitrogen (C to N) = 200:1 and 200:20, the content of AHLs increased at first and decreased afterwards, and DSF content had an increasing change which was quite different with that of AI-2. In the nitrogen eutrophic reactor, AHLs, such as C4-HSL and C6-HSL, governed the production of LB-PN to survive in this environment of unbalanced nutrient. Moreover, in oligotrophic nitrogen supply, the combination of C4-HSL, AI-2, and DSF made an increase of LB-EPS secretion, which contributed to maintain the stability of granular structure. Whereas, in nitrogen unbalanced reactors, the regulation of AI-2 and DSF made a decline of RH, granular strength and diameter to degenerate the granular sludge. The findings provide a new reference for further investigating the multi-molecular signal mechanisms of anaerobic granular sludge.
Abbreviations
- QS:
-
Quorum sensing
- AHLs:
-
Acyl-homoserine lactones
- AI-2:
-
Auto-inducers-2
- DSF:
-
Diffusible signal factor
- AnGS:
-
Anaerobic granular sludge
- COD:
-
Chemical oxygen demand
- EPS:
-
Extracellular polymeric substance
- MLVSS:
-
Mixed liquor volatile suspended solids
- MLSS:
-
Mixed liquor suspended solids
- OLR:
-
Organic loading rate
- HRT:
-
Hydraulic retention time
- OR:
-
Nitrogen oligotrophic reactor
- MR:
-
Nitrogen moderate reactor
- ER:
-
Nitrogen eutrophic reactor
- C4-HSL:
-
N-butanoyl-homoserine lactone
- UPLC-MS/MS:
-
Ultra performance liquid chromatography-tandem mass spectrometry
- HPLC:
-
High-performance liquid chromatography
- RH:
-
Relative hydrophobicity
- LB:
-
Loosely bound
- TB:
-
Tightly bound
- PN:
-
Protein
- PS:
-
Polysaccharide
- IC:
-
Integrity coefficient
- VFAs:
-
Volatile fatty acids
References
Chang, I. S., & Lee, C. H. (1998). Membrane filtration characteristics in membrane-coupled activated sludge system—the effect of physiological states of activated sludge on membrane fouling. Desalination, 120, 221–233.
Decho, A. W., Visscher, P. T., Ferry, J., Kawaguchi, T., He, L., Przekop, K. M., Norman, R. S., & Ried, R. P. (2009). Autoinducers extracted from microbial mates reveal a surprising diversity of N-acylhomoserine lactones (AHLs) and abundance changes that may relate to diel pH. Environmental Microbiology, 11(2), 409–420.
Decho, A. W., Frey, R. L., & Ferry, J. L. (2011). Chemical challenges to bacterial AHL signaling in the environment. Chemical Reviews, 111(1), 86–99.
Deng, Y., Wu, J., Eberl, L., & Zhang, L. H. (2010). Structural and functional characterization of diffusible signal factor family quorum-sensing signals produced by members of the Burkholderia cepacia complex. Appl Environ Microb, 76(14), 4675–4683.
Deng, Y., Wu, J. E., Tao, F., & Zhang, L. H. (2011). Listening to a new language DSF-based quorum sensing in gram-negative bacteria. Chemical Reviews, 111(1), 160–173.
Ding, Y. C., Feng, H. J., Huang, W. K., Li, N., Zhou, Y. Y., Wang, M. Z., Zhang, X. Q., & Shen, D. S. (2015). The effect of quorum sensing on anaerobic granular sludge in different pH conditions. Biochemical Engineering Journal, 103, 270–276.
Dubois, M., Gilles, K. A., Hamilton, J. K., Rebers, P. A., & Smith, F. (1956). Colorimetric method for determination of sugars and related substances. Analytical Chemistry, 28(3), 350–356.
Feng, H. J., Ding, Y. C., Wang, M. Z., Zhou, G. L., Zheng, X., He, H. Z., Zhang, X. Q., Shen, D. S., & Shentu, J. L. (2014). Where are signal molecules likely to be located in anaerobic granular sludge? Water Research, 50, 1–9.
Fu, B., Zhang, J., Fan, J., Wang, J., & Liu, H. (2012). Control of C/N ratio for butyric acid production from textile wastewater sludge by anaerobic digestion. Water Science and Technology, 65(5), 883–889.
Galloway, W. R. J. D., Hodgkinson, J. T., Bowden, S. D., Welch, M., & Spring, D. R. (2011). Quorum sensing in gram-negative bacteria small-molecule modulation of AHL and AI-2 quorum sensing pathways. Chemical Reviews, 111, 28–67.
Ghangrekar, M. M., Asolekar, S. R., Ranganathan, K. R., & Joshi, S. G. (1996). Experience with UASB reactor start-up under different operating conditions. Water Science and Technology, 34(5-6), 421–428.
Hao, L., Liss, S. N., & Liao, B. Q. (2016). Influence of COD:N ratio on sludge properties and their role in membrane fouling of a submerged membrane bioreactor. Water Research, 89, 132–141.
Huang, B. C., Feng, H. J., Wang, M. Z., Li, N., Cong, Y. Q., & Shen, D. S. (2013). The effect of C/N ratio on nitrogen removal in a bioelectrochemical system. Bioresource Technology, 132, 91–98.
Kapeler, J., & Gujer, W. (1994). Influences of wastewater composition and operating conditions on activated sludge bulking and scum formation. Water Science and Technology, 30(11), 181–189.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., & Randall, R. J. (1951). Protein measurement with the folin phenol reagent. The Journal of Biological Chemistry, 193, 265–275.
Magdalena, Z., Katarzyna, B., Agnieszka, C. K., Joanna, S., & Irena, W. B. (2012). Nitrogen removal from wastewater and bacterial diversity in activated sludge at different COD/N ratios and dissolved oxygen concentrations. Journal of Environmental Sciences, 24(6), 990–998.
March, J. C., & Bentley, W. E. (2004). Quorum sensing and bacterial cross-talk in biotechnology. Current Opinion in Biotechnology, 15(5), 495–502.
Maurera, C. K., Steinbacha, A., & Hartmanna, R. W. (2013). Development and validation of a UHPLC–MSMS procedure for quantification of the Pseudomonas Quinolone Signal in bacterial culture after acetylation for characterization of new quorum sensing inhibitors. J Pharmaceut Biomed Analysis, 86, 127–134.
Ren, T. T., Yu, H. Q., & Li, X. Y. (2010). The quorum-sensing effect of aerobic granules on bacterial adhesion, biofilm formation, and sludge granulation. Applied Microbiology and Biotechnology, 88(3), 789–797.
Shrout, J. D., & Nerenberg, R. (2012). Monitoring bacterial twitter: does quorum sensing determine the behavior of water and wastewater treatment biofilms? Environmental Science and Technology, 46(4), 1995–2005.
Sile, J. A., Brekelmans, J., Martín, M. A., Chica, A. F., & Martín, A. (2010). Impact of ammonia and sulphate concentration on thermophilic anaerobic digestion. Bioresource Technology, 101, 9040–9048.
Tan, C. H., Koh, K. S., Xie, C., Tay, M., Zhou, Y., Williams, R., Ng, W. J., Rice, S. A., & Kjelleberg, S. (2014). The role of quorum sensing signalling in EPS production and the assembly of a sludge community into aerobic granules. The ISME Journal, 8(6), 1186–1197.
Wang, M. Z., Zheng, X., He, H. Z., Shen, D. S., & Feng, H. J. (2012). Ecological roles and release patterns of acylated homoserine lactones in Pseudomonas sp. HF-1 and their implications in bacterial bioaugmentation. Bioresource Technology, 125, 119–126.
Wu, B., Yi, S., & Fane, A. G. (2012). Effect of substrate composition (C/N/P ratio) on microbial community and membrane fouling tendency of biomass in membrane bioreactors. Separation Science and Technology, 47(3), 440–445.
Xiong, Y., & Liu, Y. (2010). Involvement of ATP and autoinducer-2 in aerobic granulation. Biotechnology and Bioengineering, 105(1), 51–58.
Xiong, Y., & Liu, Y. (2012). Essential roles of eDNA and AI-2 in aerobic granulation in sequencing batch reactors operated at different settling times. Applied Microbiology and Biotechnology, 93(6), 2645–2651.
Yang, S. F., & Li, X. Y. (2009). Influences of extracellular polymeric substances (EPS) on the characteristics of activated sludge under non-steady-state conditions. Process Biochemistry, 44(1), 91–96.
Ye, F., Ye, Y., & Li, Y. (2011). Effect of C/N ratio on extracellular polymeric substances (EPS) and physicochemical properties of activated sludge flocs. Journal of Hazardous Materials, 188(1-3), 37–43.
Yeon, K. M., Cheong, W. S., Oh, H. S., Lee, W. N., Hwang, B. K., Lee, C. H., Beyenal, H., & Lewandowski, Z. (2009). Quorum sensing a new biofouling control paradigm in a membrane bioreactor for advanced wastewater treatment. Environmental Science and Technology, 43, 380–385.
Zhang, S. H., Yu, X., Guo, F., & Wu, Z. Y. (2011). Effect of interspecies quorum sensing on the formation of aerobic granular sludge. Water Science and Technology, 64(6), 1284–1290.
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (51208464), Program of Xinmiao Talents in Zhejiang Province (2014R408090), the Project of Education Department of Zhejiang Province (Y201430857), and an Analytical Test Plan Project from the Science and Technology Department in Zhejiang Province (2016C37052).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ding, Y., Feng, H., Zhao, Z. et al. The Effect of Quorum Sensing on Mature Anaerobic Granular Sludge in Unbalanced Nitrogen Supply. Water Air Soil Pollut 227, 334 (2016). https://doi.org/10.1007/s11270-016-3037-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-016-3037-2